Abstract
This study aimed to evaluate whether silicon application in the nutrient solution is efficient in relieving stress caused by nutrient deficiency (N, K, Ca, Mg, and S) in the forages BRS Zuri (Panicum maximum) and BRS RB331 Ipyporã (Brachiaria ruziziensis × Brachiaria brizantha). Two experiments were carried out in a greenhouse with both forages, with plants grown in a nutrient solution in pots filled with washed sand. Treatments were arranged in a completely randomized block design, with five replications and consisted of complete solution (control) and individual omission of nitrogen, potassium, calcium, magnesium, and sulfur in the absence (−Si) and presence of silicon (+Si) at a concentration of 2 mmol L−1 applied via nutrient solution as stabilized potassium sodium silicate. The dry mass of the shoot, root, and clump residual material, accumulation and use efficiency of nutrients, and the description of symptoms of nutritional deficiency were evaluated. Silicon addition in the nutrient solution favored shoot dry matter production and root dry matter production of both forages, in treatments with K, Ca, and Mg omission; the same effect was not observed for N and S. We conclude that the application of silicon attenuates the effects of potassium, calcium, and magnesium deficiency in forages BRS Zuri and BRS RB331 Ipyporã, as it increases the accumulated shoot dry mass, due to the greater efficiency in the use of these nutrients. The beneficial effect of silicon was not important in forage plants with nitrogen and sulfur deficiency.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Cultivation of grasses of the genera Panicum and Brachiaria has stood out in livestock for their ability to adapt to various environmental and management conditions (Monteiro et al. 1995) due to the high production capacity of good quality leaf dry matter (Kissmann 1997) if nutrient availability is adequate.
Problems of nutritional deficiency of forage cultivations in tropical regions, especially macronutrients, occur with high frequency due to the low soil fertility (Prado et al. 2011) and restricted use of fertilizers, harming plant growth.
Silicon is used to reduce different stresses in plants such as water deficit (Camargo et al. 2019) and arsenic toxicity (Maghsoudi et al. 2019) and also, increase the availability of nutrients like P (Wang et al. 2019). Although studies with forage grasses are scarce in relation to Si, there are beneficial effects of the element on Brachiaria brizantha plants (Sarto et al. 2016; Moro et al. 2018). In the literature, there are only studies restricted to potassium in sorghum (Chen et al. 2016) and soybean (Miao et al. 2010), nitrogen in rice (Deus et al. 2019), magnesium in corn (Hosseini et al. 2019), sulfur in barley (Maillard et al. 2018), and manganese in sorghum (de Oliveira et al. 2019). However, there are some reports that Si can favor plant growth without necessarily being under stress (Zia et al. 2017). Therefore, it is the beneficial element that reduces the deleterious effects of nutrient deficiency if this deficiency is not very severe.
The beneficial effects of Si may be related to the replacement of a portion of carbon from organic structures (Cooke and Leishman 2012) and promotion of lignin and carbohydrate crosslinking (Inanaga et al. 1995), collaborating with leaf cell wall structuring, the organ that concentrates this element in grasses (Neu et al. 2017).
The energy cost for the plant to incorporate Si into the organic chain is relatively low (Neu et al. 2017; Raven 1983; Schaller et al. 2012). In addition, Si may increase chlorophyll content (Xie et al. 2014) and activate enzymes that are important in plant metabolism (Liu et al. 2009). The sum of these effects of Si on the plant could benefit the nutritional metabolism, enhancing nutrient use efficiency for dry matter production, but it needs to be researched. The possibility of these benefits of Si increases, especially in plants accumulating this element, such as forage grasses (Melo et al. 2007) already demonstrated in previous work with Brachiaria brizantha (Melo et al. 2010) which are widely cultivated in the world. Also, Si is considered a nutrient for animals given its importance in bone formation and connective tissues (Carlisle 1972), and the ingestion of Si-enriched forage can contribute to meeting their nutritional requirements.
Therefore, it is hypothesized that Si could alleviate the effects of N, K, Ca, Mg, and S deficiency due to its contribution to nutrient use efficiency, favoring the dry matter production of two forage grown in a nutrient solution. Thus, this research may add another beneficial effect to Si, still little known, if it can alleviate the nutritional deficiency of plants accumulating the element. The practical implication would result from the possibility of adding Si to fertilizers to improve the agronomic efficiency by reducing pasture degradation, which is common in various regions of the world.
Thus, this study aimed to evaluate the effects of omitting N, K, Ca, Mg, and S from the nutrient solution in the absence and presence of silicon on their absorption and use efficiency and dry matter production of forages BRS Zuri (Panicum maximum) and BRS RB331 Ipyporã (Brachiaria ruziziensis × Brachiaria brizantha).
2 Material and Methods
2.1 Site Characteristics and Plant Material
Two experiments were carried out from July to November 2018 in a greenhouse at the São Paulo State University (UNESP/FCAV), Campus of Jaboticabal, Brazil. The forages used were BRS Zuri (Panicum maximum) and hybrid BRS RB331 Ipyporã (Brachiaria ruziziensis × Brachiaria brizantha) in the first and second experiments, respectively. Seeds were sown in trays filled with vermiculite and maintained for 20 days. After this period, seedlings were washed in distilled water, selected, and transplanted to plastic pots (2 dm3) containing sand, placing one plant per pot. This sand had a medium texture and was previously decontaminated with water, 0.1 mol L−1 hydrochloric acid solution, and deionized water.
