Abstract
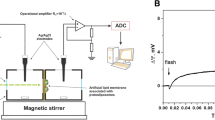
Electrophysiological approaches to the study of the activity of retinal-containing protein bacteriorhodopsin (bR) or other proteins of this family are based usually on measurements of electrical current through a planar bilayer lipid membrane (BLM) with proteoliposomes attached to the BLM surface at one side of the membrane. Here, we describe the measurements of the pumping activity of bR and channelrhodopsin 2 (ChR2) with special attention to the study of voltage dependence of the light-induced currents. Strong voltage dependence of ChR2 suggests light-triggered ion channel activity of ChR2. We also describe electrophysiological measurements with the help of collodion film instead of BLM for the measurements of fast stages of a rhodopsin photocycle as well as the estimation of the activity of proteoliposomes without a macro membrane using fluorescent probes such as oxonol VI or 9-aminoacridine.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Gushchin I, Gordeliy V (2018) Microbial rhodopsins. In: Subcellular biochemistry, pp 19–56
Ernst OP, Lodowski DT, Elstner M et al (2014) Microbial and animal rhodopsins: structures, functions, and molecular mechanisms. Chem Rev 114:126–163
Grote M, Engelhard M, Hegemann P (2014) Of ion pumps, sensors and channels—perspectives on microbial rhodopsins between science and history. Biochim Biophys Acta Bioenerg 1837:533–545
Drachev LA, Jasaitis AA, Kaulen AD et al (1974) Direct measurement of electric current generation by cytochrome oxidase, H+-ATPase and bacteriorhodopsin. Nature 249:321–324
Bamberg E, Apell HJ, Dencher NA et al (1979) Photocurrents generated by bacteriorhodopsin on planar bilayer membranes. Biophys Struct Mech 5:277–292
Drachev LA, Frolov VN, Kaulen AD et al (1976) Reconstitution of biological molecular generators of electric current. Bacteriorhodopsin. J Biol Chem 251:7059–7065
Herrmann TR, Rayfield GW (1978) The electrical response to light of bacteriorhodopsin in planar membranes. Biophys J 21:111–125
Bamberg E, Dencher NA, Fahr A et al (1981) Transmembranous incorporation of photoelectrically active bacteriorhodopsin in planar lipid bilayers. Proc Natl Acad Sci 78:7502–7506
Feldbauer K, Zimmermann D, Pintschovius V et al (2009) Channelrhodopsin-2 is a leaky proton pump. Proc Natl Acad Sci 106:12317–12322
Sineshchekov O, Govorunova EG, Wang J et al (2013) Intramolecular proton transfer in channelrhodopsins. Biophys J 104:807–817
Drachev LA, Kaulen AD, Khitrina LV et al (2005) Fast stages of photoelectric processes in biological membranes. Eur J Biochem 117:461–470
Drachev LA, Kalamkarov GR, Kaulen AD et al (1981) Fast stages of photoelectric processes in biological membranes. II Visual rhodopsin. Eur J Biochem 117:471–481
Apell H-J, Bersch B (1987) Oxonol VI as an optical indicator for membrane potentials in lipid vesicles. Biochim Biophys Acta Biomembr 903:480–494
Bennett AB, Spanswick RM (1983) Optical measurements of ΔpH and Δψ in corn root membrne vesicles: kinetic analysis of Cl− effects on a proton-translocating ATPase. J Membr Biol 71:95–107
Antonenko YN, Denisov SS, Silachev DN et al (2016) A long-linker conjugate of fluorescein and triphenylphosphonium as mitochondria-targeted uncoupler and fluorescent neuro- and nephroprotector. Biochim Biophys Acta Gen Subj 1860:2463–2473
Sone N, Yoshida M, Hirata H et al (1977) Adenosine triphosphate synthesis by electrochemical proton gradient in vesicles reconstituted from purified adenosine triphosphatase and phospholipids of thermophilic bacterium. J Biol Chem 252:2956–2960
Nichols JW, Hill MW, Bangham AD et al (1980) Measurement of net proton-hydroxyl permeability of large unilamellar liposomes with the fluorescent pH probe, 9-aminoacridine. Biochim Biophys Acta Biomembr 596:393–403
Volkov O, Kovalev K, Polovinkin V et al (2017) Structural insights into ion conduction by channelrhodopsin 2. Science 358:eaan8862
Dencher NA, Heyn MP (1982) Preparation and properties of monomeric bacteriorhodopsin. In: Methods in enzymology, pp 5–10
Nagel G, Szellas T, Huhn W et al (2003) Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc Natl Acad Sci 100:13940–13945
Gushchin I, Chervakov P, Kuzmichev P et al (2013) Structural insights into the proton pumping by unusual proteorhodopsin from nonmarine bacteria. Proc Natl Acad Sci 110:12631–12636
Kaulen AD (2000) Electrogenic processes and protein conformational changes accompanying the bacteriorhodopsin photocycle. Biochim Biophys Acta Bioenerg 1460:204–219
Kalaidzidis IV, Kaulen AD, Radionov AN et al (2001) Photoelectrochemical cycle of bacteriorhodopsin. Biochemistry (Mosc) 66:1220–1233
Siletsky SA, Mamedov MD, Lukashev EP et al (2016) Electrogenic steps of light-driven proton transport in ESR, a retinal protein from Exiguobacterium sibiricum. Biochim Biophys Acta Bioenerg 1857:1741–1750
Siletsky SA, Mamedov MD, Lukashev EP et al (2019) Elimination of proton donor strongly affects directionality and efficiency of proton transport in ESR, a light-driven proton pump from Exiguobacterium sibiricum. Biochim Biophys Acta Bioenerg 1860:1–11
Acknowledgments
We are grateful to Oleksandr Volkov for producing channelrhodopsin-2 and Christian Baeken for providing us with purple membranes.
The research was supported by the Ministry of Science and Higher Education of the Russian Federation (agreement # 075-00337-20-03, project FSMG -2020-0003). Measurements of electrogenic properties were supported by RFBR grants 19-04-00238 (to TIR) and 18-04-00503a (to SAS).
This work was supported by Russian Science Foundation (RSF) Project 21-64-00018.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Rokitskaya, T.I., Maliar, N.L., Siletsky, S.A., Gordeliy, V., Antonenko, Y.N. (2022). Electrophysiological Characterization of Microbial Rhodopsin Transport Properties: Electrometric and ΔpH Measurements Using Planar Lipid Bilayer, Collodion Film, and Fluorescent Probe Approaches. In: Gordeliy, V. (eds) Rhodopsin. Methods in Molecular Biology, vol 2501. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2329-9_12
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2329-9_12
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2328-2
Online ISBN: 978-1-0716-2329-9
eBook Packages: Springer Protocols