Abstract
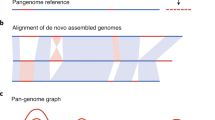
To unlock the genetic potential in crops, multi-genome comparisons are an essential tool. Decreasing costs and improved sequencing technologies have democratized plant genome sequencing and led to a vast increase in the amount of available reference sequences on the one hand and enabled the assembly of even the largest and most complex and repetitive crops genomes such as wheat and barley. These developments have led to the era of pan-genomics in recent years. Pan-genome projects enable the definition of the core and dispensable genome for various crop species as well as the analysis of structural and functional variation and hence offer unprecedented opportunities for exploring and utilizing the genetic basis of natural variation in crops. Comparing, analyzing, and visualizing these multiple reference genomes and their diversity requires powerful and specialized computational strategies and tools.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Monat C et al (2019) TRITEX: chromosome-scale sequence assembly of Triticeae genomes with open-source tools. Genome Biol 20(1):284
Mascher M et al (2017) A chromosome conformation capture ordered sequence of the barley genome. Nature 544(7651):427–433
International Wheat Genome Sequencing, C et al (2018) Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 361(6403):eaar7191
van Berkum NL et al (2010) Hi-C: a method to study the three-dimensional architecture of genomes. J Vis Exp (39)
Walkowiak S et al (2020) Multiple wheat genomes reveal global variation in modern breeding. Nature 588(7837):277–283
Milner SG et al (2019) Genebank genomics highlights the diversity of a global barley collection. Nat Genet 51(2):319–326
Jayakodi M et al (2020) The barley pan-genome reveals the hidden legacy of mutation breeding. Nature 588(7837):284–289
Himmelbach A et al (2018) Discovery of multi-megabase polymorphic inversions by chromosome conformation capture sequencing in large-genome plant species. Plant J 96(6):1309–1316
Comadran J et al (2012) Natural variation in a homolog of Antirrhinum CENTRORADIALIS contributed to spring growth habit and environmental adaptation in cultivated barley. Nat Genet 44(12):1388–1392
Rapazote-Flores P et al (2019) BaRTv1.0: an improved barley reference transcript dataset to determine accurate changes in the barley transcriptome using RNA-seq. BMC Genomics 20(1):968
Gordon Gremme VB, Michael ES, Kurtz S (2005) Engineering a software tool for gene structure prediction in higher organisms. Inf Softw Technol 47(15):965–978
Wu TD, Watanabe CK (2005) GMAP: a genomic mapping and alignment program for mRNA and EST sequences. Bioinformatics 21(9):1859–1875
Kim D, Langmead B, Salzberg SL (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12(4):357–360
Pertea M et al (2015) StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol 33(3):290–295
Ghosh S, Chan CK (2016) Analysis of RNA-Seq data using TopHat and Cufflinks. Methods Mol Biol 1374:339–361
Haas BJ et al (2013) De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat Protoc 8(8):1494–1512
Hoff KJ, Stanke M (2019) Predicting genes in single genomes with AUGUSTUS. Curr Protoc Bioinformatics 65(1):e57
Haas BJ et al (2008) Automated eukaryotic gene structure annotation using EVidenceModeler and the program to assemble spliced alignments. Genome Biol 9(1):R7
Haas BJ et al (2003) Improving the Arabidopsis genome annotation using maximal transcript alignment assemblies. Nucleic Acids Res 31(19):5654–5666
Tomato Genome, C (2012) The tomato genome sequence provides insights into fleshy fruit evolution. Nature 485(7400):635–641
Wicker T, Matthews DE, Keller B (2002) TREP: a database for Triticeae repetitive elements. Trends Plant Sci 7(12):561–562
Seppey M, Manni M, Zdobnov EM (2019) BUSCO: assessing genome assembly and annotation completeness. Methods Mol Biol 1962:227–245
Kent WJ (2002) BLAT—the BLAST-like alignment tool. Genome Res 12(4):656–664
Slater GS, Birney E (2005) Automated generation of heuristics for biological sequence comparison. BMC Bioinformatics 6:31
Galperin MY et al (2021) COG database update: focus on microbial diversity, model organisms, and widespread pathogens. Nucleic Acids Res 49(D1):D274–D281
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32(5):1792–1797
Spannagl M et al (2016) PGSB PlantsDB: updates to the database framework for comparative plant genome research. Nucleic Acids Res 44(D1):D1141–D1147
Benson G (1999) Tandem repeats finder: a program to analyze DNA sequences. Nucleic Acids Res 27(2):573–580
Ellinghaus D, Kurtz S, Willhoeft U (2008) LTRharvest, an efficient and flexible software for de novo detection of LTR retrotransposons. BMC Bioinformatics 9:18
Howe KL et al (2020) Ensembl Genomes 2020-enabling non-vertebrate genomic research. Nucleic Acids Res 48(D1):D689–D695
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Kamal, N. et al. (2022). The Barley and Wheat Pan-Genomes. In: Edwards, D. (eds) Plant Bioinformatics. Methods in Molecular Biology, vol 2443. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2067-0_7
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2067-0_7
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2066-3
Online ISBN: 978-1-0716-2067-0
eBook Packages: Springer Protocols