Abstract
The prickly-pear cactus Opuntia ficus-indica L. is grown throughout Morocco and has an important economic role as a source of food, animal feed and income for rural populations. However, currently this crop is suffering from attack by the wild cochineal Dactylopius opuntiae, which has caused huge production losses. This study was conducted to evaluate the effectiveness of five botanical extracts and a detergent for the control of nymphs and adult females of D. opuntiae in laboratory bioassays and field conditions. Under laboratory conditions, the detergent black soap at 60 g/l caused 100% mortality of D. opuntiae adult females and nymphs, 48 h after treatment. Capsicum annuum fruit extract at 10% gave 100% mortality of nymphs at 96 h after application and 56% mortality of adult females at 192 h after treatment. Under field conditions, black soap at 60 g/l applied in combination with C. annuum at 200 g/l caused the highest mortality on adult females (87.31%) at 168 h after application, and 84.9% on nymphs at 72 h after treatment. The black soap at 60 g/l had the greatest impact on mortality of D. opuntiae nymphs and adult females when applied at low to medium levels of D. opuntiae infestation. The double application of black soap at 60 g/l over a 3-day spray interval significantly increased the mortality of adult females up to 82.5% at 3 days after the second application. These findings showed that the use of the biodegradable products, black soap at 60 g/l in double application or in combination with C. annuum extract at 200 g/l, could be incorporated in the management package for the control of D. opuntiae as a safe alternative to chemical insecticides.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The prickly pear, Opuntia ficus-indica (L.) Mill. (Caryophyllales: Cactaceae), is a cactus species widespread in semi-arid and arid regions of the world. These plants serve various purposes, producing fruit and vegetables and as a forage substitute during drought for smallholder farmers. Furthermore, its industrial and medicinal products are highly demanded in the market. In many countries, the prickly pear is considered an important re-vegetation crop to control water and soil erosion in degraded areas (Yahia and Sáenz 2011). The crop has proved resilient to harsh conditions such as drought and heat and can be used to reduce the effects of desertification. Cactus pear has gradually attained economic importance in different areas, including the Mediterranean basin (Le Houérou 1996).
In Morocco, the prickly pear cactus plantations have been greatly promoted by the green Morocco Plan strategy as an alternative crop in less favorable regions. The cactus area progressed from 45,000 ha in the early 1990s to currently over 150,000 ha (MAPMDREF 2017). Unfortunately, the crop now suffers from the attack of a new sap-sucking insect pest, the wild cochineal Dactylopius opuntiae (Cockerell) (Hemiptera: Dactylopiidae). This insect was reported for the first time in Morocco in September 2014 in the Sidi Bennour region,70 km from El Jadida (Bouharroud et al. 2016). The D. opuntiae has spread over different regions, threatening the whole value chain. With such high speed of dissemination, the pest may completely wipe out cactus pear crops in the near future.
Dactylopius opuntiae is native to Mexico and was described by Cockerell in 1896 (De Lotto 1974). The females are covered with a white waxy powder that protects them from predators and, when they are crushed, they produce carminic acid that can be used in food, textile and cosmetic industries (Chávez-Moreno et al. 2009; Piña 1979). Nymphs and adult females of D. opuntiae feed on plants by sucking sap on the cladode, leading to chlorosis, drying, weakening of plants and eventually death in the case of severe infestation (Vanegas-Rico et al. 2010). The D. opuntiae is spreading rapidly in many countries, especially in the Mediterranean basin, where it has become a serious pest of prickly-pear crops (Mazzeo et al. 2018).
Because of the huge losses caused by D. opuntiae, there is an urgent need to develop an integrated pest management (IPM) approach combining several measures to control the pest population. This approach consists of multiple tools based on mechanical, physical, biological, chemical and other methods (Cavalcanti et al. 2001; Lopes et al. 2007; Santos et al. 2006). In Morocco, great progress has been made in the development of IPM options for the control of this devastating insect (Bouharroud et al. 2018; El Aalaoui et al. 2019b).The National Office of Food Safety (ONSSA) in Morocco has registered several insecticides for its control, including pyriproxyfen, spirotetramat, acetamiprid, chlorpyrifos and mineral oils such as paraffin oil, white mineral oil, petroleum oil (ONSSA-National Office of Food Safety 2019).There is increasing concern about the use of synthetic residual insecticides that, in addition to the insecticide-resistance problem, threaten human health, contaminate the environment and have negative impacts on natural enemies (Isman 2019).
The aim of this study was to investigate the potential of several alternative biodegradable products, including botanical extracts and a detergent for the control of D. opuntiae nymphs and adult females under laboratory and field conditions.
