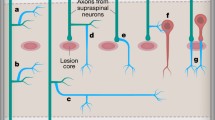
Abstract
Following injury, axolotls are able to functionally regenerate their spinal cord, regaining both motor and sensory control. In contrast, humans respond to severe spinal cord injury by forming a glial scar, which prevents further damage but also inhibits any regenerative growth, resulting in loss of function caudal to the injury site. The axolotl has become a popular system to elucidate the underlying cellular and molecular events that contribute to successful CNS regeneration. However, the experimental injuries (tail amputation and transection) that are utilized in axolotls do not mimic the blunt trauma that is often sustained in humans. Here, we report a more clinically relevant model for spinal cord injuries in the axolotl using a weight-drop technique. This reproducible model allows precise control over the severity of the injury by regulating the drop height, weight, compression, and position of the injury.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ahuja CS, Wilson JR, Nori S, Kotter MRN, Druschel C, Curt A, Fehlings MG (2017) Traumatic spinal cord injury. Nat Rev Dis Primers 3:17018. https://doi.org/10.1038/nrdp.2017.18
Sabin KZ, Jiang P, Gearhart MD, Stewart R, Echeverri K (2019) AP-1(cFos/JunB)/miR-200a regulate the pro-regenerative glial cell response during axolotl spinal cord regeneration. Commun Biol 2:91. https://doi.org/10.1038/s42003-019-0335-4
Noorimotlagh Z, Babaie M, Safdarian M, Ghadiri T, Rahimi-Movaghar V (2017) Mechanisms of spinal cord injury regeneration in zebrafish: a systematic review. Iran J Basic Med Sci 20(12):1287
Diaz Quiroz JF, Tsai E, Coyle M, Sehm T, Echeverri K (2014) Precise control of miR-125b levels is required to create a regeneration-permissive environment after spinal cord injury: a cross-species comparison between salamander and rat. Dis Model Mech 7(6):601–611. https://doi.org/10.1242/dmm.014837
Hui SP, Dutta A, Ghosh S (2010) Cellular response after crush injury in adult zebrafish spinal cord. Dev Dyn 239(11):2962–2979. https://doi.org/10.1002/dvdy.22438
Hanslik KL, Allen SR, Harkenrider TL, Fogerson SM, Guadarrama E, Morgan JR (2019) Regenerative capacity in the lamprey spinal cord is not altered after a repeated transection. PLoS One 14(1):e0204193. https://doi.org/10.1371/journal.pone.0204193
Monaghan JR, Walker JA, Page RB, Putta S, Beachy CK, Voss SR (2007) Early gene expression during natural spinal cord regeneration in the salamander Ambystoma mexicanum. J Neurochem 101(1):27–40. https://doi.org/10.1111/j.1471-4159.2006.04344.x
Walker SE, Nottrodt R, Maddalena L, Carter C, Spencer GE, Carlone RL (2018) Retinoid X receptor α downregulation is required for tail and caudal spinal cord regeneration in the adult newt. Neural Regen Res 13(6):1036–1045. https://doi.org/10.4103/1673-5374.233447
Echeverri K, Tanaka EM (2003) Electroporation as a tool to study in vivo spinal cord regeneration. Dev Dyn 226(2):418–425. https://doi.org/10.1002/dvdy.10238
Deep A, Adeeb N, Hose N, Rezaei M, Fard SA, Tubbs RS, Yashar P, Liker MA, Kateb B, Mortazavi MM (2015) Mouse models of spinal cord injury and stem cell transplantation. Transl Res Anat 1:2–10. https://doi.org/10.1016/j.tria.2015.10.001
Minakov AN, Chernov AS, Asutin DS, Konovalov NA, Telegin GB (2018) Experimental models of spinal cord injury in laboratory rats. Acta Nat 10(3):4–10
Rodrigo Albors A, Tazaki A, Rost F, Nowoshilow S, Chara O, Tanaka EM (2015) Planar cell polarity-mediated induction of neural stem cell expansion during axolotl spinal cord regeneration. elife 4:e10230. https://doi.org/10.7554/eLife.10230
Rost F, Rodrigo Albors A, Mazurov V, Brusch L, Deutsch A, Tanaka EM, Chara O (2016) Accelerated cell divisions drive the outgrowth of the regenerating spinal cord in axolotls. elife:5. https://doi.org/10.7554/eLife.20357
Thygesen MM, Lauridsen H, Pedersen M, Orlowski D, Mikkelsen TW, Rasmussen MM (2019) A clinically relevant blunt spinal cord injury model in the regeneration competent axolotl (Ambystoma mexicanum) tail. Exp Ther Med 17(3):2322–2328. https://doi.org/10.3892/etm.2019.7193
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
1 Supplementary Electronic Material (S)
Tail touch assay in an uninjured animal. The axolotl is touched with a forceps and responds by swimming away from the stimulus (MP4 4395 kb)
Tail touch assay 2 days post-injury. A spinal cord injury has been performed in the midsection of the axolotl tail. The axolotl is repeatedly touched with a forceps caudal to the lesion site but fails to respond to the stimulus (MP4 2166 kb)
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Walker, S., Santos-Ferreira, T., Echeverri, K. (2023). A Reproducible Spinal Cord Crush Injury in the Regeneration-Permissive Axolotl. In: Udvadia, A.J., Antczak, J.B. (eds) Axon Regeneration. Methods in Molecular Biology, vol 2636. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-3012-9_13
Download citation
DOI: https://doi.org/10.1007/978-1-0716-3012-9_13
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-3011-2
Online ISBN: 978-1-0716-3012-9
eBook Packages: Springer Protocols