Abstract
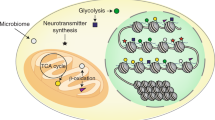
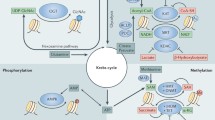
Direct links between altered metabolism, dysregulation of epigenetic processes, and cancer have been established via investigation of cancer- and syndrome-associated mutations in genes encoding key enzymes of intermediary metabolism. Here, we provide an outline for the synthesis of cell-permeable forms of the cellular metabolites (R)-2-hydroxyglutarate and (L)-2-hydroxyglutarate, and their application for the inhibition of α-ketoglutarate-dependent Jumonji histone demethylases.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Jeschke J, Collignon E, Fuks F (2016) Portraits of TET-mediated DNA hydroxymethylation in cancer. Curr Opin Genet Dev 36:16–26. doi:10.1016/j.gde.2016.01.004
Tan L, Shi YG (2012) Tet family proteins and 5-hydroxymethylcytosine in development and disease. Development 139:1895–1902. doi:10.1242/dev.070771
Ko M, An J, Rao A (2015) DNA methylation and hydroxymethylation in hematologic differentiation and transformation. Curr Opin Cell Biol 37:91–101. doi:10.1016/j.ceb.2015.10.009
Caren H, Pollard SM, Beck S (2013) The good, the bad and the ugly: epigenetic mechanisms in glioblastoma. Mol Asp Med 34:849–862. doi:10.1016/j.mam.2012.06.007
Rose NR, Woon EC, Tumber A et al (2012) Plant growth regulator daminozide is a selective inhibitor of human KDM2/7 histone demethylases. J Med Chem 55:6639–6643. doi:10.1021/jm300677j
Chowdhury R, Yeoh KK, Tian YM et al (2011) The oncometabolite 2-hydroxyglutarate inhibits histone lysine demethylases. EMBO Rep 12:463–469. doi:10.1038/embor.2011.43
Lu C, Ward PS, Kapoor GS et al (2012) IDH mutation impairs histone demethylation and results in a block to cell differentiation. Nature 483:474–478. doi:10.1038/nature10860
Xu W, Yang H, Liu Y et al (2011) Oncometabolite 2-hydroxyglutarate is a competitive inhibitor of alpha-ketoglutarate-dependent dioxygenases. Cancer Cell 19:17–30. doi:10.1016/j.ccr.2010.12.014
Zhang Y, Jurkowska R, Soeroes S et al (2010) Chromatin methylation activity of Dnmt3a and Dnmt3a/3L is guided by interaction of the ADD domain with the histone H3 tail. Nucleic Acids Res 38:4246–4253. doi:10.1093/nar/gkq147
Li BZ, Huang Z, Cui QY et al (2011) Histone tails regulate DNA methylation by allosterically activating de novo methyltransferase. Cell Res 21:1172–1181. doi:10.1038/cr.2011.92
Guo X, Wang L, Li J et al (2015) Structural insight into autoinhibition and histone H3-induced activation of DNMT3A. Nature 517:640–644. doi:10.1038/nature13899
Valinluck V, Sowers LC (2007) Endogenous cytosine damage products alter the site selectivity of human DNA maintenance methyltransferase DNMT1. Cancer Res 67:946–950. doi:10.1158/0008-5472.CAN-06-3123
Hashimoto H, Liu Y, Upadhyay AK et al (2012) Recognition and potential mechanisms for replication and erasure of cytosine hydroxymethylation. Nucleic Acids Res 40:4841–4849. doi:10.1093/nar/gks155
He YF, Li BZ, Li Z et al (2011) Tet-mediated formation of 5-carboxylcytosine and its excision by TDG in mammalian DNA. Science 333:1303–1307. doi:10.1126/science.1210944
Ito S, Shen L, Dai Q et al (2011) Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science 333:1300–1303. doi:10.1126/science.1210597
Maiti A, Drohat AC (2011) Thymine DNA glycosylase can rapidly excise 5-formylcytosine and 5-carboxylcytosine: potential implications for active demethylation of CpG sites. J Biol Chem 286:35334–35338. doi:10.1074/jbc.C111.284620
Zhang L, Lu X, Lu J et al (2012) Thymine DNA glycosylase specifically recognizes 5-carboxylcytosine-modified DNA. Nat Chem Biol 8:328–330. doi:10.1038/nchembio.914
Guo JU, Su Y, Zhong C et al (2011) Hydroxylation of 5-methylcytosine by TET1 promotes active DNA demethylation in the adult brain. Cell 145:423–434. doi:10.1016/j.cell.2011.03.022
