Abstract
Application of multiwall carbon nanotubes (MWCNTs) could have promotive effects on crop plants; however, their effects on seed germination and early growth of maize under cadmium (Cd) stress have rarely been investigated. A pot experiment was conducted with two maize varieties, Yuebaitiannuo7 and Yuecainuo2, grown under three Cd levels, i.e., 0, 100, and 200 mg L−1 (denoted as Cd0, Cd100, and Cd200, respectively) and three MWCNT levels, i.e., 0, 100, and 200 mg L−1 (denoted as M0, M100, and M200, respectively) as seed priming treatments to assess the MWCNT-induced modulations in seed germination rate, seedling growth, and related physio-biochemical attributes under Cd stress. Yuebaitiannuo7 showed higher seed germination rate and less root and shoot growth than Yuecainuo2. The MWCNT application promoted the seed germination rate of Yuecainuo2 by 11.42% at M100 and 24.76% at M200 under Cd100 at 4 days after sowing; however, these effects remained non-significant for Yuebaitiannuo7. Moreover, the MWCNTs enhanced root and shoot fresh weight and antioxidant enzyme activities, including peroxidase (POD), superoxide dismutase (SOD), and catalase (CAT) activities, and reduced the malonaldehyde (MDA) content under Cd across MWCNT treatments. Nevertheless, the toxic effects were also observed, indicating a potential risk posed by MWCNTs at high concentrations. Application of MWCNTs alleviated the Cd toxicity in growing maize plants; however, their effects were concentration dependent. Yuecainuo2 performed better than Yuebaitiannuo7 with the application of MWCNT under Cd stress.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Maize (Zea mays L.) is an important cereal crop worldwide due to its high nutritional value and is commercially used in a variety of products, including glue and edible products (Ranum et al. 2014). Even though genetic improvements in maize have led to significant enhancements in its production across China, a significant gap exists between experimental and average yield at farmers’ level (Meng et al. 2013). Therefore, it is necessary to develop some strategies to improve the crop yields with limited agro-resources.
Heavy metal pollution is a severe environmental issue (Adhikari et al. 2018; Bin et al. 2013; Huamain et al. 1999) whereas cadmium (Cd) is a major hazardous heavy metal in soil which is continuously being added by the excessive use of chemical agricultural products, and application of untreated waste water and sewage sludge which are generally considered the toxic for animal and human health and crop growth (Anjum et al. 2016; Lin et al. 2007; Yang et al. 2006). For instance, Cd treatment hindered the growth of Leucaena leucocephala and reduced its morphological attributes and dry biomass (Muhammad et al. 2010). Seed germination of Catharanthus roseus was severely inhibited under Cd stress (Nabaei and Amooaghaie 2019) with reduced photosynthetic pigments (Cui and Wang 2018). Cd toxicity reduced seed germination percentage, seedling vigor index, and the shoot and root fresh weight of maize (Chaudhary and Sharma 2009; Gowayed 2017), and an increase in Cd concentration resulted in reduced plant height, leaf area, and yield of maize (Anjum et al. 2016). Furthermore, Cd induces oxidative stress and alters the antioxidant defense system in plants (L.M. et al. 2001; Qu et al. 2018). The production of reactive oxygen species (ROS) at low Cd concentrations stimulates antioxidant enzyme activities; however, excessive ROS production at high Cd concentrations could lead to oxidative damage and growth inhibition in crop plants (Adhikari et al. 2018; Anjum et al. 2015; Cui and Wang 2018). Similarly, Cd toxicity could diminish antioxidant enzyme activities in maize (Anjum et al. 2015; Cui and Wang 2018). Overall, Cd toxicity severely affects the growth and related physio-biochemical attributes of crop plants; therefore, management strategies are needed to alleviate the toxic effects of Cd under Cd-contaminated soil.
Nanotechnology is a promising tool in agriculture and is used for the precise application of fertilizers and pesticides (Servin et al. 2015). Different nanoparticles (NPs) are used as a nutrient source to protect plants against metal stress and are applied in food quality maintenance (Lustriane et al. 2018; Wagi and Ahmed 2019; Yang et al. 2018). Likewise, multiwall carbon nanotubes (MWCNTs), a type of carbon nanoparticle (CNT), may expedite crop plants’ growth (Al-Rekaby 2018). After being absorbed through the roots, MWCNTs are transported to the stem and thus improve growth by increasing shoot and root elongation, biomass, and vigor indices of seeds of various crops (Joshi et al. 2018; Seddighinia et al. 2020). Currently, MWCNTs are known to enhance maize growth by facilitating biomass accumulation and seed germination (Dasgupta et al. 2017; Tiwari et al. 2013; Zaytseva 2016; Zhai et al. 2015). MWCNTs promote water imbibition and mineral nutrient supply in maize by stimulating ion-CNT transient-dipole interactions, thus improving seed germination and seedling growth (Dasgupta et al. 2017; Tiwari et al. 2013). Moreover, the application of MWCNTs induces the expression of antioxidant genes (Fan et al. 2018; Joshi et al. 2018; Seddighinia et al. 2020; Zhao et al. 2019) and enhanced the activities of antioxidants, such as catalase (CAT) and peroxidase (POD), in plants (Ghasempour et al. 2019). However, the efficacy of MWCNT application in maize is dependent on the application dosage, particle size, and net charge. It has also been found that root elongation in maize diminished with reduced seedling growth under MWCNT treatment (Tiwari et al. 2013; Zaytseva 2016; Zhai et al. 2015). In addition, CNTs have been recently investigated for their potential applications in phytoremediation of Cd-contaminated sites. CNTs alleviate Cd-induced toxicity in plants by increasing shoot and root length, chlorophyll content, and antioxidant enzyme activities (Oloumi et al. 2017). Similarly, MWCNTs induce plant resistance to Cd stress by promoting root and shoot growth and modulating the antioxidant defense system (Gong et al. 2019; Oloumi et al. 2018). In sum, application of MWCNTs during maize cultivation may address Cd contamination issues.
Previously, multiwall carbon nanotubes (MWCNTs) have been used to improve the growth of crop plants; however, their effects on the seed germination and early growth of maize under cadmium (Cd) toxicity have rarely been investigated. The present study was therefore conducted to assess the effects of MWCNTs on growing maize plants and the related physio-biochemical attributes under Cd toxicity to assess the efficacy of MWCNT application to alleviate Cd stress and improve maize growth.
2 Material and Methods
2.1 Experimental Description
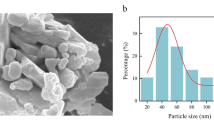
A pot experiment was conducted from July to October 2019 at the College of Agriculture, South China Agricultural University, Guangzhou, China (23° 09′ N, 113° 22′ E). Seeds of two maize varieties, Yuebaitiannuo7 and Yuecainuo2 which are widely cultivated in South China, were obtained from the Crop Research Institute, Guangdong Academy of Agricultural Sciences, Guangzhou, China. The Yuebaitiannuo7 and Yuecainuo2 are well-known and widely grown hybrid waxy maize varieties with high quality, high yield, and disease resistance in South China. The MWCNT (purity >95%) was purchased from Aladdin (Shanghai, China). The inner diameter, outer diameter, and length of the MWCNT were 3–5 nm, 8–15 nm, and ~ 50 μm, respectively.
