Abstract
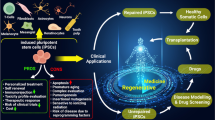
In an increasingly geriatric population, in which elderly people frequently face chronic diseases and degenerative conditions, cell therapies as part of novel regenerative medicine approaches are of great interest. Even though today’s cell therapies mostly rely on adult stem cells like the mesenchymal stem cells or primary somatic cells, pluripotent stem cells represent an enormously versatile cell model to explore possible new avenues in the field of regenerative medicine due to their capacity to grow indefinitely and to differentiate into the desired cell types. The discovery of reprogramming somatic cells into induced pluripotent stem cells augmented the pool of applicable cell entities so that researchers nowadays can resort to embryonic stem cells, but also to a plethora of patient- and disease-specific induced pluripotent stem cells. The ease of targeted genome engineering is an additional benefit that allows using pluripotent stem cells for disease modeling, drug discovery, and the development of cell therapies. However, the task is still demanding as the generation of subpopulations and a sufficient cell maturation for some cell entities have yet to be achieved. Likewise, even though for some applications the cells of interest can be produced in the large-scale dimensions and purity that are required for clinical purposes, proper integration, and function in the host tissue remain challenging. Nonetheless, the immense progress that has been made over the last decades warrants the prominent role of pluripotent stem cells in regenerative medicine as in vitro models to broaden our knowledge of disease onset/progression and treatment as well as in vivo as a substitution of damaged/aged tissue.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Joseph P, Leong D, McKee M et al (2017) Reducing the global burden of cardiovascular disease, part 1: the epidemiology and risk factors. Circ Res 121:677–694
Lee JH, Protze SI, Laksman Z et al (2017) Human pluripotent stem cell-derived atrial and ventricular cardiomyocytes develop from distinct mesoderm populations. Cell Stem Cell 21:179–194
Zhang JZ, Termglinchan V, Shao NY et al (2019) A human iPSC double-reporter system enables purification of cardiac lineage subpopulations with distinct function and drug response profiles. Cell Stem Cell 24:802–811
Devalla HD, Schwach V, Ford JW (2015) Atrial-like cardiomyocytes from human pluripotent stem cells are a robust preclinical model for assessing atrial-selective pharmacology. EMBO Mol Med 7:394–410
Laksman Z, Wauchop M, Lin E et al (2017) Modeling atrial fibrillation using human embryonic stem cell-derived atrial tissue. Sci Rep 7:5268
Lemme M, Ulmer BM, Lemoine MD et al (2018) Atrial-like engineered heart tissue: an in vitro model of the human atrium. Stem Cell Reports 11:1378–1390
Lemme M, Ulmer BM, Lemoine MD et al (2018) Atrial-like engineered heart tissue: an in vitro model of the human atrium. Stem Cell Rep 11:1378–1390
Birket MJ, Ribeiro MC, Verkerk AO et al (2015) Expansion and patterning of cardiovascular progenitors derived from human pluripotent stem cells. Nat Biotechnol 33:970–979
Liang W, Han P, Kim EH et al (2019) Canonical Wnt signaling promotes pacemaker cell specification of cardiac mesodermal cells derived from mouse and human embryonic stem cells. Stem Cells 38:352. https://doi.org/10.1002/stem.3106
Schweizer PA, Darche FF, Ullrich ND et al (2017) Subtype-specific differentiation of cardiac pacemaker cell clusters from human induced pluripotent stem cells. Stem Cell Res Ther 8:229
Gambardella L, McManus SA, Moignard V et al (2019) BNC1 regulates cell heterogeneity in human pluripotent stem cell derived-epicardium. Development 146:dev174441. https://doi.org/10.1242/dev.174441
