Abstract
Background
Natural products are one of the best candidates for controlling drug-resistant pathogens, the advantages of which include low production costs and low side effects. In this study, as potential antimicrobials, the anti-bacterial and antibiofilm activities of several Iranian native medicinal plants were screened.
Methods
The antibacterial/antifungal and anti-biofilm activities of 18 medicinal plants including Reseda lutea L., Nepeta sintenisii Bunge., Stachys turcomanica Trautv., Stachys lavandulifolia Vahl, Diarthron antoninae (Pobed.) Kit Tan., Ziziphora clinopodioides Lam., Euphorbia kopetdaghi Prokh, Euphorbia serpens Kunth., Hymenocrater calycinus Benth., Scutellaria pinnatifida A.Ham., Viola tricolor L., Hypericum helianthemoides (Spach) Boiss., Hypericum scabrum L., Convolvulus lineatus L., Scabiosa rotata M.Bieb Greuter & Burdet, Delphinium semibarbatum Bien. Ex Boiss., Glycyrrhiza triphylla Fisch. & C.A.Mey., and Ziziphus jujuba Mill., against two Gram-positive bacteria, Staphylococcus aureus, Bacillus cereus, as well as two Gram-negative bacteria, Pseudomonas aeruginosa, Escherichia coli; and Candida albicans as a fungal strain, were evaluated. The minimum inhibitory concentration (MIC) and minimum bactericidal/fungicidal concentration (MBC/MFC) values of the extracts against tested microorganisms were reported and we investigated their effect on the biofilm inhibition of Pseudomonas aeruginosa PAO1, Staphylococcus epidermis, Staphylococcus aureus and Streptococcus mutans. In addition, the effect of the extracts on the eradication of the biofilms of these bacteria was evaluated.
Results
In this study, H. scabrum was found to exhibit potentially significant activity against Gram-positive bacteria with the MIC range of 6.25–25 µg/mL. This extract also showed a significant effect on inhibiting the biofilm of S. aureus, S. mutans, and S. epidermidis and eradicating the biofilm of S. epidermidis DSMZ 3270. In addition, Hymenocrater calycinus root extract had moderate antibacterial activity against B. cereus with the MIC and MBC 62.5 µg/mL, respectively.
Conclusions
The results of this study showed that the root extracts of two plants, Hypericum scabrum and Hymenocrater calycinus, had antimicrobial and anti-biofilm effects. Based on the observed anti-biofilm effects, these two plants may be considered in future studies to find responsible antimicrobial compounds.
Similar content being viewed by others
Introduction
Drug-resistant bacteria are currently a major worldwide health concern, as they are not responding well to conventional treatments [1]. By finding novel antibiotics and chemically altering existing antimicrobial medications, it has been sought to overcome bacterial resistance to antimicrobial drugs [2]. It has been estimated that biofilms are responsible for approximately 75% of bacterial infections [3]. Depending on how genetic products function and what type of drug molecule, there are several kinds of drug resistance. Bacteria develop secondary resistance to substances when they are exposed to them repeatedly and several factors can contribute to this development [4].
Biofilms are a major concern for the food and medical industries as they have been linked to numerous hospital infections and food spoiling problems [5]. Biofilm cells are capable of irreversibly adhering to surfaces, living tissues, and indwelling medical devices such as catheters, valves, prostheses, etc. [3]. Bacterial cells embedded in a biofilm exhibit increased resistance to antimicrobial agents and other environmental stresses compared to free-floating cells. Finding new agents to combat biofilm formation and providing a substitute for routine medication would be of great importance [6]. The use of plant-based medicines is an effective alternative that may help minimize the negative effects of conventional antibiotic therapy. Additionally, plant-based formulations would enhance the amount of anti-infection chemicals available and may function in combination with existing treatments to reduce infections [7]. Medicinal plants can also be used as antibacterial and antifungal agents. Several studies have reported the anti-microbial and anti-biofilm activity of various medicinal plants [8], which highlights the need for screening herbal extracts and natural compounds to discover novel antimicrobial agents. Iran, as a vast country with diverse climatic features in Asia, has approximately 8167 identified plant species, among which are almost 1900 endemic [9, 10].
