Abstract
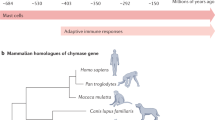
Mast cells (MCs) are tissue-based immune cells that participate to both innate and adaptive immunities as well as to tissue-remodelling processes. Their evolutionary history appears as a fascinating process, whose outline we can only partly reconstruct according to current remnant evidence. MCs have been identified in all vertebrate classes, and a cell population with the overall characteristics of higher vertebrate MCs is identifiable even in the most evolutionarily advanced fish species. In invertebrates, cells related to vertebrate MCs have been recognized in ascidians, a class of urochordates which appeared approximately 500 million years ago. These comprise the granular hemocyte with intermediate characteristics of basophils and MCs and the “test cell” (see below). Both types of cells contain histamine and heparin, and provide defensive functions. The test cell releases tryptase after stimulation with compound 48/80. A leukocyte ancestor operating in the context of a primitive local innate immunity probably represents the MC phylogenetic progenitor. This cell was likely involved in phagocytic and killing activity against pathogens and operated as a general inducer of inflammation. This early type of defensive cell possibly expressed concomitant tissue-reparative functions. With the advent of recombinase activating gene (RAG)-mediated adaptive immunity in the Cambrian era, some 550 million years ago, and the emergence of early vertebrates, MC progenitors differentiated towards a more complex cellular entity. Early MCs probably appeared in the last common ancestor we shared with hagfish, lamprey, and sharks about 450–500 million years ago.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Galli SJ, Kalesnikoff J, Grimbaldeston MA, Piliponsky AM, Williams CMM, Tsai M (2005) Mast cells as “tunable” effector and immunoregulatory cells: recent advances. Annu Rev Immunol 23:749–786
Baccari GC, Pinelli C, Santillo A, Minucci S, Rastogi RK (2011) Mast cells in nonmammalian vertebrates: an overview. Int Rev Cell Mol Biol 290:1–53
Mulero I, Sepulcre MP, Meseguer J, Garcia-Ayala A, Mulero V (2007) Histamine is stored in mast cells of most evolutionarily advanced fish and regulates the fish inflammatory response. Proc Natl Acad Sci U S A 104:19434–19439
Dobson JT, Seibert J, The EM, Da’as S, Fraser RB, Paw BH, Lin TJ, Berman JN (2008) Carboxypeptidase A5 identified a novel mast cell lineage in the zebrafish providing new insight into mast cell fate determination. Blood 112:2969–2972
Da’as S, The EM, Dobson JT, Nasrallah GK, McBride ER, Wang H, Neuberg DS, Marshall JS, Lin T-J, Berman JN (2011) Zebrafish mast cells possess an FcεRI-like receptor and participate in innate and adaptive immune responses. Dev Comp Immunol 35:125–134
Blank U, Rivera J (2004) The ins and outs of IgE-dependent mast-cell exocytosis. Trends Immunol 25:266–273
Johnson AR, Hugli TE, Müller-Eberhard HJ (1975) Release of histamine from rat mast cells by the complement peptides C3a and C5a. Immunology 28:1067
Wojtecka-Lukasik E, Maslinski S (1992) Fibronectin and fibrinogen degradation products stimulate PMN-leukocytes and mast-cell degranulation. J Physiol Pharmacol 43:173–181
Prodeus AP, Zhou X, Maurer M, Galli SJ, Carroll MC (1997) Impaired mast cell-dependent natural immunity in complement C3-deficient mice. Nature 390:172–175
Gommerman JL, Oh DY, Zhou X, Tedder TF, Maurer M, Galli SJ, Carroll MC (2000) A role for CD21/CD35 and CD19 in responses to acute septic peritonitis: a potential mechanism for mast cell activation. J Immunol 165:6915–6921
