Abstract
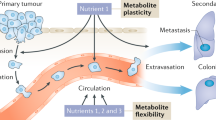
Metastasis formation is the leading cause of death in cancer patients. It has recently emerged that cancer cells adapt their metabolism to successfully transition through the metastatic cascade. Consequently, measuring and analyzing the in vivo metabolism of metastases has the potential to reveal novel treatment strategies to prevent metastasis formation. Here, we describe two different metastasis mouse models and how their metabolism can be analyzed with metabolomics and 13C tracer analysis.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Sullivan LB, Gui DY, Vander Heiden MG (2016) Altered metabolite levels in cancer: implications for tumour biology and cancer therapy. Nat Rev Cancer 16:680–693
Lorendeau D, Christen S, Rinaldi G, Fendt S-M (2015) Metabolic control of signalling pathways and metabolic auto-regulation. Biol Cell 107:251–272
Lunt SY, Fendt S-M (2018) Metabolism – a cornerstone of cancer initiation, progression, immune evasion and treatment response. Curr Opin Syst Biol 8:67–72
Elia I, Doglioni G, Fendt S-M (2018) Metabolic hallmarks of metastasis formation. Trends Cell Biol 28:673–684
Doglioni G, Parik S, Fendt S-M (2019) Interactions in the (pre)metastatic niche support metastasis formation. Front Oncol 9:219
Vander Heiden MG, DeBerardinis RJ (2017) Understanding the intersections between metabolism and cancer biology. Cell 168:657–669
Rinaldi G, Rossi M, Fendt S-M (2018) Metabolic interactions in cancer: cellular metabolism at the interface between the microenvironment, the cancer cell phenotype and the epigenetic landscape. Wiley Interdiscip Rev Syst Biol Med 10:e1397
Elia I, Schmieder R, Christen S, Fendt S-M (2015) Organ-specific cancer metabolism and its potential for therapy. Handb Exp Pharmacol 233:321–353
Elia I, Fendt S-M (2016) In vivo cancer metabolism is defined by the nutrient microenvironment. Transl Cancer Res 5:S1284–S1287
Broekaert D, Fendt SM (2019) Measuring in vivo tissue metabolism using 13C glucose infusions in mice. Methods Mol Biol 1862:67–82
Faubert B, DeBerardinis RJ (2017) Analyzing tumor metabolism in vivo. Annu Rev Cancer Biol 1:99–117
Buescher JM, Antoniewicz MR, Boros LG, Burgess SC, Brunengraber H, Clish CB, DeBerardinis RJ, Feron O, Frezza C, Ghesquiere B et al (2015) A roadmap for interpreting 13 C metabolite labeling patterns from cells. Curr Opin Biotechnol 34:189–201
Christen S, Lorendeau D, Schmieder R, Broekaert D, Metzger K, Veys K, Elia I, Buescher JM, Orth MF, Davidson SM et al (2016) Breast cancer-derived lung metastases show increased pyruvate carboxylase-dependent Anaplerosis. Cell Rep 17:837–848
Elia I, Broekaert D, Christen S, Boon R, Radaelli E, Orth MF, Verfaillie C, Grünewald TGP, Fendt S-M (2017) Proline metabolism supports metastasis formation and could be inhibited to selectively target metastasizing cancer cells. Nat Commun 8:15267
Elia I, Rossi M, Stegen S, Broekaert D, Doglioni G, van Gorsel M, Boon R, Escalona-Noguero C, Torrekens S, Verfaillie C et al (2019) Breast cancer cells rely on environmental pyruvate to shape the metastatic niche. Nature 568:117–121
Lorendeau D, Rinaldi G, Boon R, Spincemaille P, Metzger K, Jäger C, Christen S, Dong X, Kuenen S, Voordeckers K et al (2017) Dual loss of succinate dehydrogenase (SDH) and complex I activity is necessary to recapitulate the metabolic phenotype of SDH mutant tumors. Metab Eng 43:187–197
Feldman AT, Wolfe D (2014) Tissue processing and Hematoxylin and eosin staining. Methods Mol Biol 1180:31–43
Hewitson TD, Wigg B, Becker GJ (2010) Tissue preparation for Histochemistry: fixation, embedding, and antigen retrieval for light microscopy. Methods Mol Biol 611:3–18
Shrivastava A, Gupta V (2011) Methods for the determination of limit of detection and limit of quantitation of the analytical methods. Chronicles Young Sci 2:21