2.2 Experimental Design and Treatments
The experimental design was a completely randomized design, with twelve treatments and five replications. Treatments consisted of complete solution (control) and individual omission of nitrogen (−N), potassium (−K), calcium (−Ca), magnesium (−Mg), and sulfur (−S), in the absence (−Si) and presence of silicon (+Si) at a concentration of 2 mmol L−1 applied via nutrient solution. Silicon source was sorbitol-stabilized potassium sodium silicate (107 g L−1 of Si, 34.7 g L−1 of K2O, and pH 11.8). Potassium was balanced in all treatments using solution potassium chloride at 1.0 mol L−1.
2.3 Growth Conditions
The complete nutrient solution was that of Hoagland and Arnon (1950), except for Fe and Mn. Iron and manganese were applied twice the concentration, as indicated by Cavalcante et al. (2016). The nutrient solution was prepared with distilled water and then deionized (due to excess Ca in the water). Nutrient solution concentration during the first week of cultivation was maintained with 15% ionic strength and then increased by 10% every 3 days until reaching 65%, which was maintained until the end of the experimental period. The pH value of the solution was maintained at 5.0 ± 0.2 using HCl or NaOH solution, both at 1.0 mol L−1. The nutrient solution was applied in a container placed below the pots.
A standardization cutting was performed at 18 days after transplanting (DAP) at 10 cm of height in all plants of both forages to standardize plant height and induce tillering.
Three plant collections were performed during the experimental period. The plants were cut at 15 cm from the pot surface, to simulate grazing and induce the symptoms of each omitted nutrient deficiency, this being the defined moment to perform the collection.
The first collection was performed at 35 DAP for all treatments. On the same date, −N treatments were withdrawn (without and with Si) being evaluated in these treatments the root dry mass and residual material of the clump, since they presented symptoms of the omitted nutrients deficiency. The second collection of the shoot was carried out at 63 DAP for the control treatments, −K, −Ca, −Mg, and −S in the two forages. At this time, the treatments −S and −K (without and with Si) of BRS RB331 Ipyporã and −S (without and with Si) of the BRS Zuri forage showed the symptoms of the omitted nutrients deficiency; therefore, these treatments were removed and evaluated, in addition to the shoot dry mass and also the root dry mass and the clump residual dry mass. At 91 DAP, symptoms of nutrient-omitted deficiency were observed in the treatments −Ca and −Mg (without and with Si) of the BRS RB331 Ipyporã and −K, −Ca, and −Mg (without and with Si) of the BRS Zuri forage; therefore, the last collection of the shoot and root and residual material of the clump dry mass of these treatments and complete treatments (without and with Si) were performed. After collection, the plants were divided into shoot, root, and residual material of clump dry mass. The accumulated shoot dry matter was obtained from the sum of the three collections.
2.4 Performed Analyses
2.4.1 Dry Matter Production
The collected vegetative material was washed in running water, detergent solution (0.1%), hydrochloric acid solution (0.3%), and deionized water twice. Subsequently, the plant material was dried in a forced air ventilation oven (65 ± 5 °C) until reaching a constant weight, thus obtaining the dry matter of each part of the plant.
2.4.2 Chemical Analysis of Samples
Silicon (Si) content was determined in the shoot, root, and clump residual material, according to the methodology described by Korndörfer (2004). The total nitrogen (N), potassium (K), calcium (Ca), magnesium (Mg), and sulfur (S) contents were determined from acid digestion, according to the method described by Bataglia et al. (1983). From the nutrient content data (Si and dry matter), the accumulation of each element was calculated, and then, the use efficiency was calculated by the formula: (shoot dry matter)2/nutrient accumulation in the shoot, as indicated by Siddiqi and Glass (1981).
2.4.3 Visual Analysis of Symptoms
Plants were monitored for nutritional deficiency and recorded with photos taken during the onset of visual symptoms.
2.5 Statistical Analysis
The data were submitted to a bidirectional analysis of variance (ANOVA) by the F test (p ≤ 0.05) after verifying the homogeneity of the variances (Shapiro-Wilk W test). The means of the procedures with omission of nutrients (without and with Si) were compared with the complete treatment (without and with Si) by the Tukey test, with a 5% probability, using the Sisvar® statistical program (Ferreira 2011).
3 Results
3.1 Experiment 1
3.1.1 Dry Matter
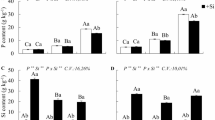
In forage plants BRS Zuri (Panicum maximum) with omission of N, K, Ca, Mg, and S in the nutrient solution, a decrease in shoot, root, and residual material of clump dry mass was observed compared with complete treatments, regardless of the Si addition (Fig. 1). It is noted that the order of limitation of forage growth occurred with the omission of N > S > Ca > K > Mg in the nutrient solution with Si, corresponding to a decrease in the dry mass production of the accumulated shoot in relation to the complete treatment equal to 99, 87, 65, 54, and 35%, respectively. Without the application Si, the order of limitation was N > S > Ca > K > Mg, with values for the reduction of accumulated dry mass of 99, 86, 66, 61, and 24%, respectively.
Shoot dry matter of the forage BRS Zuri (Panicum maximum) in the first (a), second (b), and third (c) collection and accumulated shoot dry matter (d), root dry matter (e), and clump residual dry matter (f) submitted to the treatments (complete and omitted nutrients) in the absence (-Si) and presence of silicon (+Si). Lowercase letters show differences in relation omitted and complete treatments by the F test. Uppercase letters show differences between treatments with absence (−Si) and presence of silicon (+Si) by the F test. Bars represent the standard error of the mean, n = 5
It was observed that in plants with N and S deficiency, the addition of Si in nutrient solution did not affected shoot, root, and residual material of clump dry mass (Fig. 1 a, b, d, e, and f).