2.2 Experimental Treatments
The experimental treatments consisted of two maize varieties, i.e., Yuebaitiannuo7 and Yuecainuo2; three cadmium chloride levels, i.e., 0, 100, and 200 mg L−1, denoted as Cd0, Cd100, and Cd200, respectively; and three multiwall carbon nanotube levels, i.e., 0, 100, and 200 mg L−1, denoted as M0, M100, and M200, respectively. The Cd concentration was based on previous studies of Chaudhary and Sharma (2009), who reported that Cd levels up to 200 mg L−1 resulted in a significant reduction in maize growth.
The multiwall carbon nanotube (MWCNT) treatment of seeds was performed according to Rao and Srivastava (2014). Before sowing, uniform seeds were sterilized with 3% NaClO solution for 20 min and rinsed with pure water. The MWCNT solutions were oscillated using an ultrasonic cleaner (HZS-H, China) for 15 min for better dispersion. The seeds of both maize varieties were soaked in the respective MWCNT solutions for 24 h, during which the MWCNT solutions were replaced every 12 h. After MWCNT treatment, the seeds were placed on filter paper applied with the respective Cd treatments with the endosperm facing upwards. To maintain stable Cd concentrations, filter papers were replaced every 2 days (Chaudhary and Sharma 2009). Cultivation boxes that were 16.5 cm in length, 11.5 cm in width, and 3.5 cm in height were kept indoors at 23–25 °C for 4 days.
2.3 Sampling and Measurements
2.3.1 Seed Germination Rate
Seeds were considered germinated when the radical reached 2 mm (Zaytseva 2016). The germinated seeds were counted on daily basis up to 3 days to calculate the germination rate. The seed germination rate (%) was calculated as follows:
2.3.2 Root and Shoot Growth
The root lengths of five seedlings were measured using ruler for each replicate on the fourth day after sowing. Simultaneously, root and shoot fresh weights were detected using an electronic analytical balance in 20 seedlings from each replicate and three replicates from each treatment. Shoot length and root diameter were determined by Digimatic calipers (Guilin Guanglu Measuring Instrument Co., Ltd., China) from 5 seedlings per replicate.
2.3.3 Activity of Antioxidant Enzymes and Malonaldehyde (MDA) Content
Germinated seeds were separated into roots and shoots, frozen in liquid nitrogen for 5 min, and stored at −80 °C for biochemical analyses. Crude enzyme samples were extracted according to Li et al. (2019). Briefly, fresh samples (0.3 g) were homogenized with 5 mL of sodium phosphate buffer at pH 7.8 and transferred into 10-mL centrifuge tubes to be centrifuged at 8000 rpm for 15 min. The obtained supernatant was used as a crude enzyme sample. Antioxidant enzyme activities of superoxide dismutase (SOD), POD, and CAT, as well as MDA contents were estimated according to Li et al. (2019).
For superoxide dismutase (SOD) activity, crude enzyme extract (5 μL) was added to a reaction mixture including 150 μL of 50 mM sodium phosphate buffer, 30 μL of 130 mM methionine, 30 μL of 750 μM nitroblue tetrazolium chloride, 30 μL of 100 μM ethylene diaminetetraacetic acid disodium salt, and 30 μL of 20 μM riboflavin and placed under 4000 lx light for 20 min, and the absorbance was read at 560 nm. One unit of SOD activity (U) was defined as the amount of enzyme causing a 50% reduction in the absorbance at 560 nm with the same amount of sodium phosphate-buffered solution (PBS) as a control. SOD activity is represented as units per gram fresh weight.
For peroxidase (POD) activity, 5 μL of crude enzyme extract was added to a reaction mixture composed of 100 μL of 50 mM sodium phosphate buffer, 100 μL of 0.3% hydrogen peroxide solution, and 95 μL of 0.2% guaiacol solution, and the absorbance was read at 470 nm every 30 s for 2.5 min. One unit of POD activity (U) was a reduction in absorbance per minute at 470 nm. The POD activity is depicted as units per gram fresh weight.
For catalase (CAT) activity, 0.05 mL of crude extracted enzyme solution was added to a reaction mixture composed of 1.95 mL of ultrapure water and 1 mL of 0.3% hydrogen peroxide solution, and the absorbance was promptly detected and recorded at 240 nm every 30 s for 2 min with 3 mL ultrapure water as a control. One unit of CAT activity (U) was defined as a reduction in absorbance every minute by 0.01. The CAT activity is represented as units per gram fresh weight.
For the malondialdehyde (MDA) content, 0.2 mL of crude extracted enzyme solution was added to 0.4 mL of 0.5% thiobarbituric acid solution and heated in boiling water for 30 min. The reaction mixture was cooled down and centrifuged at 8000 rpm for 10 min and the absorbance was read at 532 nm, 600 nm, and 450 nm to measure the MDA content. The final content is expressed as μmoL g−1 fresh weight.
2.4 Experimental Design and Statistical Analysis
The experiments were conducted in a completely randomized design (CRD) with 4 replicates. The experimental data were analyzed by Statistix version 8 (Analytical Software, Tallahassee, FL, USA). The difference among treatments was determined by the least significant difference (LSD) test at the 0.05 probability level. Correlations between germination rates and morphological and physiological attributes were detected by Pearson’s analyses.
3 Results
3.1 Seed Germination Rate
Seed germination rates at 2, 3, and 4 days after sowing (DAS) significantly varied for both maize varieties (Table 1), whereas Yuebaitiannuo7 showed higher seed germination rate than Yuecainuo2 (Fig. 1). On the other hand, Cd and MWCNT treatment showed no significant effect on the seed germination rate at 3 and 4 DAS for Yuebaitiannuo7. The M100 dose significantly increased the seed germination rate of Yuecainuo2 at 2 and 3 DAS compared with M0 under Cd100 (Fig. 1a, b), and the seed germination rate was significantly affected by the interaction of V × M at 4 DAS (Table 1). Compared with M0, the M100 and M200 increased the seed germination rate of Yuecainuo2 at 4 DAS under Cd100 by 11.42% and 24.76%, respectively (Fig. 1c). However, the effect of MWCNTs on the seed germination rate under Cd200 remained non-significant (P ≥ 0.05).
