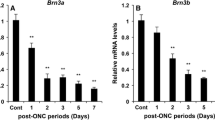
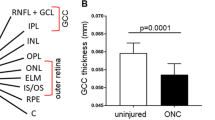
Abstract
Many human optic neuropathies lead to crippling conditions resulting in partial or complete loss of vision. While the retina is made up of several different cell types, retinal ganglion cells (RGCs) are the only cell type connecting the eye to the brain. Optic nerve crush injuries, wherein RGC axons are damaged without severing the optic nerve sheath, can serve as a model for traumatic optical neuropathies as well as some progressive neuropathies such as glaucoma. In this chapter, we describe two different surgical methods for establishing an optic nerve crush (ONC) injury in the postmetamorphic frog, Xenopus laevis. Why use the frog as an animal model? Mammals lose the ability to regenerate damaged CNS neurons, but amphibians and fish retain the ability to regenerate new RGC bodies and regrow RGC axons following an injury. In addition to presenting two different surgical ONC injury methods, we highlight their advantages and disadvantages and discuss the distinctive characteristics of Xenopus laevis as an animal model for studying CNS regeneration.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Dratviman-Storobinsky O, Hasanreisoglu M, Offen D et al (2008) Progressive damage along the optic nerve following induction of crush injury or rodent anterior ischemic optic neuropathy in transgenic mice. http://www.molvis.org/molvis/v14/a254/. Accessed 2 Mar 2021
Whitworth GB, Misaghi BC, Rosenthal DM et al (2017) Translational profiling of retinal ganglion cell optic nerve regeneration in Xenopus laevis. Dev Biol 426:360–373. https://doi.org/10.1016/j.ydbio.2016.06.003
Phipps LS, Marshall L, Dorey K, Amaya E (2020) Model systems for regeneration: Xenopus. Development 147:dev180844. https://doi.org/10.1242/dev.180844
Belrose JL, Prasad A, Sammons MA et al (2020) Comparative gene expression profiling between optic nerve and spinal cord injury in Xenopus laevis reveals a core set of genes inherent in successful regeneration of vertebrate central nervous system axons. BMC Genomics 21:540. https://doi.org/10.1186/s12864-020-06954-8
Szaro BG, Loh YP, Hunt RK (1985) Specific changes in axonally transported proteins during regeneration of the frog (Xenopus laevis) optic nerve. J Neurosci 5:192–208
Zhao Y, Szaro BG (1994) The return of phosphorylated and nonphosphorylated epitopes of neurofilament proteins to the regenerating optic nerve of Xenopus laevis. J Comp Neurol 343:158–172. https://doi.org/10.1002/cne.903430112
Tang Z, Zhang S, Lee C et al (2011) An optic nerve crush injury murine model to study retinal ganglion cell survival. J Vis Exp 2685. https://doi.org/10.3791/2685
Liu Q, Londraville RL (2003) Using the adult zebrafish visual system to study cadherin-2 expression during central nervous system regeneration. Methods Cell Sci 25:71–78. https://doi.org/10.1023/B:MICS.0000006854.18378.fc
Ananthakrishnan L, Szaro BG (2009) Transcriptional and translational dynamics of light neurofilament subunit RNAs during Xenopus laevis optic nerve regeneration. Brain Res 1250:27–40. https://doi.org/10.1016/j.brainres.2008.11.002
Watson FL, Mills EA, Wang X et al (2012) Cell type-specific translational profiling in the Xenopus laevis retina. Dev Dyn Off Publ Am Assoc Anat 241:1960–1972. https://doi.org/10.1002/dvdy.23880
McNamara S, Wlizla M, Horb ME (2018) Husbandry, general care, and transportation of Xenopus laevis and Xenopus tropicalis. In: Vleminckx K (ed) Xenopus. Springer, New York, pp 1–17
Acknowledgments
Many thanks to Mr. Dave Pfaff and Cleveland Candle for their design help with Illustrator.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Feidler, A.M., Nguyen, H.H.M., Watson, F.L. (2023). Surgical Methods in Postmetamorphic Xenopus laevis: Optic Nerve Crush Injury Model. In: Udvadia, A.J., Antczak, J.B. (eds) Axon Regeneration. Methods in Molecular Biology, vol 2636. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-3012-9_11
Download citation
DOI: https://doi.org/10.1007/978-1-0716-3012-9_11
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-3011-2
Online ISBN: 978-1-0716-3012-9
eBook Packages: Springer Protocols