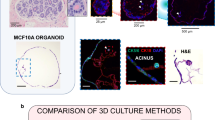
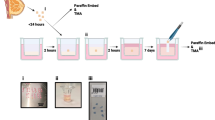
Abstract
Tissue culture has evolved considerably over the last few years, including cell culture in three dimensions, organoids, cocultures of different cell types and the use of diverse types of matrices in an attempt to mimic conditions that more closely resemble those found in the original tissue or organ. In this chapter, we describe how patient-derived breast tissue can be cultured on sponges for several days, maintaining their original architecture and with the capacity to respond to treatments. This protocol facilitates the study of the tissue responses without the need for extensive tissue manipulation, cell digestion or use of a biomaterial as scaffold, while maintaining the stroma and extracellular matrix organization. This method has the potential to improve preclinical testing by contributing to provide more accurate data reflecting cell–cell and cell–matrix interactions, tumor microenvironment, drug effects or stem cell function in normal- and pathophysiology of the breast.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Weigelt B, Ghajar CM, Bissell MJ (2014) The need for complex 3D culture models to unravel novel pathways and identify accurate biomarkers in breast cancer. Adv Drug Deliv Rev 69-70:42–51
Rosenbluth JM, Schackmann RCJ, Gray GK, Selfors LM, Li CM, Boedicker M, Kuiken HJ, Richardson A, Brock J, Garber J, Dillon D, Sachs N, Clevers H, Brugge JS (2020) Organoid cultures from normal and cancer-prone human breast tissues preserve complex epithelial lineages. Nat Commun 11(1):1711
Zhu L, Valiente M (2021) Organotypic brain cultures for metastasis research, Neuromethods book, Brain tumors. Springer Protocols, New York
Kokkinos J, Sharbeen G, Haghighi KS, Ignacio RMC, Kopecky C, Gonzales-Aloy E, Youkhana J, Timpson P, Pereira BA, Ritchie S, Pandzic E, Boyer C, Davis TP, Butler LM, Goldstein D, McCarroll JA, Phillips PA (2021) Ex vivo culture of intact human patient derived pancreatic tumour tissue. Sci Rep 11(1):1944
Centenera MM, Raj GV, Knudsen KE, Tilley WD, Butler LM (2013) Ex vivo culture of human prostate tissue and drug development. Nat Rev Urol 10:483–487
Centenera MM, Hickey TE, Jindal S, Ryan NK, Ravindranathan P, Mohammed H, Robinson JL, Schiewer MJ, Ma S, Kapur P, Sutherland PD, Hoffmann CE, Roehrborn CG, Gomella LG, Carroll JS, Birrell SN, Knudsen KE, Raj GV, Butler LM, Tilley WD (2018) A patient-derived explant (PDE) model of hormone-dependent cancer. Mol Oncol 12(9):1608–1622
Gustafsson A, Garre E, Leiva MC, Salerno S, Ståhlberg A, Landberg G (2021) Patient-derived scaffolds as a drug-testing platform for endocrine therapies in breast cancer. Sci Rep 11(1):13334
Hirata E, Sahai E (2017) Tumor microenvironment and differential responses to therapy. Cold Spring Harb Perspect Med 7(7):a026781
Boix-Montesinos P, Soriano-Teruel PM, Armiñán A, Orzáez M, Vicent MJ (2021) The past, present, and future of breast cancer models for nanomedicine development. Adv Drug Deliv Rev 173:306–330
Petersen JK, Krogsgaard J, Nielsen KM, Nørgaard EB (1984) A comparison between 2 absorbable hemostatic agents: gelatin sponge (Spongostan) and oxidized regenerated cellulose (Surgicel). Int J Oral Surg 13(5):406–410
Jensen SS, Yazdi PM, Hjørting-Hansen E, Bosshardt DD, von Arx T (2010) Haemostatic effect and tissue reactions of methods and agents used for haemorrhage control in apical surgery. Int Endod J 43(1):57–63
Klangjorhor J, Nimkingratana P, Settakorn J, Pruksakorn D, Leerapun T, Arpornchayanon O, Rojanasthien S, Kongtawelert P, Pothacharoen P (2012) Hyaluronan production and chondrogenic properties of primary human chondrocyte on gelatin based hematostatic spongostan scaffold. J Orthop Surg Res 7:40
Ribatti D, Nico B, Vacca A, Presta M (2006) The gelatin sponge-chorioallantoic membrane assay. Nat Protoc 1(1):85–91
Ricciardelli C, Lokman NA, Sabit I, Gunasegaran K, Bonner WM, Pyragius CE, Macpherson AM, Oehler MK (2018) Novel ex vivo ovarian cancer tissue explant assay for prediction of chemosensitivity and response to novel therapeutics. Cancer Lett 421:51–58
Leiva MC, Garre E, Gustafsson A, Svanström A, Bogestål Y, Håkansson J, Ståhlberg A, Landberg G (2021) Breast cancer patient-derived scaffolds as a tool to monitor chemotherapy responses in human tumor microenvironments. J Cell Physiol 236(6):4709–4724
Kähkönen TE, Toriseva M, Petruk N, Virta AR, Maher A, Eigéliené N, Kaivola J, Boström P, Koskivuo I, Nees M, Tuomela JM, Ivaska KK, Härkönen PL (2021) Effects of FGFR inhibitors TKI258, BGJ398 and AZD4547 on breast cancer cells in 2D, 3D and tissue explant cultures. Cell Oncol (Dordr) 44(1):205–218
Acknowledgments
We are grateful for the generosity of many women and patients that collaborated in this project. This work was supported by grants from the Ministry of Science and Innovation (SAF2017-84934-R) and the Department of Industry and Innovation Technology of the Government of the Autonomous Community of the Basque Country.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Gris-Cárdenas, I., Rábano, M., Vivanco, M.d.M. (2022). Patient-Derived Explant Cultures of Normal and Tumor Human Breast Tissue. In: Vivanco, M.d. (eds) Mammary Stem Cells. Methods in Molecular Biology, vol 2471. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2193-6_17
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2193-6_17
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2192-9
Online ISBN: 978-1-0716-2193-6
eBook Packages: Springer Protocols