Material and methods
Preparation of plant extracts
Five plants known for their insecticidal effect were collected from their natural habitats in different Moroccan localities (Table 1). In the ICARDA Entomology Laboratory at Rabat, different parts of plants were rinsed, dried at room temperature 26 ± 2 °C and a relative humidity of 75 ± 5%, and then powdered using an electric blender. One hundred grams of each sample were macerated in 1 l of distilled water for 72 h, and then filtered through Whatman No. 1 filter paper. After filtration, the extracts were collected in a round-bottom flask and frozen at −80 °C for 24 h, and then freeze-dried using a Labconco Free-zone 6 instrument (Kansas City, US).
Laboratory bioassays
All experiments were conducted in the laboratory at the Khemis Zemamra Experimental Station of the National Institute of Agronomic Research (32°37'48"N, 8°42'0"W) in 2018.
Insecticidal effects of botanical extracts on D. opuntiae nymphs and adult females
The insecticidal activity of botanical extracts was determined by direct contact application in laboratory conditions at temperature of 26 ± 2 °C, relative humidity of 75% and photoperiod of 14/10 h of light/dark. A completely randomized design was adopted using five replicates per concentration for each treatment. Five concentrations for each plant extract (0.625, 1.25, 2.5, 5 and 10%) were applied using a 1-l hand sprayer.
Ten first instar nymphs of D. opuntiae and 10 females of the same age were deposited separately on unsprayed and no infested cladodes of the same size placed in Petri dishes (9 cm in diameter), including a check treated with water only. Artificial infestation on cladode surfaces was performed by fixing each stage of D. opuntiae using an entomological brush. All D. opuntiae stages were collected from cactus pear plants used for rearing in cages. Nymph mortality was recorded after 24, 48, 72 and 96 h. Mortality of adult females was counted every 24 h for 8 days after applying treatments. Mortality was examined using a binocular microscope (Motic). The dead nymphs showed no movement, and had color modifications, while the dead females revealed a desiccation of their bodies and dark brown color.
Insecticidal effects of insecticidal soap on D. opuntiae nymphs and adult females
The insecticidal activity of black soap was determined by contact application using a completely randomized design with five replicates. Four concentrations were selected for their efficacy against D. opuntiae nymphs and mature females: 30, 60,100 and 150 g/l. The insecticidal soap solution was applied using a 1-l hand sprayer. Mortality of nymphs and adult females was recorded 24, 48 and 72 h after treatments.
Field trials
Experimental site
The promising plant extracts that showed significant efficacy against D. opuntiae females under laboratory conditions were selected to assess their efficacy in natural field infestations in various trials during September–November 2018. The field trials were conducted in Jamaat Riah rural commune in Settat Province (33°09′41.0″N, 7°22′51.6″W).
Experiment 1: Exposure of D. opuntiae to low doses of biopesticides
The first experiment included four treatments – black soap (30 g/l), C. annuum extract (10%), U.dioica extract (10%) and control treated only with water – applied using a 2-l hand sprayer A randomized complete block design was used with four replicates. Female mortality was recorded at 72 and 168 h after treatments on three infested cladodes selected for each treatment, and mortality on nymphs was recorded 72 h after treatments.
Experiment 2: Toxicity of 60 g/l soap with three different concentrations of C. annuum extract
The second trial consisted of four treatments using the synergy of the black soap (60 g/l) with three different concentrations of C. annuum:50 g macerated in 1 l of water (50 g/l), 100 and 200 g/l for 72 h and then filtered. A randomized complete block design was used with four replicates. Female mortality was recorded at 72 and 168 h after treatment using four cladodes per plot, and nymph mortality was recorded 72 h after treatment.
Experiment 3: Toxicity of 60 g/l soap and 200 g/l C. annuum and their combination
The trial was conducted following a randomized complete block design with four replicates and four treatments: black soap at 60 g/l, C. annuum extract at 200 g/l macerated for 72 h and filtrated, their combination, and water as a control treatment. Mortality of nymphs and females was recorded as described previously.
Experiment 4: toxicity of 60 g/l soap at different levels of D. opuntiae infestation
The most efficient concentration of the black soap (60 g/l) obtained from laboratory bioassays was tested under natural field conditions. The black soap at 60 g/l was applied on cladodes with different degrees of infestation using a rating scale modified from Silva (1991). The levels of infestation were low (1–25%), medium (26–50%) and high (51–75%). The experiment was conducted in a randomized complete block design with three replicates. The four treatments used in the trial included untreated plot (control) and black soap at 60 g/l applied at the three levels of D. opuntiae infestation, using three cladodes for each plot. Mortality of females was recorded 72 and168 h after treatment, and mortality of nymphs was recorded 72 h after treatment.