In the course of our screening study to discover new antimicrobial compounds from Iranian medicinal plants [11], we have screened 18 plants which are based on their traditional use. R. lutea, Z. clinopodioides., E. serpens, H. calycinus, and C. lineatus are used as antimicrobials by local people. Additionally, the antimicrobial activity of some other plant species in this study has demonstrated, N. sintenisii [12], S. lavandulifolia [13], S. pinnatifida [14], V. tricolor L. [15], H. helianthemoides [16], H. scabrum [17], G. triphylla [18], and Z. jujuba [19].
It was reported that using a binary solvent mixture with one polar and a non-polar component (MeOH- CH2Cl2, 1:1) could extract diverse bioactive compounds from plant materials [20, 21]. Therefore, in our study, the preparation of the extracts was performed using this combination.
Materials and methods
Tryptic soy agar (TSA) and Sabouraud dextrose broth (SDB) were supplied from HiMedia, India. Glucose and crystal violet, dimethyl sulfoxide (DMSO), and 2,3,5-triphenyl tetrazolium chloride (TTC) were provided from Merck, Germany. Mueller Hinton broth (MHB) was purchased from Liofilchem, Italy, and cloxacillin was obtained from Farabi, Iran. All solvents used were of analytical grade and provided by Dr. Mojallali Industrial Chemical Complex Co, Iran.
Extract preparation
All the plant species were collected from various locations and identified by Ms. Souzani and have been deposited at the Herbarium of the School of Pharmacy, Mashhad University of Medical Sciences, Iran. The dried plant parts were powdered and extracted with dichloromethane/methanol (1:1) using the maceration method at 20–22 °C for 24 h (3 times). The obtained extracts were then filtered and dried under vacuum by rotary evaporator. The extracts were kept at -20 °C until being used.
Bacterial strains
To screen the antimicrobial activity of the extracts, five microorganisms were used as representative bacteria and fungi including Staphylococcus aureus (ATCC 25293) and Bacillus cereus (PTCC 1023), as Gram-positive bacteria, as well as Pseudomonas aeruginosa (PTCC 1074), and Escherichia coli (PTCC 1330) as Gram-negative bacteria; and Candida albicans (PTCC 5027) as fungal strain. All microorganisms were purchased from the Persian Type Culture Collection (PTCC). Biofilm-producer strains of bacteria including P. aeruginosa PAO1 (Nottingham wild type), Streptococcus mutans (ATCC 35668), and Staphylococcus epidermidis (DSMZ 3270) were also used in further assessments. While the bacterial strains were cultured overnight at 37 °C in Tryptic soy agar (TSA), the fungi were cultured for 3 days at 25 °C in sabouraud dextrose agar (SDA), according to the United States Pharmacopeia (USP).
Minimum inhibitory concentration (MIC) and minimum bactericidal/fungicidal concentration (MBC/MFC)
The procedure was performed in 96-well plates according to the microdilution method of CLSI standard. The extracts were dissolved in DMSO at a concentration of 25 mg/200–500 μL and subsequently, the volume reached 5 mL. This reference concentration (5 mg/mL) was used to prepare the first concentration of the assessment (1 mg/mL). The other concentrations (500–0.97 µg/mL) were prepared by the serially twofold dilution method. In the next step, 180 μL of various concentrations of the extracts were inoculated into the wells. Subsequently, 20 μL of bacterial/fungal suspension (106 \(\frac{CFU}{ml}\)) was added to each well, separately. To evaluate the accuracy of the test, three wells were filled with MHB containing 2.5% DMSO as a negative control (sterility control), and three wells were specified for each microorganism to check its ability to grow in the culture medium (positive control). The plates were incubated at 37 °C for 16 h for bacterial strains and 48 h at 25 °C for the fungus. Finally, 20 μL TTC (5 mg/mL) was added to each well as a coloring indicator and incubated for another 1 h for bacteria and 24 h for the fungus [3]. Each test was done in triple replication. The MIC was considered as the lowest concentration of an extract that inhibited visible growth (absence of changing in color to red).
