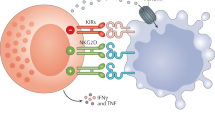
Abstract
Natural killer (NK) cells can kill virus-infected cells and tumor cells without prior sensitization and secrete numerous cytokines and chemokines that modulate the activity of different cells of the immune system. The recognition of target cells is mediated by germ line-encoded receptors, and the activity of NK cells can be further regulated by soluble factors such as cytokines and Toll-like receptor ligands. Thus, NK cells display an exciting potential as a powerful immunotherapeutic tool against malignant diseases, and different strategies are being tested aiming to overcome tumor-induced NK cell suppression and restore NK-cell mediated antitumor activity. This section describes different flow cytometry-based protocols to study NK cell effector functions, which can be used to evaluate the immunomodulatory ability of different therapeutic compounds.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Diefenbach A, Colonna M, Romagnani C (2017) The ILC world revisited. Immunity 46:327–332
Vivier E, Tomasello E, Baratin M, Walzer T, Ugolini S (2008) Functions of natural killer cells. Nat Immunol 9:503–510
Lanier LL (2008) Up on the tightrope: natural killer cell activation and inhibition. Nat Immunol 9:495–502
Fauriat C, Long EO, Ljunggren HG, Bryceson YT (2010) Regulation of human NK-cell cytokine and chemokine production by target cell recognition. Blood 115:2167–2176
Long EO, Kim HS, Liu D, Peterson ME, Rajagopalan S (2013) Controlling natural killer cell responses: integration of signals for activation and inhibition. Annu Rev Immunol 31:227–258
Lanier LL (2005) NK cell recognition. Annu Rev Immunol 23:225–274
Zwirner NW, Domaica CI (2010) Cytokine regulation of natural killer cell effector functions. Biofactors 36:274–288
Sivori S, Carlomagno S, Pesce S, Moretta A, Vitale M, Marcenaro E (2014) TLR/NCR/KIR: which one to use and when? Front Immunol 5:105
Caligiuri MA (2008) Human natural killer cells. Blood 112:461–469
Cooper MA, Fehniger TA, Caligiuri MA (2001) The biology of human natural killer-cell subsets. Trends Immunol 22:633–640
Montaldo E, Del Zotto G, Della Chiesa M, Mingari MC, Moretta A, De Maria A, Moretta L (2013) Human NK cell receptors/markers: a tool to analyze NK cell development, subsets and function. Cytometry A 83:702–713
Wilk AJ, Blish CA (2018) Diversification of human NK cells: lessons from deep profiling. J Leukoc Biol 103:629–641
Freud AG, Mundy-Bosse BL, Yu J, Caligiuri MA (2017) The broad Spectrum of human natural killer cell diversity. Immunity 47:820–833
Horowitz A, Strauss-Albee DM, Leipold M, Kubo J, Nemat-Gorgani N, Dogan OC, Dekker CL, Mackey S, Maecker H, Swan GE, Davis MM, Norman PJ, Guethlein LA, Desai M, Parham P, Blish CA (2013) Genetic and environmental determinants of human NK cell diversity revealed by mass cytometry. Sci Transl Med 5:208ra145
Lopez-Verges S, Milush JM, Schwartz BS, Pando MJ, Jarjoura J, York VA, Houchins JP, Miller S, Kang SM, Norris PJ, Nixon DF, Lanier LL (2011) Expansion of a unique CD57(+)NKG2Chi natural killer cell subset during acute human cytomegalovirus infection. Proc Natl Acad Sci U S A 108:14725–14732
Peng H, Tian Z (2017) Diversity of tissue-resident NK cells. Semin Immunol 31:3–10
Morvan MG, Lanier LL (2016) NK cells and cancer: you can teach innate cells new tricks. Nat Rev Cancer 16:7–19
Guillerey C, Huntington ND, Smyth MJ (2016) Targeting natural killer cells in cancer immunotherapy. Nat Immunol 17:1025–1036
Childs RW, Carlsten M (2015) Therapeutic approaches to enhance natural killer cell cytotoxicity against cancer: the force awakens. Nat Rev Drug Discov 14:487–498
Dall’Ozzo S, Tartas S, Paintaud G, Cartron G, Colombat P, Bardos P, Watier H, Thibault G (2004) Rituximab-dependent cytotoxicity by natural killer cells: influence of FCGR3A polymorphism on the concentration-effect relationship. Cancer Res 64:4664–4669
Kawaguchi Y, Kono K, Mimura K, Sugai H, Akaike H, Fujii H (2007) Cetuximab induce antibody-dependent cellular cytotoxicity against EGFR-expressing esophageal squamous cell carcinoma. Int J Cancer 120:781–787
Kim GG, Donnenberg VS, Donnenberg AD, Gooding W, Whiteside TL (2007) A novel multiparametric flow cytometry-based cytotoxicity assay simultaneously immunophenotypes effector cells: comparisons to a 4 h 51Cr-release assay. J Immunol Methods 325:51–66
Alter G, Malenfant JM, Altfeld M (2004) CD107a as a functional marker for the identification of natural killer cell activity. J Immunol Methods 294:15–22
Schuerwegh AJ, Stevens WJ, Bridts CH, De Clerck LS (2001) Evaluation of monensin and brefeldin a for flow cytometric determination of interleukin-1 beta, interleukin-6, and tumor necrosis factor-alpha in monocytes. Cytometry 46:172–176
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Domaica, C.I., Sierra, J.M., Zwirner, N.W., Fuertes, M.B. (2020). Immunomodulation of NK Cell Activity. In: Katz, S., Rabinovich, P. (eds) Cell Reprogramming for Immunotherapy. Methods in Molecular Biology, vol 2097. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0203-4_9
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0203-4_9
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0202-7
Online ISBN: 978-1-0716-0203-4
eBook Packages: Springer Protocols