Abstract
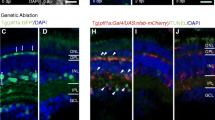
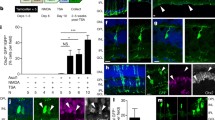
Zebrafish regenerate functional retinal neurons after injury. Regeneration takes place following photic, chemical, mechanical, surgical, or cryogenic lesions, as well as after lesions that selectively target specific neuronal cell populations. An advantage of chemical retinal lesion for studying the process of regeneration is that the lesion is topographically widespread. This results in the loss of visual function as well as a regenerative response that engages nearly all stem cells (Müller glia). Such lesions can therefore be used to further our understanding of the process and mechanisms underlying re-establishment of neuronal wiring patterns, retinal function, and visually mediated behaviors. Widespread chemical lesions also permit the quantitative analysis of gene expression throughout the retina during the period of initial damage and over the duration of regeneration, as well as the study of growth and targeting of axons of regenerated retinal ganglion cells. The neurotoxic Na+/K+ ATPase inhibitor ouabain specifically offers a further advantage over other types of chemical lesions in that it is scalable; the extent of damage can be targeted to include only inner retinal neurons, or all retinal neurons, simply by adjusting the intraocular concentration of ouabain that is used. Here we describe the procedure through which these “selective” vs. “extensive” retinal lesions can be generated.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Massoz L, Dupont MA, Manfroid I (2021) Zebra-fishing for regenerative awakening in mammals. Biomedicine 9(1). https://doi.org/10.3390/biomedicines9010065
Gao H, Luodan A, Huang X, Chen X, Xu H (2021) Muller glia-mediated retinal regeneration. Mol Neurobiol 58:2342. https://doi.org/10.1007/s12035-020-02274-w
Konar GJ, Ferguson C, Flickinger Z, Kent MR, Patton JG (2020) miRNAs and Muller glia reprogramming during retina regeneration. Front Cell Dev Biol 8:632632. https://doi.org/10.3389/fcell.2020.632632
Lahne M, Nagashima M, Hyde DR, Hitchcock PF (2020) Reprogramming Muller glia to regenerate retinal neurons. Annu Rev Vis Sci 6:171–193. https://doi.org/10.1146/annurev-vision-121219-081808
Yurco P, Cameron DA (2005) Responses of Muller glia to retinal injury in adult zebrafish. Vis Res 45(8):991–1002
Fausett BV, Goldman D (2006) A role for alpha1 tubulin-expressing Muller glia in regeneration of the injured zebrafish retina. J Neurosci 26(23):6303–6313
Bernardos RL, Barthel LK, Meyers JR, Raymond PA (2007) Late-stage neuronal progenitors in the retina are radial Muller glia that function as retinal stem cells. J Neurosci 27(26):7028–7040
Thummel R, Kassen SC, Montgomery JE, Enright JM, Hyde DR (2008) Inhibition of Muller glial cell division blocks regeneration of the light-damaged zebrafish retina. Dev Neurobiol 68(3):392–408
Nagashima M, Barthel LK, Raymond PA (2013) A self-renewing division of zebrafish Muller glial cells generates neuronal progenitors that require N-cadherin to regenerate retinal neurons. Development 140(22):4510–4521. https://doi.org/10.1242/dev.090738
Fausett BV, Gumerson JD, Goldman D (2008) The proneural basic helix-loop-helix gene ascl1a is required for retina regeneration. J Neurosci 28(5):1109–1117
Qin Z, Barthel LK, Raymond PA (2009) Genetic evidence for shared mechanisms of epimorphic regeneration in zebrafish. Proc Natl Acad Sci U S A 106(23):9310–9315
Ramachandran R, Reifler A, Parent JM, Goldman D (2010) Conditional gene expression and lineage tracing of tuba1a expressing cells during zebrafish development and retina regeneration. J Comp Neurol 518(20):4196–4212. https://doi.org/10.1002/cne.22448
Ramachandran R, Zhao XF, Goldman D (2011) Ascl1a/Dkk/beta-catenin signaling pathway is necessary and glycogen synthase kinase-3beta inhibition is sufficient for zebrafish retina regeneration. Proc Natl Acad Sci U S A 108(38):15858–15863
Powell C, Grant AR, Cornblath E, Goldman D (2013) Analysis of DNA methylation reveals a partial reprogramming of the Muller glia genome during retina regeneration. Proc Natl Acad Sci U S A 110(49):19814–19819. https://doi.org/10.1073/pnas.1312009110
Powell C, Elsaeidi F, Goldman D (2012) Injury-dependent Muller glia and ganglion cell reprogramming during tissue regeneration requires Apobec2a and Apobec2b. J Neurosci 32(3):1096–1109. https://doi.org/10.1523/JNEUROSCI.5603-11.2012