The MBC/MFC was determined according to the method explained by Sánchez et al. [3] with slight modifications. Briefly, 10 µl from the wells with no color changing was inoculated on the surface of TSA and incubated at 35 °C for 24 h and 25 °C for 72 h for bacteria and fungi, respectively. After incubation, the formation of colonies was investigated. The MBC was considered the lowest concentration with no microbial growth.
Assessment of anti-biofilm activity
The anti-biofilm activities of the extracts were investigated in two steps, before biofilm formation (inhibiting biofilm formation) and after biofilm formation (eradication of biofilm structures). The extract concentration was applied at half of their MICs to avoid growth inhibition. Briefly, 180 µL of glucose-enriched MHB culture was mixed with the extracts to reach \(\frac{1}{2}\) MIC. Subsequently, 20 µL of the cell suspension, containing 1 × 106 CFU/mL of Pseudomonas aeruginosa PAO1, Staphylococcus epidermis, Staphylococcus aureus and Streptococcus mutans were added to each well, separately. In the control groups, four wells were dedicated to the formation of biofilms as the positive control and the negative control is the sterile culture medium to ensure the sterility of the culture medium during the experiment. The plates were incubated at 37°C for 48 h for mature biofilm formation. However, the culture media was refreshed every 12 h. Biofilms constructed at 96-well bottom-flat plates were washed three times with sterile 0.9% NaCl solution to discard non-adhered bacteria and allowed to dry. The total biomass was measured by crystal violet staining (0.03% w/v). Sterile normal saline was used to remove extra crystal violet, and ethanol 95% (200 μl) was employed to extract the bound crystal violet [22]. The experiments were done in triplicate for each extract. Medium with and without microorganisms in the absence of extracts were used as positive and negative controls, respectively. To evaluate the eradication of biofilm, first, 180 µl MHB enriched with glucose was added to the wells. Then, 20 µl of 1 × 106 CFU/mL from microbial suspension of Staphylococcus epidermis was added to each well. The plates were incubated for 48 h at 37°C. To investigate the effect of the extract on the eradication of immature and mature biofilm, it was operated in two ways. In the first method, after 24 h, the previous culture medium was replaced with a new nutrient culture medium. Then, after 12 h, the medium was replaced with MHB containing the extract with a concentration equal to \(\frac{1}{2}\) MIC. In the second method, after 24 h, we replaced the previous culture medium with MHB containing an extract with a concentration of \(\frac{1}{2}\) MIC. This process was repeated 12 h later. The biofilm staining process was done as well as previously. The optical density of each well stained with crystal violet is measured at 590 nm as a biofilm formation indicator [23].
Statistical analysis
Statistical analysis was done using SPSS software version 22. One-way analysis of variance (ANOVA) was used to compare the results. A comparison of the means was performed using a Duncan test at a 95% confidence level (P < 0.05).
Results
Antimicrobial activities
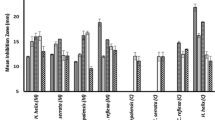
In this study, the antibacterial, antifungal, and anti-biofilm activity of 18 medicinal plants from 11 various families were assayed. Table 1 presents the MIC and MBC/MFC values of the extracts against tested microorganisms. In our experiment, 16 extracts with MIC > 1000 µg/mL could not show their antimicrobial activities. The root extract of Hymenocrater calycinus was not effective against Gram-negative bacteria at the tested concentrations but inhibited the growth of C. albicans and, at lower concentrations, Gram-positive bacteria. The extracts from roots and aerial parts of H. scabrum possess higher antimicrobial effects against Gram-positive bacteria, especially S. aureus, and with lower concentrations, it inhibited C. albicans. Extracts prepared from the root of H. scabrum demonstrated strong growth inhibitory and bactericidal effects against S. aureus, and S. mutans with MIC and MBC of 6.25 and 12.5 µg/mL, respectively. In addition, H. calycinus root extract showed potent antibacterial activities against B. cereus with both MIC and MBC of 62.5 µg/mL. The extracts from other plant species did not show potent antibacterial and antifungal properties. Therefore, only the roots of H. scabrum and H. calycinus were chosen for further anti-biofilm investigations.