Marshall JS (2004) Mast-cell responses to pathogens. Nat Rev Immunol 4:787–799
Enerbäck L (1966) Mast cells in rat gastrointestinal mucosa. 1. Effects of fixation. Acta Pathol Microbiol Scand 66:289–302
Enerbäck L (1966) Mast cells in rat gastrointestinal mucosa. 2. Dye-binding and metachromatic properties. Acta Pathol Microbiol Scand 66:303–312
Bienenstock J, Befus AD, Denburg J, Goodacre R, Pearce F, Shanahan F (1983) Mast cell heterogeneity. Monogr Allergy 18:124–128
Galli SJ (1990) Biology of disease. New insights into “the riddle of mast cells”: microenvironmental regulation of mast cell development and phenotypic heterogeneity. Lab Invest 62:5–33
Dvorak AM (2005) Ultrastructural studies of human basophils and mast cells. J Histochem Cytochem 53:1043–1070
Enerbäck L (1986) Mast cell heterogeneity: the evolution of the concept of a specific mucosal mast cell. In: Befus AD, Bienenstock J, Denburg JA (eds) Mast cell differentiation and heterogeneity. Raven, New York, NY, pp 1–26
Irani AM, Schechter NM, Craig SS, De Blois G, Schwartz LB (1986) Two types of human mast cells that have distinct neutral protease composition. Proc Natl Acad Sci U S A 83:4464–4468
Chiu H, Lagunoff D (1971) Histochemical comparison of frog and rat mast cells. J Histochem Cytochem 19:369–375
Chieffi Baccari G, De Paulis A, Di Matteo L, Gentile M, Marone G, Minacci S (1998) In situ characterization of mast cells in the frog Rana esculenta. Cell Tissue Res 292:151–162
Izzo Vitiello I, Chieffi Baccari G, Di Matteo L, Rusciani A, Chieffi P, Minucci S (1997) Number of mast cells in the Harderian gland of the lizard Podarcis sicula sicula (Raf): the annual cycle and its relation to environmental factors and estradiol administration. Gen Comp Endocrinol 107:394–400
Baccari GC, Chieffi G, Di Matteo L, Dafnis D, De Rienzo G, Minucci S (2000) Morphology of the Harderian gland of the Gecko, Tarentola mauritanica. J Morphol 244:137–142
Sayed BA, Christy A, Quirion MR, Brown MA (2008) The master switch: the role of mast cells in autoimmunity and tolerance. Annu Rev Immunol 26:705–739
Metz M, Siebenhaar F, Maurer M (2008) Mast cell functions in the innate skin immune system. Immunobiology 213:251–269
Reite OB, Evensen O (2006) Inflammatory cells of teleostean fish: a review focusing on mast cells/eosinophilic granule cells and rodlet cells. Fish Shellfish Immunol 20:192–208
Sottovia-Filho D (1974) Morphology and histochemistry of the mast cells of snakes. J Morphol 142:109–116
Matsuyama T, Iida T (1999) Degranulation of eosinophilic granular cells with possible involvement in neutrophil migration to site of inflammation in tilapia. Dev Comp Immunol 23:451–457
Silphaduang U, Noga EJ (2001) Antimicrobials: peptide antibiotics in mast cells of fish. Nature 414:268–269
Silphaduang U, Colorni A, Noga EJ (2006) Evidence for widespread distribution of piscidin antimicrobial peptides in teleost fish. Dis Aquat Organ 72:241–252
Campagna S, Saint N, Molle G, Aumelas A (2007) Structure and mechanism of action of the antimicrobial peptide piscidin. Biochemistry 46:1771–1778
Murray HM, Gallant JW, Douglas SE (2003) Cellular localization of pleurocidin gene expression and synthesis in winter flounder gill using immunohistochemistry and in situ hybridization. Cell Tissue Res 312:197–202
De Barros CM, Andrade LR, Allodi S, Viskov C, Mourier PA, Cavalcante MCM, Straus AH, Tahahashi HK, Pomin VH, Carvalho VF, Martins MA, Pavão MSG (2007) The hemolymph of the ascidian Styela plicata (Chordata-Tunicata) contains heparin inside basophil-like cells and a unique sulfated galactoglucan in the plasma. J Biol Chem 282:1615–1626
