Abstract
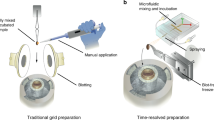
With the advent of direct electron detectors, cryo-EM has become a popular choice for molecular structure determination. Among its advantages over X-ray crystallography are (1) no need for crystals, (2) much smaller sample volumes, and (3) the ability to determine multiple structures or conformations coexisting in one sample. In principle, kinetic experiments can be done using standard cryo-EM, but mixing and freezing grids require several seconds. However, many biological processes are much faster than that time scale, and the ensuing short-lived states of the molecules cannot be captured. Here, we lay out a detailed protocol for how to capture such intermediate states on the millisecond time scale with time-resolved cryo-EM.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Fischer N, Konevega AL, Wintermeyer W, Rodnina MV, Stark H (2010) Ribosome dynamics and tRNA movement by time-resolved electron cryomicroscopy. Nature 466(7304):329–333. https://doi.org/10.1038/nature09206
Chen B, Kaledhonkar S, Sun M, Shen BX, Lu ZH, Barnard D, Lu TM, Gonzalez RL, Frank J (2015) Structural dynamics of ribosome subunit association studied by mixing-spraying time-resolved cryogenic electron microscopy. Structure 23(6):1097–1105. https://doi.org/10.1016/j.str.2015.04.007
Feng X, Fu Z, Kaledhonkar S, Jia Y, Shah B, Jin A, Liu Z, Sun M, Chen B, Grassucci RA, Ren Y, Jiang H, Frank J, Lin Q (2017) A fast and effective microfluidic spraying-plunging method for high-resolution single-particle cryo-EM. Structure 25(4):663–670 e663. https://doi.org/10.1016/j.str.2017.02.005
Fu Z, Kaledhonkar S, Borg A, Sun M, Chen B, Grassucci RA, Ehrenberg M, Frank J (2016) Key intermediates in ribosome recycling visualized by time-resolved cryoelectron microscopy. Structure 24(12):2092–2101. https://doi.org/10.1016/j.str.2016.09.014
Lu ZH, Shaikh TR, Barnard D, Meng X, Mohamed H, Yassin A, Mannella CA, Agrawal RK, Lu TM, Wagenknecht T (2009) Monolithic microfluidic mixing-spraying devices for time-resolved cryo-electron microscopy. J Struct Biol 168(3):388–395. https://doi.org/10.1016/j.jsb.2009.08.004
Shaikh TR, Yassin AS, Lu ZH, Barnard D, Meng X, Lu TM, Wagenknecht T, Agrawal RK (2014) Initial bridges between two ribosomal subunits are formed within 9.4 milliseconds, as studied by time-resolved cryo-EM. Proc Natl Acad Sci U S A 111(27):9822–9827. https://doi.org/10.1073/pnas.1406744111
White HD, Thirumurugan K, Walker ML, Trinick J (2003) A second generation apparatus for time-resolved electron cryo-microscopy using stepper motors and electrospray. J Struct Biol 144(1–2):246–252. https://doi.org/10.1016/j.jsb.2003.09.027
White HD, Walker ML, Trinick J (1998) A computer-controlled spraying-freezing apparatus for millisecond time-resolution electron cryomicroscopy. J Struct Biol 121(3):306–313. https://doi.org/10.1006/jsbi.1998.3968
Tivol WF, Briegel A, Jensen GJ (2008) An improved cryogen for plunge freezing. Microsc Microanal 14(5):375–379. https://doi.org/10.1017/s1431927608080781
Suloway C, Pulokas J, Fellmann D, Cheng A, Guerra F, Quispe J, Stagg S, Potter CS, Carragher B (2005) Automated molecular microscopy: the new Leginon system. J Struct Biol 151(1):41–60. https://doi.org/10.1016/j.jsb.2005.03.010
Zheng SQ, Palovcak E, Armache JP, Verba KA, Cheng YF, Agard DA (2017) MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat Methods 14(4):331–332. https://doi.org/10.1038/nmeth.4193
Grant T, Grigorieff N (2015) Measuring the optimal exposure for single particle cryo-EM using a 2.6 angstrom reconstruction of rotavirus VP6. elife 4:e06980. https://doi.org/10.7554/eLife.06980
Rohou A, Grigorieff N (2015) CTFFIND4: fast and accurate defocus estimation from electron micrographs. J Struct Biol 192(2):216–221. https://doi.org/10.1016/j.jsb.2015.08.008
Zhang K (2016) Gctf: real-time CTF determination and correction. J Struct Biol 193(1):1–12. https://doi.org/10.1016/j.jsb.2015.11.003
Scheres SHW (2012) RELION: implementation of a Bayesian approach to cryo-EM structure determination. J Struct Biol 180(3):519–530. https://doi.org/10.1016/j.jsb.2012.09.006
Acknowledgments
This research has been supported by the HHMI and NIH R01 GM29169 and GM55440 (to J.F.) and NIH AR40964 and NIH Fogarty Senior International Fellowship (to H.W.).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Kaledhonkar, S., Fu, Z., White, H., Frank, J. (2018). Time-Resolved Cryo-electron Microscopy Using a Microfluidic Chip. In: Marsh, J. (eds) Protein Complex Assembly. Methods in Molecular Biology, vol 1764. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7759-8_4
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7759-8_4
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7758-1
Online ISBN: 978-1-4939-7759-8
eBook Packages: Springer Protocols