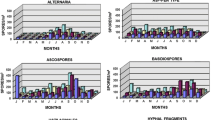
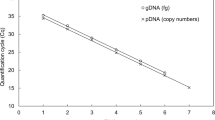
Abstract
The inoculum of H. fraxineus consists mainly of ascospores released from apothecia which are growing on fallen leaves infected during the previous year. The ascospores can be detected in various manners due to their high concentration in the air during the main sporulation season, which corresponds to astronomic summer. This methodology is focused on one of the methods which have been successfully used. It employs a cheap, but highly efficient rotating arm air sampler and a specific quantitative real-time PCR method for the quantification of the air samples. The methodology is accompanied by lots of detailed theoretical and practical notes for its smooth application, including mentioning other alternatives.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kowalski T (2006) Chalara fraxinea sp. nov. associated with dieback of ash (Fraxinus excelsior) in Poland. For Pathol 36:264–270. https://doi.org/10.1111/j.1439-0329.2006.00453.x
Pautasso M, Aas G, Queloz V, Holdenrieder O (2013) European ash (Fraxinus excelsior) dieback – a conservation biology challenge. Biol Conserv 158:37–49. https://doi.org/10.1016/j.biocon.2012.08.026
Vasaitis R, Enderle R (2017) Dieback of European ash (Fraxinus spp.) -consequences and guidelines for sustainable management the report on European Cooperation in Science & Technology (COST) Action FP1103 FRAXBACK
Kowalski T, Holdenrieder O (2009) Pathogenicity of Chalara fraxinea. For Pathol 39:1–7. https://doi.org/10.1111/j.1439-0329.2008.00565.x
Gross A, Zaffarano PL, Duo A, Grünig CR (2012) Reproductive mode and life cycle of the ash dieback pathogen Hymenoscyphus pseudoalbidus. Fungal Genet Biol 49:977–986. https://doi.org/10.1016/j.fgb.2012.08.008
Fones HN, Mardon C, Gurr SJ (2016) A role for the asexual spores in infection of Fraxinus excelsior by the ash-dieback fungus Hymenoscyphus fraxineus. Sci Rep 6:1–10. https://doi.org/10.1038/srep34638
Cleary MR, Arhipova N, Gaitnieks T et al (2013) Natural infection of Fraxinus excelsior seeds by Chalara fraxinea. For Pathol 43:83–85. https://doi.org/10.1111/efp.12012
Chandelier A, Gerarts F, San Martin G et al (2016) Temporal evolution of collar lesions associated with ash dieback and the occurrence of Armillaria in Belgian forests. For Pathol 46:289–297. https://doi.org/10.1111/efp.12258
Kräutler K, Treitler R, Kirisits T (2015) Hymenoscyphus fraxineus can directly infect intact current-year shoots of Fraxinus excelsior and artificially exposed leaf scars. For Pathol 45:274–280. https://doi.org/10.1111/efp.12168
Hietala AM, Timmermann V, BØrja I et al (2013) The invasive ash dieback pathogen Hymenoscyphus pseudoalbidus exerts maximal infection pressure prior to the onset of host leaf senescence. Fungal Ecol 6:302–308. https://doi.org/10.1016/j.funeco.2013.03.008
Timmermann V, Børja I, Hietala AM et al (2011) Ash dieback: pathogen spread and diurnal patterns of ascospore dispersal, with special emphasis on Norway. EPPO Bull 41:14–20. https://doi.org/10.1111/j.1365-2338.2010.02429.x
Chandelier A, Helson M, Dvorak M et al (2014) Detection and quantification of airborne inoculum of Hymenoscyphus pseudoalbidus using real-time PCR assays. Plant Pathol 63:1296–1305. https://doi.org/10.1111/ppa.12218
Husson C, Caël O, Grandjean JP et al (2012) Occurrence of Hymenoscyphus pseudoalbidus on infected ash logs. Plant Pathol 61:889–895. https://doi.org/10.1111/j.1365-3059.2011.02578.x
Aguayo J, Husson C, Chancerel E et al (2021) Combining permanent aerobiological networks and molecular analyses for large-scale surveillance of forest fungal pathogens: a proof-of-concept. Plant Pathol 70:181–194. https://doi.org/10.1111/ppa.13265
