Abstract
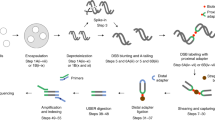
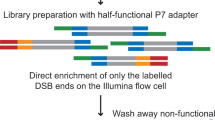
DNA double-strand breaks (DSBs) are major DNA lesions that are constantly formed during physiological processes such as DNA replication, transcription, and recombination, or as a result of exogenous agents such as ionizing radiation, radiomimetic drugs, and genome editing nucleases. Unrepaired DSBs threaten genomic stability by leading to the formation of potentially oncogenic rearrangements such as translocations. In past few years, several methods based on next-generation sequencing (NGS) have been developed to study the genome-wide distribution of DSBs or their conversion to translocation events. We developed Breaks Labeling, Enrichment on Streptavidin, and Sequencing (BLESS), which was the first method for direct labeling of DSBs in situ followed by their genome-wide mapping at nucleotide resolution (Crosetto et al., Nat Methods 10:361–365, 2013). Recently, we have further expanded the quantitative nature, applicability, and scalability of BLESS by developing Breaks Labeling In Situ and Sequencing (BLISS) (Yan et al., Nat Commun 8:15058, 2017). Here, we first present an overview of existing methods for genome-wide localization of DSBs, and then focus on the BLESS and BLISS methods, discussing different assay design options depending on the sample type and application.
Similar content being viewed by others
References
Jackson SP, Bartek J (2009) The DNA-damage response in human biology and disease. Nature 461:1071–1078
van Gent DC, Hoeijmakers JH, Kanaar R (2001) Chromosomal stability and the DNA double-strandedbreak connection. Nat Rev Genet 2:196–206
McKinnon PJ, Caldecott KW (2007) DNA strand break repair and human genetic disease. Annu Rev Genomics Hum Genet 8:37–55
Porteus MH, Carroll D (2005) Gene targeting using zinc finger nucleases. Nat Biotechnol 23:967–973
Joung JK, Sander JD (2013) TALENs: a widely applicable technology for targeted genome editing. Nat Rev Mol Cell Biol 14:49–55
Hsu PD, Lander ES, Zhang F (2014) Development and applications of CRISPR-Cas9 for genome engineering. Cell 157:1262–1278
Zetsche B, Gootenberg JS, Abudayyeh OO et al (2015) Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell 163:759–771
Tsai SQ, Keith Joung J (2016) Defining and improving the genome-wide specificities of CRISPR-Cas9nucleases. Nat Rev Genet 17:300–312
Szilard RK, Jacques P-E, Laramée L et al (2010) Systematic identification of fragile sites via genome-wide location analysis of gamma-H2AX. Nat Struct Mol Biol 17:299–305
Iacovoni JS, Caron P, Lassadi I et al (2010) High-resolution profiling of gammaH2AX around DNA double strand breaks in the mammalian genome. EMBO J 29:1446–1457
Madabhushi R, Gao F, Pfenning AR et al (2015) Activity-induced DNA breaks govern the expression of neuronal early-response genes. Cell 161:1592–1605
Crosetto N, Mitra A, Silva MJ et al (2013) Nucleotide-resolution DNA double-strand break mapping by next-generation sequencing. Nat Methods 10:361–365
Ran FA, Cong L, Yan WX et al (2015) In vivo genome editing using Staphylococcus aureus Cas9. Nature 520:186–191
Slaymaker IM, Gao L, Zetsche B et al (2016) Rationally engineered Cas9 nucleases with improved specificity. Science 351:84–88
Lensing SV, Marsico G, Hänsel-Hertsch R et al (2016) DSBCapture: in situ capture and sequencing of DNA breaks. Nat Methods 13:855–857
Canela A, Sridharan S, Sciascia N et al (2016) DNA breaks and end resection measured genome-wide by end sequencing. Mol Cell 63:898–911
Yan WX, Mirzazadeh R, Garnerone S et al (2017) BLISS is a versatile and quantitative method for genome-wide profiling of DNA double-strand breaks. Nat Commun 8:15058
Kim D, Bae S, Park J et al (2015) Digenome-seq: genome-wide profiling of CRISPR-Cas9 off-targeteffects in human cells. Nat Methods 12:237–243. 1 p following 243
Kim D, Kim J, Hur JK et al (2016) Genome-wide analysis reveals specificities of Cpf1 endonucleases in human cells. Nat Biotechnol 34:863–868
Tsai SQ, Zheng Z, Nguyen NT et al (2015) GUIDE-seq enables genome-wide profiling of off-targetcleavage by CRISPR-Cas nucleases. Nat Biotechnol 33:187–197
Kleinstiver BP, Tsai SQ, Prew MS et al (2016) Genome-wide specificities of CRISPR-Cas Cpf1 nucleases in human cells. Nat Biotechnol 34:869–874
Wang X, Wang Y, Wu X et al (2015) Unbiased detection of off-target cleavage by CRISPR-Cas9 and TALENs using integrase-defective lentiviral vectors. Nat Biotechnol 33:175–178
Frock RL, Hu J, Meyers RM et al (2015) Genome-wide detection of DNA double-stranded breaks induced by engineered nucleases. Nat Biotechnol 33:179–186
Wei P-C, Chang AN, Kao J et al (2016) Long neural genes harbor recurrent DNA break clusters in neural stem/progenitor cells. Cell 164:644–655
Kivioja T, Vähärautio A, Karlsson K et al (2012) Counting absolute numbers of molecules using unique molecular identifiers. Nat Methods 9:72–74
Baslan T, Kendall J, Rodgers L et al (2012) Genome-wide copy number analysis of single cells. Nat Protoc 7:1024–1041
Naumova N, Imakaev M, Fudenberg G et al (2013) Organization of the mitotic chromosome. Science 342:948–953
Dunnett CW (1955) A multiple comparison procedure for comparing several treatments with a control. J Am Stat Assoc 50:1096–1121
Zhang Y, Liu T, Meyer CA et al (2008) Model-based analysis of ChIP-Seq (MACS). Genome Biol 9:R137
Heinz S, Benner C, Spann N et al (2010) Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol Cell 38:576–589
Zang C, Schones DE, Zeng C et al (2009) A clustering approach for identification of enriched domains from histone modification ChIP-Seq data. Bioinformatics 25:1952–1958
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Mirzazadeh, R., Kallas, T., Bienko, M., Crosetto, N. (2018). Genome-Wide Profiling of DNA Double-Strand Breaks by the BLESS and BLISS Methods. In: Muzi-Falconi, M., Brown, G. (eds) Genome Instability. Methods in Molecular Biology, vol 1672. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7306-4_14
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7306-4_14
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7305-7
Online ISBN: 978-1-4939-7306-4
eBook Packages: Springer Protocols