Effect of MWCNTs on germination rates at 2 DAS (a), 3 DAS (b), and 4 DAS (c) in maize varieties under Cd stress. Vertical bars represent the mean values. Capped bars above the means are the SEs (standard errors) of four replicates. Lowercase letters indicate comparisons among the treatments by LSD test at P = 0.05. DAS, days after sowing; Cd, cadmium; CdCl2, cadmium chloride; MWCNT, multiwalled carbon nanotube; Cd 0, 0 mg L−1 CdCl2; Cd 100, 100 mg L−1 CdCl2; Cd 200, 200 mg L−1 CdCl2; M 0, 0 mg L−1 MWCNTs; M 100, 100 mg L−1 MWCNTs; M 200, 200 mg L−1 MWCNTs
3.2 Root and Shoot Growth
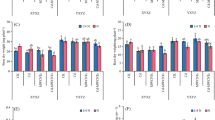
Cadmium and Variety×Cadmium significantly affected the root fresh weight (Table 1); for instance, Cd200 decreased the root fresh weight of Yuebaitiannuo7 and Yuecainuo2 by 56.50% and 79.14%, respectively (Fig. 2a). Furthermore, M and V × M significantly affected the root fresh weight (Table 1), whereas M200 increased the root fresh weight of Yuebaitiannuo7 by 8.37% under Cd0, compared with Cd0M0. In contrast, the root fresh weight of Yuecainuo2 was reduced by 20.14% at M100Cd0, compared with Cd0M0 (Fig. 2a). The Cd × M and V × Cd × M significantly affected the root fresh weight (Table 1), whereas M100 increased the root fresh weight of Yuebaitiannuo7 by 13.73% at Cd100 and 51.76% at Cd200, respectively, compared with M0. However, the effect of MWCNTs on the root fresh weight of Yuecainuo2 under Cd stress remained non-significant (Fig. 2a).
Effect of MWCNTs on the fresh weight of roots (a) and shoots (b) under Cd stress. Vertical bars represent the mean values. Capped bars above the means are the SEs (standard errors) of four replicates. Lowercase letters indicate comparisons among the treatments by LSD test at P = 0.05. Cd, cadmium; CdCl2, cadmium chloride; MWCNTs, multiwalled carbon nanotubes; Cd 0, 0 mg L−1 CdCl2; Cd 100, 100 mg L−1 CdCl2; Cd 200, 200 mg L−1 CdCl2; M 0, 0 mg L−1 MWCNTs; M 100, 100 mg L−1 MWCNTs; M 200, 200 mg L−1 MWCNTs
Cadmium and Variety×Cadmium significantly affected the shoot fresh weight (Table 1), whereas cadmium treatment decreased the shoot fresh weight of Yuecainuo2 by 36.65% at Cd100 and 63.35% at Cd200 compared with that at Cd0M0. Nonetheless, the effect of Cd on the shoot fresh weight of Yuebaitiannuo7 was non-significant (Fig. 2b). The V × M significantly affected the shoot fresh weight (Table 1). The M200 significantly increased the shoot fresh weight of Yuebaitiannuo7 by 31.15% under Cd0, compared with Cd0M0. In contrast, the M200 significantly reduced the shoot fresh weight of Yuecainuo2 by 20.07% under Cd0 compared with Cd0M0 (Fig. 2b). The Cd × M and V × Cd × M significantly affected the shoot fresh weight (Table 1), whereas M200 significantly decreased the shoot fresh weight of Yuebaitiannuo7 under Cd100 by 11.93%. The M100 significantly decreased the shoot fresh weight of Yuecainuo2 by 21.43% at Cd100, but the shoot fresh weight was increased by 39.43% at Cd200, compared with M0 (Fig. 2b).
Cadmium and Variety×Cadmium significantly affected the root length (Table 1). For instance, Cd100 significantly reduced the root length of Yuebaitiannuo7 and Yuecainuo2 by 60.10% and 61.70%, respectively, under M0 compared with Cd0M0 (Fig. 3a). Moreover, M, Cd × M and V × Cd × M significantly affected root length (Table 1); i.e., M100 and M200 significantly reduced the root length of Yuecainuo2 by 24.58% and 42.78%, respectively, under Cd100 compared with M0 (Fig. 3a).
Effect of MWCNTs on the length of roots (a) and shoots (b) and root diameter (c) under Cd stress. Vertical bars represent the mean values. Capped bars above the means are the SEs (standard errors) of four replicates. Lowercase letters indicate comparisons among the treatments by LSD test at P = 0.05. Cd, cadmium; CdCl2, cadmium chloride; MWCNTs, multiwalled carbon nanotubes; Cd 0, 0 mg L−1 CdCl2; Cd 100, 100 mg L−1 CdCl2; Cd 200, 200 mg L−1 CdCl2; M 0, 0 mg L−1 MWCNTs; M 100, 100 mg L−1 MWCNTs; M 200, 200 mg L−1 MWCNTs
Variety, Cadmium and Variety×Cadmium significantly affected the shoot length (Table 1). Cd100 and Cd200 substantially reduced the shoot length of Yuecainuo2 by 17.14% and 34.32%, respectively, under M0 compared with Cd0M0, whereas the effect of Cd on the shoot length of Yuebaitiannuo7 under M0 remained non-significant (Fig. 3b). The Cd × M significantly affected the shoot length (Table 1); for example, M200 significantly increased the shoot length of Yuebaitiannuo7 by 12.79% under Cd0, compared with Cd0M0. Conversely, MWCNT application decreased the shoot length of Yuecainuo2 by 15.66% at M100 and 25.58% at M200 under Cd100 compared with M0 (Fig. 3b). The root diameter remained similar for both maize varieties under Cd and MWCNT treatments (Fig. 3c).
3.3 Activity of Antioxidant Enzymes and Malonaldehyde Content
Substantial differences were noted regarding superoxide dismutase (SOD) activity for both maize cultivars (Table 1), whereas the SOD activity of Yuebaitiannuo7 remained higher than that of Yuecainuo2 (Fig. 4). Cd and V × Cd significantly affected the SOD activity in the roots of both maize varieties (Table 1). Cd significantly decreased the SOD activity in the roots of Yuecainuo2 by 69.24% at Cd100, compared with Cd0M0 (Fig. 4a). V × M significantly affected the root SOD activity (Table 1). The M200 significantly reduced the root SOD activity in Yuebaitiannuo7 by 41.09% under Cd0, compared with M0 (Fig. 4a). Moreover, Cd × M and V × Cd × M significantly affected the root SOD activity (Table 1). M200 significantly increased the SOD activity in the roots of Yuecainuo2 by 146.55% at Cd100, but decreased by 35.43% at Cd200, compared with M0 (Fig. 4a). Cd and V × Cd significantly affected the SOD activity in shoots (Table 1); e.g., Cd100 decreased the SOD activity in the shoots of Yuebaitiannuo7 by 12.30% but increased SOD activity in the shoots of Yuecainuo2 by 51.66% under M0, compared with Cd0M0 (Fig. 4b). Furthermore, application of MWCNTs significantly affected the SOD activity in shoots (Table 1); for example, the M200 significantly decreased the SOD activity in the shoots of Yuebaitiannuo7 by 28.89% under Cd0 compared with Cd0M0. The MWCNT treatment significantly increased the SOD activity in the shoots of Yuecainuo2 by 89.22% at M100 under Cd0 compared with Cd0M0 (Fig. 4b). Cd × M and V × Cd × M significantly affected the SOD activity in shoots (Table 1). MWCNT treatment significantly increased the SOD activity in the shoots of Yuebaitiannuo7 under Cd stress. However, MWCNT treatment decreased SOD activity in the shoots of Yuecainuo2 by 29.13% at M100 under Cd100 compared with that under M0 (Fig. 4b).