The addition of Si in plants without deficiency (complete), compared with plants without Si addition, provided an increase in shoot dry mass of 23, 36, 21, and 30%, for the first, second, third collection, and accumulated shoots, respectively. The addition of Si in K deficient plants compared with Si absence provided an increase in the shoot dry mass of 51, 39, and 39% for the second, third collection, and accumulated shoots, respectively. The addition of Si in the deficient of Ca plants compared with Si absence provided an increase in the shoot dry mass of 25, 38, and 33%, for the first, third collection, and accumulated shoots, respectively. In relation to Mg, the addition of Si in plants deficient compared with Si absence caused an increase in the shoot dry mass of 20, 17, and 18%, for second, third, and accumulated shoots, respectively.
3.1.2 Nutrient Accumulation
Plants grown with omission of N, K, Ca, Mg, and S had a decrease in the accumulation of these nutrients in shoot, root, and residual material of clump dry matter in relation to the complete treatment, regardless of Si addition in the nutrient solution (Table 1).
The addition of Si provided an increase in the accumulation of this element in the shoot, root, and residual material of clump dry mass in all treatments in relation to the complete treatment (Table 2). Regarding the shoot dry mass in the first sampling, the addition of Si provided an increase in Si accumulation of 57, 48, 31, 36, 21, and 49%, in the complete, −N, −K, −Ca, −Mg, and −S treatments, respectively. In the second collection, increases of 66, 64, 55, 63, and 84%, were observed for the complete, −K, −Ca, −Mg, and −S treatments, respectively. In the third collection, increases of 69, 77, 84, and 66% were observed for −K, −Ca, and − Mg, treatments, respectively.
3.1.3 Use Efficiency
Nutrient omission decreased nitrogen use efficiency in plants with N omission, with and without Si addition in the first collection, reduction that was 50% (Table 3). The efficiency of using S with the omission of S, with the addition of Si for the first and second collection, reached a reduction of only 24 and 30%, respectively, but without the addition of Si in the second collection, this reduction was high, reaching 49%.
In the cultivation of plants without omission of nutrients, note that an addition of nutrient solution increased the efficiency of use of N, Mg, and S in the first collection by 37, 26, and 30%, respectively and in the second collection by 18, 20, and 30%, for N, K, and S, respectively and N with a value of 29% in the third collection.
In relation to treatments with omission of nutrients, in the first collection, the application of Si compared with the absence promoted an increase in the efficiency of use of the omitted nutrients in 40, 28, 24, 7.8, and 25%, for the nutrients N, K, Ca, Mg, and S, respectively. In the second collection, the addition of Si in plants deficient in relation to its absence caused an increase in the efficiency of use of the omitted nutrients by 41, 33, 16, and 49%, for the nutrients K, Ca, Mg, and S, respectively. In the third collection, the application of Si in deficient plants compared with its absence caused an increase of 27, 57, and 52%, in treatments K, Ca, and Mg, respectively.
3.2 Experiment 2
3.2.1 Dry Matter
The cultivation of the forage BRS RB331 Ipyporã (Brachiaria ruziziensis × Brachiaria brizantha) with omission of N, K, Ca, Mg, and S in the nutrient solution showed a decreased production of shoot dry matter in the different collections, accumulated shoot dry matter, root, and clump residual material, regardless of Si addition, in relation to the complete treatment (Fig. 2). It is noted that the order of limitation of forage growth occurred with the omission of N > S > K > Ca > Mg in the nutrient solution with Si, corresponding to a decrease in the dry mass production of the accumulated shoot in relation to the complete treatment equal to 99, 90, 81, 39, and 37%, respectively. Without the application, the order of limitation was N > S > K > Ca > Mg, with values for the reduction of accumulated dry mass of 99, 87, 81, 27, and 22%, respectively.
Shoot dry matter of the forage BRS RB331 Ipyporã (Brachiaria ruziziensis × Brachiaria brizantha) in the first (a), second (b), and third (c) collection and accumulated shoot dry matter (d), root dry matter (e), and clump residual dry matter (f) submitted to the treatments (complete and omitted nutrients) in the absence (−Si) and presence of silicon (+Si). Lowercase letters show differences in relation omitted and complete treatments by the F test. Uppercase letters show differences between treatments with absence (−Si) and presence of silicon (+Si) by the F test. Bars represent the standard error of the mean, n = 5
Silicon application did not influence the production of shoot dry matter, accumulated dry matter, root, and clump residual material in treatments with N and S omission (Fig. 2a, b, d, e, and f).
However, the application of Si in plants without deficiency (complete) in relation to its absence caused an increase in the dry mass of the aerial part from 32, 25, 34, and 30% to first, second, third collection, and accumulated dry mass, respectively. In K deficient plants, the addition of Si in relation to its absence promoted an increase in the dry mass of the aerial part of Si by 31, 30, and 31%, for the first, second collection, and accumulated dry mass, respectively. The addition of Si in plants with Ca deficiency in relation to its presence allowed an increase in the dry mass of the aerial part of 21, 14, 19, and 17%, for the first, third collection, and accumulated dry mass, respectively. The addition of Si in plants with Mg deficiency in relation to their absence promotes an increase in dry mass of the aerial part of 16, 13, and 14%, for the second, third collection, and accumulated dry mass, respectively.
3.2.2 Nutrient Accumulation
Omission of N, K, Ca, Mg, and S in forage plants led to a decreased accumulation of these nutrients in the shoot dry matter in the first, second, and third collections, accumulated shoots, roots, and clump residual material (Table 4) when compared with the complete treatment, regardless of the addition of Si in the nutrient solution. Note that deficient plants cause reduction to the shoot dry matter were influenced by the application of Si first and third collection with an increase of 71 and 80%, respectively. This effect was observed in relation to K in the second collection with an increase of 41%.