Biofilm formation and eradication
The ability of H. scabrum and H. calycinus against biofilm formation was assessed against biofilm producer strains, P. aeruginosa PAO1, S. epidermidis, S. aureus, and S. mutans. by calculating the optical density ratio (ODr). The results indicated that: (I) both extracts did not show anti-biofilm activity against P. aeruginosa in the level of biofilm formation. However, they were able to significantly inhibit the biofilm formation of Gram-positive bacteria (S. epidermidis (P < 0.05), S. aureus (P < 0.05), and S. mutans (P < 0.05), compared to the positive control (Table 2 and Fig. 1). (II) H. scabrum showed a significant biofilm eradication (P < 0.05) effect against immature and mature-S. epidermidis biofilm (Table 3 and Fig. 2).
Discussion
The present study is an effort to expand our knowledge of the antimicrobial and anti-biofilm effects of some Iranian medicinal plants against bacterial and fungal pathogens. The nature of the organic solvent used for extraction plays an important role in isolating the optimum amounts of various secondary metabolites from the tested plants. In our study, the dichloromethane/methanol (1:1) mixture was speculated to be the best solvent mixture with the ability to extract responsible bioactive metabolites with various polarities. In this study, significant antimicrobial activity against Gram-positive bacteria especially S. aureus, S. mutans, and S. epidermis was observed in the various parts of H. scabrum. Many Hypericum species, most notably H. perforatum, and their extracts have been shown to possess a range of therapeutic qualities, including anti-inflammatory, antiviral, antimicrobial, antioxidant, antitumoral, and wound-healing effects [24]. The antimicrobial activity of H. scabrum previously was reported by several authors [25,26,27]. For example, Ergin et al. reported that MeOH, n-hexane, and dichloromethane extracts from H. scabrum root were effective against S. aureus (MIC = 312.5–625 µg/mL) [28]. Also, our present observations are in agreement with another previous study by Keser et al., [29] who reported the potent antimicrobial activity of H. scabrum against E. coli, Proteus vulgaris, P. aeruginosa, Listeria monocytogenes, Klebsielle pneumonia and Bacillus subtilis. It was reported that H. scabrum contains a variety of secondary metabolites such as flavonoids [30], xanthones [31], and especially phloroglucinols [32, 33]. It was found that lipophilic fractions of Hypericum species contain phloroglucinol derivatives, which have been demonstrated to have antifungal and antibacterial properties against pathogens like S. aureus, B. cereus, B. subtilis, and Nocardia gardenen [34]. Such compounds could be the cause of the antimicrobial activity, even at relatively low quantities [34]. Furthermore, phloroglucinol and its derivatives are frequently used to treat microbial diseases caused by bacteria, fungi, and viruses [35]. Phytochemical analysis of ethyl acetate extract of the aerial part of H. scabrum led to the isolation of eight flavonoids. They have shown moderate to no antimicrobial activity against S. aureus, E. coli, and C. albicans [36]. Thus, phloroglucinols might be responsible for the potent antimicrobial activity of H. scabrum. In our study, also, H. calycinus root extract showed good antibacterial activities against B. cereus (MIC = 62.5µg/ml). The primary component of H. calycinus extract, rosmarinic acid, demonstrated antifungal properties against C. albicans [37]. For rosmarinic acid, a wide range of biological actions have been noted. Astringent, anti-oxidative, anti-inflammatory, anti-mutagen, antibacterial, and anti-viral are the major effects [37]. Biofilms are the primary etiological factors in 80% of Human microbial infections. Moreover, biofilms