Experiment 5: Comparative toxicity of 60 g/l soap applied in single and double applications
The trial was conducted in a cactus area of about 20 m2 under a high level of D. opuntiae infestation. One side of the plot received one application of black soap (60 g/l) and other side received a second application 72 h after the first treatment. Four cladodes were selected randomly on both sides in order to compare the mortality of adult female D. opuntiae at 72 and 168 h after treatment for each side.
Data analysis
Data were analyzed using one-way analysis of variance (ANOVA). Mortality percentages were transformed into arcsine √% before the statistical analysis. The means were compared using Fisher’s least significant differences (LSD) test at P < 0.05 using Genstat (19th Edition, VSN International, UK).
Results
Laboratory bioassays
Effect of botanical extracts on D. opuntiae nymphs and adult females
Mean percent mortality of D. opuntiae nymphs and adult females exposed to five aqueous extract are presented in Tables 2 and 3. The statistical analysis showed a significant difference in nymph mortality rates among all the tested products during the different exposure periods.
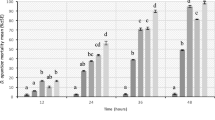
At 48 h after application, the highest percentage mortality (96%) of nymphs was recorded for the C. annuum extract at 10%. The M. officinarum at 10% and C. annuum at 5% were the second most effective extracts, both resulting in 74% nymph mortality. The M. officinarum at 5% produced 60% mortality on nymphs.
At 72 h after treatment, there were significant differences among botanical extracts (P < 0.001). The mortality percentages compared using LSD showed that C. annuum at 10% was separated as a different group from the other extracts. The C. annuum showed the highest percentage of nymph mortality (100%) at 10% and about 84% at 5%.
At 96 h after treatment, the highest percentage mortality of nymphs (100%) occurred for C. annuum at 10%, and extracts of C. annuum at 5% and U. dioica at 10% both resulted in 92% mortality. The increasing doses of the extracts significantly increased the mortality of nymphs that were exposed to different extracts for different periods.
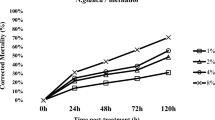
The bioassays showed a significant difference (P < 0.001) between the treatments through various exposure periods on D. opuntiae adult females (Table 3). The C. annuum and U. dioica extracts expressed the highest toxic activity on D. opuntiae adult females among all tested extracts. The moderate percentage mortality (56%) occurred at 192 h for C. annuum at 10%. However, the U. dioica extract at10% caused the low mortality rate (26%) at 144–192 h after application.
Insecticidal effects of black soap on D. opuntiae nymphs and adult females
The ANOVA showed no significant differences in mortality of. Opuntiae nymphs and adult females caused by black soap treatment at all doses tested for various exposure times. However, the mortality means significantly differed from the check (P < 0.001; Table 4). The results showed high toxicity of the black soap at different doses on D. opuntiae females and nymphs. At 6 h post-application, the mortality of nymphs exceeded 92% at the lowest dose (30 g/l). After 48 h, the highest mortality (100%) of nymphs occurred for all tested doses (i.e. 60, 100 and 150 g/l).
For adult females, the toxicity of black soap at different doses ranged from 96% at 24 h after treatment at 30 g/l to 100% at 48 h after application for the other tested doses (i.e. 60, 100 and 150 g/l).
Field trials
Insecticidal effects of C. annum and U. dioica and black soap solution on D. opuntiae nymphs and adult females
The mortality of D. opuntiae nymphs and adult females after exposure to botanical extracts and black soap is presented in Table 5.There were significant differences in mortality for both nymphs and adult females(P < 0.001).Data from the first trial showed that the black soap at 30 g/l and C. annuum at 10% caused the highest nymph mortality with 73 and 70% at 72 h after application, respectively. However, the highest mortality observed was only 33% of adult females with black soap at 30 g/l at 168 h after application.
The second field trial showed significant differences in mortality of nymphs and adult females between black soap at 60 g/l applied in combination with different doses of C. annuum extract and the control (P < 0.001). At 72 h after treatment, the highest toxicities against nymphs (82.50%) and adult females (80.38%) were recorded for the black soap (60 g/l) in combination with the highest dose (200 g/l) of C. annuum extract, 168 h after treatment The lowest mortalities of adult females of 53.75 and 40.00% occurred for black soap at 60 g/l combined with the lowest doses of C. annuum at 100 and 50 g/l, respectively.
