Abstract
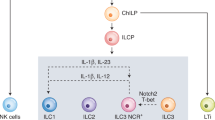
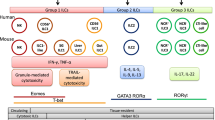
Innate lymphoid cells (ILCs) are important for both tissue immunity and tissue homeostasis. They are classified into three groups: Group 1 ILCs include NK cells, which are important in eliciting immunity against intracellular pathogens; group 2 ILCs protect against parasitic helminths; and group 3 ILCs protect against extracellular pathogens. The role of ILCs in cancer immunity remains unclear. In this chapter, we discuss methods for isolating and characterizing tumor-infiltrating ILC subsets within the tumor microenvironment in an experimental murine model of B16 melanoma. The chapter also highlights the expression of PD-1 on the various ILC subsets within the tumor microenvironment.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Salimi M, Barlow JL, Saunders SP et al (2013) A role for IL-25 and IL-33-driven type-2 innate lymphoid cells in atopic dermatitis. J Exp Med 210(13):2939–2950
Geremia A, Arancibia-Carcamo CV, Fleming MP et al (2011) IL-23-responsive innate lymphoid cells are increased in inflammatory bowel disease. J Exp Med 208(6):1127–1133
Spits H, Artis D, Colonna M et al (2013) Innate lymphoid cells—a proposal for uniform nomenclature. Nat Rev Immunol 13(2):145–149
Crellin NK, Trifari S, Kaplan CD et al (2010) Regulation of cytokine secretion in human CD127(+) LTi-like innate lymphoid cells by Toll-like receptor 2. Immunity 33(5):752–764
Herberman RB, Nunn ME, Holden HT et al (1975) Natural cytotoxic reactivity of mouse lymphoid cells against syngeneic and allogeneic tumors. II. Characterization of effector cells. Int J Cancer 16(2):230–239
Weizman OE, Adams NM, Schuster IS et al (2017) ILC1 confer early host protection at initial sites of viral infection. Cell 171(4):795–808.e12
Klose CSN, Kiss EA, Schwierzeck V et al (2014) Differentiation of type 1 ILCs from a common progenitor to all helper-like innate lymphoid cell lineages. Cell 157(2):340–356
Neill DR, Wong SH, Bellosi A et al (2010) Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature 464(7293):1367–1370
Huang Y, Guo L, Qiu J et al (2015) IL-25-responsive, lineage-negative KLRG1(hi) cells are multipotential ‘inflammatory’ type 2 innate lymphoid cells. Nat Immunol 16(2):161–169
Sanos SL, Bui VL, Mortha A et al (2009) RORgammat and commensal microflora are required for the differentiation of mucosal interleukin 22-producing NKp46+ cells. Nat Immunol 10(1):83–91
Cella M, Fuchs A, Vermi W et al (2009) A human natural killer cell subset provides an innate source of IL-22 for mucosal immunity. Nature 457(7230):722–725
Buonocore S, Ahern PP, Uhlig HH et al (2010) Innate lymphoid cells drive interleukin-23-dependent innate intestinal pathology. Nature 464(7293):1371–1375
Sonnenberg GF, Monticelli LA, Ellosi MM et al (2011) CD4(+) lymphoid tissue-inducer cells promote innate immunity in the gut. Immunity 34(1):122–134
Takatori H, Kanno Y, Watford WT et al (2009) Lymphoid tissue inducer-like cells are an innate source of IL-17 and IL-22. J Exp Med 206(1):35–41
Morvan MG, Lanier LL (2016) NK cells and cancer: you can teach innate cells new tricks. Nat Rev Cancer 16(1):7–19
Martini M, Testi MG, Passetto M et al (2010) IFN-gamma-mediated upmodulation of MHC class I expression activates tumor-specific immune response in a mouse model of prostate cancer. Vaccine 28(20):3548–3557
Nakajima C, Uekusa Y, Iwasaki M et al (2001) A role of interferon-gamma (IFN-gamma) in tumor immunity: T cells with the capacity to reject tumor cells are generated but fail to migrate to tumor sites in IFN-gamma-deficient mice. Cancer Res 61(8):3399–3405
Beatty G, Paterson Y (2001) IFN-gamma-dependent inhibition of tumor angiogenesis by tumor-infiltrating CD4+ T cells requires tumor responsiveness to IFN-gamma. J Immunol 166(4):2276–2282
Larmonier N, Cathelin D, Larmonier C et al (2007) The inhibition of TNF-alpha anti-tumoral properties by blocking antibodies promotes tumor growth in a rat model. Exp Cell Res 313(11):2345–2355
Jovanovic IP, Pejnovic N, Radosavljevic GD et al (2014) Interleukin-33/ST2 axis promotes breast cancer growth and metastases by facilitating intratumoral accumulation of immunosuppressive and innate lymphoid cells. Int J Cancer 134(7):1669–1682
Molofsky AB, Nussbaum JC, Liang HE et al (2013) Innate lymphoid type 2 cells sustain visceral adipose tissue eosinophils and alternatively activated macrophages. J Exp Med 210(3):535–549
Huber S, Gagliani N, Zenewicz LA et al (2012) IL-22BP is regulated by the inflammasome and modulates tumorigenesis in the intestine. Nature 491(7423):259–263
Chan IH, Jain R, Tessmer MS et al (2014) Interleukin-23 is sufficient to induce rapid de novo gut tumorigenesis, independent of carcinogens, through activation of innate lymphoid cells. Mucosal Immunol 7(4):842–856
Schatton T, Murphy GF, Frank NY et al (2008) Identification of cells initiating human melanomas. Nature 451(7176):345–349
Acknowledgments
We would like to thank the Academy of Medical Sciences, Springboard Award [SBF003\1129] supported by Wellcome Trust and Newcastle University Research Fellowship for supporting this work. G.M. is supported by a Medical Research Council [MRC] Discovery Medicine North Ph.D. studentship. M.P. is supported by a Wellcome Trust Vacation Scholarship [216503/Z/19/Z].
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Mallett, G., Patterson, W., Payne, M., Amarnath, S. (2020). Isolation and Characterization of Innate Lymphoid Cells within the Murine Tumor Microenvironment. In: Amarnath, S. (eds) Innate Lymphoid Cells . Methods in Molecular Biology, vol 2121. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0338-3_14
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0338-3_14
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0337-6
Online ISBN: 978-1-0716-0338-3
eBook Packages: Springer Protocols