Effect of MWCNTs on SOD activity in roots (a) and shoots (b) of maize varieties under Cd stress. Vertical bars represent the mean values. Capped bars above the means are the SEs (standard errors) of four replicates. Lowercase letters indicate comparisons among the treatments by LSD test at P = 0.05. Cd, cadmium; CdCl2, cadmium chloride; MWCNTs, multiwalled carbon nanotubes; SOD, superoxide dismutase; FW, fresh weight; Cd 0, 0 mg L−1 CdCl2; Cd 100, 100 mg L−1 CdCl2; Cd 200, 200 mg L−1 CdCl2; M 0, 0 mg L−1 MWCNTs; M 100, 100 mg L−1 MWCNTs; M 200, 200 mg L−1 MWCNTs
Variety, Cadmium and Variety×Cadmium significantly affected peroxidase (POD) activity in roots (Table 1). Cd treatment significantly increased POD activity in the roots of Yuecainuo2 46.41% at Cd200 under M0 compared with Cd0M0 (Fig. 5a). M treatment significantly affected the POD activity in roots of both maize verities (Table 1); for example, M200 significantly decreased the POD activity in the roots of Yuebaitiannuo7 by 17.91% under Cd0 compared with Cd0M0 (Fig. 5a). Cd × M and V × Cd × M significantly affected the POD activity in roots (Table 1); e.g., M100 significantly reduced the POD activity by 26.19% under Cd100 compared with that under Cd0M0 (Fig. 5a). Cd and V × Cd significantly affected the POD activity in shoots (Table 1). Cd200 significantly increased POD activity in the shoots of Yuecainuo2 by 29.29% under M0 compared with Cd0M0 (Fig. 5b). M and V × M significantly affected the shoot POD activity (Table 1). M100 significantly increased the POD activity in the shoots of Yuebaitiannuo7 by 59.98% under Cd0, compared with Cd0M0 (Fig. 5b). Cd × M significantly affected the POD activity in the shoots of both maize verities (Table 1). M100 significantly increased the POD activity in the shoots of Yuebaitiannuo7 by 16.25% under Cd200 compared with M0. M200 significantly decreased the POD activity in the shoots of Yuecainuo2 by 32.33% at Cd200 compared with M0 (Fig. 5b).
Effect of MWCNTs on POD activity in roots (a) and shoots (b) of maize varieties under Cd stress. Vertical bars represent the mean values. Capped bars above the means are the SEs (standard errors) of four replicates. Lowercase letters indicate comparisons among the treatments by LSD test at P = 0.05. Cd, cadmium; CdCl2, cadmium chloride; MWCNTs, multiwalled carbon nanotubes; POD, peroxidase; FW, fresh weight; Cd 0, 0 mg L−1 CdCl2; Cd 100, 100 mg L−1 CdCl2; Cd 200, 200 mg L−1 CdCl2; M 0, 0 mg L−1 MWCNTs; M 100, 100 mg L−1 MWCNTs; M 200, 200 mg L−1 MWCNTs
Variety, Cadmium and Variety×Cadmium significantly affected the catalase (CAT) activity in roots (Table 1). The CAT activity in the roots of Yuebaitiannuo7 was increased by 171.79% at Cd200 under M0 compared with that under Cd0M0. The CAT activity in the roots of Yuecainuo2 was increased by 163.13% at Cd100 under M0 compared with Cd0M0 (Fig. 6a). M200 significantly decreased CAT activity in the roots of Yuebaitiannuo7 by 39.56% at Cd0, compared with Cd0M0. Cd × M and V × Cd × M significantly affected the root CAT activity (Table 1). M100 significantly reduced the CAT activity in the roots of Yuebaitiannuo7 by 17.31% under Cd200 compared with M0. M100 significantly reduced the CAT activity in the roots of Yuecainuo2 by 35.63% under Cd100 compared with M0. M100 significantly increased the CAT activity in the roots of Yuecainuo2 by 56.68% under Cd200, compared with M0 (Fig. 6a). Variety, Cd and V × Cd significantly affected the CAT activity in shoots (Table 1). The CAT activity of Yuebaitiannuo7 in shoots was significantly higher than Yuecainuo2 (Fig. 6b). Compared with Cd0M0, the Cd200 significantly increased the CAT activity in the shoots of Yuebaitiannuo7 and Yuecainuo2 by 46.94% and 117.16% under M0, respectively (Fig. 6b). M and V × M were significantly affected the CAT activity in shoots (Table 1). M100 significantly increased the CAT activity in the shoots of Yuebaitiannuo7 under Cd0 by 13.54%, compared with Cd0M0 (Fig. 6b). Cd × M and V × Cd × M significantly affected the CAT activity in shoots (Table 1). The CAT activity in the shoots of Yuebaitiannuo7 was improved by 14.24% at M100 under Cd200, while M200 decreased the CAT activity by 14.16% compared with M0. M200 significantly increased the CAT activity in the shoots of Yuecainuo2 under Cd 200 by 44.74% compared with M0 (Fig. 6b).
Effect of MWCNTs on CAT activity in roots (a) and shoots (b) of maize varieties under Cd stress. Vertical bars represent the mean values. Capped bars above the means are the SEs (standard errors) of four replicates. Lowercase letters indicate comparisons among the treatments by LSD test at P = 0.05. Cd, cadmium; CdCl2, cadmium chloride; MWCNTs, multiwalled carbon nanotubes; CAT, catalase; FW, fresh weight; Cd 0, 0 mg L−1 CdCl2; Cd 100, 100 mg L−1 CdCl2; Cd 200, 200 mg L−1 CdCl2; M 0, 0 mg L−1 MWCNTs; M 100, 100 mg L−1 MWCNTs; M 200, 200 mg L−1 MWCNTs
Variety×Multiwall carbon nanotube, Cadmium×Multiwall carbon nanotube, and Variety×Cadmium×Multiwall carbon nanotube significantly affected the malondialdehyde (MDA) contents in the roots of both maize varieties (Table 1). The MDA content in roots of Yuebaitiannuo7 was decreased by 16.45% under Cd0 at M100, compared with Cd0M0. M200 increased the MDA content in the roots of Yuecainuo2 by 35.33% under Cd0 (Fig. 7a). M200 significantly reduced the MDA content in the roots of Yuebaitiannuo7 under Cd100 by 21.72%, compared with M0. M200 significantly increased the MDA content in the roots of Yuebaitiannuo7 by 23.96% under Cd200. M200 significantly decreased the MDA content in the roots of Yuecainuo2 by 42.64% under Cd100, compared with M0. In contrast, the MDA content in the roots of Yuecainuo2 was increased by 34.63% at M200 under Cd200, compared with M0 (Fig. 7a). Variety, Cd and V × Cd significantly affected the MDA content in shoots (Table 1). The MDA content in the shoots of Yuebaitiannuo7 was significantly increased compared with Cd0M0 by 54.66% at Cd100M0. Similarly, the MDA content in the shoots of Yuecainuo2 was significantly increased compared with Cd0M0 by 40.24% at Cd100M0 (Fig. 7b). V × M significantly affected the MDA content in shoots (Table 1). M100 significantly increased the MDA content in the shoots of Yuebaitiannuo7 under Cd0 by 27.40% compared with Cd0M0 (Fig. 7b). Cd × M and V × Cd × M significantly affected the MDA content in shoots (Table 1). M100 significantly increased the MDA content in the shoots of Yuebaitiannuo7 by 11.66% under Cd200, compared with M0. M200 significantly decreased the MDA content in the shoots of Yuecainuo2 by 24.97% under Cd100 (Fig. 7b).
