Abstract
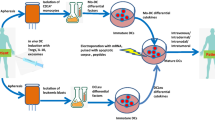
Cancer immunotherapy based on dendritic cell (DC) vaccination has promising alternatives for the treatment of cancer. A central tenet of DC-based cancer immunotherapy is the generation of antigen-specific cytotoxic T lymphocyte (CTL) response. Tumor-associated antigens (TAA) and DC play pivotal roles in this process. DCs are well known to be the most potent antigen-presenting cells and have the most powerful antigen-presenting capacity. DCs pulsed with various TAA have been shown to be effective in producing specific antitumor effects both in vitro and in vivo. Several types of tumor antigens have been applied in cancer treatment including tumor RNA, lysates, apoptotic bodies, heat shock protein, peptides from TAA, and allogeneic tumor cells. Among them, the use of immunogenic HLA-A*0201-specific epitopes from multiple TAA enhances induction of antigen-specific CTL and associated therapeutic efficacy in HLA-A*0201+ cancer patients. The current chapter provides a detailed protocol of generating multiple peptide cocktail-pulsed DC to elicit CTL with a broad spectrum of immune responses against the related tumor antigens.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Steinman RM (1991) The dendritic cell system and its role in immunogenicity. Annu Rev Immunol 9:271–296
Fong L, Engleman EG (2000) Dendritic cells in cancer immunotherapy. Annu Rev Immunol 18:245–273
Nestle FO, Farkas A, Conrad C (2005) Dendritic-cell-based therapeutic vaccination against cancer. Curr Opin Immunol 17:163–169
Yang DH et al (2010) Alpha-type 1-polarized dendritic cells loaded with apoptotic allogeneic myeloma cell line induce strong CTL responses against autologous myeloma cells. Ann Hematol 89:795–801
Lee HJ et al (2012) Identification of novel HLA-A*0201-restricted epitopes from anterior gradient-2 as a tumor-associated antigen against colorectal cancer. Cell Mol Immunol 9:175–183
Lee HJ et al (2012) In vitro induction of anterior gradient-2-specific cytotoxic T lymphocytes by dendritic cells transduced with recombinant adenoviruses as a potential therapy for colorectal cancer. Exp Mol Med 44:60–67
Nguyen-Pham TN et al (2012) Cellular immunotherapy using dendritic cells against multiple myeloma. Korean J Hematol 47:17–27
Mayordomo J et al (1995) Bone marrow-derived dendritic cells pulsed with synthetic tumour peptides elicit protective and therapeutic antitumour immunity. Nat Med 1:1297–1302
Nair SK et al (2000) Induction of cytotoxic T cell responses and tumor immunity against unrelated tumors using telomerase reverse transcriptase RNA transfected dendritic cells. Nat Med 6:1011–1017
Hsu FJ et al (1996) Vaccination of patients with B-cell lymphoma using autologous antigen-pulsed dendritic cells. Nat Med 2:52–58
Nestle FO et al (1998) Vaccination of melanoma patients with peptide- or tumor lysate-pulsed dendritic cells. Nat Med 4:328–332
Ahmad M et al (2004) Escape from immunotherapy: possible mechanisms that influence tumor regression/progression. Cancer Immunol Immunother 53:844–854
Waeckerle-Men Y et al (2006) Dendritic cell-based multi-epitope immunotherapy of hormone-refractory prostate carcinoma. Cancer Immunol Immunother 55:1524–1533
Bae J et al (2012) Myeloma-specific multiple peptides able to generate cytotoxic T lymphocytes: a potential therapeutic application in multiple myeloma and other plasma cell disorders. Clin Cancer Res 18:4850–4860
Fuessel S et al (2006) Vaccination of hormone-refractory prostate cancer patients with peptide cocktail-loaded dendritic cells: results of a phase I clinical trial. Prostate 66:811–821
Mailliard RB et al (2004) alpha-type-1 polarized dendritic cells: a novel immunization tool with optimized CTL-inducing activity. Cancer Res 64:5934–5937
Cella M et al (1996) Ligation of CD40 on dendritic cells triggers production of high levels of interleukin-12 and enhances T cell stimulatory capacity: T-T help via APC activation. J Exp Med 184:747–752
Fanslow WC et al (1994) Structural characteristics of CD40 ligand that determine biological function. Semin Immunol 6:267–278
Kalinski P et al (1999) Final maturation of dendritic cells is associated with impaired responsiveness to IFN-gamma and to bacterial IL-12 inducers: decreased ability of mature dendritic cells to produce IL-12 during the interaction with Th cells. J Immunol 162:3231–3236
Nijman HW et al (1993) Identification of peptide sequences that potentially trigger HLA-A2.1-restricted cytotoxic T lymphocytes. Eur J Immunol 23:1215–1219
Kalinski P et al (2010) Generation of stable Th1/CTL-, Th2-, and Th17-inducing human dendritic cells. Methods Mol Biol 595:117–133
Qian J et al (2007) Dickkopf-1 (DKK1) is a widely expressed and potent tumor-associated antigen in multiple myeloma. Blood 110:1587–1594
Acknowledgments
This study was financially supported by grant no. 2011-0005285 from General Researcher Program Type II of the National Research Foundation of Korea; grant no. RTI05-01-01 from the Regional Technology Innovation Program of the Ministry of Commerce, Industry and Energy; grant no. A000200058 from the Regional Industrial Technology Development program of the Ministry of Knowledge and Economy; grant no. 1120390 from the National R&D Program for Cancer Control, Ministry for Health and Welfare; and grant no. 2011-0030034 from Leading Foreign Research Institute Recruitment Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (MEST), Republic of Korea.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Lee, HJ., Choi, NR., Vo, MC., Hoang, MD., Lee, YK., Lee, JJ. (2014). Generation of Multiple Peptide Cocktail-Pulsed Dendritic Cells as a Cancer Vaccine. In: Lawman, M., Lawman, P. (eds) Cancer Vaccines. Methods in Molecular Biology, vol 1139. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-0345-0_2
Download citation
DOI: https://doi.org/10.1007/978-1-4939-0345-0_2
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-0344-3
Online ISBN: 978-1-4939-0345-0
eBook Packages: Springer Protocols