Guadix JA, Orlova VV, Giacomelli E et al (2017) Human pluripotent stem cell differentiation into functional epicardial progenitor cells. Stem Cell Rep 9:1754–1764
Iyer D, Gambardella L, Bernard WG et al (2016) Robust derivation of epicardium and its differentiated smooth muscle cell progeny from human pluripotent stem cells. Development 143:904
Piquereau J, Ventura-Clapier R (2018) Maturation of cardiac energy metabolism during perinatal development. Front Physiol 9:959
Parikh SS, Blackwell DJ, Gomez-Hurtado N et al (2017) Thyroid and glucocorticoid hormones promote functional T-tubule development in human-induced pluripotent stem cell-derived cardiomyocytes. Circ Res 121:1323–1330
Correia C, Koshkin A, Duarte P et al (2017) Distinct carbon sources affect structural and functional maturation of cardiomyocytes derived from human pluripotent stem cells. Sci Rep 7:8590
Horikoshi Y, Yan Y, Terashvili M et al (2019) Fatty acid-treated induced pluripotent stem cell-derived human cardiomyocytes exhibit adult cardiomyocyte-like energy metabolism phenotypes. Cell 8(9):1095. https://doi.org/10.3390/cells8091095
Lin B, Lin X, Stachel M et al (2017) Culture in glucose-depleted medium supplemented with fatty acid and 3,3′,5-triiodo-l-thyronine facilitates purification and maturation of human pluripotent stem cell-derived cardiomyocytes. Front Endocrinol (Lausanne) 8:253
Kadari A, Mekala S, Wagner N et al (2015) Robust generation of cardiomyocytes from human iPS cells requires precise modulation of BMP and WNT signaling. Stem Cell Rev Rep 11:560–569
Moretti A, Bellin M, Welling A et al (2010) Patient-specific induced pluripotent stem-cell models for long-QT syndrome. N Engl J Med 363:1397–1409
Benzoni P, Campostrini G, Landi S et al (2019) Human iPSC modeling of a familial form of atrial fibrillation reveals a gain of function of if and ICaL in patient-derived cardiomyocytes. Cardiovasc Res 116:1147. https://doi.org/10.1093/cvr/cvz217
Shafaattalab S, Lin E, Christidi E (2019) Ibrutinib displays atrial-specific toxicity in human stem cell-derived cardiomyocytes. Stem Cell Rep 12:996–1006
Shiba Y, Fernandes S, Zhu WZ et al (2012) Human ES-cell-derived cardiomyocytes electrically couple and suppress arrhythmias in injured hearts. Nature 489:322–325
Liu YW, Chen B, Yang X et al (2018) Erratum: human embryonic stem cell-derived cardiomyocytes restore function in infarcted hearts of non-human primates. Nat Biotechnol 36:899
Liu YW, Chen B, Yang X et al (2018) Human embryonic stem cell-derived cardiomyocytes restore function in infarcted hearts of non-human primates. Nat Biotechnol 36:597–605
Romagnuolo R, Masoudpour H, Porta-Sanchez A et al (2019) Human embryonic stem cell-derived cardiomyocytes regenerate the infarcted pig heart but induce ventricular tachyarrhythmias. Stem Cell Rep 12:967–981
Menasche P, Vanneaux V, Hagege A et al (2018) Transplantation of human embryonic stem cell-derived cardiovascular progenitors for severe ischemic left ventricular dysfunction. J Am Coll Cardiol 71:429–438
Martin U (2017) Therapeutic application of pluripotent stem cells: challenges and risks. Front Med (Lausanne) 4:229
Benito-Kwiecinski S, Lancaster MA (2019) Brain organoids: human neurodevelopment in a dish. Cold Spring Harb Perspect Biol 12:a035709. https://doi.org/10.1101/cshperspect.a035709
Sakaguchi H, Kadoshima T, Soen M et al (2015) Generation of functional hippocampal neurons from self-organizing human embryonic stem cell-derived dorsomedial telencephalic tissue. Nat Commun 6:8896
Qian X, Nguyen HN, Song MM et al (2016) Brain-region-specific organoids using mini-bioreactors for modeling ZIKV exposure. Cell 165:1238–1254
Qian X, Jacob F, Song MM et al (2018) Generation of human brain region-specific organoids using a miniaturized spinning bioreactor. Nat Protoc 13:565–580