Si application reflected an increase in the accumulation of this element in the shoot dry matter, root, and clump residual material (Table 5). In the first collection of shoot dry matter, an addition of Si provided an increase in the accumulation of Si by 67, 70, 76, 51, 43, and 46%, in the complete treatments, −N, −K, −Ca, −Mg, and −S, respectively. In the second collection, we found increments of 73, 64, 58, 84, and 69% for the complete treatments, −K, −Ca, −Mg, and −S. In the third collection, the Si increments were 75, 55, and 74% in the complete treatments, −Ca and − Mg, respectively.
3.2.3 Use Efficiency
Only in Mg omission, a decrease in use and efficiency of this macronutrient was observed in the plant, when compared with the complete treatment without and with Si, a decrease of 41 and 52%, respectively, result observed only for the first collection (Table 6).
Addition of Si in the nutrient solution increased the efficiency of use of all nutrients in the first collection of the complete treatment, providing increases of 28, 35, 69, 26, and 30% for the elements N, K, Ca, Mg, and S, respectively. In the second collection, increases were observed only in N and S, with an increase of 22 and 29%, respectively. In the third collection, increases were observed in N, K, and S treatments, corresponding to 30, 43, and 37%, respectively, showed in increase in the efficiency of use of these nutrients under its deficiency.
Addition of Si nutrient solution increased the efficiency of use of N, K, Ca, Mg, and S in the aerial part of the plants in the treatments where these elements were omitted, a fact that was observed in all collections. In the first collection, addition of Si compared with treatments without Si promoted an increase in the use of omitted nutrients by 18, 44, 81, 15, and 23%, for nutrients N, K, Ca, Mg, and S, respectively. In the second collection, the addition of Si in the plants deficient in relation to its absence caused an increase in the efficiency of use of the omitted nutrients by 17, 12, and 12%, for K, Ca, and Mg, respectively. In the third collection, the application of Si in plants deficient in relation to its absence caused an increase of 52 and 28% in the treatments with omission Ca and Mg, respectively.
4 Discussion
4.1 Complete Nutrient Solution
Silicon (Si) addition in the complete nutrient solution favored shoot dry matter production of all collections and root dry matter production of both forages. These effects are possibly due to the fact that Si contributes to increasing nutrient absorption, mainly K in the first collection (Table 1); N, K, Ca, Mg, and S in the second collection; K and Mg in the third collection for the forage BRS Zuri; and N, K, Ca, Mg, and S in all collections for BRS RB331 Ipyporã (Table 4). The higher absorption of these nutrients led to an increase in N use efficiency in all collections in treatments with Mg and S omission in the first collection and K and S in the second collection for the cultivar BRS Zuri (Table 3), and N, K, Ca, Mg, and S in the first collection, N and S in the second collection, and N, K, and S in the third collection for forage BRS RB331 Ipyporã (Table 6), favoring their dry matter production.
The beneficial effects of Si observed in cultivated plants even without the imposition of nutritional disorders can be attributed to the energy gain that the element can provide, especially in plants that accumulate large amounts of Si, such as Poaceae group (Mitani and Ma 2005). This was because Si can replace part of the carbon in the formation of some low energy cost structural compounds (Cooke and Leishman 2012), especially in the cell wall (Melo et al. 2010), being in grasses the highest concentration in leaf cell walls (Neu et al. 2017), which may enhance the efficiency of nutrient use, favoring biomass production.
Thus, Si addition benefits forage growth even in plants without nutritional deficiency. Other authors have reported similar results on rice cultivation (Zia et al. 2017). Thus, reports that reinforce the importance of Si only under stress (Chen et al. 2016) should be reviewed, especially in hyperaccumulator plants of this element, such as forage grasses.
4.2 Nitrogen (N)
Nitrogen deficiency rapidly manifested, this nutrient being the one that most limited the development, in both forages regardless of the presence or absence of Si seen in Fig. 3. The N omitted in the nutrient solution impaired dry matter production due to a low N absorption by plants, inducing the characteristic symptoms of this nutrient, as reported by several authors in forages (Avalhaes et al. 2009; Prado et al. 2011).
Forage BRS Zuri (Panicum maximum) grown in complete nutrient solution with Si (+Si) and N omission in the presence of Si (a), complete nutrient solution without Si (−Si) and N omission (−N) in the absence of Si (b) and forage BRS RB331 Ipyporã (Brachiaria ruziziensis × Brachiaria brizantha) grown in complete nutrient solution with Si and N omission in the presence of Si (c), and complete nutrient solution without Si and N omission in the absence of Si (d)
The deficiency symptoms were characterized by an intense chlorosis, initially in the leaves of the lower part and less accentuated in the upper part, which happened due to the high phloem mobility of this nutrient (Prado 2008), and with the evolution of the symptoms, the chlorotic regions turn necrotic. The loss of pigmentation in leaves under N deficiency is because the redistribution process due to proteolysis of the enzyme Rubisco and other chloroplast proteins, releasing the N present in these compounds to meet the demand for new organs (Feller et al. 2008).
Add to that, the fact that the concentration of N in the leaf tissue directly interferes in the pigments and consequently in the photosynthetic activity because it is a structural nutrient of the chlorophyll molecule. This decrease reflects in a reduction in the photosynthetic rate, in respiration, in the activity of enzymes associated with photosynthesis (Zhang et al. 2013), and in the water use efficiency (Akram et al. 2011). All these effects caused a reduction in the plant dry mass, due to the reduction in the amount of soluble proteins, limiting the formation of new tissues in the plant. The beneficial effect of Si in attenuating the damage caused by N deficiency has been described in rice plants (Deus et al. 2019), demonstrating that this beneficial element has the potential to mitigate nutritional deficiency depending on the level of severity of stress.
