Abstract
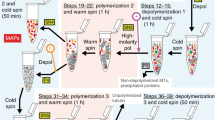
Tubulin/microtubule plays crucial role in eukaryotic cell division. Polymerization of αβ-tubulin heterodimers forms the microtubules, which is essential for the segregation of chromosomes during cell division and organelle positioning. Our method of tubulin purification from the goat brain includes isolation of goat brain, multiple cycles of polymerization (warming at 37 °C)-depolymerization (cooling at 4 °C) followed by centrifugation process. The purified tubulin from goat brain is highly functional and successfully used in different applications including reconstitution of cell like environments and understanding molecular mechanisms. Toward the end of the chapter, we have discussed, how this purified tubulin can be used for reconstitution of intracellular microtubule-associated events or function. To enable our reconstitution approach, we have developed various micropatterned-based platform and their fabrication methodology with single ligand and dual-ligand functionalizations, which are also demonstrated. These chemically functionalized micropatterned platforms are extremely useful for immobilization of tubulin/microtubule onto the localized defined area, which will be helpful in mimicking cellular phenomena like kinesin-driven transport, microtubule dynamics, etc.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Goodson HV, Jonasson EM (2018) Microtubules and microtubule-associated proteins. Cold Spring Harb Perspect Biol 10:a022608
Keating TJ, Borisy GG (1999) Centrosomal and non-centrosomal microtubules. Biol Cell 91:321–329
Alberts B (2015) Molecular biology of the cell sixth edition Ch. 16. Garland Science, Taylor and Francis Group, London, pp 889–962
Mitchison T, Kirschner M (1984) Dynamic instability of microtubule growth. Nature 312:237–242
Horio T, Hotani H (1986) Visualization of the dynamic instability of individual microtubules by dark-field microscopy. Nature 321:605–607
Aldaz H, Rice LM, Stearns T, Agard DA (2005) Insights into microtubule nucleation from the crystal structure of human gamma-tubulin. Nature 435:523–527
Stearns T, Kirschner M (1994) In vitro reconstitution of centrosome assembly and function: the central role of gamma-tubulin. Cell 76:623–637
Heald R, Tournebize R, Blank T, Sandaltzopoulos R, Becker P, Hyman A, Karsenti E (1996) Self-organization of microtubules into bipolar spindles around artificial chromosomes in Xenopus egg extracts. Nature 382:420–425
Doxsey S, McCollum D, Theurkauf W (2005) Centrosomes in cellular regulation. Annu Rev Cell Dev Biol 21:411–434
Brouhard GJ, Stear JH, Noetzel TL, Al-Bassam J, Kinoshita K, Harrison SC, Howard J, Hyman AA (2008) XMAP215 is a processive microtubule polymerase. Cell 132:79–88
Verhey KJ, Kaul N, Soppina V (2011) Kinesin assembly and movement in cells. Annu Rev Biophys 40:267–288
Ishikawa T (2017) Axoneme structure from motile cilia. Cold Spring Harb Perspect Biol 9:a028076
Singla V, Reiter JF (2006) The primary cilium as the cell's antenna: signaling at a sensory organelle. Science 313:629–633
Ibañez-Tallon I, Gorokhova S, Heintz N (2002) Loss of function of axonemal dynein Mdnah5 causes primary ciliary dyskinesia and hydrocephalus. Hum Mol Genet 11:715–721
Beswick RW, Ambrose HE, Wagner SD (2006) Nocodazole, a microtubule depolymerising agent, induces apoptosis of chronic lymphocytic leukaemia cells associated with changes in Bcl-2 phosphorylation and expression. Leuk Res 30:427–436
Horwitz SB (1994) Taxol (paclitaxel): mechanisms of action. Ann Oncol 5:S3–S6
Bieling P, Telley IA, Surrey T (2010) A minimal midzone protein module controls formation and length of antiparallel microtubule overlaps. Cell 142:420–432
Bhagawati M, Ghosh S, Reichel A, Froehner K, Surrey T, Piehler J (2009) Organization of Motor Proteins into functional micropatterns fabricated by a Photoinduced Fenton reaction. Angew Chem 121:9352–9355. Angew Chem Int Ed 48:9188–9191
Piehler J, Brecht A, Valiokas R, Liedberg B, Gauglitz G (2000) A high-density poly(ethylene glycol) polymer brush for immobilization on glass-type surfaces. Biosens Bioelectron 15:473–481
Bieling P, Laan L, Schek H, Munteanu EL, Sandblad L, Dogterom M, Brunner D, Surrey T (2007) Reconstitution of a microtubule plus-end tracking system in vitro. Nature 450:1100–1105
Biswas A, Saha A, Jana B, Kurkute P, Mondal G, Ghosh S (2013) A biotin micropatterned surface generated by Photodestruction serves as a novel platform for microtubule organisation and DNA hybridisation. Chembiochem 14:689–694
Biswas A, Saha A, Ghosh D, Jana B, Ghosh S (2014) Co-and distinct existence of Tris-NTA and biotin functionalities on individual and adjacent micropatterned surfaces generated by photo-destruction. Soft Matter 10:2341–2345
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Ghosh, S., Garg, S., Mukherjee, N., Ghosh, S. (2022). Facile Method of Tubulin Purification from Goat Brain for Reconstitution of Microtubule-Associated Intracellular Function. In: Inaba, H. (eds) Microtubules. Methods in Molecular Biology, vol 2430. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1983-4_2
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1983-4_2
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1982-7
Online ISBN: 978-1-0716-1983-4
eBook Packages: Springer Protocols