Effect of MWCNTs on MDA content in roots (a) and shoots (b) of maize varieties under Cd stress. Vertical bars represent the mean values. Capped bars above the means are the SEs (standard errors) of four replicates. Lowercase letters indicate comparisons among the treatments by LSD test at P = 0.05. Cd, cadmium; CdCl2, cadmium chloride; MWCNTs, multiwalled carbon nanotubes; MDA, malondialdehyde; FW, fresh weight; Cd 0, 0 mg L−1 CdCl2; Cd 100, 100 mg L−1 CdCl2; Cd 200, 200 mg L−1 CdCl2; M 0, 0 mg L−1 MWCNTs; M 100, 100 mg L−1 MWCNTs; M 200, 200 mg L−1 MWCNTs
3.4 Correlation Analysis
The germination rate at 4 days after sowing (DAS) showed a significant and positive correlation with root superoxide dismutase (SOD) activity, shoot SOD activity, and root peroxidase (POD) activity but a significant negative correlation with shoot malondialdehyde (MDA) content. Moreover, shoot fresh weight was significantly and positively associated with root length, shoot length, and root SOD activity but negatively associated with root and shoot CAT activity (Table 2).
4 Discussion
Toxic effects of cadmium (Cd) on the early growth of crop plants have been widely reported (Gowayed 2017; Gozubenli 2010; Kilic et al. 2017); however, improvements in seed germination at Cd low concentrations, i.e., 2–20 mg L−1, have also been reported previously (Liu et al. 2010). Presence of Cd could inhibit the seed germination in various plant species such as Trigonella foenum-graecum (Espanany et al. 2015), Catharanthus roseus (Nabaei and Amooaghaie 2019) and Leucaena leucocephala (Muhammad et al. 2010), and Oryza sativa (Kanu et al. 2019) etc. It has been reported that root and shoot elongation, biomass accumulation, and seedling dry weight decreased under Cd stress (L.M. et al. 2001; Muhammad et al. 2010; Shen et al. 2018). In the present study, Cd100 significantly reduced the germination rate in Yuecainuo2 whereas no significant effects were noted for Yuebaitiannuo7 in this regard (Fig. 1). Our findings are in accordance with Meng et al. (2016) who reported that Cd inhibited the maize seed germination and concluded that such effects might be genotype specific; i.e., the same Cd level could be toxic for one variety but not for the other(s). Cd treatment has been reported to cause the inhibition in water and nutrient uptake, chromosomal abnormalities, and interference with cell division, thus inhibiting seed germination (Gowayed 2017; Gozubenli 2010; Hemachandra and Pathiratne 2015). In this study, the growth of Yuebainuo2 was reduced in terms of reduced root-shoot length and weight under Cd toxicity (Figs. 2 and 3). These findings corroborate with previous reports regarding the effects of Cd toxicity on the growth of maize (Chaudhary and Sharma 2009; Liu et al. 2010; Meng et al. 2016). The presence of Cd could result in inactivation of -SH enzymes including amylase, nitrogen metabolism- and chlorophyll-generation-related enzymes. Decreased amylase activity would inhibit reserve starch hydrolyzation (Chaudhary and Sharma 2009), and a decrease in nitrogen metabolism enzymes would suppress nitrate assimilation (Boussama et al. 1999), resulting in poor seedling growth. Moreover, Chaudhary and Sharma (2009) found a substantial reduction in chlorophyll content whereas Kilic et al. (2017) observed a decline in the stomatal index and CO2 assimilation as well as reduced leaf area in maize, restraining photosynthesis in maize seedlings, under Cd toxicity. Nonetheless, the effects of Cd toxicity were more prominent in Yuecainuo2 than Yuebaitiannuo7 owing to the better tolerance of Yuebaitiannuo7 to Cd stress conditions (Figs. 1, 2, and 3) with regard to germination rate and growth indexes. Specifically, the shoot fresh weight of Yuecainuo2 significantly decreased higher than Yuebaitiannuo7 under Cd200M0 compared with Cd0M0 (Fig. 2b). Furthermore, Cd100 led to substantial reduction in root weight and length under M0, while it showed no significant effect on shoot dry weight for Yuebaitiannuo7, suggesting that Cd-induced inhibition was more prominent in roots than in shoots (Figs. 2 and 3). Previously, Meng et al. (2016) explained that Cd-induced inhibition in roots may be linked to the excessive production of ethylene in roots rather than shoots under Cd toxicity. In addition, this possibility may be due to the direct contact of roots, triggering more Cd accumulation in roots than in shoots. Therefore, to prevent maize seeds from exhibiting Cd toxicity by directly contacting and absorbing Cd2+, seed pretreatment could be a feasible option to alleviate Cd toxicity in maize.
