Abstract
Pregnancy and neonatal outcomes in women diagnosed with endometriosis were investigated using data collected from the Korea National Health Insurance Claims Database and the National Health Screening Program for Infants and Children. A total of 3,778,561 women who gave birth between 2007 and 2015 were identified, and 1,938,424 primiparous women and their newborns were included in this study. Women with a diagnosis of endometriosis had a significantly higher incidence of multiple pregnancy, cesarean section, breech presentation, postpartum hemorrhage, placental abruption, placenta previa, and stillbirth than women without endometriosis (P < 0.0001). The rates of preterm birth and low birth weight were also increased in women with endometriosis (P < 0.0001). After adjusting for age, the estimated risks of these outcomes remained significant. Women previously diagnosed with endometriosis have an increased risk of adverse pregnancy and neonatal outcomes. Particular attention and information regarding these conditions should be provided to women with endometriosis during the preconception or antenatal periods.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endometriosis is characterized by the development of endometrial cells outside the uterus. The estimated prevalence of endometriosis is 10% in women of reproductive age, and up to 30–50% among women with pelvic pain or infertility [1, 2]. Endometriosis is a benign condition, but its course is progressive, chronic, and highly recurrent. General symptoms include severe dysmenorrhea, chronic pelvic pain, reduced fertility, or infertility, which may adversely affect both quality of life and reproduction in affected women.
Endometriosis is associated with impaired fertility and causes difficulty in conceiving. The monthly fecundity rate is 15–20% in healthy women, but 2–10% in women with endometriosis [3,4,5]. Although the primary pathogenic mechanisms underlying endometriosis remain unclear, complex biological factors have been suggested to play a role in its development and progression, including changes in hormonal, immunological, and inflammatory molecules. In addition, a key finding in pelvic endometriosis is the presence of adhesions in the pelvic structures causing tissue damage and anatomical distortions. These biochemical and anatomical changes are considered to have a negative effect on fertility.
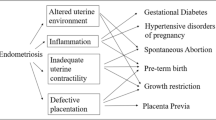
Hormones and the immune system play an important role in female reproductive function, including embryo implantation, pregnancy maintenance, and the labor process [6, 7]. Moreover, notable physiological changes occur in the anatomy and structure of pelvic organs during pregnancy due to fetal development. Considering that endometriosis is associated with the alteration and dysfunction of many hormonal and biochemical markers, as well as scarring, adhesions, and fibrotic changes in pelvic organ tissues, its effect on pregnancy and neonatal complications in women who have endometriosis should be further clarified. Recent studies reported that some adverse outcomes, such as an increased risk of preterm birth, abnormal placentation, and preeclampsia, are higher in pregnant women diagnosed with endometriosis [8,9,10]. However, there are discrepancies between the findings of different studies, which may be attributed to the evaluation of different populations, confounders, or small sample sizes [11,12,13].
This study investigated pregnancy and neonatal outcomes in primiparous women who had been previously diagnosed with endometriosis using data from a large population obtained from two national health programs.
Materials and Methods
Dataset Characteristics
This study merged datasets from the Korea National Health Insurance (KNHI) claims database and the National Health Screening Program for Infants and Children (NHSP-IC). With regard to the KNHI claims database, 97% of the Korean population is required to enroll in the KNHI program, while the remaining 3% is treated under the Medical Aid Program [14]. Therefore, this database contains all claim information for individuals, along with disease occurrences, with the exception of medical conditions and treatments that are not covered by insurance.
As a part of the KNHI system, a National Health Screening Program for Infants and Children (NHSP-IC) was started in 2007 comprising 7 consecutive health examinations in different age groups (4–9 months, 9–18 months, 18–30 months, 30–42 months, 42–54 months, 54–66 months, and 66–80 months). Data from this program included physical and anthropometric examinations and developmental screenings. This study protocol was approved by the Institutional Review Board of the Korea University Medical Center.
Dataset
An enrollment flowchart for the study is shown in Fig. 1. All women who gave birth between January 1, 2007, and December 31, 2015, were identified, and 1,840,137 multiparous cases and cases with missing data were excluded (dataset 1). Data on pregnancy outcomes were analyzed from dataset 1 using relevant codes from the International Classification of Diseases, 10th Revision (ICD-10) (World Health Organization, Geneva, Switzerland). The NHSP-IC database includes data on preterm birth, neonatal sex, and birth weight. To further analyze the neonatal outcomes related to these variables, women were excluded from the dataset if their offspring did not undergo at least 1 of the 7 consecutive NHSP-IC health examinations or if they had missing data (dataset 2).