Fernandez CA, Des RC, Previs SF, David F, Brunengraber H (1996) Correction of 13C mass Isotopomer distributions for natural stable isotope abundance. J Mass Spectrom 31:255–262
Millard P, Letisse F, Sokol S, Portais J-C (2012) IsoCor: correcting MS data in isotope labeling experiments. Bioinformatics 28:1294–1296
Pierozan P, Jernerén F, Ransome Y, Karlsson O (2017) The choice of euthanasia method affects metabolic serum biomarkers. Basic Clin Pharmacol Toxicol 121:113–118
Overmyer KA, Thonusin C, Qi NR, Burant CF, Evans CR (2015) Impact of anesthesia and euthanasia on metabolomics of mammalian tissues: studies in a C57BL/6J mouse model. PLoS One 10:e0117232
Brooks SP, Lampi BJ, Bihun CG (1999) The influence of euthanasia methods on rat liver metabolism. Contemp Top Lab Anim Sci 38(6):19–24
Hui S, Ghergurovich JM, Morscher RJ, Jang C, Teng X, Lu W, Esparza LA, Reya T, Le Zhan L, Yanxiang Guo J et al (2017) Glucose feeds the TCA cycle via circulating lactate. Nature 551:115–118
Davidson SM, Papagiannakopoulos T, Olenchock BA, Heyman JE, Keibler MA, Luengo A, Bauer MR, Jha AK, O’Brien JP, Pierce KA et al (2016) Environment impacts the metabolic dependencies of Ras-driven non-small cell lung cancer. Cell Metab 23:517–528
Faubert B, Li KY, Cai L, Hensley CT, Kim J, Zacharias LG, Yang C, Do QN, Doucette S, Burguete D et al (2017) Lactate metabolism in human lung tumors. Cell 171:358–371.e9
Courtney KD, Bezwada D, Mashimo T, Pichumani K, Vemireddy V, Funk AM, Wimberly J, McNeil SS, Kapur P, Lotan Y et al (2018) Isotope tracing of human clear cell renal cell carcinomas demonstrates suppressed glucose oxidation in vivo. Cell Metab 28:793–800
Fan TWM, Lane AN, Higashi RM, Farag MA, Gao H, Bousamra M, Miller DM (2009) Altered regulation of metabolic pathways in human lung cancer discerned by 13Cstable isotope-resolved metabolomics (SIRM). Mol Cancer 8:1–19
Sellers K, Fox MP, Bousamra M, Slone SP, Higashi RM, Miller DM, Wang Y, Yan J, Yuneva MO, Deshpande R et al (2015) Pyruvate carboxylase is critical for non–small-cell lung cancer proliferation. J Clin Invest 125:687–698
Hensley CT, Faubert B, Yuan Q, Lev-Cohain N, Jin E, Kim J, Jiang L, Ko B, Skelton R, Loudat L et al (2016) Metabolic heterogeneity in human lung tumors. Cell 164:681–694
Tardito S, Oudin A, Ahmed SU, Fack F, Keunen O, Zheng L, Miletic H, Sakariassen PØ, Weinstock A, Wagner A et al (2015) Glutamine synthetase activity fuels nucleotide biosynthesis and supports growth of glutamine-restricted glioblastoma. Nat Cell Biol 17:1556–1568
Marin-Valencia I, Yang C, Mashimo T, Cho S, Baek H, Yang X-L, Rajagopalan KN, Maddie M, Vemireddy V, Zhao Z et al (2012) Analysis of tumor metabolism reveals mitochondrial glucose oxidation in genetically diverse human Glioblastomas in the mouse brain in vivo. Cell Metab 15:827–837
Maher EA, Marin-Valencia I, Bachoo RM, Mashimo T, Raisanen J, Hatanpaa KJ, Jindal A, Jeffrey FM, Choi C, Madden C et al (2012) Metabolism of [U-13C]glucose in human brain tumors in vivo. NMR Biomed 25:1234–1244
Yuneva MO, Fan TWM, Allen TD, Higashi RM, Ferraris DV, Tsukamoto T, Matés JM, Alonso FJ, Wang C, Seo Y et al (2012) The metabolic profile of tumors depends on both the responsible genetic lesion and tissue type. Cell Metab 15:157–170
Kucejova B, Duarte J, Satapati S, Fu X, Ilkayeva O, Newgard CB, Brugarolas J, Burgess SC (2016) Hepatic mTORC1 opposes impaired insulin action to control mitochondrial metabolism in obesity. Cell Rep 16:508–519
Satapati S, Kucejova B, Duarte JAG, Fletcher JA, Reynolds L, Sunny NE, He T, Nair LA, Livingston KA, Livingston K et al (2015) Mitochondrial metabolism mediates oxidative stress and inflammation in fatty liver. J Clin Invest 125:4447–4462