The application of multiwall carbon nanotubes (MWCNTs) enhanced the seed germination and seedling growth of crops, including maize, peanut, garlic, and soybean, at lower concentrations but reduced early growth at higher concentrations (Lahiani et al. 2013; Rao and Srivastava 2014; Tiwari et al. 2013; Zaytseva 2016). In the present study, the enhancement of MWCNTs under Cd treatment affected both maize varieties differently; for example, application of MWCNTs under Cd treatment significantly increased the seed germination rate of Yuecainuo2; however, the effect remained nonsignificant for Yuebaitiannuo7 (Fig. 1). The application of MWCNTs induced cell wall and seed coat perforation and the expression of water channel proteins (aquaporins), enhancing water absorption and thus promoting seed germination (Lahiani et al. 2013; Tiwari et al. 2013). It has also been reported that MoS2/SH-MWCNT nanocomposites are able to adsorb Cd (II) by ion exchange and electrostatic interactions to alleviate heavy metal stress in plants (Gusain et al. 2019). In this study, the presence of MWCNTs could alleviate Cd toxicity by promoting root and shoot fresh weight (Fig. 2). Specifically, M100 enhanced the root fresh weight of Yuebaitiannuo7 under Cd100 compared with M0 (Fig. 2a). These results are in accordance with a previous study showing that application of MWCNTs could promote the growth of roots and shoots of maize (Rao and Srivastava 2014; Tiwari et al. 2013). Gong et al. (2019) suggested that the application of MWCNTs adsorbed and immobilized Cd as a consequence of their negatively charged surface and functional groups. Moreover, the application of MWCNTs is reported to show a greater promoting effect on shoots than on roots under Cd200 (Figs. 2 and 3). For instance, the presence of MWCNTs significantly increased the shoot fresh weight of Yuecainuo2 under Cd200 but showed no significant effect on root fresh weight (Fig. 2). M100 significantly reduced the root length of Yuebaitiannuo7 than M0 under Cd200 but showed no significant effect on shoot length (Fig. 3). These results might be due to the MWCNT-induced protective effects on roots and shoots under Cd toxicity. The application of MWCNTs may further facilitate the solubility of Cd, thus encouraging vacuolar compartmentation of Cd and limiting the translocation of Cd from roots to shoots in ramie crop (Gong et al. 2019). In addition, roots that directly contact Cd2+ ions might absorb them by ion interactions to diminish the Cd2+ concentration and alleviate Cd toxicity in shoots. Recently, Gong et al. (2019) found that the application of MWCNTs improved the morphological traits of ramie crop under Cd stress conditions. In contrast, Oloumi et al. (2018) indicated that the application of MWCNTs enhanced Cd accumulation in canola, sunflower, and cannabis seedlings. Hence, the ability of MWCNTs to mitigate Cd stress is largely dependent on crop genotype, crop growth stage, and the concentration applied. For example, in present study, application of MWCNTs significantly improved the shoot fresh weight of Yuecainuo2 but showed no effect on Yuebaitiannuo7 under Cd200 (Fig. 2b). There was a more significant alteration in Yuecainuo2 under treatments with regard to germination rate and growth indexes, indicating that Yuecainuo2 is more suitable for MWCNT application (Figs. 1, 2, 3, 4, 5, 6, and 7). Nevertheless, more maize varieties should be to further evaluate the genotypic-specific effects of MWCNT application. Moreover, no significant alteration in the germination rate of Yuebaitiannuo7 at 4 DAS was noted, but the presence of MWCNTs promoted the root fresh weight under Cd100. In addition, the M100 significantly increased the root fresh weight of Yuebaitiannuo7 by 51.76% and M200 by 20.84% under Cd200 and the root length of Yuecainuo2 decreased with increased concentrations of MWCNT under Cd100, indicating the potential risk of the concurrent effects of MWCNTs and Cd (if applied in excess) (Figs. 2 and 3). In short, the pretreatment of maize seeds with MWCNTs when sown in Cd-contaminated soil is a promising strategy to improve the seed germination rate and seedling growth; however, suitable MWCNT concentrations and corresponding crop genotypes should be used to avoid MWCNT stress.
Plants are equipped with an enzymatic antioxidant defense system to counter oxidative toxicity under metal stress conditions (Ashraf et al. 2017a; Ashraf et al. 2017b; Ashraf and Tang 2017). For example, SOD catalyzes the transformation of O2.− to H2O2, and POD and CAT promote the conversion of H2O2 into H2O (Gong et al. 2019). Simultaneously, plants may suffer from oxidative toxicity in vivo because lipid oxidation may lead to metabolic disorders and loss of cellular integrity (Janero 1990). Antioxidant enzyme activities are related to plant resilience under stress conditions (Noreen et al. 2021). Chaudhary and Sharma (2009) reported increased POD and CAT activities under Cd treatment, indicating that the presence of Cd enhanced the antioxidant defense system. In contrast, Gowayed (2017) reported that SOD and CAT activities decreased under high Cd levels, revealing a threshold at which heavy metals promote the antioxidant defense system in plants. In the present study, higher POD activity in shoots and CAT activity in roots and an increased MDA content in shoots under Cd treatment revealed that ROS triggered endogenous antioxidant defenses in growing maize plants. On the other hand, reduced SOD and POD activities in roots and CAT activity in shoots under 200 mg L−1 Cd treatment indicated that excessive Cd toxicity reduced the ROS-quenching efficiency of the antioxidant defense system in growing maize plants (Figs. 4, 5, 6, and 7). The MWCNT treatment under Cd stress increased the SOD activity in roots and shoots (Fig. 4) and decreased the MDA content in shoots (Fig. 7), revealing the potential of MWCNTs to alleviate Cd toxicity by enhancing plant antioxidant ability. Correlation analysis revealed that SOD activity in roots and shoots and POD activity in roots showed a significant and positive correlation with the germination rate, confirming that MWCNTs alleviated oxidative stress with elevated SOD and POD activities to enhance the germination rate and seedling growth under Cd stress (Fig. 4). Moreover, the decreased activity of POD in shoots (Fig. 5) and increased content of MDA in roots (Fig. 7) under MWCNT treatment across Cd treatments indicated that the application of MWCNTs might increase antioxidant stress under combined application of MWCNTs and Cd. Moreover, in present study, the CAT activity in roots was increased at Cd200M100 than Cd0M0 in Yuecainuo2 (Fig. 6), which indicated that MWCNT could promote the antioxidant activities in growing maize plants under Cd toxic conditions. Gong et al. (2019) reported that the application of MWCNTs enhanced the MDA contents, which is in accordance with the present study in which an increased MDA content was observed in roots under MWCNTs across Cd treatments (Fig. 7). Furthermore, there were stronger alterations in Yuecainuo2 in SOD and CAT activities in roots and shoots and POD activity and MDA content in roots, indicating that Yuecainuo2 had a more effective antioxidant defense system which further modulated by MWCNT application (Figs. 4, 5, 6, and 7). Although the application of MWCNTs improved the early growth of maize plants under Cd toxicity, however, further investigation focusing on more MWCNT-induced physiological and molecular changes in maize under Cd exposure is needed.
5 Conclusions
The seed germination rate and early growth of maize were observed to decrease under cadmium toxicity. Multiwall carbon nanotube could alleviate the cadmium toxicity by increasing the seed germination rate and morphological attributes and modulating the antioxidant defense system. Therefore, multiwall carbon nanotube is a potential seed-soaking reagent and seed-coating material which can be used in agriculture production systems to address the issues of heavy metal–polluted soils. Comparatively, Yuecainuo2 is more suitable for multiwall carbon nanotube investigation than Yuebaitiannuo7, with less tolerance to cadmium toxicity and higher alteration under cadmium across multiwall carbon nanotube treatment. Further studies are recommended to determine the suitable dosages, corresponding varieties and necessary transformations to improve the crop performance with multiwall carbon nanotube application under heavy metal polluted soils.