Therefore, Si addition to the nutrient solution in nitrogen-deficient forage plants did not increase N accumulation (Tables 1 and 4), although it increased N use efficiency (Tables 3 and 6), which was not sufficient to influence growth and attenuates the severe symptoms of this macronutrient deficiency (Fig. 3).
It is evident that either Si was not efficient in mitigating very severe N and S deficiencies, suggesting that in new research, it is important to supply 20 to 30% of the indication by Hoagland and Arnon’s solution (1950) to induce moderate use.
4.3 Potassium (K)
Potassium omission decreased biomass production (Figs. 1 and 2), being the third macronutrient that most limited the growth of both forages, impairing their physiological function, since K is strongly linked to regulation in stomatal opening, loading of photoassimilates in the phloem, maintenance of the membrane potential, in the synthesis of nucleic acids, carbohydrates and proteins and, mainly, in the activation of more than 60 enzymes (Prado 2008; Mak et al. 2014).
K deficiency caused marginal chlorosis in the older leaves, followed by necrosis, demonstrating the high phloem mobility that this nutrient presents (Prado 2008). Marginal chlorosis due to K deficiency is due to a drop in protein synthesis, causing the accumulation of putrescine polyamine, which in high concentrations accentuates cellular imbalance and marginal necrosis of plant tissues (Pathak et al. 2014).
However, the addition of Si in the culture solution reduced the damage caused by this nutritional deficiency. This beneficial effect of Si on biomass production is due to increased nutrient uptake, especially Ca and Mg in the second collection and Mg in the third collection for the forage BRS Zuri, as well as N and Mg in the first collection and N, K, Ca, and Mg in the second collection for BRS RB331 Ipyporã. Moreover, plants with K omission that received Si showed an increase in nutrient use efficiency as a function of forage and collection. An increase in K use efficiency was observed in all collections of both forages and also other nutrients, such as N and S for BRS Zuri (Table 3) and N for BRS RB331 Ipyporã (Table 6) probably due to the fact that the role of K+ in the osmotic function can be partially replaced by other cations, such as Mg2+, Na+, or Ca2+ (Winkler and Zotz 2010), which contributed to a higher dry matter production of these forages. Similar effects in mitigating the effects of K deficiency have also been reported in soybean (Miao et al. 2010) and sorghum plants (Chen et al. 2016).
The Si supply increased the efficiency of the use of K probably due to the role that the beneficial element plays in improving the metabolism of plants, favoring an increase in chlorophyll levels and photosynthetic rates (Chen et al. 2016), mainly by activating enzymes involved in secondary defense metabolism, such as superoxide dismutase, catalase, and ascorbate peroxidase, which reduce the accumulation of reactive oxygen species and inhibit the degradation of photosynthetic pigments (de Oliveira et al. 2019; Liu et al. 2009). Adds up, to this is the fact that Si increases the efficient use of water (Gao et al. 2005) which is impaired in K deficient plants (Prado 2008), benefiting the plant’s metabolism. Therefore, relief of K deficiency with the use of Si was evidenced visually due to a decrease in severity of deficiency symptoms of this macronutrient (Fig. 4).
Forage BRS Zuri (Panicum maximum) grown in complete nutrient solution with Si (+Si) and K omission in the presence of Si (a), complete nutrient solution without Si (−Si) and K omission (−K) in the absence of Si (b), forage BRS RB331 Ipyporã (Brachiaria ruziziensis × Brachiaria brizantha) grown in complete nutrient solution with Si and K omission in the presence of Si (c), and complete nutrient solution without Si and K omission in the absence of Si (d)
4.4 Calcium (Ca)
Calcium deficiency impaired dry matter production in both forages, regardless of the Si in the nutrient solution, being the fourth macronutrient that most limited the development of forages. Ca deficiency (Fig. 5) showed very similar symptoms in both varieties, especially young leaves tearing, a fact also reported by Avalhaes et al. (2009), who evaluated macronutrient omission in elephant grass (Pennisetum purpureum Shum cv. Mott) grown in the nutrient solution.
Forage BRS Zuri (Panicum maximum) grown in complete nutrient solution with Si (+Si) and Ca omission in the presence of Si (a), complete nutrient solution without Si (−Si) and Ca omission (−Ca) in the absence of Si (b), forage BRS RB331 Ipyporã (Brachiaria ruziziensis × Brachiaria brizantha) grown in complete nutrient solution with Si and Ca omission in the presence of Si (c), and complete nutrient solution without Si and Ca omission in the absence of Si (d)
This nutritional disorder occurred because calcium forms calcium pectates in the cell wall, responsible for cell structure, one of the damages caused by the deficiency of this macronutrient is the less expansion and lack of firmness of the cell wall, which is governed by the bonds of Ca+2 to the pectates (Hepler and Winship 2010; Malavolta et al. 1997). Si supply in the nutrient solution in plants under Ca deficiency decreased the effects of stress caused by Ca suppression, reflecting an increase of shoot dry matter production of both forages (Figs. 1 and 2) and root dry matter production in the forage BRS Zuri (Fig. 1). In addition, Ca deficient plants that receive Si in the nutrient solution presented an increase in Ca use efficiency in all collections of both forage, which contributed to a higher dry matter production.
Ca deficient forages showed the benefit of element, especially due to the increased use efficiency of Ca because it was the only deficient element in the nutrient solution. This effect possibly occurred because Si structurally assists in cell wall components, in which Ca is involved in its formation. Reports have indicated that Si forms complexes with structural cell polymers, such as pectin and callose (Boylston et al. 1990) and crosslinking with lignins and carbohydrates via associations with phenolic acids or aromatic rings (Inanaga et al. 1995), collaborating with the structuring of the cell wall. In addition, plants that received Si had a reduction in lipid peroxidation due to the lower accumulation of reactive oxygen species (de Oliveira et al. 2019); it also decreases as activities of enzymes that degrade the cell wall while preserving its integrity (Kochanová et al. 2014).