Definitions and Pregnancy Outcomes
Maternal diseases were identified by searching relevant diagnosis codes in the ICD-10. A maternal diagnosis of endometriosis before pregnancy was made if the patients were diagnosed with endometriosis (ICD-10 code N80). Data on pregnancy outcomes were extracted from dataset 1 using ICD-10 codes, which included information on multiple pregnancies, type of delivery, breech presentation, preeclampsia, postpartum hemorrhage (PPH), placental abruption, placenta previa, and stillbirth. Data on neonatal outcomes, including preterm birth, neonatal sex, and birth weight, were extracted from dataset 2. Preterm birth was defined as having a gestational age < 37 weeks, low birth weight (LBW) was defined as a birth weight < 2,500 g, and large for gestational age (LGA) was defined as a birth weight > 4,000 g.
Statistical Analysis
Continuous and categorical variables were expressed as means ± standard deviations and percentages, respectively. Clinical characteristics were compared using the t test for continuous variables and the chi-square test for categorical variables. Multivariable logistic regression analysis was used to estimate the adjusted odds ratio (OR) and 95% confidence intervals (CIs) for an association between a history of endometriosis and adverse pregnancy outcomes. All tests were two-sided, and P < .05 was considered statistically significant. Statistical analyses were performed using the SAS software version 9.4 for Windows (SAS Inc., Cary, NC, USA).
Results
A total of 3,778,561 births (either vaginal or cesarean) were identified between 2007 and 2015 in the KNHI database. From these, we excluded 1,838,802 multiparous cases (not first pregnancy and delivery) and 1,335 cases involving missing data on maternal age. Dataset 1 contained 1,938,424 cases, including 1,893,996 without endometriosis (97.71%) and 44,283 with endometriosis (2.29%), and was used for analyzing pregnancy outcomes (Fig. 1). The mean age at the time of pregnancy was significantly higher in women with endometriosis than in the control group (31.7 ± 3.7 years vs. 29.9 ± 3.8 years) (P < 0.0001).
The results showed that women with a previous diagnosis of endometriosis had a significantly higher incidence of multiple pregnancy, cesarean section, breech presentation, preeclampsia, PPH, placental abruption, placenta previa, and stillbirth (P < 0.0001) (Table 1). Dataset 2 contained data on 1,765,549 births and was used to analyze neonatal outcomes. The incidence of preterm birth and LBW was significantly higher in women with endometriosis than in the control group (Table 2). The percentage of LGA infants was higher among women without endometriosis (P < 0.0001).
Regression analyses were performed to assess the risk of obstetric and neonatal outcomes in the affected and control groups, and data are described with crude ORs and age-adjusted ORs (AORs) with 95% confidence intervals (CIs). Women with endometriosis had a significantly higher risk of obstetric complications, which remained significant after adjusting for age (Table 3). The estimated risk of multiple pregnancy, breech presentation, placenta previa, or stillbirth was more than twofold higher in the affected group than in the control group. Furthermore, neonatal complications, such as preterm birth (AOR, 1.657; 95% CI 1.588–1.729) and LBW (AOR, 1.635; 95% CI 1.577–1.696), were significantly increased in women with endometriosis. In the sub-population analysis for the risks of obstetric and neonatal events in only singleton birth cases (n = 1,897,399), all outcome variables were found to be significant with the exception of preeclampsia, which was estimated to be AOR = 1.025 (95% CI 0.965–1.089) (Table 4).
Discussion
The results of the present population study indicated that women with a previous diagnosis of endometriosis had adverse consequences, including an increased risk of pregnancy and neonatal complications, and this risk remained significant after adjusting for maternal age. The risk of multiple pregnancy, breech presentation, and placenta previa was more than twofold higher in women with endometriosis than in the control group. In addition, the estimated risk of stillbirth was more than 1.5-fold higher in the affected group.