The third trial showed that black soap at 60 g/l and the combination of black soap at 60 g/l with C. annuum at 200 g/l showed the greatest mortality rates of nymphs at 72 h after application, with 87.53 and 84.90%, respectively. At 168 h after treatment, the combination of 60 g/l black soap and 200 g/l C. annuum extract showed the greatest percentage mortality on adult females with 87.31%. The comparison of means grouped this combination separately from the other treatments in terms of mortality on adult females.
Insecticidal effects of black soap on nymphs and adult females at different D. opuntiae infestation levels
The percentage of mortality of nymph and adult females for black soap applied at 60 g/l significantly differed among the various levels of D. opuntiae infestation (P < 0.001; Table 6). The black soap at 60 g/l had a high toxicity effect on nymphs and adult females when applied at low to medium levels of D. opuntiae infestation. The highest mortality of nymphs was 91.11 and 88.89% at 72 h after application of black soap at 60 g/l on cladodes with low and medium levels of D. opuntiae infestation, respectively. At 168 h after treatment, the mortality of 98.33% of adult females occurred for application of black soap at 60 g/l on cladodes with low infestation. The lowest mortality of adult females was for black soap at 60 g/l at medium and high levels of infestation with 72.22 and 59.83%, respectively.
Comparative toxicity of 60 g/l black soap applied in single and double applications
The mortality of nymphs and adult females after exposure to a single and double applications of black soap at 60 g/l under the high infestation level of D. opuntiae is presented in Table 7.There was a significant difference in mortality rates between the black soap at 60 g/l and the control for different exposure periods (Table 7).The mortality of adult females was 57.50% at 72 h after the first application; whereas, mortality increased significantly, at 72 h after the second application of black soap at 60 g/l, to about 82.5%. At 168 h after the second treatment, the mortality of adult females reached 85.0%.
Discussion
The present study was conducted to evaluate the insecticidal effect of various biodegradable products, including botanicals extracts, and a detergent for the control of D. opuntiae nymphs and adult females. Among all the tested bio-insecticides, black soap at 60 g/l, C. annuum extract at 10% and their combination were the most effective against both nymphs and adult females.
The black soap at the tested concentrations of 60, 100 and 150 g/l effectively reduced the numbers of D. opuntiae adult females and nymphs. Depending on the level of D. opuntiae infestation, the black soap at 60 g/l had an excellent insecticidal effect on nymphs and adult females when applied at low to medium levels of D. opuntiae infestation. The field experiments showed the high potential of black soap at 60 g/l, when applied in double sprays over a 72-h spray interval, without any evident phytotoxicity for the treated plants. This insecticidal effect seemed to be greater at the beginning of the D. opuntiae infestation. The first application of black soap caused 57.5% mortality of adult females, with a strong degradation of the cuticular wax. The second application significantly increased mortality of adult females to 85% most probably caused by asphyxia after obstruction of spiracles, dehydration of the body and increased exposure to UV radiation. The removal of the waxy and fatty substances from female bodies exposes them to the UV radiation in sunlight, causing extreme dehydration and quick death.
Potassium salts of fatty acids (PSFA) are commonly referred to as soap salts and are used as insecticides, herbicides, fungicides and algaecides (NPIC - National Pesticide Information Center 2001). Moroccan black soap is a traditional product manufactured from natural fatty acids derived from olive oil. Soaps and oils may have been among the first chemicals used to control insects (Puritch 1981). Several authors have reported high insecticidal effects of soap on a range of insect pests, mostly against soft-bodied insects, aphids, mealybugs and thrips as well as spider mites. These effects were usually achieved with the highest concentration tested, in most cases under or equal to 2%, and with the greatest number of sprays (Curkovic 2016). The present study corroborates the findings of many earlier studies using insecticidal soap in different formulations and doses against two different cactus cochineal species, D. opuntiae and D. coccus.
Mendoza et al. (2004) reported that laundry soap (detergent Roma® and Peak Plus) at 0.5–3.0% caused a mortality rate exceeding 50% among D. opuntiae adult females and first- and second-instar nymphs,10 days after each application. They showed that the first application removed the cuticular wax, which caused insect dehydration and thus death. Additionally, repeated application increased the mortality rates for each concentration of the tested products.
Carvalho et al. (2006) confirmed excellent control (100%) 7 days after application with laundry soap on carmine cochineal D. coccus colonies, when applied at concentrations of 1–5%. The present findings corroborate those of Carvalho and Lopes (2007) that direct application of powder laundry soap at 2% and neutral detergent at 5% caused a significant reduction (98–100%) of D. opuntiae colonies. In addition, Brito et al. (2008) reported a high reduction rate (83.80%) for D. opuntiae colonies at 8 days after application of 5% soap powder. A recent study revealed that application of a commercial insecticidal soap (HAMPER, with PSFA, of 500 g/l) at 60 cm3/hl showed a mortality rate of 88% on first- and second-instars of D. opuntiae, 24 h after spraying (El Aalaoui et al. 2019a). However, mortality was only 46.98% for adult females at 120 h after application.