4.5 Magnesium (Mg)
It was evident that the omission of Mg decreased the production of biomass in both forages. This occurred because an omission of Mg is harmful to plants, due to the physiological role that is presented in the enzyme and in the chemical composition of chlorophyll (Prado 2008), where about 10 to 20% of the Mg present in the plant is linked to these pigments. This way, low amounts of this element can promote the degradation of chlorophyll molecules in older leaves, being translocated to newer tissues (Verbruggen and Hermans 2013), inducing visual symptoms that seriously affect the internal chloride in older leaves (Fig. 6), similar to what has already been described by Prado et al. (2011) in Tanzania grass grown in nutrient solution. However, Si application to the nutrient solution mitigated the effects caused by nutritional stress due to Mg deficiency since there was an increase in shoot biomass production in the third collection in the forage BRS Zuri (Fig. 1) and first and second collections in the forage BRS RB331 Ipyporã (Fig. 2). The benefit provided by Si addition to the nutrient solution in Mg deficient plants did not occur by increasing its absorption (Tables 1 and 4), but by increasing the use efficiency of this macronutrient in all collections of both forages (Tables 3 and 6), which reflected in higher forage dry matter production.
Forage BRS Zuri (Panicum maximum) grown in complete nutrient solution with Si (+Si) and Mg omission in the presence of Si (a), complete nutrient solution without Si (−Si) and Mg omission (−Mg) in the absence of Si (b), forage BRS RB331 Ipyporã (Brachiaria ruziziensis × Brachiaria brizantha) grown in complete nutrient solution with Si and Mg omission in the presence of Si (c), and complete nutrient solution without Si and Mg omission in the absence of Si (d)
In addition, Si use may have contributed to mitigating Mg deficiency stress due to the indirect association of this beneficial element in increasing chlorophyll content (Xie et al. 2014) and photosynthetic rates (Chen et al. 2016; Zuccarini 2008), which can contribute to a better use efficiency of Mg absorbed and converted into dry matter production. This effect of Si was visually evidenced, as there was relief from the severity of symptoms of nutritional deficiency (Fig. 6). Hosseini et al. (2019) found that Si application to corn plants exposed to Mg deficiency regulated primary metabolites and increased the levels of phytohormones (cytokines), which maintain growth and development of plants with deficiency of this nutrient.
4.6 Sulfur (S)
Sulfur was the second nutrient to present deficiency symptoms after its omission in both forages. The omission of this element in the nutrient solution caused a decrease in the values of all studied variables in both forages when compared with the complete treatment. This result is related to the function of this nutrient in the plant, being part of important organic compounds such as cystine, cysteine, and methionine and all proteins, also having enzymatic and structural functions (Prado 2008).
Si addition to the solution did not interfere with the reported physiological damage in plants grown under S omission in both forages, as the deficiency of this macronutrient occurred drastically, and there was no time for Si to exert its physiological effects and alleviate this nutritional disorder. Also, Si addition to the nutrient solution did not increase nutrient accumulation in S deficient forage plants (Tables 1 and 4). However, it increased S use efficiency in all collections of both forages Tables (3 and 6), but it was not enough to increase forage dry matter production and also did not alleviate the severity of the symptoms of deficiency of this nutrient (Fig. 7). This unprecedented finding indicates that it was not important to mitigate the effects of S deficiency, possibly because this nutrient is especially associated with the constitution of essential amino acids guaranteeing protein synthesis and the fact that there are no reports that Si would have effects on this metabolic pathway.
Forage BRS Zuri (Panicum maximum) grown in complete nutrient solution with Si (+Si) and S omission in the presence of Si (a), complete nutrient solution without Si (−Si) and S omission (−S) in the absence of Si (b), forage BRS RB331 Ipyporã (Brachiaria ruziziensis × Brachiaria brizantha) grown in complete nutrient solution with Si and S omission in the presence of Si (c), and complete nutrient solution without Si and S omission in the absence of Si (d)
The results of this study confirm the hypothesis only for K, Ca, and Mg, indicating that their deficiency in forages can be alleviated with Si supply. The practical implication of these unpublished data extends the benefits of Si in plant nutrition and opens the possibility of its use in forages. This practice can strengthen the sustainability of these species because they are cultivated in soils with low natural fertility or insufficient fertilization. In addition, Si enrichment in forages is an additional effect on animal production, as it is considered essential for its metabolism (Carlisle 1972).
It is pertinent to emphasize the need for further studies evaluating the effects of Si on forage plants with non-severe N and S deficiency and to expand these studies for other forage species. In addition, it is important to deepen studies that may indicate the biological role of Si on a biochemical level and the efficiency of carbon use in plants with nutritional deficiency.
5 Conclusions
We conclude that the application of silicon attenuates the effects of potassium, calcium, and magnesium deficiency in forages BRS Zuri and BRS RB331 Ipyporã, as it increases the accumulated shoot dry mass, due to the greater efficiency in the use of these nutrients.
The beneficial effect of silicon was not important in forage plants with nitrogen and sulfur deficiency.
The supply of Si in solution (fertigation) may be a new strategy in forages in the Poaceae group, for being Si accumulator and for attenuating a nutritional deficiency that benefits the production of fodder.