Grosdidier M, Aguayo J, Marçais B et al (2017) Detection of plant pathogens using real-time PCR: how reliable are late Ct values? Plant Pathol 66:359–367. https://doi.org/10.1111/ppa.12591
D’Amato G, Lobefalo G (1989) Allergenic pollens in the southern Mediterranean area. J Allergy Clin Immunol 83:116–122
Hirst JM (1952) An automatic volumetric spore trap. Ann Appl Biol 39:257–265
Dvořák M, Rotková G, Botella L (2016) Detection of airborne inoculum of Hymenoscyphus fraxineus and H. albidus during seasonal fluctuations associated with absence of apothecia. Forests 7:1–13. https://doi.org/10.3390/f7010001
Dhingra OD, Sinclair JB (1995) Basic plant pathology methods, 2nd edn. CRC Press
Lacey ME, West JS (2006) The air Spora – a manual for catching and identifying airborne biological particles. Springer
Perkins WA, Leighton PA (1957) The rotorod sampler. Second semi-annual report no. CML 186, aerosol laboratory
McCartney HA, Fitt BDL, Schmechel D (1997) Sampling bioaerosols in plant pathology. J Aerosol Sci 28:349–364
Čermáková V, Kudláček T, Rotková G et al (2017) Hymenoscyphus fraxineus mitovirus 1 naturally disperses through the airborne inoculum of its host, Hymenoscyphus fraxineus, in the Czech Republic. Biocontrol Sci Tech 27:992–1008. https://doi.org/10.1080/09583157.2017.1368455
Chandelier A, André F, Laurent F (2010) Detection of Chalara fraxinea in common ash (Fraxinus excelsior) using real time PCR. For Pathol 40:87–95. https://doi.org/10.1111/j.1439-0329.2009.00610.x
Botella L, Bačová A, Dvořák M et al (2019) Detection and quantification of the air inoculum of Caliciopsis pinea in a plantation of Pinus radiata in Italy. iForest - Biogeosc For 12:193–198. https://doi.org/10.3832/ifor2866-012
Dvořák M, Janoš P, Botella L et al (2017) Spore dispersal patterns of fusarium circinatum on an infested Monterey pine forest in North-Western Spain. Forests 8:432. https://doi.org/10.3390/f8110432
UCCE Napa Viticulture Advisor (2020) How to build a rotating arm spore trap for powdery mildew. https://www.youtube.com/watch?v=D0kJZbNw7tc
Noll KE (1970) A rotary inertial impactor for sampling giant particles in the atmosphere. Atmos Environ 4:9–19
Kowalski T, Holdenrieder O (2009) The teleomorph of Chalara fraxinea, the causal agent of ash dieback. For Pathol 39:304–308. https://doi.org/10.1111/j.1439-0329.2008.00589.x
Dvořák M, Rotková G (2015) Spore dispersal of Hymenoscyphus fraxineus in the Czech Republic. In: Joint IUFRO Working Party Meetings 7–12 June, 2015 in Uppsala, Sweden. Swedish University of Agricultural Sciences, p 30
Koukol O, Haňáčková Z, Dvořák M, Havrdová L (2016) Unseen, but still present in Czechia: Hymenoscyphus albidus detected by real-time PCR, but not by intensive sampling. Mycol Prog 15:1–9. https://doi.org/10.1007/s11557-015-1149-5
Acknowledgment
This work was supported by using the laboratories of Mendel University in Brno, Phytophthora Research Centre (funded by project CZ.02.1.01/0.0/0.0/15_003/0000453), and laboratories of the University of South Bohemia in České Budějovice, Faculty of Agriculture and Technologies, Department of Genetics and Agricultural Biotechnology. Furthermore, this work was financed from a HORIZON 2020 project (agreement no. 771271): Holistic Management of Emerging Forest Pests and Diseases (HOMED).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Dvořák, M. (2022). Detection of Airborne Inoculum of Hymenoscyphus fraxineus: The Causal Agent of Ash Dieback. In: Luchi, N. (eds) Plant Pathology. Methods in Molecular Biology, vol 2536. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2517-0_8
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2517-0_8
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2516-3
Online ISBN: 978-1-0716-2517-0
eBook Packages: Springer Protocols