Abstract
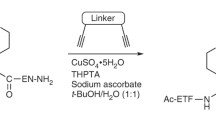
Cyclization confers several advantages to peptides, cumulatively serving to make them more drug-like. In this protocol, cyclic peptides are generated via bis-alkylation of cysteine-containing peptides using α,α′-dibromo-m-xylene. The reactions are robust and high yielding. Multiple reaction platforms for the application of this versatile strategy are described herein: the cyclization of solid-phase-synthesized peptides, both in solution and on resin, as well as the cyclization of in vitro translated mRNA-peptide fusion libraries on oligo(dT) resin.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Pomilio AB, Battista ME, Vitale AA (2006) Naturally-occurring cyclopeptides: structures and bioactivity. Curr Org Chem 10:2075–2121. doi:10.2174/138527206778742669
Millward SW, Takahashi TT, Roberts RW (2005) A general route for post-translational cyclization of mRNA display libraries. J Am Chem Soc 127:14142–14143. doi:10.1021/ja054373h
Gudmundsson OS, Pauletti GM, Wang W, Shan DX, Zhang HJ, Wang BH, Borchardt RT (1999) Coumarinic acid-based cyclic prodrugs of opioid peptides that exhibit metabolic stability to peptidases and excellent cellular permeability. Pharm Res 16:7–15. doi:10.1023/A:1018828207920
Piserchio A, Salinas GD, Li T, Marshall J, Spaller MR, Mierke DF (2004) Targeting specific PDZ domains of PSD-95: structural basis for enhanced affinity and enzymatic stability of a cyclic peptide. Chem Biol 11:469–473. doi:10.1016/j.chembiol.2004.03.013
Kwon Y, Kodadek T (2007) Quantitative comparison of the relative cell permeability of cyclic and linear peptides. Chem Biol 14:671–677. doi:10.1016/j.chembiol.2007.05.006
Rezai T, Yu B, Millhauser GL, Jacobson MP, Lokey RS (2006) Testing the conformational hypothesis of passive membrane permeability using synthetic cyclic peptide diastereomers. J Am Chem Soc 128:2510–2511. doi:10.1021/ja0563455
Dimaio J, Nguyen TMD, Lemieux C, Schiller PW (1982) Synthesis and pharmacological characterization in vitro of cyclic enkephalin analogs—effect of conformational constraints on opiate receptor selectivity. J Med Chem 25:1432–1438. doi:10.1021/jm00354a008
Khan AR, Parrish JC, Fraser ME, Smith WW, Bartlett PA, James MNG (1998) Lowering the entropic barrier for binding conformationally flexible inhibitors to enzymes. Biochemistry 37:16839–16845. doi:10.1021/bi9821364
Raffler NA, Schneider-Mergener J, Famulok M (2003) A novel class of small functional peptides that bind and inhibit human α-thrombin isolated by mRNA display. Chem Biol 10:69–79. doi:10.1016/S1074-5521(02)00309-5
Guillen Schlippe YV, Hartman MCT, Josephson K, Szostak JW (2012) In vitro selection of highly modified cyclic peptides that act as tight binding inhibitors. J Am Chem Soc 134:10469–10477. doi:10.1021/ja301017y
Timmerman P, Beld J, Puijk WC, Meloen RH (2005) Rapid and quantitative cyclization of multiple peptide loops onto synthetic scaffolds for structural mimicry of protein surfaces. Chembiochem 6:821–824. doi:10.1002/cbic.200400374
Heinis C, Rutherford T, Freund S, Winter G (2009) Phage-encoded combinatorial chemical libraries based on bicyclic peptides. Nat Chem Biol 5:502–507. doi:10.1038/nchembio.184
Timmerman P, Puijk WC, Meloen RH (2007) Functional reconstruction and synthetic mimicry of a conformational epitope using CLIPS™ technology. J Mol Recognit 20:283–299. doi:10.1002/jmr.846
Dewkar GK, Carneiro PB, Hartman MC (2009) Synthesis of novel peptide linkers: simultaneous cyclization and labeling. Org Lett 11:4708–4711. doi:10.1021/ol901662cc
Roberts RW, Szostak JW (1997) RNA-peptide fusions for the in vitro selection of peptides and proteins. Proc Natl Acad Sci U S A 94:12297–12302
Shimizu Y, Inoue A, Tomari Y, Suzuki T, Yokogawa T, Nishikawa K, Ueda T (2001) Cell-free translation reconstituted with purified components. Nat Biotechnol 19:751–755
Ma Z, Hartman MT (2012) In vitro selection of unnatural cyclic peptide libraries via mRNA display. J Am Chem Soc 805:367–390. doi:10.1007/978-1-61779-379-0_21
Takahashi TT, Roberts RW (2009) In vitro selection of protein and peptide libraries using mRNA display. Methods Mol Biol 535:293–314. doi:10.1007/978-1-59745-557-2_17
Teixeira A, Benckhuijsen WE, de Koning PE, Valentijn ARPM, Drijfhout JW (2002) The use of Dodt as a non-malodorous scavenger in Fmoc-based peptide synthesis. Protein Pept Lett 9:379–385. doi:10.2174/0929866023408481
Acknowledgments
This work was supported by grants from the Concern Foundation and the NIH (1R01CA166264).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this protocol
Cite this protocol
Hacker, D.E., Almohaini, M., Anbazhagan, A., Ma, Z., Hartman, M.C.T. (2015). Peptide and Peptide Library Cyclization via Bromomethylbenzene Derivatives. In: Derda, R. (eds) Peptide Libraries. Methods in Molecular Biology, vol 1248. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-2020-4_8
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2020-4_8
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-2019-8
Online ISBN: 978-1-4939-2020-4
eBook Packages: Springer Protocols