Nelson CM, Ackerman KM, O’Hayer P, Bailey TJ, Gorsuch RA, Hyde DR (2013) Tumor necrosis factor-alpha is produced by dying retinal neurons and is required for Muller glia proliferation during zebrafish retinal regeneration. J Neurosci 33(15):6524–6539. https://doi.org/10.1523/JNEUROSCI.3838-12.2013
Lenkowski JR, Qin Z, Sifuentes CJ, Thummel R, Soto CM, Moens CB, Raymond PA (2013) Retinal regeneration in adult zebrafish requires regulation of TGFbeta signaling. Glia 61(10):1687–1697. https://doi.org/10.1002/glia.22549
Wan J, Zhao XF, Vojtek A, Goldman D (2014) Retinal injury, growth factors, and cytokines converge on beta-catenin and pStat3 signaling to stimulate retina regeneration. Cell Rep 9(1):285–297. https://doi.org/10.1016/j.celrep.2014.08.048
Nagashima M, D’Cruz TS, Danku AE, Hesse D, Sifuentes C, Raymond PA, Hitchcock PF (2020) Midkine-a is required for cell cycle progression of Muller glia during neuronal regeneration in the vertebrate retina. J Neurosci 40(6):1232–1247. https://doi.org/10.1523/JNEUROSCI.1675-19.2019
White DT, Sengupta S, Saxena MT, Xu Q, Hanes J, Ding D, Ji H, Mumm JS (2017) Immunomodulation-accelerated neuronal regeneration following selective rod photoreceptor cell ablation in the zebrafish retina. Proc Natl Acad Sci U S A 114(18):E3719–E3728. https://doi.org/10.1073/pnas.1617721114
Mitchell DM, Lovel AG, Stenkamp DL (2018) Dynamic changes in microglial and macrophage characteristics during degeneration and regeneration of the zebrafish retina. J Neuroinflammation 15(1):163. https://doi.org/10.1186/s12974-018-1185-6
Mitchell DM, Sun C, Hunter SS, New DD, Stenkamp DL (2019) Regeneration associated transcriptional signature of retinal microglia and macrophages. Sci Rep 9(1):4768. https://doi.org/10.1038/s41598-019-41298-8
Conedera FM, Pousa AMQ, Mercader N, Tschopp M, Enzmann V (2019) Retinal microglia signaling affects Muller cell behavior in the zebrafish following laser injury induction. Glia 67(6):1150–1166. https://doi.org/10.1002/glia.23601
Fimbel SM, Montgomery JE, Burket CT, Hyde DR (2007) Regeneration of inner retinal neurons after intravitreal injection of ouabain in zebrafish. J Neurosci 27(7):1712–1724
Sherpa T, Fimbel SM, Mallory DE, Maaswinkel H, Spritzer SD, Sand JA, Li L, Hyde DR, Stenkamp DL (2008) Ganglion cell regeneration following whole-retina destruction in zebrafish. Dev Neurobiol 68(2):166–181
Sherpa T, Lankford T, McGinn TE, Hunter SS, Frey RA, Sun C, Ryan M, Robison BD, Stenkamp DL (2014) Retinal regeneration is facilitated by the presence of surviving neurons. Dev Neurobiol 74(9):851–876. https://doi.org/10.1002/dneu.22167
Powell C, Cornblath E, Elsaeidi F, Wan J, Goldman D (2016) Zebrafish Muller glia-derived progenitors are multipotent, exhibit proliferative biases and regenerate excess neurons. Sci Rep 6:24851. https://doi.org/10.1038/srep24851
D’Orazi FD, Zhao XF, Wong RO, Yoshimatsu T (2016) Mismatch of synaptic patterns between neurons produced in regeneration and during development of the vertebrate retina. Curr Biol 26(17):2268–2279. https://doi.org/10.1016/j.cub.2016.06.063
McGinn TE, Mitchell DM, Meighan PC, Partington N, Leoni DC, Jenkins CE, Varnum MD, Stenkamp DL (2018) Restoration of dendritic complexity, functional connectivity, and diversity of regenerated retinal bipolar neurons in adult zebrafish. J Neurosci 38(1):120–136. https://doi.org/10.1523/JNEUROSCI.3444-16.2017
McGinn TE, Galicia CA, Leoni DC, Partington N, Mitchell DM, Stenkamp DL (2019) Rewiring the regenerated zebrafish retina: reemergence of bipolar neurons and cone-bipolar circuitry following an inner retinal lesion. Front Cell Dev Biol 7:95. https://doi.org/10.3389/fcell.2019.00095
Maier W, Wolburg H (1979) Regeneration of the goldfish retina after exposure to different doses of ouabain. Cell Tissue Res 202(1):99–118
Raymond PA, Reifler MJ, Rivlin PK (1988) Regeneration of goldfish retina: rod precursors are a likely source of regenerated cells. J Neurobiol 19(5):431–463. https://doi.org/10.1002/neu.480190504
Mensinger AF, Powers MK (1999) Visual function in regenerating teleost retina following cytotoxic lesioning. Vis Neurosci 16(2):241–251
Kastner R, Wolburg H (1982) Functional regeneration of the visual system in teleosts. Comparative investigations after optic nerve crush and damage of the retina. Z Naturforsch C Biosci 37(11–12):1274–1280. https://doi.org/10.1515/znc-1982-11-1229