Satapati S, Sunny NE, Kucejova B, Fu X, He TT, Méndez-Lucas A, Shelton JM, Perales JC, Browning JD, Burgess SC (2012) Elevated TCA cycle function in the pathology of diet-induced hepatic insulin resistance and fatty liver. J Lipid Res 53:1080–1092
Mashimo T, Pichumani K, Vemireddy V, Hatanpaa KJ, Singh DK, Sirasanagandla S, Nannepaga S, Piccirillo SG, Kovacs Z, Foong C et al (2014) Acetate is a bioenergetic substrate for human glioblastoma and brain metastases. Cell 159:1603–1614
Kim C-W, Addy C, Kusunoki J, Anderson NN, Deja S, Fu X, Burgess SC, Li C, Ruddy M, Chakravarthy M et al (2017) Acetyl CoA carboxylase inhibition reduces hepatic Steatosis but elevates plasma triglycerides in mice and humans: a bedside to bench investigation. Cell Metab 26:394–406.e6
Kennedy KM, Scarbrough PM, Ribeiro A, Richardson R, Yuan H, Sonveaux P, Landon CD, Chi J-T, Pizzo S, Schroeder T et al (2013) Catabolism of exogenous lactate reveals it as a legitimate metabolic substrate in breast cancer. PLoS One 8:e75154
Rauckhorst AJ, Gray LR, Sheldon RD, Fu X, Pewa AD, Feddersen CR, Dupuy AJ, Gibson-Corley KN, Cox JE, Burgess SC et al (2017) The mitochondrial pyruvate carrier mediates high fat diet-induced increases in hepatic TCA cycle capacity. Mol Metab 6:1468–1479
Vatner DF, Majumdar SK, Kumashiro N, Petersen MC, Rahimi Y, Gattu AK, Bears M, Camporez J-PG, Cline GW, Jurczak MJ et al (2015) Insulin-independent regulation of hepatic triglyceride synthesis by fatty acids. Proc Natl Acad Sci U S A 112:1143–1148
DeLany JP, Windhauser MM, Champagne CM, Bray GA (2000) Differential oxidation of individual dietary fatty acids in humans. Am J Clin Nutr 72:905–911
Sidossis LS, Coggan AR, Gastaldelli A, Wolfe RR (1995) Pathway of free fatty acid oxidation in human subjects. Implications for tracer studies. J Clin Invest 95:278–284
Blaak EE, Wagenmakers AJM (2002) The fate of [U-(13)C]palmitate extracted by skeletal muscle in subjects with type 2 diabetes and control subjects. Diabetes 51:784–789
Gallego S, Hermansson M, Liebisch G, Hodson L, Ejsing C, Gallego SF, Hermansson M, Liebisch G, Hodson L, Ejsing CS (2018) Total fatty acid analysis of human blood samples in one minute by high-resolution mass spectrometry. Biomol Ther 9:7
Ducker GS, Chen L, Morscher RJ, Ghergurovich JM, Esposito M, Teng X, Kang Y, Rabinowitz JD (2016) Reversal of cytosolic one-carbon flux compensates for loss of the mitochondrial Folate pathway. Cell Metab 23:1140–1153
Neinast MD, Jang C, Hui S, Murashige DS, Chu Q, Morscher RJ, Li X, Zhan L, White E, Anthony TG et al (2018) Quantitative analysis of the whole-body metabolic fate of branched-chain amino acids. Cell Metab 29(2):417–429
Strong JM, Anderson LW, Monks A, Chisena CA, Cysyk RL (1983) A 13C tracer method for quantitating de novo pyrimidine biosynthesis in vitro and in vivo. Anal Biochem 132:243–253
Busch R, Kim Y-K, Neese RA, Schade-Serin V, Collins M, Awada M, Gardner JL, Beysen C, Marino ME, Misell LM et al (2006) Measurement of protein turnover rates by heavy water labeling of nonessential amino acids. Biochim Biophys Acta 1760:730–744
Pinnick KE, Gunn PJ, Hodson L (2019) Measuring human lipid metabolism using deuterium labeling: in vivo and in vitro protocols. Methods Mol Biol 1862:83–96
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Altea-Manzano, P., Broekaert, D., Duarte, J.A.G., Fernández-García, J., Planque, M., Fendt, SM. (2020). Analyzing the Metabolism of Metastases in Mice. In: Nagrath, D. (eds) Metabolic Flux Analysis in Eukaryotic Cells. Methods in Molecular Biology, vol 2088. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0159-4_6
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0159-4_6
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0158-7
Online ISBN: 978-1-0716-0159-4
eBook Packages: Springer Protocols