In Morocco, Joutei and Bassy (2010) confirmed the high insecticidal potential of black soap at 3%, with significant reductions in the population level of green peach aphid (Myzus persicae Sulzer) and without any negative effects on beneficial insects.
In order to provide direct contact with D. opuntiae, the insecticidal soap must be applied at shorter application intervals and with complete possible coverage of the different parts of the cactus plant.
The modes of action for soaps and detergents against pests are not well understood; they are known to act at multiple sites not a specific target site (Sparks and Nauen 2015).Wax removal, repellency or cell membrane disruption, arthropod dislodging and drowning have been mentioned as lethal mechanisms involved in use of soaps and detergents (Butler et al. 1993; Curkovic 2016).
The application of insecticidal soap is considered more selective than conventional insecticides, being compatible with biological control due to low adverse impacts on beneficial fauna (Curkovic 2016). Brito et al. (2008) studied the selectivity of several biodegradable products including soap powder at 5% to control D. opuntiae. They found that this application did not significantly affect the populations of natural enemies, such as ladybug larvae Zagreus bimaculosus and Cycloneda sanguinea (Linnaeus 1763) (Coleoptera: Coccinellidae) and Baccha sp. (Diptera: Sirphidae). A similar study was conducted by El Aalaoui et al. (2019a) using several chemical and biological insecticides against Cryptolaemus montrouzieri adults and larvae. The application of the insecticidal soap (HAMPER, 500 g/l PSFA) at 20–60 cm3/hl was less harmful to the predator C. montrouzieri, causing mortality rates of 27.06–44.71 and 25.00–41.67% for adults and larva, respectively.
Comparison of insecticidal efficacy of the different extracts showed that the C. annuum fruit extract at 10% was the most effective among the five aqueous extracts tested on D. opuntiae adult females and nymphs. The mortality under controlled conditions reached 100% on nymphs at 72 h and 56% on adult females at 192 h after treatment. However, C. annuum extract was less toxic under field infestation when applied directly to cladodes with colonies protected by a thicker wax. The current study showed that the first application of black soap at 60 g/l allowed removal of the cuticular wax, which exposed the D. opuntiae nymphs and adult females to high contact toxicity with 200 g/l C. annuum extract. This explains the higher percentage mortality following soap application compared to a single application of C. annuum extract.
The insecticidal properties of C. annuum result from the combination of several alkaloid compounds called capsaicinoids. Among these components, capsaicin and dihydrocapsaicin contribute approximately 80–90% of the total pungency in most C. annuum fruit (Zewdie and Bosland 2001). The amount of capsaicin in C. annuum fruit comprises more than 70% of their pungency (Titze et al. 2002). According to many studies, capsaicin (8-methyl-N-vanillyl-6 nonemide) has broad-spectrum insecticidal activity against various species of insect pests. In addition, these alkaloid compounds are biodegradable and ecofriendly (Isman 1999). The capsaicin’s toxicity appears to be through metabolic disruption, membrane damage and nervous system dysfunction (Copping 2001). Several insecticides and rodenticides based on capsaicin have been registered by the Environmental Protection Agency of the USA (Gudeva et al. 2013).
Edelson et al. (2002) also reported good efficacy of “Capsyn”, a formulation containing 21 g/l of capsaicinoids, mixed with “M-Pede” containing 490 g/l PSFA on Myzus persicae (Sulzer). This formulation provided the greatest mortality (79%) among all tested mixtures. The authors reported that capsaicin extracts alone showed low levels of toxicity, but acted synergistically in mixtures with other insecticides and provided greater than expected levels of mortality. Likewise, C. annuum extract has shown its insecticidal effect against rice grain insects Angoumois grain moth (Sitotroga cerealella) (Prakash and Rao 2006). Similarly, Madhumathy et al. (2007) reported that 0.024% C. annum extract had strong toxicity (>96%) against fourth-instar larvae of two mosquito disease vector species Culexquinque fasciatus and Anopheles stephensi (Diptera: Culicidae).
Diets containing capsaicin showed some growth-retarding activity on the larvae of spiny bollworm Earias insulana (Lepidoptera: Noctuidae) (Weissenberg et al. 1986). Extracts of C. annum were also proven as repellents of some species of stored product beetles, such as Sitophilus zeamais Motschulsky (Coleoptera: Curculinidae) and Tribolium castaneum (Herbst) (Coleoptera: Tenebriouidae) (Ho et al. 1997). In Malaysia, Mutalib et al. (2017) reported that the ethanolic extract of C. annuum showed excellent repellent activity and strong insecticidal activity against the ant pest Tapinoma sessile.