Church M, Levi-Schaffer F (1997) The human mast cell. J Allergy Clin Immunol 99:155–160
Humphries DE, Wong GW, Friend DS, Gurish MF, Qiu WT, Huang C, Sharpe AH, Stevens RL (1999) Heparin is essential for the storage of specific granule proteases in mast cells. Nature 400:769–772
Forsberg E, Pejler G, Ringvall M, Lunderius C, Tomasini-Johansson B, Kusche-Gullberg M, Eriksson I, Ledin J, Hellman L, Kjellen L (1999) Abnormal mast cells in mice deficient in a heparin-synthesizing enzyme. Nature 400:773–776
Reite OB (1965) A phylogenic approach to the functional significance of tissue mast cell histamine. Nature 206:1034–1035
Takaya K (1969) The relationship between mast cells and histamine in phylogeny with special reference to reptiles and birds. Arch Histol Jpn 30:401–420
Takaya K, Fujito T, Endo K (1967) Mast cells free of histamine in Rana catesbiana. Nature 215:776–777
Cavalcante MCM, De Aandrade LR, Du Bocage Santos-Pinto C, Straus AH, Takahashi HK, Allodi S, Pavão MSG (2002) Colocalization of heparin and histamine in the intracellular granules of test cells from the invertebrate Styela plicata (chordata-tunicata). J Struct Biol 137:313–321
Cavalcante MCM, Mourãom PA, Pavão MS (1999) Isolation and characterization of a highly sulfated heparin sulfate from ascidian test cells. Biochim Biophys Acta 1428:77–87
Gianguzza M, Dolcemascolo G (1978) On the ultrastructure of the follicle cells of Ascidia malaca during oogenesis. Acta Embryol Exp 2:197–211
Cavalcante MCM, Allodi S, Valente AP, Straus AH, Takahashi HK, Mourão PAS, Pavão MSG (2000) Occurrence of heparin in the invertebrate Styela plicata (Tunicata) is restricted to cell layers facing the outside environment. J Biol Chem 275:36189–36196
Pancer Z, Cooper MD (2006) The evolution of adaptive immunity. Annu Rev Immunol 24:497–518
Féger F, Varadaradjalou S, Gao Z, Abraham SN, Arock M (2002) The role of mast cells in host defense and their subversion by bacterial pathogens. Trends Immunol 23:151–157
Knight PA, Wright SH, Lawrence CE, Paterson YY, Miller HR (2000) Delayed expulsion of the nematode Trichinella spiralis in mice lacking the mucosal mast cell-specific granule chymase, mouse mast cell protease-1. J Exp Med 192:1849–1856
Terenina NB, Asatrian AM, Movsessian SO (1997) Neurochemical changes in rats infected with Trichinella spiralis and T. pseudospiralis. Dokl Biol Sci 355:412–413
Sher A, Hein A, Moser G, Caulfield JP (1979) Complement receptors promote the phagocytosis of bacteria by rat peritoneal mast cells. Lab Invest 41:490–499
Gilfillan AM, Tkaczyc C (2006) Integrated signalling pathways for mast-cell activation. Nat Rev Immunol 6:218–230
Miyazawa S, Azumi K, Nonaka M (2001) Cloning and characterization of integrin alpha subunits from the solitary ascidian, Halocynthia roretzi. J Immunol 166:1710–1715
Zhu Y, Thangamani S, Ho B, Ding JL (2005) The ancient origin of the complement system. EMBO J 24:382–394
Von Köckritz-Blickwede M, Goldmann O, Thulin P, Heinemann K, Norrby-Teglund A, Rohde M, Medina E (2008) Phagocytosis-independent antimicrobial activity of mast cells by means of extracellular trap formation. Blood 111:3070–3080
Di Nardo A, Vitiello A, Gallo RL (2003) Cutting edge: mast cell antimicrobial activity is mediated by expression of cathelicidin antimicrobial peptide. J Immunol 170:2274–2278