Endometriosis can develop in women of any age but is more prevalent in those who are in their reproductive years. Therefore, its effect on fertility and clinical outcomes in assisted reproductive technology (ART) has been extensively explored over the past few years. Recently, there has been growing concern regarding obstetric and neonatal outcomes in women previously diagnosed with endometriosis. A population study from a Danish cohort showed that women with endometriosis had an increased risk of severe preeclampsia, PPH, placenta previa, placental abruption, small for gestational age, and premature rupture of the membrane (PROM) [9]. Another study analyzed 82,793 cases of singleton births and found that the risk of preeclampsia, cesarean section, and preterm birth was higher in women with endometriosis, independent of ART [15]. Despite the discrepancies in outcome parameters between studies, the increased risk of preterm birth, PROM, and abnormal placentation in women with endometriosis has been repeatedly demonstrated [8, 9, 15,16,17,18]. Chronic inflammation is a key phenomenon in the pathogenesis of endometriosis and may be a mechanism underlying obstetrical complications [19, 20]. Proinflammatory cytokines are responsible for remodeling the uterine cervix, and their alteration can stimulate cervical weakening and membrane rupture, thus promoting uterine contraction, which is the established mechanism for preterm birth following PROM [21,22,23]. Accordingly, if the dysregulation of inflammatory molecules is induced by endometriosis in the uteroplacental environment, the risk of PROM or preterm labor is increased in women with endometriosis [19]. Previous studies have assessed the role of metalloproteinases, COX-2, and prostaglandins to clarify the mechanisms underlying preterm birth in endometriosis or adenomyosis [19, 24, 25].
Data on the risk of preeclampsia are conflicting [9, 15, 16, 26]. In theory, dysfunction in the uterine junctional zone (JZ) is associated with the development of gestational hypertension in preexisting endometriosis [27, 28]. The uterine JZ constitutes the inner one-third of the myometrium, which is highly specialized and thickened in women with endometriosis in comparison with women with a normal uterus [20, 29, 30]. The full conversion of myometrial spiral arteries into large uteroplacental vessels is essential in normal placentation [20]. Therefore, certain remodeling defects of the myometrial spiral arteries in an altered uterine JZ can induce deferred placentation and subsequent abnormal placentation, which may increase the risk of placenta previa or preeclampsia in cases of preexisting endometriosis [20, 28, 31]. The results obtained in this study showed a marginal and significant increase of preeclampsia in women with endometriosis, but this was not significant in analysis including only singleton birth cases. This finding suggests a potential effect of multiple pregnancies contributing to the risk of gestational hypertension and may explain the discrepancy in the results regarding the risk estimation of preeclampsia among previous studies.
Few studies have previously evaluated the risk of PPH in women with endometriosis, showing discordant results [9, 15]. Some have found that the occurrence of spontaneous hemoperitoneum during pregnancy is associated with preexisting endometriosis, and endometriosis seems to play a role in the pathogenesis of this condition [20, 32]. With respect to possible mechanisms, ectopic endometrial implants may induce histological changes in decidualization during pregnancy [20, 32,33,34], and these decidualized endometriotic lesions can transform into well-vascularized tissue characterized by increased vascular permeability, remodeling, and angiogenesis [35]. Therefore, these ectopic endometrial foci located near the pelvic vessels could result in vessel rupture and unpredictable bleeding [20]. This phenomenon may cause bleeding at the time of birth and during the postpartum period and could explain the higher rate of PPH in affected women. Otherwise, the increased risk of PPH in women with endometriosis might also be attributable to incomplete placental detachment or a retained placenta secondarily caused by abnormal placentation.
The increased risk of multiple pregnancies in endometriosis may indicate that more women with endometriosis are seeking ART for conception. However, the direct effect of ART on the analyzed outcomes was not assessed because ART is not covered by the National Health Insurance in our country, and this could constitute a potential limitation of this study. However, endometriosis is a common cause of infertility, and studies have shown that women with endometriosis require ART 10 times more often than women without the disease [28, 36].
Another limitation of our study was the lack of information on surgical findings and the stage of endometriosis in the affected women; thus, the relationship between obstetric outcomes and disease severity was not determined. In addition, we could not obtain information regarding anthropometric or lifestyle parameters such as smoking and alcohol intake from the database.