Several studies have reported the use of botanical extracts for control of D. opuntiae. In Brazil, Borges et al. (2013) found that neem (Azadirachta indica A. Juss.) extract reduced the infestation rate of D. opuntiae on prickly pear under greenhouse conditions.
Vigueras et al. (2009) tested several botanical extracts on D. opuntiae under laboratory conditions. They showed mortality rates of 82, 92, 95, 98 and 99% for extracts of Chenopodium ambrosioides L., Mentha piperita L., Mentha viridis L., Tagetes erecta L. and Tagetes florida L. at 72 h following treatment against second instars of D. opuntiae. They also reported that the volatile compounds present in the different extracts consisted of alkanes, alcohol, aldehydes and terpenoids, especially monoterpenoids, which may be responsible for the insecticidal toxicity. Pérez-Ramirez et al. (2014) showed that the terpenoids eugenol, 1–8 cineol and menthol successfully led to the reduction in D. opuntiae nymphs fixed to healthy cactus pear cladodes. Similarly, Lopes et al. (2009) reported that orange oil obtained from citrus juice with sodium tetraborohydrate decahydrate, with lowest dose at 0.3% caused a mortality rate > 90% on D. opuntiae nymphs and adult females within 48 h after treatment in field conditions. This application showed no side effects on the ladybugs Cycloneda sanguinea L. and Scymnus intrusus Casey, but was lethal to larvae of Baccha sp. at 0.7%.
In a recent study, Lopes et al. (2018) found that leaf and pod extracts of Libidibia ferrea var. ferrea exhibited greater insecticidal effect (81%) on second-instar nymphs and 97% on adult females of D. opuntiae. In the same study, Agave sisalana leaf showed insecticidal activity only on adult females with mortality rates ranging within 51–97%.
Conclusion
The findings of the current study showed that double applications of the black soap solution at 60 g/l or when applied in combination with C. annuum fruit extract at 200 g/l could be used as one IPM component for the control of D. opuntiae in Morocco. Black soap and C. annuum extract represents a viable, effective and ecofriendly alternative to synthetic pesticides. They are also less expensive, easy to produce and available to smallholder farmers.
References
Borges LR, Santos DC, Gomes EWF, Cavalcanti VALB, Silva IMM, Falcão HM, da Silva DMP (2013) Use of biodegradable products for the control of Dactylopius opuntiae (Hemiptera: Dactylopiidae) in cactus pear. Acta Hortic 995:379–386
Bouharroud R, Amarraque A, Qessaoui R (2016) First report of the Opuntia cochineal scale Dactylopius opuntiae (Hemiptera: Dactylopiidae) in Morocco. EPPO Bull 46:308–310
Bouharroud R, Sbaghi M, Boujghagh M, El Bouhssini M (2018) Biological control of the prickly pear cochineal Dactylopius opuntiae Cockerell (Hemiptera: Dactylopiidae). EPPO Bull 48:300–306
Brito CH, Lopes EB, Albuquerque IC, Batista JL (2008) Avaliação de productos Alternatives e pesticidas no combate a cochonilha-do-carmimna Paraíba. In: Congresso Brasileiro de Entomologia. Anais. Sociedade Entomológica do Brasil, Uberlândia
Butler GD, Henneberry TJ, Stansly PA, Schuster DJ (1993) Insecticidal effects of selected soaps, oils and detergents on the sweet potato whitefly. Fla Entomol 76:161–167
Carvalho RA, Lopes EB (2007) Alternative control methods of the cochineal Dactylopius opuntiae in northeastern Brazil. In: XVI international plant protection congress. Glasgow, Scotland, 16 p 2
Carvalho RA, Lopes EB, Silva AC, Leandro RS, Campos VB (2006) Controle alternativo da cochonilha-do-carmim em palma forrageira no cariri paraibano. Available in: www.mma.gov.br/port/sbf/invasoras/capa/docs/paineis/ss>
Cavalcanti V, Sena RC, Coutinho JLB, Arruda GP, Rodrigues FB (2001) Controle das cochonilhas da palma forrageira. Recife: Empresa Pernambucana de Pesquisa Agropecuária. IPA Responde 39:1–2
