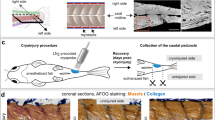
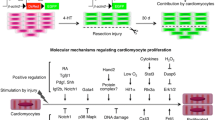
Abstract
Zebrafish extraocular muscles regenerate after severe injury. Injured myocytes dedifferentiate to a mesenchymal progenitor state and reenter the cell cycle to proliferate, migrate, and redifferentiate into functional muscles. A dedifferentiation process that begins with a multinucleated syncytial myofiber filled with sarcomeres and ends with proliferating mononucleated myoblasts must include significant remodeling of the protein machinery and organelle content of the cell. It turns out that autophagy plays a key role early in this process, to degrade the sarcomeres as well as the excess nuclei of the syncytial multinucleated myofibers. Because of the robustness of the zebrafish reprogramming process, and its relative synchrony, it can serve as a useful in vivo model for studying the biology of autophagy. In this chapter, we describe the surgical muscle injury model as well as the experimental protocols for assessing and manipulating autophagy activation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Becker T, Wullimann MF, Becker CG, Bernhardt RR, Schachner M (1997) Axonal regrowth after spinal cord transection in adult zebrafish. J Comp Neurol 377(4):577–595
Poss KD, Wilson LG, Keating MT (2002) Heart regeneration in zebrafish. Science 298(5601):2188–2190. https://doi.org/10.1126/science.1077857
Raya A, Koth CM, Buscher D, Kawakami Y, Itoh T, Raya RM, Sternik G, Tsai HJ, Rodriguez-Esteban C, Izpisua-Belmonte JC (2003) Activation of Notch signaling pathway precedes heart regeneration in zebrafish. Proc Natl Acad Sci U S A 100(Suppl 1):11889–11895. https://doi.org/10.1073/pnas.1834204100
Hitchcock PF, Raymond PA (2004) The teleost retina as a model for developmental and regeneration biology. Zebrafish 1(3):257–271. https://doi.org/10.1089/zeb.2004.1.257
Goldman D (2014) Muller glial cell reprogramming and retina regeneration. Nat Rev Neurosci 15(7):431–442. https://doi.org/10.1038/nrn3723
Poss KD, Shen J, Nechiporuk A, McMahon G, Thisse B, Thisse C, Keating MT (2000) Roles for Fgf signaling during zebrafish fin regeneration. Dev Biol 222(2):347–358. https://doi.org/10.1006/dbio.2000.9722
Pfefferli C, Jazwinska A (2015) The art of fin regeneration in zebrafish. Regeneration (Oxf) 2(2):72–83. https://doi.org/10.1002/reg2.33
Kan NG, Junghans D, Izpisua Belmonte JC (2009) Compensatory growth mechanisms regulated by BMP and FGF signaling mediate liver regeneration in zebrafish after partial hepatectomy. FASEB J 23(10):3516–3525. https://doi.org/10.1096/fj.09-131730
Berberoglu MA, Gallagher TL, Morrow ZT, Talbot JC, Hromowyk KJ, Tenente IM, Langenau DM, Amacher SL (2017) Satellite-like cells contribute to pax7-dependent skeletal muscle repair in adult zebrafish. Dev Biol 424(2):162–180. https://doi.org/10.1016/j.ydbio.2017.03.004
Saera-Vila A, Kasprick DS, Junttila TL, Grzegorski SJ, Louie KW, Chiari EF, Kish PE, Kahana A (2015) Myocyte dedifferentiation drives extraocular muscle regeneration in adult zebrafish. Invest Ophthalmol Vis Sci 56(8):4977–4993. https://doi.org/10.1167/iovs.14-16103
Louie KW, Saera-Vila A, Kish PE, Colacino JA, Kahana A (2017) Temporally distinct transcriptional regulation of myocyte dedifferentiation and Myofiber growth during muscle regeneration. BMC Genomics 18(1):854. https://doi.org/10.1186/s12864-017-4236-y
Saera-Vila A, Kish PE, Kahana A (2016) Fgf regulates dedifferentiation during skeletal muscle regeneration in adult zebrafish. Cell Signal 28(9):1196–1204. https://doi.org/10.1016/j.cellsig.2016.06.001