De Y, Chen Q, Schmidt AP, Anderson GM, Wang JM, Wooters J, Oppenheim JJ, Chertov O (2000) LL-37, the neutrophil granule- and epithelia cell-derived cathelicidin, utilizes formyl peptide receptor-like 1 (FPRL1) as a receptor to chemoattract human peripheral blood neutrophils, monocytes, and T cells. J Exp Med 192:1069
Scott MG, Davidson DJ, Gold MR, Bowdish D, Hancock RE (2002) The human antimicrobial peptide LL-37 is a multifunctional modulator of innate immune responses. J Immunol 169:3883–3891
Galli SJ, Nakae S, Tsai M (2005) Mast cells in the development of adaptive immune responses. Nat Immunol 6:135–142
Galli SJ, Grimbaldeston M, Tsai M (2008) Immunomodulatory mast cells: negative, as well as positive, regulators of immunity. Nat Rev Immunol 8:445–454
Sayed BA, Brown MA (2007) Mast cells as modulators of T-cell responses. Immunol Rev 217:53–64
Frossi B, Gri G, Tripodo C, Pucillo C (2010) Exploring a regulatory role for mast cells: ‘MCregs’? Trends Immunol 31:97–102
Ritter U, Meissner A, Ott J, Kömer H (2003) Analysis of the maturation process of dendritic cells deficient for TNF and lymphotoxin-α reveals an essential role for TNF. J Luekoc Biol 74:216–222
Merluzzi S, Frossi B, Gri G, Parusso S, Tripodo C, Pucillo C (2010) Mast cells enhance proliferation of B lymphocytes and drive their differentiation toward IgA-secreting plasma cells. Blood 115:2810–2817
Hershko AY, Rivera J (2010) Mast cell and T cell communication; amplification and control of adaptive immunity. Immunol Lett 128:98–104
Nakae S, Suto H, Iikura M, Kakurai M, Sedgwick JD, Tsai M, Galli SJ (2006) Mast cells enhance T cell activation: importance of mast cell costimulatory molecules and secreted TNF. J Immunol 176:2238–2248
Kambayashi T, Allenspach EJ, Chang JT, Zou T, Shoag JE, Reiner SL, Caton AJ, Koretzky GA (2009) Inducible MHC class II expression by mast cells supports effector and regulatory T cell activation. J Immunol 182:4686–4695
Galli SJ, Tsai M, Piliponski AM (2008) The development of allergic inflammation. Nature 454:445–454
Nakae S, Suto H, Hakurai M, Sedgwick JD, Tsai M, Galli SJ (2005) Mast cells enhance T cell activation: importance of mast cell-derived TNF. Proc Natl Acad Sci U S A 102:6467–6472
Laird DJ, De Tomaso AW, Cooper MD, Weissman IL (2000) 50 million years of chordate evolution: seeking the origins of adaptive immunity. Proc Natl Acad Sci U S A 97:6924–6926
Bachelet I, Levi-Schaffer F (2007) Mast cells as effector cells: a costimulating question. Trends Immunol 28:360–365
Rihet P, Demeure CE, Bourgois A, Prata A, Dessein AJ (1991) Evidence for an association between human resistance to Schistosoma mansoni and high anti-larval IgE levels. Eur J Immunol 21:2679–2686
King CL, Xianli J, Malthotra I, Liu S, Mahmoud AA, Oettgen HC (1997) Mice with a targeted deletion of the IgE gene have increased worm burdens and reduced granulomatous inflammation following primary infection with Schistosoma mansoni. J Immunol 158:294–300
Warr GW, Magon KE, Higgins DA (1995) IgY: clues to the origins of modern antibodies. Immunol Today 16:392–398
Caldwell DJ, Danforth HD, Morris BC, Ameiss KA, McElroy AP (2004) Participation of the intestinal epithelium and mast cells in local mucosal immune responses in commercial poultry. Poult Sci 83:591–599
Bengtén E, Clem LW, Miller NW, Warr GW, Wilson M (2006) Channel catfish immunoglobulins: repertoire and expression. Dev Comp Immunol 30:77–92
McNeil HP, Adachi R, Stevens RI (2007) Mast cell-restricted tryptase: structure and function in inflammation and pathogen disease. J Biol Chem 282:20785–20789
