Abstract
Th9 cells are associated with atopic and inflammatory diseases, and their increased levels and function correlate with the severity of symptoms in various inflammatory disorders including asthma, food allergy, atopic dermatitis, ulcerative colitis, and psoriatic arthritis. Thus, clinical trials are warranted to evaluate the role of Th9 cells in allergic diseases with the goal of controlling these ailments.
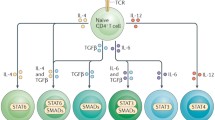
Circulating T cells (naïve or memory CD4+ T cells) purified from human blood and expanded using anti-CD3 and anti-CD28 antibodies can be treated with appropriate cytokines in order to polarize them to the Th9 phenotype as evidenced by their production of IL-9. When treated in vitro with cholecalciferol or 1,25(OH)2 vitamin D3, cells polarized under Th9 conditions significantly downregulate production of IL-9. The percentage of polarized Th9 memory cells from patients treated with steroids or other modalities can be monitored during clinical trials and compared to control populations.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Palmer MT, Lee YK, Maynard CL, Oliver JR, Bikle DD, Jetten AM, Weaver CT (2011) Lineage-specific effects of 1,25-dihydroxyvitamin D(3) on the development of effector CD4 T cells. J Biol Chem 286(2):997–1004. doi:10.1074/jbc.M110.163790
Chang HC, Sehra S, Goswami R, Yao W, Yu Q, Stritesky GL, Jabeen R, McKinley C, Ahyi AN, Han L, Nguyen ET, Robertson MJ, Perumal NB, Tepper RS, Nutt SL, Kaplan MH (2010) The transcription factor PU.1 is required for the development of IL-9-producing T cells and allergic inflammation. Nat Immunol 11(6):527–534. doi:10.1038/ni.1867
Goswami R, Jabeen R, Yagi R, Pham D, Zhu J, Goenka S, Kaplan MH (2012) STAT6-dependent regulation of Th9 development. J Immunol 188(3):968–975. doi:10.4049/jimmunol.1102840
Jabeen R, Goswami R, Awe O, Kulkarni A, Nguyen ET, Attenasio A, Walsh D, Olson MR, Kim MH, Tepper RS, Sun J, Kim CH, Taparowsky EJ, Zhou B, Kaplan MH (2013) Th9 cell development requires a BATF-regulated transcriptional network. J Clin Invest 123(11):4641–4653. doi:10.1172/JCI69489
Keating P, Munim A, Hartmann JX (2014) Effect of vitamin D on T-helper type 9 polarized human memory cells in chronic persistent asthma. Ann Allergy Asthma Immunol 112(2):154–162. doi:10.1016/j.anai.2013.11.015
Knoops L, Louahed J, Renauld JC (2004) IL-9-induced expansion of B-1b cells restores numbers but not function of B-1 lymphocytes in xid mice. J Immunol 172(10):6101–6106
Yao W, Tepper RS, Kaplan MH (2011) Predisposition to the development of IL-9-secreting T cells in atopic infants. J Allergy Clin Immunol 128(6):1357–1360.e1355. doi:10.1016/j.jaci.2011.06.019
Erpenbeck VJ, Hohlfeld JM, Volkmann B, Hagenberg A, Geldmacher H, Braun A, Krug N (2003) Segmental allergen challenge in patients with atopic asthma leads to increased IL-9 expression in bronchoalveolar lavage fluid lymphocytes. J Allergy Clin Immunol 111(6):1319–1327
Shimbara A, Christodoulopoulos P, Soussi-Gounni A, Olivenstein R, Nakamura Y, Levitt RC, Nicolaides NC, Holroyd KJ, Tsicopoulos A, Lafitte JJ, Wallaert B, Hamid QA (2000) IL-9 and its receptor in allergic and nonallergic lung disease: increased expression in asthma. J Allergy Clin Immunol 105(1 Pt 1):108–115
Brough HA, Cousins DJ, Munteanu A, Wong YF, Sudra A, Makinson K, Stephens AC, Arno M, Ciortuz L, Lack G, Turcanu V (2014) IL-9 is a key component of memory TH cell peanut-specific responses from children with peanut allergy. J Allergy Clin Immunol 134(6):1329–1338.e1310. doi:10.1016/j.jaci.2014.06.032
Ma L, Xue HB, Guan XH, Shu CM, Zhang JH, Yu J (2014) Possible pathogenic role of T helper type 9 cells and interleukin (IL)-9 in atopic dermatitis. Clin Exp Immunol 175(1):25–31. doi:10.1111/cei.12198
Gerlach K, Hwang Y, Nikolaev A, Atreya R, Dornhoff H, Steiner S, Lehr HA, Wirtz S, Vieth M, Waisman A, Rosenbauer F, McKenzie AN, Weigmann B, Neurath MF (2014) TH9 cells that express the transcription factor PU.1 drive T cell-mediated colitis via IL-9 receptor signaling in intestinal epithelial cells. Nat Immunol 15(7):676–686. doi:10.1038/ni.2920