constructed on surgical implants and tissue surfaces are an important threat to human health. Biofilm infections are extremely difficult to treat with common antibiotics, resulting in chronic infections and non-healing wounds. As a result, it has been necessary to develop new antibacterial agents that are effective against biofilm [38]. For instance, New biofilm-inhibiting substances are urgently needed to combat refractory infections like methicillin-resistant S. aureus (MRSA) and vancomycin-methicillin-resistant S. aureus because biofilms play crucial roles in antibiotic resistance [39]. In our study, the extract of H. scabrum demonstrated significant biofilm inhibition against S. epidermidis and S. aureus and the extract could eradicate S. epidermidis at 60% after 24h. Interestingly, the extracts outperformed common antibiotics (streptomycin sulfate and nystatin) concerning their antimicrobial effects. Pereira et all., found that Hypericum brasiliense extract inhibited and eradicated S. aureus HU25 planktonic cells and their biofilm formation [40]. The anti-biofilm activity of H. scabrum could be related to the higher proportion of polyphenols and flavonoids including catechin, quercetin and vanillic acid in the plant, which have been found to possess anti-biofilm activities [41, 42]. The mechanism underlying the anti-biofilm properties of the plant might involve decreasing polysaccharide intercellular adhesion production and altering the composition of exopolysaccharides in S. epidermidis [42]. Taken together and based on our current findings, it is possible to employ H. scabrum as a source of effective antimicrobial substances to combat infections brought on by vulnerable organisms.
Conclusions
Our findings established that among 18 Iranian medicinal plants, both H. scabrum and H. calycinus root extracts had well-potent antimicrobial activities against some tested pathogens. The root of H. scabrum extract could inhibit biofilm formation and eradicate the pre-formed biofilm of S. epidermidis. However, the clinical application of such anti-microbial compounds needs further both in vitro and in vivo investigations.
Availability of data and materials
The data supported during the present research are available from the corresponding author upon reasonable request.
Abbreviations
- MIC:
-
Minimum Inhibitory Concentration
- MBC/MFC:
-
Minimum Bactericidal/Fungicidal Concentration
- TSA:
-
Tryptic Soy Agar
- SDB:
-
Sabouraud Dextrose Broth
- DMSO:
-
Dimethyl Sulfoxide
- TTC:
-
2,3,5-Triphenyl Tetrazolium Chloride
- MHB:
-
Mueller Hinton Broth
- PTCC:
-
Persian Type Culture Collection
- ATCC:
-
American Type Culture Collection
- ODr:
-
Optical Density Ratio
- MRSA:
-
Methicillin-resistant S. aureus
References
Saeloh D, Visutthi M. Efficacy of Thai plant extracts for antibacterial and anti-biofilm activities against pathogenic bacteria. Antibiotics. 2021;10(12):1470–8.
Nourbakhsh F, Lotfalizadeh M, Badpeyma M, Shakeri A, Soheili V. From plants to antimicrobials: Natural products against bacterial membranes. Phytother Res. 2022;36(1):33–52. https://doi.org/10.1002/ptr.7275.
Sánchez E, Morales CR, Castillo S, Leos-Rivas C, García-Becerra L, Martínez DMO. Antibacterial and antibiofilm activity of methanolic plant extracts against nosocomial microorganisms. Evid Based Complement Alternat Med. 2016;2016:1–8.
Lahiri D, Dash S, Dutta R, Nag M. Elucidating the effect of anti-biofilm activity of bioactive compounds extracted from plants. J Biosci. 2019;44(2):52–70. https://doi.org/10.1007/s12038-019-9868-4.
Al shahwany A, Tawfeeq H, Hamed S, Imad S. Antibacterial and anti-biofilm activity of three phenolic plant extracts and silver nanoparticles on Staphylococcus aureus and Klebsiella pneumoniae. J biotechnol biomed. 2016;4(4):12–8.