Lindahl G, Lindstedt G, Lindstedt S (1967) Metabolism of 2-amino-5-hydroxyadipic acid in the rat. Arch Biochem Biophys 119:347–352
Chalmers RA, Lawson AM, Watts RW et al (1980) D-2-hydroxyglutaric aciduria: case report and biochemical studies. J Inherit Metab Dis 3:11–15
Rzem R, Veiga-da-Cunha M, Noel G et al (2004) A gene encoding a putative FAD-dependent L-2-hydroxyglutarate dehydrogenase is mutated in L-2-hydroxyglutaric aciduria. Proc Natl Acad Sci U S A 101:16849–16854. doi:10.1073/pnas.0404840101
Achouri Y, Noel G, Vertommen D et al (2004) Identification of a dehydrogenase acting on D-2-hydroxyglutarate. Biochem J 381(Pt 1):35–42. doi:10.1042/BJ20031933
Struys EA, Salomons GS, Achouri Y et al (2005) Mutations in the D-2-hydroxyglutarate dehydrogenase gene cause D-2-hydroxyglutaric aciduria. Am J Hum Genet 76:358–360. doi:10.1086/427890
Dang L, White DW, Gross S et al (2009) Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 462:739–744. doi:10.1038/nature08617
Losman JA, Kaelin WG Jr (2013) What a difference a hydroxyl makes: mutant IDH, (R)-2-hydroxyglutarate, and cancer. Genes Dev 27:836–852. doi:10.1101/gad.217406.113
Gross S, Cairns RA, Minden MD et al (2010) Cancer-associated metabolite 2-hydroxyglutarate accumulates in acute myelogenous leukemia with isocitrate dehydrogenase 1 and 2 mutations. J Exp Med 207:339–344. doi:10.1084/jem.20092506
Ward PS, Patel J, Wise DR et al (2010) The common feature of leukemia-associated IDH1 and IDH2 mutations is a neomorphic enzyme activity converting alpha-ketoglutarate to 2-hydroxyglutarate. Cancer Cell 17:225–234. doi:10.1016/j.ccr.2010.01.020
Borger DR, Tanabe KK, Fan KC et al (2012) Frequent mutation of isocitrate dehydrogenase (IDH)1 and IDH2 in cholangiocarcinoma identified through broad-based tumor genotyping. Oncologist 17:72–79. doi:10.1634/theoncologist.2011-0386
Cairns RA, Iqbal J, Lemonnier F et al (2012) IDH2 mutations are frequent in angioimmunoblastic T-cell lymphoma. Blood 119:1901–1903. doi:10.1182/blood-2011-11-391748
Yan H, Parsons DW, Jin G et al (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360:765–773. doi:10.1056/NEJMoa0808710
Mardis ER, Ding L, Dooling DJ et al (2009) Recurring mutations found by sequencing an acute myeloid leukemia genome. N Engl J Med 361:1058–1066. doi:10.1056/NEJMoa0903840
Parsons DW, Jones S, Zhang X et al (2008) An integrated genomic analysis of human glioblastoma multiforme. Science 321:1807–1812. doi:10.1126/science.1164382
Jin G, Reitman ZJ, Duncan CG et al (2013) Disruption of wild-type IDH1 suppresses D-2-hydroxyglutarate production in IDH1-mutated gliomas. Cancer Res 73:496–501. doi:10.1158/0008-5472.CAN-12-2852
Rendina AR, Pietrak B, Smallwood A et al (2013) Mutant IDH1 enhances the production of 2-hydroxyglutarate due to its kinetic mechanism. Biochemistry 52:4563–4577. doi:10.1021/bi400514k
Ward PS, Lu C, Cross JR et al (2013) The potential for isocitrate dehydrogenase mutations to produce 2-hydroxyglutarate depends on allele specificity and subcellular compartmentalization. J Biol Chem 288:3804–3815. doi:10.1074/jbc.M112.435495
Losman JA, Looper RE, Koivunen P et al (2013) (R)-2-hydroxyglutarate is sufficient to promote leukemogenesis and its effects are reversible. Science 339:1621–1625. doi:10.1126/science.1231677
Shim EH, Livi CB, Rakheja D et al (2014) L-2-Hydroxyglutarate: an epigenetic modifier and putative oncometabolite in renal cancer. Cancer Discov 4:1290–1298. doi:10.1158/2159-8290.CD-13-0696
Rzem R, Vincent MF, Van Schaftingen E et al (2007) L-2-hydroxyglutaric aciduria, a defect of metabolite repair. J Inherit Metab Dis 30:681–689. doi:10.1007/s10545-007-0487-0