Chávez-Moreno CK, Tecante A, Casas A (2009) The Opuntia (Cactaceae) and Dactylopius (Hemiptera: Dactylopiidae) in Mexico: a historical perspective of use, interaction and distribution. Biodivers Conserv 18:3337–3355
Copping LG (2001) The BioPesticide manual, second ed. British Crop Protection Council, Farnham
Curkovic T (2016) Detergents and soaps as tools for IPM in agriculture, in: integrated Pest management (IPM): environmentally sound Pest management. IntechOpen. https://doi.org/10.5772/64343
De Lotto G (1974) On the status and identity of the cochineal insects (Homoptera: Coccoidea: Dactylopiidae). J Entomol Soc South Afr 37:167–193
Edelson JV, Duthie J, Roberts W (2002) Toxicity of biorational insecticides: activity against the green peach aphid, Myzus persicae (Sulzer). Pest Manag Sci 58:255–260
El Aalaoui M, Bouharroud R, Sbaghi M, El Bouhssini M, Hilali L, Dari K (2019a) Comparative toxicity of different chemical and biological insecticides against the scale insect Dactylopius opuntiae and their side effects on the predator Cryptolaemus montrouzieri. Arch Phytopathol Pflanzenschutz 52:155–169
El Aalaoui M, Bouharroud R, Sbaghi M, El Bouhssini M, Hilali L (2019b) Predatory potential of eleven native Moroccan adult ladybird species on different stages of Dactylopius opuntiae (Cockerell) (Hemiptera: Dactylopiidae). EPPO Bull 49:374–379. https://doi.org/10.1111/epp.12565
Gudeva LK, Mitrev S, Maksimova V, Spasov D (2013) Content of capsaicin extracted from hot pepper (Capsicum annuum ssp. microcarpum L.) and its use as an ecopesticide. Hem Ind 67:671–675
Ho S, Ma Y, Tan HT, Halid H (1997) Repellency of some plant extracts to the stored products beetles, Tribolium castaneum (Herbst) and Sitophilus zeamais Motsch. Proceedings of the symposium on pest management for stored food and feed. Southeast Asian regional Centre for Tropical Biology. Bio Trop 59:209–215
Isman MB (1999) Pesticides based on plant essential oils. Pestic Outlook 2:68–72
Isman MB (2019) Botanical insecticides in the twenty-first century—fulfilling their promise? Annu Rev Entomol:65. https://doi.org/10.1146/annurev-ento-011019-025010
Joutei AB, Bassy F (2010) Essais de lutte chimique et biologique contre les acariens (Tetranychus urticae Koch) et les pucerons (Myzus persicae Sulzer) sur cultures maraîchères dans la région du Saïs. In: Himmi O. (editor). Entomologie et developpement durable. Travaux de l’Institut Scientifique, Rabat, Série Zoologie, n° 47
Le Houérou HN (1996) The role of cacti (Opuntia spp.) in erosion control, land reclamation, rehabilitation and agricultural development in the Mediterranean Basin. J Arid Environ 33:135–159
Lopes EB, Brito CH, Guedes CC, Santos DC, Araújo E, Batista JL, Araújo LF, Vasconcelos MF, Coelho RSB, Cavalcanti VALB (2007) Palma Forrageira: Cultivo, Uso Atual e Perspectivas de utilização no Semi-Arido Nordestino. In: VI Congresso Internacional de Palma e Cochonilha e VI Encontro Geral da FAOCACTUSNET. EMEPA/FAEPA. João Pessoa – Paraíba, Brazil, p130
Lopes EB, Brito CH, Albuquerque IC, Batista JL (2009) Desempenho do oleo de laranja no controle da cochonilha-do-carmim em palma gigante. Engenharia Ambiental 6:252–258
Lopes RS, Oliveira LG, Costa AF, Correia MTS, Lima EAL, Lima VLM (2018) Efficacy of Libidibia ferrea var. ferrea and Agave sisalana extracts against Dactylopius opuntiae (Hemiptera: Coccoidea). J Agric Sci 10:255–267
Madhumathy AP, Aivazi AA, Vijayan VA (2007) Larvicidal efficacy of Capsicum annum against Anopheles stephensi and Culex quinquefasciatus. J Vect Borne Dis 44:223–226
Mazzeo G, Nucifora S, Russo A, Suma P (2018) Dactylopius opuntiae, a new prickly pear cactus pest in the Mediterranean: an overview. Entomol Exp Appl 167:59–72. https://doi.org/10.1111/eea.12756
Mendoza CP, Hernández RN, Cázares CL, Hernández HG (2004) Efectividad biológica de productos biodegradables para el control de la cochinilla silvestre Dactylopius opuntiae (Cockerell) (Homoptera: Dactylopiidae). Acta Zool Mex 20:99–106