Ciccia F, Guggino G, Ferrante A, Raimondo S, Bignone R, Rodolico V, Peralta S, Van Tok M, Cannizzaro A, Schinocca C, Ruscitti P, Cipriani P, Giacomelli R, Alessandro R, Dieli F, Rizzo A, Baeten D, Triolo G (2016) IL-9 over-expression and Th9 polarization characterize the inflamed gut, the synovial tissue and the peripheral blood of patients with psoriatic arthritis. Arthritis Rheumatol. doi:10.1002/art.39649
Szymczak I, Pawliczak R (2016) The active metabolite of vitamin D3 as a potential immunomodulator. Scand J Immunol 83(2):83–91. doi:10.1111/sji.12403
Kim G, Bae JH (2016) Vitamin D and atopic dermatitis: a systematic review and meta-analysis. Nutrition. doi:10.1016/j.nut.2016.01.023
Chambers ES, Nanzer AM, Pfeffer PE, Richards DF, Timms PM, Martineau AR, Griffiths CJ, Corrigan CJ, Hawrylowicz CM (2015) Distinct endotypes of steroid-resistant asthma characterized by IL-17A(high) and IFN-gamma(high) immunophenotypes: Potential benefits of calcitriol. J Allergy Clin Immunol 136(3):628–637.e624. doi:10.1016/j.jaci.2015.01.026
Takami M, Fujimaki K, Nishimura MI, Iwashima M (2015) Cutting edge: AhR is a molecular target of calcitriol in human T cells. J Immunol 195(6):2520–2523. doi:10.4049/jimmunol.1500344
Litonjua AA, Carey VJ, Laranjo N, Harshfield BJ, McElrath TF, O'Connor GT, Sandel M, Iverson RE Jr, Lee-Paritz A, Strunk RC, Bacharier LB, Macones GA, Zeiger RS, Schatz M, Hollis BW, Hornsby E, Hawrylowicz C, Wu AC, Weiss ST (2016) Effect of prenatal supplementation with vitamin D on asthma or recurrent wheezing in offspring by age 3 years: The VDAART Randomized Clinical Trial. JAMA 315(4):362–370. doi:10.1001/jama.2015.18589
Jiao J, Castro M (2015) Vitamin D and asthma: current perspectives. Curr Opin Allergy Clin Immunol 15(4):375–382. doi:10.1097/ACI.0000000000000187
Gold DR, Litonjua AA, Carey VJ, Manson JE, Buring JE, Lee IM, Gordon D, Walter J, Friedenberg G, Hankinson JL, Copeland T, Luttmann-Gibson H (2016) Lung VITAL: rationale, design, and baseline characteristics of an ancillary study evaluating the effects of vitamin D and/or marine omega-3 fatty acid supplements on acute exacerbations of chronic respiratory disease, asthma control, pneumonia and lung function in adults. Contemp Clin Trials 47:185–195. doi:10.1016/j.cct.2016.01.003
Parker JM, Oh CK, LaForce C, Miller SD, Pearlman DS, Le C, Robbie GJ, White WI, White B, Molfino NA, M-CT G (2011) Safety profile and clinical activity of multiple subcutaneous doses of MEDI-528, a humanized anti-interleukin-9 monoclonal antibody, in two randomized phase 2a studies in subjects with asthma. BMC Pulm Med 11:14. doi:10.1186/1471-2466-11-14
White B, Leon F, White W, Robbie G (2009) Two first-in-human, open-label, phase I dose-escalation safety trials of MEDI-528, a monoclonal antibody against interleukin-9, in healthy adult volunteers. Clin Ther 31(4):728–740. doi:10.1016/j.clinthera.2009.04.019
Oh CK, Leigh R, McLaurin KK, Kim K, Hultquist M, Molfino NA (2013) A randomized, controlled trial to evaluate the effect of an anti-interleukin-9 monoclonal antibody in adults with uncontrolled asthma. Respir Res 14:93. doi:10.1186/1465-9921-14-93
Ramming A, Druzd D, Leipe J, Schulze-Koops H, Skapenko A (2012) Maturation-related histone modifications in the PU.1 promoter regulate Th9-cell development. Blood 119(20):4665–4674. doi:10.1182/blood-2011-11-392589
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Keating, P., Hartmann, J.X. (2017). Isolation and Purification of Th9 Cells for the Study of Inflammatory Diseases in Research and Clinical Settings. In: Goswami, R. (eds) Th9 Cells. Methods in Molecular Biology, vol 1585. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-6877-0_19
Download citation
DOI: https://doi.org/10.1007/978-1-4939-6877-0_19
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-6876-3
Online ISBN: 978-1-4939-6877-0
eBook Packages: Springer Protocols