References
Akram M, Ashraf MY, Jamil M, Iqbal RM, Nafees M, Khan MA (2011) Nitrogen application improves gas exchange characteristics and chlorophyll fluorescence in maize hybrids under salinity conditions. Russ J Plant Physiol 58:394–401. https://doi.org/10.1134/S1021443711030022
Avalhaes CC, Prado RM, Oliveira Gondim AR, Alves AU, Correia MAR (2009) Rendimento e crescimento da beterraba em função da adubação com fósforo. Sci Agrár 10:75–80. https://doi.org/10.5380/rsa.v10i1.13173
Bataglia OC, Furlani AMC, Teixeira JPF, Gallo JR (1983) Métodos de análise química de plantas. Boletim técnico 78. Campinas, Brasil
Boylston EK, Hebert JJ, Hensarling TP, Bradow JM, Thibodeaux DP (1990) Role of silicon in developing cotton fibers. J Plant Nutr 13:131–148. https://doi.org/10.1080/01904169009364063
Carlisle EM (1972) Silicon: an essential element for the chick. Sci 178:619–621. https://doi.org/10.1126/science.178.4061.619
Cavalcante VS, de Mello PR, de Lima VR, Campos CNS (2016) Iron concentrations in sugar cane (Saccharum officinarum L.) cultivated in nutrient solution. Agrocienc 50:867–875 Available via DIALOG. http://www.redalyc.org/articulo.oa?id=30248292007. Accessed 20 Jun 2019
Chen D, Cao B, Wang S, Liu P, Deng X, Yin L, Zhang S (2016) Silicon moderated the K deficiency by improving the plant-water status in sorghum. Sci Rep 6:22882. https://doi.org/10.1038/srep22882
Cooke J, Leishman MR (2012) Tradeoffs between foliar silicon and carbon-based defences: evidence from vegetation communities of contrasting soil types. Oikos 121:2052–2060. https://doi.org/10.1111/j.1600-0706.2012.20057.x
de Camargo MS, Bezerra BKL, Holanda LA, Oliveira AL, Vitti AC, Silva MA (2019) Silicon fertilization improves physiological responses in sugarcane cultivars grown under water deficit. J Soil Sci Plant Nutr 19:81–91. https://doi.org/10.1007/s42729-019-0012-1
de Oliveira RLL, Prado R d M, Felisberto G, Checchio MV, Gratão PL (2019) Silicon mitigates manganese deficiency stress by regulating the physiology and activity of antioxidant enzymes in Sorghum plants. J Soil Sci Plant Nutr 19:1–11. https://doi.org/10.1007/s42729-019-00051-w
Deus ACF, Prado RDM, Alvarez RDCF, de Oliveira RLL, Felisberto G (2019) Role of silicon and salicylic acid in the mitigation of nitrogen deficiency stress in rice plants. Silicon 1–9. https://doi.org/10.1007/s12633-019-00195-5
Feller U, Anders I, Demirevska K (2008) Degradation of rubisco and other chloroplast proteins under abiotic stress. Gen Appl Plant Physiol 34:5–18
Ferreira DF (2011) Sisvar: a computer statistical analysis system. Ciênc e Agrotec 35:1039–1042. https://doi.org/10.1590/S1413-70542011000600001
Gao X, Wang CZL, Zhang F (2005) Silicon improves water use efficiency in maize plants. J Plant Nutr 27:1457–1470. https://doi.org/10.1081/PLN-200025865
Hepler PK, Winship LJ (2010) Calcium at the cell wall-cytoplast interface. J Integr Plant Biol 52:147–160. https://doi.org/10.1111/j.1744-7909.2010.00923.x
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Circular. California agricultural experiment station 347(2nd edit)
Hosseini SA, Naseri Rad S, Ali N, Yvin JC (2019) The ameliorative effect of silicon on maize plants grown in Mg-deficient conditions. Int J Mol Sci 20:969. https://doi.org/10.3390/ijms20040969
Inanaga S, Okasaka A, Tanaka S (1995) Does silicon exist in association with organic compounds in rice plant? J Soil Sci Plant Nutr 41:111–117. https://doi.org/10.1080/00380768.1995.10419564
Kissmann KG (1997) Plantas infestantes e nocivas, 2ª edn. São Paulo, Brasil
Kochanová Z, Jasková K, Sedláková B, Luxová M (2014) Silicon improves salinity tolerance and affects ammonia assimilation in maize roots. Biologia 69:1164–1171. https://doi.org/10.2478/s11756-014-0411-7
Korndörfer GH (2004) Análise de silício: solo, planta e fertilizante. Uberlândia, Brasil
Liu JJ, Lin SH, Xu PL, Wang XJ, Bai JG (2009) Effects of exogenous silicon on the activities of antioxidant enzymes and lipid peroxidation in chilling-stressed cucumber leaves. Agr Sci China 8:1075–1086. https://doi.org/10.1016/S1671-2927(08)60315-6
Maghsoudi K, Arvin MJ, Ashraf M (2019) Mitigation of arsenic toxicity in wheat by the exogenously applied salicylic acid, 24-Epi-Brassinolide and silicon. J Soil Sci Plant Nutr 1–12. https://doi.org/10.1007/s42729-019-00147-3
Maillard A, Ali N, Schwarzenberg A, Jamois F, Yvin JC, Hosseini SA (2018) Silicon transcriptionally regulates sulfur and ABA metabolism and delays leaf senescence in barley under combined sulfur deficiency and osmotic stress. Environ Exp Bot 155:394–410. https://doi.org/10.1016/j.envexpbot.2018.07.026
Mak M, Babla M, Xu SC, O’Carrigan A, Liu XH, Gong YM, Holford P, Chen ZH (2014) Leaf mesophyll K+, H+ and Ca2+ fluxes are involved in drought-induced decrease in photosynthesis and stomatal closure in soybean. Environ Exp Bot 98:1–12. https://doi.org/10.1016/j.envexpbot.2013.10.003