Mutalib NA, Firdaus Azis TM, Mohamad S, Azizan NI, Sidek HS, Roziana MH, Razali Z (2017) The repellent and lethal effects of black pepper (Piper nigrum), chilli pepper (Capsicum annuum) and cinnamon (Cinnamomum zeylanicum) extracts towards the odorous house ant (Tapinoma sessile). J Eng Appl Sci 12:2710–2714
NPIC - National Pesticide Information Center (2001) Potassium salts of fatty acids (technical fact sheet). http://npic.orst.edu/factsheets/psfatech.pdf [accessed on January 2012]
ONSSA-National Office of Food Safety (2019) (http://eservice.onssa.gov.ma/IndPesticide.aspx)
Pérez-Ramirez A, Castrejón-Ayala F, Jiménez-Pérez A (2014) Potential of terpenoids and mealybug extract to deter the establishment of Dactylopius opuntiae (Hemiptera, Dactylopiidae) crawlers on Opuntia ficus indica. Florida Entomol 97:269–271
Piña LI (1979) Principales países productores de grana fina y algunos aspectos biológicos sobre la producción de este colorante. Tecnología L.A.N.F.I. Revista de los. Laboratorios Nacionales de Fomento Industrial 5:14–16
Prakash A, Rao J (2006) Exploitation of newer botanicals as rice grain protectants against Angoumois grain moth Sitotroga cerealella. Entomon 31:1–8
Puritch GS (1981) Pesticidal soaps and adjuvants -what are they and how do they work? Proc. 23rd Ann. Lower Mainland Hort. Impr. Grower’s Short Course, Abbotsford, B.C., Canada
Santos DC, Farias I, Lira MA, Santos MVF, Arruda GP, Coelho RSB, Dias FM, Melo JN (2006) Manejo e utilização da palma forrageira (Opuntia e Nopalea). Empresa Pernambucana de Pesquisa Agropecuária. Recife, IPA, (Documento 30). Brazil, p 48. Retrieved from http://www.ipa.br/publicacoes_tecnicas/Pal01.pdf
Silva SQ (1991) Proposta para avaliação do controle biológico da cochonilha Diaspis echinocacti (Bouché, 1833) (Homoptera, Diaspididae) da palma forrageira em Pernambuco. Recife. Universidade Federal Rural de Pernambuco, Recife, p 103 Dissertação Mestrado
Sparks TC, Nauen R (2015) IRAC: mode of action classification and insecticide resistance management. Pestic Biochem Physiol 121:122–128
Titze KP, Mueller-Seitz E, Petz M (2002) Pungency in paprika (Capsicum annuum). 2. Heterogeneity of capsaicinoid content in individual fruits from one plant. J Agric Food Chem 50:1264–1266
Vanegas-Rico JM, Lomeli-Flores JR, Rodríguez-leyva E, Mora-aguilera GY, Valdez JM (2010) Enemigos naturales de Dactylopius opuntiae (Cockerell) en Opuntia ficus-indica (L.) Miller en el centro de México. Acta Zool Mex 26:415–433
Vigueras AL, Cibrián-Tovar J, Pelayo-Ortiz C (2009) Use of botanical extracts to control wild cochineal (Dactylopius opuntiae Cockerell) on cactus pear. Acta Hortic (811):229–234
Weissenberg M, Klein M, Meisner J, Ascher KRS (1986) Larval growth inhibition of the spiny bollworm, Earias insulana, by some steroidal secondary plant compounds. Entomol Exp Appl 42:213–217
Yahia EM, Sáenz C (2011) Cactus pear (Opuntia species). In: Yahia EM (ed) Postharvest Biology and Technology of Tropical and Subtropical Fruits. Woodhead Publishing, Sawston UK, pp 290–331e
Zewdie Y, Bosland PW (2001) Capsaicinoid profiles are not good chemotaxonomic indicators for Capsicum species. Biochem Syst Ecol 29:161–169. https://doi.org/10.1016/S0305-1978(00)00041-7
Acknowledgments
The authors thank Dr. Zakaria Kehel for statistical help with data analysis, Dr. Karim El Fakhouri and Dr. Abdelhadi Sabraoui for their help with field work.
Funding
This research was partially funded by FAO under the project TCP/MOR/3604.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have NO competing financial interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ramdani, C., Bouharroud, R., Sbaghi, M. et al. Field and laboratory evaluations of different botanical insecticides for the control of Dactylopius opuntiae (Cockerell) on cactus pear in Morocco. Int J Trop Insect Sci 41, 1623–1632 (2021). https://doi.org/10.1007/s42690-020-00363-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-020-00363-w