References
Adhikari S et al (2018) Sulfate improves cadmium tolerance by limiting cadmium accumulation, modulation of sulfur metabolism and antioxidant defense system in maize. Environ Exp Bot 153:143–162. https://doi.org/10.1016/j.envexpbot.2018.05.008
Al-Rekaby LS (2018) Influence of multiwalled carbon nanotubes and biostimulators on growth and content of bioactive constituents of karkade (Hibiscus sabdariffa L.). J Bot 2018:1–11. https://doi.org/10.1155/2018/9097363
Anjum SA et al (2015) Cadmium toxicity in maize (Zea mays L.): consequences on antioxidative systems, reactive oxygen species and cadmium accumulation. Environ Sci Pollut Res 22:17022–17030. https://doi.org/10.1007/s11356-015-4882-z
Anjum SA, Tanveer M, Hussain S, ullah E, Wang L, Khan I, Samad RA, Tung SA, Anam M, Shahzad B (2016) Morpho-physiological growth and yield responses of two contrasting maize cultivars to cadmium exposure CLEAN. Soil, Air, Water 44:29–36. https://doi.org/10.1002/clen.201400905
Ashraf U, Tang X (2017) Yield and quality responses, plant metabolism and metal distribution pattern in aromatic rice under lead (Pb) toxicity. Chemosphere 176:141–155. https://doi.org/10.1016/j.chemosphere.2017.02.103
Ashraf U, Hussain S, Anjum SA, Abbas F, Tanveer M, Noor MA, Tang X (2017a) Alterations in growth, oxidative damage, and metal uptake of five aromatic rice cultivars under lead toxicity. Plant Physiol Biochem 115:461–471. https://doi.org/10.1016/j.plaphy.2017.04.019
Ashraf U, Kanu AS, Deng Q, Mo Z, Pan S, Tian H, Tang X (2017b) Lead (Pb) toxicity; physio-biochemical mechanisms, grain yield, quality, and Pb distribution proportions in scented rice. Front Plant Sci 8:259. https://doi.org/10.3389/fpls.2017.00259
Bin HE, Yun ZJ, Shi JB, Jiang GB (2013) Research progress of heavy metal pollution in China: sources, analytical methods, status, and toxicity. Sci Bull 58:6–12. https://doi.org/10.1007/s11434-012-5541-0
Boussama N, Ouariti O, Suzuki A, Ghorbal MH (1999) Cd-stress on nitrogen assimilation. J Plant Physiol 155:310–317. https://doi.org/10.1016/S0176-1617(99)80110-2
Chaudhary S, Sharma YK (2009) Interactive studies of potassium and copper with cadmium on seed germination and early seedling growth in maize (Zea mays L.). J Environ Biol 30:427–432
Cui Y, Wang Q (2018) Physiological responses of maize to elemental sulphur and cadmium stress. Plant Soil Environ 52:523–529. https://doi.org/10.17221/3542-PSE
Dasgupta N, Tiwari DK, Francis E, Torres P, Cendejas LM, Lara-Romero J, Villaseñor-Mora C (2017) Plant responses to nano and micro structured carbon allotropes: water imbibition by maize seeds upon exposure to multiwalled carbon nanotubes and activated carbon advances in Nano Research 5:245-251 https://doi.org/10.12989/anr.2017.5.3.245
Espanany A, Fallah S, Tadayyon A (2015) The effect of halopriming and salicylic acid on the germination of fenugreek (Trigonella foenum-graecum) under different cadmium concentrations. Notulae Scientia Biologicae 7:322–329. https://doi.org/10.15835/nsb739563
Fan X et al (2018) Multiwall carbon nanotubes modulate paraquat toxicity in Arabidopsis thaliana. Environ Pollut 233:633–641. https://doi.org/10.1016/j.envpol.2017.10.116
Ghasempour M, Iranbakhsh A, Ebadi M, Oraghi Ardebili Z (2019) Multi-walled carbon nanotubes improved growth, anatomy, physiology, secondary metabolism, and callus performance in Catharanthus roseus: an in vitro study. 3 Biotech 9:404. https://doi.org/10.1007/s13205-019-1934-y
Gong XM et al (2019) Roles of multiwall carbon nanotubes in phytoremediation: cadmium uptake and oxidative burst in Boehmeria nivea (L.) Gaudich. Environ Sci-Nano 6:851–862. https://doi.org/10.1039/c8en00723c
Gowayed S (2017) Impact of zinc oxide nanoparticles on germination and antioxidant system of maize (Zea mays L.) seedling under cadmium stress. J Plant Prod Sci 6:1–11. https://doi.org/10.21608/jpps.2017.7389
Gozubenli H (2010) Seed vigor of maize grown on the contaminated soils by cadmium. Asian J Plant Sci 9:168–171. https://doi.org/10.3923/ajps.2010.168.171
Gusain R, Kumar N, Fosso-Kankeu E, Ray SS (2019) Efficient removal of Pb(II) and Cd(II) from industrial mine water by a hierarchical MoS2/SH-MWCNT nanocomposite ACS. Omega 4:13922–13935. https://doi.org/10.1021/acsomega.9b01603
Hemachandra CK, Pathiratne A (2015) Assessing toxicity of copper, cadmium and chromium levels relevant to discharge limits of industrial effluents into inland surface waters using common onion, Allium cepa bioassay. Bull Environ Contam Toxicol 94:199–203. https://doi.org/10.1007/s00128-014-1373-8
Huamain C, Chunrong Z, Cong T, Yongguan Z (1999) Heavy metal pollution in soils in China: status and countermeasures. AMBIO 28:130–134
Janero DR (1990) Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic Biol Med 9:515–540. https://doi.org/10.1016/0891-5849(90)90131-2
Joshi A, Kaur S, Singh P, Dharamvir K, Nayyar H, Verma G (2018) Tracking multi-walled carbon nanotubes inside oat (Avena sativa L.) plants and assessing their effect on growth, yield, and mammalian (human) cell viability. Appl Nanosci 8:1399–1414. https://doi.org/10.1007/s13204-018-0801-1
Kanu AS, Ashraf U, Mo ZW, Sabir SUR, Baggie I, Charley CS, Tang XR (2019) Calcium amendment improved the performance of fragrant rice and reduced metal uptake under cadmium toxicity. Environ Sci Pollut Res 26:24748–24757. https://doi.org/10.1007/s11356-019-05779-7
Kilic S, Karaboyaci M, Sencan A, Kilic M (2017) Ecotoxicological responses of morphological and physiological parameters of cadmium-stressed maize seeds. Bangladesh J Bot 46:211–216
L.M. S, H.C. D, Gómez M, M.C. RP, L.A. DR (2001) Cadmium-induced changes in the growth and oxidative metabolism of pea plants. J Exp Bot 52:364. https://doi.org/10.1093/jexbot/52.364.2115
Lahiani MH, Dervishi E, Chen J, Nima Z, Gaume A, Biris AS, Khodakovskaya MV (2013) Impact of carbon nanotube exposure to seeds of valuable crops ACS. Appl Mater Interfaces 5:7965–7973. https://doi.org/10.1021/am402052x
Li S, Jiang H, Wang J, Wang Y, Mo Z (2019) Responses of plant growth, physiological, gas exchange parameters of super and non-super rice to rhizosphere temperature at the tillering stage. Sci Rep 9:10618. https://doi.org/10.1038/s41598-019-47031-9