Malavolta E, Vitti GC, Oliveira SA (1997) Avaliação do estado nutricional das plantas: princípios e aplicações, 2ª edn. Piracicaba, Brasil
Melo SPD, Monteiro FA, Manfredini D (2007) Silicate and phosphate combinations for marandu palisade grass growing on an oxisol. Sci Agric 64:275–281. https://doi.org/10.1590/S0103-90162007000300010
Melo SPD, Monteiro FA, Bona FDD (2010) Silicon distribution and accumulation in shoot tissue of the tropical forage grass Brachiaria brizantha. Plant Soil 336:241–249. https://doi.org/10.1007/s11104-010-0472-5
Miao BH, Han XG, Zhang WH (2010) The ameliorative effect of silicon on soybean seedlings grown in potassium-deficient medium. Ann Bot 105:967–973. https://doi.org/10.1093/aob/mcq063
Mitani N, Ma JF (2005) Uptake system of silicon in different plant species. J Exp Bot 56:1255–1261. https://doi.org/10.1093/jxb/eri121
Monteiro FA, Ramos AKB, De Carvalho DD, de Abreu JBR, Daiub JAS, Da Silva JEP, Natale W (1995) Cultivo de Brachiaria brizantha Stapf. cv. Marandu em solução nutritiva com omissões de macronutrientes. Sci Agric 52:135–141. https://doi.org/10.1590/S0103-90161995000100022
Moro AL, Pacheco AC, Moro E (2018) Physiological and biochemical alterations of Urocholoa brizantha submitted to water deficit and silicate fertilization. J Agric Sci 10:8. https://doi.org/10.5539/jas.v10n8p166
Neu S, Schaller J, Dudel EG (2017) Silicon availability modifies nutrient use efficiency and content, C: N: P stoichiometry, and productivity of winter wheat (Triticum aestivum L.). Sci Rep 7:40829. https://doi.org/10.1038/srep40829
Pathak MR, Silva JAT, Wani SH (2014) Polyamines in abiotic stress tolerance through transgenic approaches. GM Crops Food 5:87–96. https://doi.org/10.4161/gmcr.28774
Prado RM (2008) Manual de nutrição de plantas forrageiras. Jaboticabal, Brasil
Prado RM, Hojo RH, Avalhães CC, Vale DW, Pimentel UV (2011) Desempenho do capim-tanzânia cultivado em solução nutritiva com à omissão de macronutrientes. Sci Agrar Paran10:58. https://doi.org/10.1818/sap.v10i1.5270
Raven JA (1983) The transport and function of silicon in plants. Biol Rev 58:179–207. https://doi.org/10.1111/j.1469-185X.1983.tb00385.x
Sarto MVM, Do Carmo LM, Rampim L, Rosset JS, Inagaki AM, Bassegio D (2016) Effects of silicon (Si) fertilization on gas exchange and production Brachiaria. Aust J Crop Sci 10:307–313. https://doi.org/10.21475/ajcs.2016.10.03.p6864
Schaller J, Brackhage C, Gessner MO, Bauker E, Gertdudel E (2012) Silicon supply modifies C:N:P stoichiometry and growth of Phragmites australis. Plant Biol 14:392–396. https://doi.org/10.1111/j.1438-8677.2011.00537.x
Siddiqi MY, Glass AD (1981) Utilization index: a modified approach to the estimation and comparison of nutrient utilization efficiency in plants. J Plant Nutr 4:289–302. https://doi.org/10.1080/01904168109362919
Verbruggen N, Hermans C (2013) Physiological and molecular responses to magnesium nutritional imbalance in plants. Plant Soil 368:87–99
Wang L, Ashraf U, Chang C, Abrar M, Cheng X (2019) Effects of silicon and phosphatic fertilization on rice yield and soil fertility. J Soil Sci Plant Nutr:1–9. https://doi.org/10.1007/s42729-019-00145-5
Winkler U, Zotz G (2010) ‘And then there were three’: highly efficient uptake of potassium by foliar trichomes of epiphytic bromeliads. Ann Bot 106:421–427. https://doi.org/10.1093/aob/mcq120
Xie Z, Song F, Xu H, Shao H, Song R (2014) Effects of silicon on photosynthetic characteristics of maize (Zea mays L.) on alluvial soil. Sci World J 2014:1–6. https://doi.org/10.1155/2014/718716
Zhang XC, Yu XF, Ma YF (2013) Effect of nitrogen application and elevated CO2 on photosynthetic gas exchange and electron transport in wheat leaves. Photosynthetica 51:593–602. https://doi.org/10.1007/s11099-013-0059-5
Zia Z, Bakhat HF, Saqib ZA, Shah GM, Fahad S, Ashraf MR, Hammad HM, Naseem W, Shahid M (2017) Effect of water management and silicon on germination, growth, phosphorus and arsenic uptake in rice. Ecotoxicol Environ Saf 144:11–18. https://doi.org/10.1016/j.ecoenv.2017.06.004
Zuccarini P (2008) Effects of silicon on photosynthesis, water relations and nutrient uptake of Phaseolus vulgaris under NaCl stress. Biol Plant 52:157–160. https://doi.org/10.1007/s10535-008-0034-3
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Buchelt, A.C., Teixeira, G.C.M., Oliveira, K.S. et al. Silicon Contribution Via Nutrient Solution in Forage Plants to Mitigate Nitrogen, Potassium, Calcium, Magnesium, and Sulfur Deficiency. J Soil Sci Plant Nutr 20, 1532–1548 (2020). https://doi.org/10.1007/s42729-020-00245-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-020-00245-7