Arora DS, Mahajan H. Major phytoconstituents of Prunus cerasoides responsible for antimicrobial and antibiofilm potential against some reference strains of pathogenic bacteria and clinical isolates of MRSA. Appl Biochem Biotechnol. 2019;188(4):1185–204.
Luciano-Montalvo C, Boulogne I, Gavillán-Suárez J. A screening for antimicrobial activities of Caribbean herbal remedies. BMC Complement Altern Med. 2013;13(1):126–34. https://doi.org/10.1186/1472-6882-13-126.
Teanpaisan R, Kawsud P, Pahumunto N, Puripattanavong J. Screening for antibacterial and antibiofilm activity in Thai medicinal plant extracts against oral microorganisms. J Tradit Complement Med. 2017;7(2):172–7.
Nadaf M, Amiri MS, Joharchi MR, Omidipour R, Moazezi M, Mohaddesi B, et al. Ethnobotanical diversity of trees and shrubs of Iran: A comprehensive review. Int J Plant Biol. 2023. https://doi.org/10.3390/ijpb14010011.
Kiani M, Mohammadi S, Babaei A, Sefidkon F, Naghavi MR, Ranjbar M, et al. Iran supports a great share of biodiversity and floristic endemism for Fritillaria spp. (Liliaceae): A review. Plant Divers. 2017;39(5):245–62. https://doi.org/10.1016/j.pld.2017.09.002.
Shakeri A, Sharifi JM, Fazly Bazzaz SB, Emami A, Soheili V, Sahebkar A, et al. Bioautography detection of antimicrobial compounds from the essential oil of Salvia Pachystachys. Curr Bioact Compd. 2018;14(1):80–5. https://doi.org/10.2174/1573407212666161014132503.
Shakeri A, Khakdan F, Soheili V, Sahebkar A, Shaddel R, Asili J. Volatile composition, antimicrobial, cytotoxic and antioxidant evaluation of the essential oil from Nepeta sintenisii Bornm. Ind Crops Prod. 2016;84:224–9. https://doi.org/10.1016/j.indcrop.2015.12.030.
Işcan G, Demirci B, Demirci F, Göger F, Kirimer N, Köse YB, et al. Antimicrobial and antioxidant activities of Stachys lavandulifolia subsp. lavandulifolia essential oil and its infusion. Nat Product Commun. 2012;7(9):1241–4.
Delazar A, Nazemiyeh H, Afshar FH, Barghi N, Esnaashari S, Asgharian P. Chemical compositions and biological activities of Scutellaria pinnatifida A. Hamilt aerial parts. Res Pharm Sci. 2017;12(3):187–95. https://doi.org/10.4103/1735-5362.207199.
Witkowska-Banaszczak E, Bylka W, Matławska I, Goślińska O, Muszyński Z. Antimicrobial activity of Viola tricolor herb. Fitoterapia. 2005;76(5):458–61. https://doi.org/10.1016/j.fitote.2005.03.005.
Ghasemi Pirbalouti A, Fatahi-Vanani M, Craker L, Shirmardi H. Chemical composition and bioactivity of essential oils of Hypericum helianthemoides, Hypericum perforatum and Hypericum scabrum. Pharm Biol. 2014;52(2):175–81. https://doi.org/10.3109/13880209.2013.821663.
Esmaeilzadehkashi M, Soheili V, Asili J, Davoodi J, Soleimanpour S, Karimi G, et al. Screening of some Iranian medicinal plants for anti-tuberculosis, anti-bacterial, and cytotoxic activities. S Afr J Bot. 2023;154:260–4. https://doi.org/10.1016/j.sajb.2023.01.036.
Shakeri A, Akhtari J, Soheili V, Taghizadeh SF, Sahebkar A, Shaddel R, et al. Identification and biological activity of the volatile compounds of Glycyrrhiza triphylla Fisch. & C.AMey. Microb Pathog. 2017;109:39–44. https://doi.org/10.1016/j.micpath.2017.05.022.