Muguruma K, Nishiyama A, Kawakami H et al (2015) Self-organization of polarized cerebellar tissue in 3D culture of human pluripotent stem cells. Cell Rep 10:537–550
Meinhardt A, Eberle D, Tazaki A et al (2014) 3D reconstitution of the patterned neural tube from embryonic stem cells. Stem Cell Rep 3:987–999
Lancaster MA, Renner M, Martin CA et al (2013) Cerebral organoids model human brain development and microcephaly. Nature 501:373–379
Lancaster MA, Knoblich JA (2014) Generation of cerebral organoids from human pluripotent stem cells. Nat Protoc 9:2329–2340
Mansour AA, Goncalves JT, Bloyd CW et al (2018) Erratum: an in vivo model of functional and vascularized human brain organoids. Nat Biotechnol 36:772
Mansour AA, Goncalves JT, Bloyd CW (2018) An in vivo model of functional and vascularized human brain organoids. Nat Biotechnol 36:432–441
Xiang Y, Yoshiaki T, Patterson B (2018) Generation and fusion of human cortical and medial ganglionic Eminence brain organoids. Curr Protoc Stem Cell Biol 47(1). https://doi.org/10.1002/cpsc.61
Song L, Yuan X, Jones Z et al (2019) Assembly of human stem cell-derived cortical spheroids and vascular spheroids to model 3-D brain-like tissues. Sci Rep 9:5977
Yoon SJ, Elahi LS, Pasca AM et al (2019) Reliability of human cortical organoid generation. Nat Methods 16:75–78
Amin ND, Pasca SP (2018) Building models of brain disorders with three-dimensional organoids. Neuron 100:389–405
Kamao H, Mandai M, Okamoto S et al (2014) Characterization of human induced pluripotent stem cell-derived retinal pigment epithelium cell sheets aiming for clinical application. Stem Cell Rep 2:205–218
Matsumoto E, Koide N, Hanzawa H et al (2019) Fabricating retinal pigment epithelial cell sheets derived from human induced pluripotent stem cells in an automated closed culture system for regenerative medicine. PLoS One 14:e0212369
Kawamata S, Kanemura H, Sakai N et al (2015) Design of a tumorigenicity test for induced pluripotent stem cell (iPSC)-derived cell products. J Clin Med 4:159–171
Hori K, Kuwabara J, Tanaka Y (2019) A simple and static preservation system for shipping retinal pigment epithelium cell sheets. J Tissue Eng Regen Med 13:459–468
Kitahata S, Tanaka Y, Hori K et al (2019) Critical functionality effects from storage temperature on human induced pluripotent stem cell-derived retinal pigment epithelium cell suspensions. Sci Rep 9:2891
Takagi S, Mandai M, Gocho K et al (2019) Evaluation of transplanted autologous induced pluripotent stem cell-derived retinal pigment epithelium in exudative age-related macular degeneration. Ophthalmol Retina 3:850–859
Mandai M, Watanabe A, Kurimoto Y et al (2017) Autologous induced stem-cell-derived retinal cells for macular degeneration. N Engl J Med 376:1038–1046
Pagliuca FW, Millman JR, Gurtler M et al (2014) Generation of functional human pancreatic beta cells in vitro. Cell 159:428–439
Cogger KF, Sinha A, Sarangi F et al (2017) Glycoprotein 2 is a specific cell surface marker of human pancreatic progenitors. Nat Commun 8:331
Millman JR, Xie C, Van DA et al (2016) Corrigendum: generation of stem cell-derived beta-cells from patients with type 1 diabetes. Nat Commun 7:12,379
Millman JR, Xie C, Van DA (2016) Generation of stem cell-derived beta-cells from patients with type 1 diabetes. Nat Commun 7:11,463
Acknowledgments
I.S.S. is funded by the German Federal Ministry of Education and Research (BMBF, support code 02NUK049A).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Schroeder, I.S. (2021). Pluripotent Stem Cells for Cell Therapy. In: Stock, P., Christ, B. (eds) In Vitro Models for Stem Cell Therapy. Methods in Molecular Biology, vol 2269. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1225-5_2
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1225-5_2
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1224-8
Online ISBN: 978-1-0716-1225-5
eBook Packages: Springer Protocols