Nonetheless, the evaluated population was homogenous and derived from two datasets of the national cohort program, which is a strength of the study. Moreover, our data are representative of the entire population and free from potential selection bias, as most (97%) of the population is enrolled and managed under the centralized KNHI system.
In conclusion, our results indicate that women with a previous diagnosis of endometriosis have significant perinatal and neonatal complications, including abnormal placentation, PPH, stillbirth, PROM, preterm birth, and LBW. These results underscore the need for health care providers to inform women with a previous diagnosis of endometriosis about the potential obstetrical complications related to the disease and provide them with recommendations during the preconception or antenatal period.
References
Giudice LC, Kao LC. Endometriosis. Lancet. 2004;364:1789–99.
Cramer DW, Missmer SA. The epidemiology of endometriosis. Ann. N. Y. Acad. Sci. 2002;955:11–22 discussion 34–6, 396–406.
Eskenazi B, Warner ML. Epidemiology of endometriosis. Obstet. Gynecol. Clin. N. Am. 1997;24:235–58.
Dunselman GA, Vermeulen N, Becker C, et al. ESHRE guideline: management of women with endometriosis. Hum. Reprod. 2014;29:400–12.
Tomassetti C, D’Hooghe T. Endometriosis and infertility: insights into the causal link and management strategies. Best Pract Res Clin Obstet Gynaecol. 2018;51:25–33.
Mor G, Cardenas I. The immune system in pregnancy: a unique complexity. Am. J. Reprod. Immunol. 2010;63:425–33.
Bouman A, Heineman MJ, Faas MM. Sex hormones and the immune response in humans. Hum. Reprod. Update. 2005;11:411–23.
Conti N, Cevenini G, Vannuccini S, Orlandini C, Valensise H, Gervasi MT, Ghezzi F, di Tommaso M, Severi FM, Petraglia Fet al. Women with endometriosis at first pregnancy have an increased risk of adverse obstetric outcome. J. Matern. Fetal Neonatal Med. 2015;28:1795–1798.
Berlac JF, Hartwell D, Skovlund CW, Langhoff-Roos J, Lidegaard O. Endometriosis increases the risk of obstetrical and neonatal complications. Acta Obstet. Gynecol. Scand. 2017;96:751–60.
Harada T, Taniguchi F, Onishi K, Kurozawa Y, Hayashi K, Harada T. Obstetrical complications in women with endometriosis: a cohort study in Japan. PLoS One. 2016;11:e0168476.
Brosens IA, De Sutter P, Hamerlynck T, et al. Endometriosis is associated with a decreased risk of pre-eclampsia. Hum. Reprod. 2007;22:1725–9.
Benaglia L, Bermejo A, Somigliana E, Scarduelli C, Ragni G, Fedele L, Garcia-Velasco JAet al. Pregnancy outcome in women with endometriomas achieving pregnancy through IVF. Hum. Reprod. 2012;27:1663–1667.
Mekaru K, Masamoto H, Sugiyama H, et al. Endometriosis and pregnancy outcome: are pregnancies complicated by endometriosis a high-risk group? Eur. J. Obstet. Gynecol. Reprod. Biol. 2014;172:36–9.
Kang EJ, Seo JH, Kim LY, et al. Pregnancy-associated risk factors of postpartum breast cancer in Korea: a nationwide health insurance database study. PLoS One. 2016;11:e0168469.
Glavind MT, Forman A, Arendt LH, Nielsen K, Henriksen TB. Endometriosis and pregnancy complications: a Danish cohort study. Fertil. Steril. 2017;107:160–6.
Zullo F, Spagnolo E, Saccone G, et al. Endometriosis and obstetrics complications: a systematic review and meta-analysis. Fertil. Steril. 2017;108:667–672.e5.
Lalani S, Choudhry AJ, Firth B, Bacal V, Walker M, Wen SW, Singh S, Amath A, Hodge M, Chen Iet al. Endometriosis and adverse maternal, fetal and neonatal outcomes, a systematic review and meta-analysis. Hum. Reprod. 2018;33:1854–1865.
Chen I, Lalani S, Xie RH, Shen M, Singh SS, Wen SW. Association between surgically diagnosed endometriosis and adverse pregnancy outcomes. Fertil. Steril. 2018;109:142–7.