Woodbury RG, Neurath H (1980) Structure, specificity and localization of the serine proteases of connective tissues. FEBS Lett 114:189–196
Wernersson S, Reimer JM, Poorafshar M, Karlson U, Wermenstam N, Bengten E, Wilson M, Pilström L, Hellman L (2006) Granzyme-like sequences in bony fish shed light on the emergence of hematopoietic serine proteases during vertebrate evolution. Dev Comp Immunol 30:901–918
Huang C, Sali A, Stevens RL (1998) Regulation and function of mast cell proteases in inflammation. J Clin Immunol 18:169–183
Vanderslice P, Ballinger SM, Tam EK, Glodstein SM, Crail CS, Caughey GM (1990) Human mast cell tryptase: multiple cDNAs and genes reveal a multigene serine protease family. Proc Natl Acad Sci U S A 87:3811–3815
Miller JS, Westin EH, Schwartz LB (1989) Cloning and characterization of complementary DNA for human tryptase. J Clin Invest 84:1188–1195
Miller JS, Moxley G, Schwartz LB (1990) Cloning and characterization of a second complementary DNA for human tryptase. J Clin Invest 86:864–870
Thakurdas SM, Melicoff E, Sansores-Garcia L, Moreira DC, Petrova Y, Stevens RL, Adachi R (2007) The mast cell-restricted tryptase mMCP-6 has a critical immunoprotective role in bacterial infections. J Biol Chem 282:20809–20815
Shin K, Watts GF, Oettgen HC, Friends DS, Pemberton AD, Gurish MF, Lee DM (2008) Mouse mast cell tryptase mMCP-6 is a critical link between adaptive and innate immunity in the chronic phase of Trichinella spiralis infection. J Immunol 180:4885–4891
Orinska Z, Maurer M, Mirghomizadeh F, Bulanova E, Metz M, Nashkevich N, Schiemann F, Schulmistrat J, Budagian V, Giron-Michel V, Brandt E, Paus R, Bulfone-Paus S (2007) IL-15 constrains mast cell-dependent antibacterial defences by suppressing chymase activities. Nat Med 13:927–934
Ruoss SJ, Hartmann T, Caughey GH (1991) Mast cell tryptase is a mitogen for cultured fibroblasts. J Clin Invest 88:493–499
Cairns JA, Walls AF (1997) Mast cell tryptase stimulate the synthesis of type I collagen in human lung fibroblasts. J Clin Invest 99:1313–1321
Gruber BL, Kew RR, Jelaska A, Marchese MJ, Garlick J, Ren S, Schwartz WB, Korn JH (1997) Human mast cells activate fibroblasts. J Immunol 158:2310–2317
Blair RJ, Meng H, Marchese MJ, Ren S, Schwartz LB, Tonnesen MG, Gruber BL (1997) Tryptase is a novel, potent angiogenic factor. J Clin Invest 99:2691–2700
Triverdi NN, Tong Q, Raman K, Bhagwandin VJ, Caughey GH (2007) Mast cell α and β tryptases changed rapidly during primate speciation and evolved from γ-like transmembrane peptidases in ancestral vertebrates. J Immunol 179:6072–6079
Hirose E, Shirae M, Saito Y (2003) Ultrastructures and classification of circulating hemocytes in 9 botryllid ascidians (Chordata: Ascidiacea). Zoolog Sci 20:647–656
Hirose E, Taneda Y, Ishii T (1997) Two modes of tunic cuticle formation in a colonial ascidian Aplidium yamazii, responding to wounding. Dev Comp Immunol 21:25–34
Acknowledgments
This study was supported by MIUR local funds to the Department of Experimental and Clinical Medicine, Anatomy Section, University of Udine.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this protocol
Cite this protocol
Crivellato, E., Travan, L., Ribatti, D. (2015). The Phylogenetic Profile of Mast Cells. In: Hughes, M., McNagny, K. (eds) Mast Cells. Methods in Molecular Biology, vol 1220. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-1568-2_2
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1568-2_2
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-1567-5
Online ISBN: 978-1-4939-1568-2
eBook Packages: Springer Protocols