Wang Z, Yang Q, Zhang H, He Y, Wang R, Lu X. Isolation, identification, and antibacterial activities of flavonoids from Jujube (Zizipus Jujuba Mill.) Fruit. Int J Fruit Sci. 2023;23(1):51–61. https://doi.org/10.1080/15538362.2023.2186149.
Cequier-Sánchez E, Rodríguez C, Ravelo ÁG, Zárate R. Dichloromethane as a solvent for lipid extraction and assessment of lipid classes and fatty acids from samples of different natures. J Agric Food Chem. 2008;56(12):4297–303. https://doi.org/10.1021/jf073471e.
Mbaoji FN, Onwuka AM, Onu S, Peter IE, Nweze JA, Okonta LE. Evaluation of methanol-dichloromethane extract of Stemonocoleus micranthus Harms (Fabaceae) stem bark for anti-inflammatory and immunomodulatory activities. Evid Based Complementary Altern Med. 2020;2020:1738163–72. https://doi.org/10.1155/2020/1738163.
Muruzović MŽ, Mladenović KG, Stefanović OD, Vasić SM, Čomić LR. Extracts of Agrimonia eupatoria L. as sources of biologically active compounds and evaluation of their antioxidant, antimicrobial, and antibiofilm activities. J Food Drug Anal. 2016;24(3):539–47.
Saxena S, Banerjee G, Garg R, Singh M. Comparative study of biofilm formation in Pseudomonas aeruginosa isolates from patients of lower respiratory tract infection. J Clin Diagnostic Res. 2014;8(5):DC09-DC11.
Aygül A, Şerbetçi T. The antibacterial and antivirulent potential of Hypericum lydium against Staphylococcus aureus: Inhibition of growth, biofilm formation, and hemolytic activity. Eur J Integr Med. 2020;35:101061–7. https://doi.org/10.1016/j.eujim.2020.101061.
Dastan SD. Chemical and functional composition and biological activities of Anatolian Hypericum scabrum L. J Mol Struct. 2023;1275: 134561. https://doi.org/10.1016/j.molstruc.2022.134561.
GhasemiPirbalouti A, Rahnama GH, Malekpoor F, Broujeni H. Variation in antibacterial activity and phenolic content of Hypericum scabrum L. populations. J Med Plant Res. 2011;5:4119–25.
Shafaghat A. Antioxidant, antimicrobial activities and fatty acid components of flower, leaf, stem and seed of Hypericum scabrum. Nat Prod Commun. 2011;6(11):1739–42. https://doi.org/10.1177/1934578X1100601142.
Ergin KN, Karakaya S, Göger G, Sytar O, Demirci B, Duman H. Anatomical and phytochemical characteristics of different parts of Hypericum scabrum L. extracts, essential oils, and their antimicrobial potential. Molecules. 2022;27(4):1228–47.
Keser S, Keser F, Kaygili O, Tekin S, Demir E, Turkoglu I, et al. Phytochemical compounds and antiradical, antimicrobial, and cytotoxic activities of the extracts from Hypericum scabrum L. Flowers Nat Prod Res. 2020;34(5):714–9. https://doi.org/10.1080/14786419.2018.1493735.
Ayan AK, Radušienė J, Çirak C, Ivanauskas L, Janulis V. Secondary metabolites of Hypericum scabrum and Hypericum bupleuroides. Pharm Biol. 2009;47(9):847–53. https://doi.org/10.1080/13880200902942436.
Teng LP, Zeng H, Yang CY, Wang HB, Zhou ZB. Three new xanthones from Hypericum scabrum and their quorum sensing inhibitory activities against Chromobacterium violaceum. Molecules. 2022;27(17):5519–29. https://doi.org/10.3390/molecules27175519.
Yang JB, Liu RD, Ren J, Wei Q, Wang AG, Su YL. Two new prenylated phloroglucinol derivatives from Hypericum scabrum. J Asian Nat Prod Res. 2016;18(5):436–42. https://doi.org/10.1080/10286020.2015.1123693.