Schatz L, Segal HL (1969) Reduction of alpha-ketoglutarate by homogeneous lactic dehydrogenase X of testicular tissue. J Biol Chem 244:4393–4397
Oldham WM, Clish CB, Yang Y et al (2015) Hypoxia-mediated increases in l-2-hydroxyglutarate coordinate the metabolic response to reductive stress. Cell Metab 22:291–303. doi:10.1016/j.cmet.2015.06.021
Intlekofer AM, Dematteo RG, Venneti S et al (2015) Hypoxia induces production of L-2-hydroxyglutarate. Cell Metab 22:304–311. doi:10.1016/j.cmet.2015.06.023
Koivunen P, Lee S, Duncan CG et al (2012) Transformation by the (R)-enantiomer of 2-hydroxyglutarate linked to EGLN activation. Nature 483:484–488. doi:10.1038/nature10898
Rzem R, Van Schaftingen E, Veiga-da-Cunha M (2006) The gene mutated in l-2-hydroxyglutaric aciduria encodes l-2-hydroxyglutarate dehydrogenase. Biochimie 88:113–116. doi:10.1016/j.biochi.2005.06.005
Shim EH, Sudarshan S (2015) Another small molecule in the oncometabolite mix: L-2-Hydroxyglutarate in kidney cancer. Oncoscience 2:483–486
Hirata M, Sasaki M, Cairns RA et al (2015) Mutant IDH is sufficient to initiate enchondromatosis in mice. Proc Natl Acad Sci U S A 112:2829–2834. doi:10.1073/pnas.1424400112
Reitman ZJ, Duncan CG, Poteet E et al (2014) Cancer-associated isocitrate dehydrogenase 1 (IDH1) R132H mutation and d-2-hydroxyglutarate stimulate glutamine metabolism under hypoxia. J Biol Chem 289:23318–23328. doi:10.1074/jbc.M114.575183
Saha SK, Parachoniak CA, Ghanta KS et al (2014) Mutant IDH inhibits HNF-4alpha to block hepatocyte differentiation and promote biliary cancer. Nature 513:110–114. doi:10.1038/nature13441
Huang J, Chin R, Diep S et al. (2015) Compositions and methods for treating aging and age-related diseases and symptoms. U.S. Patent PCT/US2015/015304
Pusch S, Schweizer L, Beck AC et al (2014) D-2-Hydroxyglutarate producing neo-enzymatic activity inversely correlates with frequency of the type of isocitrate dehydrogenase 1 mutations found in glioma. Acta Neuropathol Commun 2:19. doi:10.1186/2051-5960-2-19
Balduf H, Kirchmaier AL (Unpublished)
Hnasko TS, Hnasko RM (2015) The western blot. Methods Mol Biol 1318:87–96. doi:10.1007/978-1-4939-2742-5_9
Burnette WN (1981) “Western blotting”: electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem 112:195–203
Burnette WN (2009) Western blotting: remembrance of past things. Methods Mol Biol 536:5–8. doi:10.1007/978-1-59745-542-8_2
Mahmood T, Yang PC (2012) Western blot: technique, theory, and trouble shooting. N Am J Med Sci 4:429–434. doi:10.4103/1947-2714.100998
Shechter D, Dormann HL, Allis CD et al (2007) Extraction, purification and analysis of histones. Nat Protoc 2:1445–1457. doi:10.1038/nprot.2007.202
Acknowledgments
This work was supported by the W.M. Keck Foundation (A.L.K.), Purdue University Center for Cancer Research (http://www.cancerresearch.purdue.edu) Innovative Pilot and Shared Resource Grants (A.L.K.), and a Bird Stair Fellowship (H.B.). This research was also supported by the National Cancer Institute (http://www.cancer.gov) [CCSG CA23168] for data acquired in the Purdue Computational and Medicinal Chemistry Resource.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Balduf, H.T., Pepe, A., Kirchmaier, A.L. (2017). Synthesis and Application of Cell-Permeable Metabolites for Modulating Chromatin Modifications Regulated by α-Ketoglutarate-Dependent Enzymes. In: Stefanska, B., MacEwan, D. (eds) Epigenetics and Gene Expression in Cancer, Inflammatory and Immune Diseases. Methods in Pharmacology and Toxicology. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-6743-8_5
Download citation
DOI: https://doi.org/10.1007/978-1-4939-6743-8_5
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-6741-4
Online ISBN: 978-1-4939-6743-8
eBook Packages: Springer Protocols