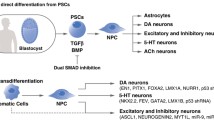
Abstract
Human embryonic stem cell (hESC) and human-induced pluripotent stem cell (hiPSC) technologies have a critical role in regenerative strategies for personalized medicine. Both share the ability to differentiate into almost any cell type of the human body. The study of their properties and clinical applications requires the development of robust and reproducible cell culture paradigms that direct cell differentiation toward a specific phenotype in vitro and in vivo. Our group evaluated the potential of mouse ESCs (mESCs), hESCs, and hiPSCs (collectively named pluripotent stem cells, PSCs) to analyze brain microenvironments through the use of embryoid body (EB)-derived cells from these cell sources. EB are cell aggregates in 3D culture conditions that recapitulate embryonic development. Our approach focuses on studying the midbrain dopaminergic phenotype and transplanting EB into the substantia nigra pars compacta (SNpc) in a Parkinson’s disease rodent model. Here, we describe cell culture protocols for EB generation from PSCs that show significant in vivo differentiation toward dopaminergic neurons.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Chin MH, Mason MJ, Xie W, Volinia S, Singer M, Peterson C et al (2009) Induced pluripotent stem cells and embryonic stem cells are distinguished by gene expression signatures. Cell Stem Cell 5(1):111–123. https://pubmed.ncbi.nlm.nih.gov/19570518/. [cited 2021 Apr 12]
Giandomenico SL, Sutcliffe M, Lancaster MA (2020) Generation and long-term culture of advanced cerebral organoids for studying later stages of neural development. Nat Protoc 16(2) https://pubmed.ncbi.nlm.nih.gov/33328611/. [cited 2021 Mar 18]
Wu J, Izpisua Belmonte JC (2016) Stem cells: a renaissance in human biology research. In: Cell, vol 165. Cell Press, pp 1572–1585. https://doi.org/10.1016/j.cell.2016.05.043. [cited 2021 Apr 12]
Martin GR (1981) Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci U S A 78(12 II):7634–7638. https://pubmed.ncbi.nlm.nih.gov/6950406/. [cited 2021 Mar 30]
Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz AA, Swiergiel JJ, Marshall VS et al (1998) Embryonic stem cell lines derived from human blastocysts. Science 282:1145–1147
Landry DW, Zucker HA (2004) Embryonic death and the creation of human embryonic stem cells. J Clin Invest 114:1184–1186. https://pubmed.ncbi.nlm.nih.gov/15520846/. [cited 2021 Mar 30]
Schüle B, Pera RAR, Langston JW (2009) Can cellular models revolutionize drug discovery in Parkinson’s disease? Biochim Biophys Acta 1792:1043–1051. https://pubmed.ncbi.nlm.nih.gov/19733239/. [cited 2021 Apr 29]
Paik DT, Chandy M, Wu JC (2020) Patient and disease–specific induced pluripotent stem cells for discovery of personalized cardiovascular drugs and therapeutics. Pharmacol Rev 72(1):320–342
Arenas E, Denham M, Villaescusa JC (2015) How to make a midbrain dopaminergic neuron. Development 142(11):1918–1936. https://pubmed.ncbi.nlm.nih.gov/26015536/. [cited 2021 Apr 12]
Doss MX, Sachinidis A (2019) Current challenges of iPSC-based disease modeling and therapeutic implications. Cell 8(5):403
Maya-Espinosa G, Collazo-Navarrete O, Millán-Aldaco D, Palomero-Rivero M, Guerrero-Flores G, Drucker-Colín R et al (2015) Mouse embryonic stem cell-derived cells reveal niches that support neuronal differentiation in the adult rat brain. Stem Cells 33(2):491–502
Collazo-Navarrete O, Hernández-García D, Guerrero-Flores G, Drucker-Colín R, Guerra-Crespo M, Covarrubias L (2019) The substantia nigra is permissive and gains inductive signals when lesioned for dopaminergic differentiation of embryonic stem cells. Stem Cells Dev 28(16):1104–1115
Vierbuchen T, Ostermeier A, Pang ZP, Kokubu Y, Südhof TC, Wernig M (2010) Direct conversion of fibroblasts to functional neurons by defined factors. Nature 463(7284):1035–1041. https://www.nature.com/articles/nature08797. [cited 2021 Apr 22]