Petraglia F, Arcuri F, de Ziegler D, Chapron C. Inflammation: a link between endometriosis and preterm birth. Fertil. Steril. 2012;98:36–40.
Leone Roberti Maggiore U, Ferrero S, Mangili G, et al. A systematic review on endometriosis during pregnancy: diagnosis, misdiagnosis, complications and outcomes. Hum. Reprod. Update. 2016;22:70–103.
Osman I, Young A, Ledingham MA, Thomson AJ, Jordan F, Greer IA, Norman JEet al. Leukocyte density and pro-inflammatory cytokine expression in human fetal membranes, decidua, cervix and myometrium before and during labour at term. Mol. Hum. Reprod. 2003;9:41–45.
King AE, Kelly RW, Sallenave JM, Bocking AD, Challis JR. Innate immune defences in the human uterus during pregnancy. Placenta. 2007;28:1099–106.
Xu P, Alfaidy N, Challis JR. Expression of matrix metalloproteinase (MMP)-2 and MMP-9 in human placenta and fetal membranes in relation to preterm and term labor. J. Clin. Endocrinol. Metab. 2002;87:1353–61.
Bieglmayer C, Hofer G, Kainz C, Reinthaller A, Kopp B, Janisch H. Concentrations of various arachidonic acid metabolites in menstrual fluid are associated with menstrual pain and are influenced by hormonal contraceptives. Gynecol. Endocrinol. 1995;9:307–12.
Karck U, Reister F, Schafer W, Zahradnik HP, Breckwoldt M. PGE2 and PGF2 alpha release by human peritoneal macrophages in endometriosis. Prostaglandins. 1996;51:49–60.
Perez-Lopez FR, Calvo-Latorre J, Alonso-Ventura V, Bueno-Notivol J, Martinez-Dominguez SJ, Chedraui P. Systematic review and meta-analysis regarding the association of endometriosis and preeclampsia in women conceiving spontaneously or through assisted reproductive technology. Pregnancy Hypertens. 2018;14:213–21.
Brosens I, Pijnenborg R, Vercruysse L, Romero R. The “great obstetrical syndromes” are associated with disorders of deep placentation. Am. J. Obstet. Gynecol. 2011;204:193–201.
Falconer H. Pregnancy outcomes in women with endometriosis. Semin. Reprod. Med. 2013;31:178–82.
Benagiano G, Brosens I, Habiba M. Structural and molecular features of the endomyometrium in endometriosis and adenomyosis. Hum. Reprod. Update. 2014;20:386–402.
Exacoustos C, Luciano D, Corbett B, et al. The uterine junctional zone: a 3-dimensional ultrasound study of patients with endometriosis. Am. J. Obstet. Gynecol. 2013;209:248.e1–7.
Brosens IA, Robertson WB, Dixon HG. The role of the spiral arteries in the pathogenesis of preeclampsia. Obstet Gynecol Annu. 1972;1:177–91.
Lier MCI, Malik RF, Ket JCF, Lambalk CB, Brosens IA, Mijatovic V. Spontaneous hemoperitoneum in pregnancy (SHiP) and endometriosis - a systematic review of the recent literature. EurJ Obstet Gynecol Reprod Biol. 2017;219:57–65.
Bailleux M, Bernard JP, Benachi A, Deffieux X. Ovarian endometriosis during pregnancy: a series of 53 endometriomas. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017;209:100–4.
Ueda Y, Enomoto T, Miyatake T, Fujita M, Yamamoto R, Kanagawa T, Shimizu H, Kimura Tet al. A retrospective analysis of ovarian endometriosis during pregnancy. Fertil. Steril. 2010;94:78–84.
Plaisier M. Decidualisation and angiogenesis. Best Pract Res Clin Obstet Gynaecol. 2011;25:259–71.
Stephansson O, Kieler H, Granath F, Falconer H. Endometriosis, assisted reproduction technology, and risk of adverse pregnancy outcome. Hum. Reprod. 2009;24:2341–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study protocol was approved by the Institutional Review Board of the Korea University Medical Center.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Yi, K.W., Cho, G.J., Park, K. et al. Endometriosis Is Associated with Adverse Pregnancy Outcomes: a National Population-Based Study. Reprod. Sci. 27, 1175–1180 (2020). https://doi.org/10.1007/s43032-019-00109-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-019-00109-1