Zhou ZB, Mou PY, Huang YY, Zeng H, Huang ZL, Wei X. Bioactive polycyclic polyprenylated acylphloroglucinols from Hypericum scabrum. Fitoterapia. 2022;161:105249. https://doi.org/10.1016/j.fitote.2022.105249.
Dall’Agnol R, Ferraz A, Bernardi AP, Albring D, Nör C, Sarmento L, et al. Antimicrobial activity of some Hypericum species. Phytomedicine. 2003;10(6–7):511–6. https://doi.org/10.1078/094471103322331476.
Khan F, Tabassum N, Bamunuarachchi NI, Kim YM. Phloroglucinol and its derivatives: Antimicrobial properties toward microbial pathogens. J Agric Food Chem. 2022;70(16):4817–38. https://doi.org/10.1021/acs.jafc.2c00532.
Jiang L, Numonov S, Bobakulov K, Qureshi MN, Zhao H, Aisa HA. Phytochemical profiling and evaluation of pharmacological activities of Hypericum scabrum L. Molecules. 2015. https://doi.org/10.3390/molecules200611257.
Gohari AR, Saeidnia S, Shahverdi AR, Yassa N, Malmir M, Mollazade K, et al. Phytochemistry and antimicrobial compounds of Hymenocrater calycinus. Eur Asia J Bio Sci. 2009;3(9):64–8.
Kamer AMA, Abdelaziz AA, Al-Monofy KB, Al-Madboly LA. Antibacterial, antibiofilm, and anti-quorum sensing activities of pyocyanin against methicillin-resistant Staphylococcus aureus: in vitro and in vivo study. BMC Microbiol. 2023;23(1):116–34. https://doi.org/10.1186/s12866-023-02861-6.
Lee J-H, Park J-H, Cho HS, Joo SW, Cho MH, Lee J. Anti-biofilm activities of quercetin and tannic acid against Staphylococcus aureus. Biofouling. 2013;29(5):491–9. https://doi.org/10.1080/08927014.2013.788692.
Pereira RFA, Andre LSP, Pinheiro FR, Scaffo J, Corrêa AL, Ribeiro TAN, et al. Hypericum brasiliense: bactericidal and antibiofilm activity against Staphylococcus aureus in vitro. Nat Prod Res. 2022;36(10):2643–7. https://doi.org/10.1080/14786419.2021.1915306.
Gonçalves I, Matamá T, Cavaco-Paulo A, Silva C. Laccase coating of catheters with poly (catechin) for biofilm reduction. Biocatal. 2014;32(1):2–12.
Mu Y, Zeng H, Chen W. Quercetin inhibits biofilm formation by decreasing the production of EPS and altering the composition of EPS in Staphylococcus epidermidis. Front Microbiol. 2021;12:631058–65.
Acknowledgements
We thank Mashhad University of Medical Sciences, School of Pharmacy, Mashhad, Iran.
Funding
This work was financially supported by grants from the Mashhad University of Medical Sciences Research Council (grant number 991842).
Author information
Authors and Affiliations
Contributions
M.H and A.T carried out experiments and analyzed data. B.J, V.S and A.Sh wrote the manuscript, conceived and designed research, reviewed and edited the draft. All authors read and approved the final published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This article does not contain any studies with human participants or animals. All the plant species were collected from various locations by Dr. Abolfazl Shakeri (Assistant Professor) following proper guidelines and legislation procedures and identified by Ms. Souzani (MSc of Systematic Botany, Department of Pharmacognosy, School of Pharmacy) and have been deposited at the Herbarium of School of Pharmacy, Mashhad University of Medical Sciences, Iran. No specific license was required for the collection of the plants.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Hamidi, M., Toosi, A.M., Javadi, B. et al. In vitro antimicrobial and antibiofilm screening of eighteen Iranian medicinal plants. BMC Complement Med Ther 24, 135 (2024). https://doi.org/10.1186/s12906-024-04437-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12906-024-04437-x