Abstract
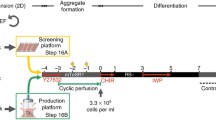
Human induced pluripotent stem cells (hiPSCs) can be expanded at limitless scale in vitro and give rise to various organotypic cells, cardiomyocytes (CMs) among them. Advanced protocols shape the differentiation process of pluripotent stem cells by controlled growth factor application. Modulating the Wnt signaling pathway is effective to direct hiPSCs to CMs (hiPSC-CMs) and native growth factors were replaced by small chemical compounds. Here, we describe a refined protocol for scalable generation of hiPSC-CMs that manipulates porcupine and tankyrase sub-pathways of Wnt signaling for tight inhibition of non-canonical Wnt signaling. The approach results in a differentiation efficiency toward hiPSC-CMs of 87 ± 0.9% in stirred bioreactor cultures and yields about 70 million hiPSC-CMs per 100 mL serum free cardiac differentiation medium. The differentiation protocol is easily adapted from 3D to 2D culture and vice versa and has been demonstrated to work with different hiPSC lines.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Hashmi S, Ahmad HR (2019) Molecular switch model for cardiomyocyte proliferation. Cell Regen 8:12–20. https://doi.org/10.1016/j.cr.2018.11.002
Richardson WJ, Clarke SA, Quinn TA, Holmes JW (2015) Physiological implications of myocardial scar structure. Compr Physiol 5:1877–1909. https://doi.org/10.1002/cphy.c140067
Takahashi K et al (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131:861–872. https://doi.org/10.1016/j.cell.2007.11.019
Brade T, Pane LS, Moretti A, Chien KR, Laugwitz K-L (2013) Embryonic heart progenitors and cardiogenesis. Cold Spring Harb Perspect Med 3:a013847–a013847. https://doi.org/10.1101/cshperspect.a013847
Sa S, McCloskey K, Activin A (2012) BMP4 signaling for efficient cardiac differentiation of H7 and H9 human embryonic stem cells. J Stem Cells Regen Med 8:198–202
Kattman SJ et al (2011) Stage-specific optimization of activin/nodal and BMP signaling promotes cardiac differentiation of mouse and human pluripotent stem cell lines. Cell Stem Cell 8:228–240. https://doi.org/10.1016/j.stem.2010.12.008
Lian X et al (2012) Robust cardiomyocyte differentiation from human pluripotent stem cells via temporal modulation of canonical Wnt signaling. Proc Natl Acad Sci U S A 109:E1848–E1857. https://doi.org/10.1073/pnas.1200250109
Lian X et al (2013) Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/beta-catenin signaling under fully defined conditions. Nat Protoc 8:162–175. https://doi.org/10.1038/nprot.2012.150
Burridge PW et al (2014) Chemically defined generation of human cardiomyocytes. Nat Methods 11:855–860. https://doi.org/10.1038/nmeth.2999
Burridge PW, Holmstrom A, Wu JC (2015) Chemically defined culture and cardiomyocyte differentiation of human pluripotent stem cells. Curr Protoc Hum Genet 87:21.3.1–21.3.15. https://doi.org/10.1002/0471142905.hg2103s87
Chen VC et al (2015) Development of a scalable suspension culture for cardiac differentiation from human pluripotent stem cells. Stem Cell Res 15:365–375. https://doi.org/10.1016/j.scr.2015.08.002
Kempf H, Kropp C, Olmer R, Martin U, Zweigerdt R (2015) Cardiac differentiation of human pluripotent stem cells in scalable suspension culture. Nat Protoc 10:1345–1361. https://doi.org/10.1038/nprot.2015.089
Hamad S et al (2019) Generation of human induced pluripotent stem cell-derived cardiomyocytes in 2D monolayer and scalable 3D suspension bioreactor cultures with reduced batch-to-batch variations. Theranostics 9:7222–7238. https://doi.org/10.7150/thno.32058
Ovchinnikov DA et al (2015) Isolation of contractile cardiomyocytes from human pluripotent stem-cell-derived cardiomyogenic cultures using a human NCX1-EGFP reporter. Stem Cells Dev 24:11–20. https://doi.org/10.1089/scd.2014.0195
Takagi H et al (2012) Nucleosome exclusion from the interspecies-conserved central AT-rich region of the Ars insulator. J Biochem 151:75–87. https://doi.org/10.1093/jb/mvr118
Tajima S et al (2006) Ars insulator identified in sea urchin possesses an activity to ensure the transgene expression in mouse cells. J Biochem 139:705–714. https://doi.org/10.1093/jb/mvj075
Li X et al (2005) piggyBac internal sequences are necessary for efficient transformation of target genomes. Insect Mol Biol 14:17–30. https://doi.org/10.1111/j.1365-2583.2004.00525.x
Yusa K, Zhou L, Li MA, Bradley A, Craig NL (2011) A hyperactive piggyBac transposase for mammalian applications. Proc Natl Acad Sci U S A 108:1531–1536. https://doi.org/10.1073/pnas.1008322108
Acknowledgments
This work was supported by “Marga und Walter Boll Stiftung” (Kerpen, Germany).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Hamad, S., Derichsweiler, D., Hescheler, J., Pfannkuche, K. (2021). Scalable Generation of Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes. In: Nagy, A., Turksen, K. (eds) Induced Pluripotent Stem (iPS) Cells. Methods in Molecular Biology, vol 2454. Humana, New York, NY. https://doi.org/10.1007/7651_2021_395
Download citation
DOI: https://doi.org/10.1007/7651_2021_395
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2118-9
Online ISBN: 978-1-0716-2119-6
eBook Packages: Springer Protocols