Lin R, Wang X, Luo Y, Du W, Guo H, Yin D (2007) Effects of soil cadmium on growth, oxidative stress and antioxidant system in wheat seedlings (Triticum aestivum L.). Chemosphere 69:89–98. https://doi.org/10.1016/j.chemosphere.2007.04.041
Liu WS, Zhou C, Guo PJ, Shi-You LI, Yang YU (2010) Influence of heavy metal cadmium on maize seed germination and growth of embryo. Hubei Agric Sci 49:842–844. https://doi.org/10.14088/j.cnki.issn0439-8114.2010.04.026
Lustriane C, Dwivany FM, Suendo V, Reza M (2018) Effect of chitosan and chitosan-nanoparticles on post harvest quality of banana fruits. J Plant Biotechnol 45:36–44. https://doi.org/10.5010/JPB.2018.45.1.036
Meng G, Tang T, Jing Z, Ying Z, Chen Y, Yang Y, Li Z (2016) Analysis on cadmium tolerance of different maize varieties during seed germination stage. Mol Plant Breed 14:3166–3171. https://doi.org/10.13271/j.mpb.014.003166
Meng QF, Hou P, Wu L, Chen XP, Cui ZL, Zhang FS (2013) Understanding production potentials and yield gaps in intensive maize production in China. Field Crop Res 143:91–97. https://doi.org/10.1016/j.fcr.2012.09.023
Muhammad S, Iqbal MZ, Mohammad A (2010) Effect of lead and cadmium on germination and seedling growth of Leucaena leucocephala. J Appl Sci Environ Manag 12:61–66. https://doi.org/10.4314/jasem.v12i3.55497
Nabaei M, Amooaghaie R (2019) Interactive effect of melatonin and sodium nitroprusside on seed germination and seedling growth of Catharanthus roseus under cadmium stress. Russ J Plant Physiol 66:128–139. https://doi.org/10.1134/S1021443719010126
Noreen S et al (2021) Foliar fertigation of ascorbic acid and zinc improves growth, antioxidant enzyme activity and harvest index in barley (Hordeum vulgare L.) grown under salt stress. Plant Physiol Biochem 158:244–254. https://doi.org/10.1016/j.plaphy.2020.11.007
Oloumi H, Mousavi EA, Daneshmand F (2017) Changes in Cd/Pb accumulation and growth and physiological indices on Sorghum bicolor sp. seedlings exposed to carbon nano tubes. Thai J Agric Sci 50:96–107
Oloumi H, Mousavi EA, Nejad RM (2018) Multi-wall carbon nanotubes effects on plant seedlings growth and cadmium/lead uptake in vitro Russian. J Plant Physiol 65:260–268. https://doi.org/10.1134/S102144371802019x
Qu DY, Gu WR, Li LJ, Jing L, Li CF, Shi W, University NA (2018) Regulation of chitosan on the ascorbate-glutathione cycle in Zea mays seedling leaves under cadmium stress. Plant Sci J 36:291–299. https://doi.org/10.11913/PSJ.2095-0837.2018.20291
Ranum P, Pena-Rosas JP, Garcia-Casal MN (2014) Global maize production, utilization, and consumption Annals of the New York Academy of Sciences 1312:105-112 https://doi.org/10.1111/nyas.12396
Rao DP, Srivastava A (2014) Enhancement of seed germination and plant growth of wheat, maize, peanut and garlic using multiwalled carbon nanotubes. Eur Chem Bull 3:502–504. https://doi.org/10.17628/ecb.2014.3.502-504
Seddighinia FS, Iranbakhsh A, Ardebili ZO, Satari TN, Soleimanpour S (2020) Seed priming with cold plasma and multi-walled carbon nanotubes modified growth, tissue differentiation, anatomy, and yield in bitter melon (Momordica charantia). J Plant Growth Regul 39:87–98. https://doi.org/10.1007/s00344-019-09965-2
Servin A et al (2015) A review of the use of engineered nanomaterials to suppress plant disease and enhance crop yield. J Nanopart Res 17:92. https://doi.org/10.1007/s11051-015-2907-7
Shen X, Huang R, Guo Z, Junjie XU, Guo S, Zhu J, Wang Z (2018) Effects of cadmium stress on seedling growth of Carya illinoinensis. J Nucl Agric Sci 32:1627–1638. https://doi.org/10.11869/j.issn.100-8551.2018.08.1627
Tiwari DK, Dasgupta-Schubert N, Villaseñor Cendejas LM, Villegas J, Carreto Montoya L, Borjas García SE (2013) Interfacing carbon nanotubes (CNT) with plants: enhancement of growth, water and ionic nutrient uptake in maize (Zea mays) and implications for nanoagriculture. Appl Nanosci 4:577–591. https://doi.org/10.1007/s13204-013-0236-7
Wagi S, Ahmed A (2019) Green production of AgNPs and their phytostimulatory impact. Asia Pac J Risk Insur 8:885–894. https://doi.org/10.1515/gps-2019-0059
Yang K-Y et al (2018) Remodeling of root morphology by CuO and ZnO nanoparticles: effects on drought tolerance for plants colonized by a beneficial pseudomonad. Botanique 96:175–186. https://doi.org/10.1139/cjb-2017-0124
Yang Z, Liu S, Zheng D, Feng S (2006) Effects of cadium, zinc and lead on soil enzyme activities. J Environ Sci 018:1135–1141. https://doi.org/10.1016/S1001-0742(06)60051-X
Zaytseva O (2016) Differential impact of multi-walled carbon nanotubes on germination and seedling development of Glycine max, Phaseolus vulgaris and Zea mays. Eur Chem Bull 5:202–210. https://doi.org/10.17628/ecb.2016.5.202-210
Zhai G, Gutowski SM, Walters KS, Yan B, Schnoor JL (2015) Charge, size, and cellular selectivity for multiwall carbon nanotubes by maize and soybean. Environ Sci Technol 49:7380–7390. https://doi.org/10.1021/acs.est.5b01145
Zhao G et al (2019) Nitrate reductase-dependent nitric oxide is crucial for multi-walled carbon nanotube-induced plant tolerance against salinity. Nanoscale 11:10511–10523. https://doi.org/10.1039/c8nr10514f
Funding
This research was funded by the National Key R&D Program Project, 2018YFD0200706 and 2018YFD0100106, and the Project of the Guangdong Province Science and Technology Program, 2018B020202008 and Guangdong Basic and Applied Basic Research Foundation, 2019A1515010305 and Guangdong Agricultural Seed Industry Common Key Technical Innovation Team, 2020KJ106 and project of rural revitalization in Guangdong Province, Yue Nong Nong Han [2020]100 and the Special Financial Fund of Foshan–Guangdong Agricultural Science and Technology Demonstration City Project in 2021.
Author information
Authors and Affiliations
Contributions
Conceptualization, W.L.; investigation, X.Z.; formal analysis, J.C., W. Y., H.X. J.L., G.L., and W.L.; writing–original draft, J.C., W. Y., H.X. J.L., G.L., and W.L.; writing–review and editing, Z.M., W.L., and U.A. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, J., Zeng, X., Yang, W. et al. Seed Priming with Multiwall Carbon Nanotubes (MWCNTs) Modulates Seed Germination and Early Growth of Maize Under Cadmium (Cd) Toxicity. J Soil Sci Plant Nutr 21, 1793–1805 (2021). https://doi.org/10.1007/s42729-021-00480-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-021-00480-6