Kurz-Isler G, Wolburg H (1982) Morphological study on the regeneration of the retina in the rainbow trout after ouabain-induced damage: evidence for dedifferentiation of photoreceptors. Cell Tissue Res 225(1):165–178. https://doi.org/10.1007/BF00216226
Eastlake K, Heywood WE, Tracey-White D, Aquino E, Bliss E, Vasta GR, Mills K, Khaw PT, Moosajee M, Limb GA (2017) Comparison of proteomic profiles in the zebrafish retina during experimental degeneration and regeneration. Sci Rep 7:44601. https://doi.org/10.1038/srep44601
Thomas JL, Morgan GW, Dolinski KM, Thummel R (2018) Characterization of the pleiotropic roles of Sonic Hedgehog during retinal regeneration in adult zebrafish. Exp Eye Res 166:106–115. https://doi.org/10.1016/j.exer.2017.10.003
Lees GJ, Lehmann A, Sandberg M, Hamberger A (1990) The neurotoxicity of ouabain, a sodium-potassium ATPase inhibitor, in the rat hippocampus. Neurosci Lett 120(2):159–162. https://doi.org/10.1016/0304-3940(90)90027-7
Fraser B, DuVal MG, Wang H, Allison WT (2013) Regeneration of cone photoreceptors when cell ablation is primarily restricted to a particular cone subtype. PLoS One 8(1):e55410. https://doi.org/10.1371/journal.pone.0055410
D’Orazi FD, Suzuki SC, Darling N, Wong RO, Yoshimatsu T (2020) Conditional and biased regeneration of cone photoreceptor types in the zebrafish retina. J Comp Neurol 528(17):2816–2830. https://doi.org/10.1002/cne.24933
Medrano MP, Bejarano CA, Battista AG, Venera GD, Bernabeu RO, Faillace MP (2017) Injury-induced purinergic signalling molecules upregulate pluripotency gene expression and mitotic activity of progenitor cells in the zebrafish retina. Purinergic Signal 13(4):443–465. https://doi.org/10.1007/s11302-017-9572-5
Easter SS Jr, Johns PR, Baumann LR (1977) Growth of the adult goldfish eye—I: optics. Vis Res 17(3):469–477. https://doi.org/10.1016/0042-6989(77)90041-4
Collery RF, Veth KN, Dubis AM, Carroll J, Link BA (2014) Rapid, accurate, and non-invasive measurement of zebrafish axial length and other eye dimensions using SD-OCT allows longitudinal analysis of myopia and emmetropization. PLoS One 9(10):e110699. https://doi.org/10.1371/journal.pone.0110699
Duval MG, Chung H, Lehmann OJ, Allison WT (2013) Longitudinal fluorescent observation of retinal degeneration and regeneration in zebrafish using fundus lens imaging. Mol Vis 19:1082–1095
Sifuentes CJ, Kim JW, Swaroop A, Raymond PA (2016) Rapid, dynamic activation of Muller glial stem cell responses in zebrafish. Invest Ophthalmol Vis Sci 57(13):5148–5160. https://doi.org/10.1167/iovs.16-19973
Schroeter EH, Wong RO, Gregg RG (2006) In vivo development of retinal ON-bipolar cell axonal terminals visualized in nyx::MYFP transgenic zebrafish. Vis Neurosci 23(5):833–843. https://doi.org/10.1017/S0952523806230219
Allison WT, Barthel LK, Skebo KM, Takechi M, Kawamura S, Raymond PA (2010) Ontogeny of cone photoreceptor mosaics in zebrafish. J Comp Neurol 518(20):4182–4195. https://doi.org/10.1002/cne.22447
Sherpa T, Hunter SS, Frey RA, Robison BD, Stenkamp DL (2011) Retinal proliferation response in the buphthalmic zebrafish, bugeye. Exp Eye Res 93(4):424–436. https://doi.org/10.1016/j.exer.2011.06.001
Lindsey AE, Powers MK (2007) Visual behavior of adult goldfish with regenerating retina. Vis Neurosci 24(3):247–255
Acknowledgments
Retinal regeneration studies in our laboratories using the approaches described have been supported by NIH R01 EY012146 (DLS), NIH R01 EY030467 (DMM), and NIH R21 EY026814 (DLS and DMM). We are also grateful for support in the form of pilot grants and other funding from Idaho INBRE (NIH P20 GM103408). We thank past and present members of the Stenkamp and Mitchell laboratories, particularly Ruth Frey for zebrafish husbandry and Lindsey Barrett for the images shown in Fig. 3a, b.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Mitchell, D.M., Stenkamp, D.L. (2023). Generating Widespread and Scalable Retinal Lesions in Adult Zebrafish by Intraocular Injection of Ouabain. In: Udvadia, A.J., Antczak, J.B. (eds) Axon Regeneration. Methods in Molecular Biology, vol 2636. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-3012-9_12
Download citation
DOI: https://doi.org/10.1007/978-1-0716-3012-9_12
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-3011-2
Online ISBN: 978-1-0716-3012-9
eBook Packages: Springer Protocols