Boya P, Reggiori F, Codogno P (2013) Emerging regulation and functions of autophagy. Nat Cell Biol 15(7):713–720. https://doi.org/10.1038/ncb2788
Mizushima N, Levine B (2010) Autophagy in mammalian development and differentiation. Nat Cell Biol 12(9):823–830. https://doi.org/10.1038/ncb0910-823
Wong E, Cuervo AM (2010) Autophagy gone awry in neurodegenerative diseases. Nat Neurosci 13(7):805–811. https://doi.org/10.1038/nn.2575
Eng CH, Abraham RT (2011) The autophagy conundrum in cancer: influence of tumorigenic metabolic reprogramming. Oncogene 30(47):4687–4696. https://doi.org/10.1038/onc.2011.220
Pan H, Cai N, Li M, Liu GH, Izpisua Belmonte JC (2013) Autophagic control of cell ‘stemness’. EMBO Mol Med 5(3):327–331. https://doi.org/10.1002/emmm.201201999
Wang S, Xia P, Rehm M, Fan Z (2015) Autophagy and cell reprogramming. Cell Mol Life Sci 72(9):1699–1713. https://doi.org/10.1007/s00018-014-1829-3
Saera-Vila A, Kish PE, Louie KW, Grzegorski SJ, Klionsky DJ, Kahana A (2016) Autophagy regulates cytoplasmic remodeling during cell reprogramming in a zebrafish model of muscle regeneration. Autophagy 12(10):1864–1875. https://doi.org/10.1080/15548627.2016.1207015
Saera-Vila A, Louie KW, Sha C, Kelly RM, Kish PE, Kahana A (2018) Extraocular muscle regeneration in zebrafish requires late signals from insulin-like growth factors. PLoS One 13(2):e0192214. https://doi.org/10.1371/journal.pone.0192214
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (2002) Short protocols in molecular biology, 5th edn. Wiley, New York
Hu Z, Zhang J, Zhang Q (2011) Expression pattern and functions of autophagy-related gene atg5 in zebrafish organogenesis. Autophagy 7(12):1514–1527
Higashijima S, Okamoto H, Ueno N, Hotta Y, Eguchi G (1997) High-frequency generation of transgenic zebrafish which reliably express GFP in whole muscles or the whole body by using promoters of zebrafish origin. Dev Biol 192(2):289–299
He C, Bartholomew CR, Zhou W, Klionsky DJ (2009) Assaying autophagic activity in transgenic GFP-Lc3 and GFP-Gabarap zebrafish embryos. Autophagy 5(4):520–526
Nüsslein-Volhard C, Dahm R (2002) Zebrafish: a practical approach, vol 975. Oxford University Press, Oxford
Westerfield M (2007) The zebrafish book. A guide for the laboratory use of zebrafish (Danio rerio), 5th edn. University of Oregon Press, Eugene
Fodor E, Sigmond T, Ari E, Lengyel K, Takacs-Vellai K, Varga M, Vellai T (2017) Methods to study autophagy in zebrafish. Methods Enzymol 588:467–496. https://doi.org/10.1016/bs.mie.2016.10.028
Mathai BJ, Meijer AH, Simonsen A (2017) Studying autophagy in zebrafish. Cell 6(3). https://doi.org/10.3390/cells6030021
Acknowledgments
This work was funded by R01 EY022633 from the National Eye Institute (A.K.), the Alfred Taubman Medical Research Institute (A.K.), the Alliance for Vision Research (A.K.), and an unrestricted departmental grant from Research to Prevent Blindness, Inc.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media New York
About this protocol
Cite this protocol
Saera-Vila, A., Kish, P.E., Kahana, A. (2018). Autophagy in Zebrafish Extraocular Muscle Regeneration. In: Turksen, K. (eds) Autophagy in Differentiation and Tissue Maintenance. Methods in Molecular Biology, vol 1854. Humana Press, New York, NY. https://doi.org/10.1007/7651_2018_160
Download citation
DOI: https://doi.org/10.1007/7651_2018_160
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8747-4
Online ISBN: 978-1-4939-8748-1
eBook Packages: Springer Protocols