Zhang SC, Wernig M, Duncan ID, Brüstle O, Thomson JA (2001) In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nat Biotechnol 19(12):1129–1133. https://www.nature.com/articles/nbt1201-1129. [cited 2021 Apr 22]
Kawasaki H, Mizuseki K, Nishikawa S, Kaneko S, Kuwana Y, Nakanishi S et al (2000) Induction of midbrain dopaminergic neurons from ES cells by stromal cell-derived inducing activity. Neuron 28(1):31–40. https://pubmed.ncbi.nlm.nih.gov/11086981/. [cited 2021 Apr 22]
Watanabe K, Kamiya D, Nishiyama A, Katayama T, Nozaki S, Kawasaki H et al (2005) Directed differentiation of telencephalic precursors from embryonic stem cells. Nat Neurosci 8(3):288–296. https://www.nature.com/articles/nn1402. [cited 2021 Apr 28]
Lee SH, Lumelsky N, Studer L, Auerbach JM, McKay RD (2000) Efficient generation of midbrain and hindbrain neurons from mouse embryonic stem cells. Nat Biotechnol 18(6):675–679. https://www.nature.com/articles/nbt0600_675. [cited 2021 Apr 22]
Kim JH, Auerbach JM, Rodríguez-Gómez JA, Velasco I, Gavin D, Lumelsky N et al (2002) Dopamine neurons derived from embryonic stem cells function in an animal model of Parkinson’s disease. Nature 418(6893):50–56. https://pubmed.ncbi.nlm.nih.gov/12077607/. [cited 2021 Apr 26]
Friling S, Andersson E, Thompson LH, Jönsson ME, Hebsgaard JB, Nanou E et al (2009) Efficient production of mesencephalic dopamine neurons by Lmx1a expression in embryonic stem cells. Proc Natl Acad Sci U S A 106(18):7613–7618. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2671325&tool=pmcentrez&rendertype=abstract
Sánchez-Danés A, Consiglio A, Richaud Y, Rodríguez-Pizà I, Dehay B, Edel M et al (2012) Efficient generation of A9 midbrain dopaminergic neurons by lentiviral delivery of LMX1A in human embryonic stem cells and induced pluripotent stem cells. Hum Gene Ther 23(1):56–69. http://www.liebertonline.com/doi/abs/10.1089/hum.2011.054
Zhu F, Gamboa M, Farruggio AP, Hippenmeyer S, Tasic B, Schüle B et al (2014) DICE, an efficient system for iterative genomic editing in human pluripotent stem cells. Nucleic Acids Res 42(5):e34. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3950688/pdf/gkt1290.pdf
Jozefczuk J, Drews K, Adjaye J (2012) Preparation of mouse embryonic fibroblast cells suitable for culturing human embryonic and induced pluripotent stem cells. J Vis Exp 64:1–5. http://www.jove.com/video/3854/. [cited 2021 Mar 18]
Miyamoto D, Nakazawa K (2016) Differentiation of mouse iPS cells is dependent on embryoid body size in microwell chip culture. J Biosci Bioeng 122(4):507–512. https://pubmed.ncbi.nlm.nih.gov/27090344/. [cited 2021 Mar 18]
Antonchuk J (2013) Formation of embryoid bodies from human pluripotent stem cells using AggreWell™ plates. Methods Mol Biol 946:523–533. https://pubmed.ncbi.nlm.nih.gov/23179853/. [cited 2021 Apr 29]
Baizabal JM, Covarrubias L (2009) The embryonic midbrain directs neuronal specification of embryonic stem cells at early stages of differentiation. Dev Biol 325(1):49–59. https://doi.org/10.1016/j.ydbio.2008.09.024
Acknowledgments
This work was supported by IN211419 DGAPA-PAPIIT and CONACYT No. A1-S-10064. We are grateful to Brandt Bertrand for his help editing the manuscript and Marcela Palomero-Rivero and Francisco Pérez-Eugenio for their critical comments.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Guerra-Crespo, M., Collazo-Navarrete, O., Ramos-Acevedo, R., Morato-Torres, C.A., Schüle, B. (2021). Embryoid Body Formation from Mouse and Human Pluripotent Stem Cells for Transplantation to Study Brain Microenvironment and Cellular Differentiation. In: Turksen, K. (eds) Embryonic Stem Cell Protocols . Methods in Molecular Biology, vol 2520. Humana, New York, NY. https://doi.org/10.1007/7651_2021_433
Download citation
DOI: https://doi.org/10.1007/7651_2021_433
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2436-4
Online ISBN: 978-1-0716-2437-1
eBook Packages: Springer Protocols