Abstract
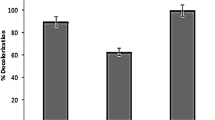
The present study evaluated the efficiency of combined biological and AOPs treatment (Bio–AOP) using Aeromonas hydrophila SK16 and AOPs-H2O2 (4%) for the remediation of the textile dyes. Bio–AOP treatment showed 100% decolorization of Reactive Red 180 (RR 180), Reactive Black 5 (RB 5) and Remazol Red (RR), while 72% decolorization was observed in individual treatments. Combined treatment significantly reduced BOD and COD of RR 180–78 and 68%, RB 5–52 and 83% and RR—42 and 47%, respectively as compare to individual treatment. Significant increased levels of tyrosinase, laccase, lignin peroxidase, riboflavin reductase and azoreductase were observed in A. hydrophila SK16. Fourier-transform infrared spectroscopy and high-performance liquid chromatography analysis showed noteworthy biotransformation of textile dyes. Possible metabolic pathway of degradation of dyes were predicted based on GC–MS analysis. This study indicates that the Bio–AOP treatment is more efficient than an individual treatment of textile wastewater.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Textile industries are using structurally different varieties of reactive dyes for accomplishment of many shades of color, stronghold profiles, comfort to use, less energy utilization and excellent colors. The more commonly used dyes are anthraquinone, azo dyes (N=N) and phthalocyanine. These coloring agents are become serious threats to all life form as they are carcinogenic and mutagenic in nature [28]. More than 100,000 commercial dyes are available in market whose annual production is more than one million tons. Out of all the dyes used for dying and printing, around 20% of dyes are being discharged into the environmental sink because of its low level of affinity with the fabrics [20]. The aromatic amines produced after Azo dyes biotransformation has mutagenic and carcinogenic effects [12]. The discharge of these effluent containing dyes into the water resources results in alteration of pH, reduction in light penetration and gas solubility, as well as raise in the COD, BOD, TOC of water resources that causes inimical effects on all life form including animals and plants [34, 53]. So, the management of textile effluent is inevitable before final discharge to the ecosystem.
Many studies have been conducted on the consequence of biological, chemical and physical treatments of textile wastewater. Treatment of textile wastewater with already existing physicochemical methods is inefficient due to its inability to reduce COD, TOC, BOD, color, pH and metals. These techniques are costly and generate huge quantity of toxicants and sludge [7]. Textile wastewater treatment using biological method is an ecofriendly and low cost treatment methods [5]. Many biological agents such as bacteria, yeasts, fungi, algae and actimonycetes are capable of degrading azo dyes, among which bacterial cells represent an inexpensive and promising tool for the removal of different azo dyes from textile effuluent [42]. Different taxanomic group of bacteria repored for dye degradation [6] (Aeromonas hydrophila); [54] (Bacillus cereus); [17] (Halomonas sp.) and [18] (Serratia liquefaciens). All biological treatments involve use of biocatalysts to oxidize recalcitrant pollutants. But, they have some drawbacks like low biodegradability [50] and no efficient removal of refractory compounds [44].
AOPs are set of chemical treatment planned to remove organic and some inorganic waste from water by hydroxyl radical (·OH) oxidation [32]. The ·OH production from H2O2 catalyzes with the help of UV light by hemolytic O–O bond cleavage. The most current developments in AOPs is the use of solar radiation, as UV photon source, decreasing artificial energy requirement for the application of these methods [51]. These reactive species (·OH) are potent oxidant can react with any kind of pollutant dissolved in water matrix. Initially they attack on larger molecules to breakdown into fragments then subsequently attack until the ultimate mineralization.
At an initial stage, AOPs aimed to improve biological treatment [8]. In the later stage, the oxidation process is used for the removal of those contaminants which are not completely degraded in the biological treatment process [39]. The prebiological treatment responsible for breakdown of strong bonds like azo and utilize biodegradable compounds. Later advance oxidation treatment deal with toxic aromatic amines as well as other recalcitrant metabolites produced by bioremediation process [35]. In addition to these the use of AOPs as pretreatment could results stable intermediates formation that are more toxic to biological system as well as less biodegradable than the original molecule [33]. Moreover, high quantity use of oxidants used for oxidation also lead to creation of condition that are toxic to microorganisms [21]. Pretreatment of AOPs is become expensive as they required lots of chemicals, therefore it was suggested that prebiological treated wastewater is beneficial to use for AOPs treatment [1, 40, 43]. Other oxidation techniques like Fenton oxidation process and sonolysis are also available. But, sonolysis require high energy inputs and use of Fenton oxidation technique combined with biological process is limited due to low pH 2 and 3. At the same condition use of UV/H2O2 is best option instead of these techniques for wastewater treatment [52].
The combinatorial approach executed as bioremediation couple with advance oxidation process with Aeromonas hydrophila SK16 was thought to have additional benefit of faster treatment efficiency. Both bioremediation and AOP involve oxidation process to degrade waste. The several studies have been reported about successive oxidative treatment with chemicals and organisms [45]. The pilot scale wastewater treatment by Photo-Fenton oxidation coupled with bioremediation showed 94% mineralization of effluent as well as found to reduced 96% toxicity [55]. The study carried out by Mandal et al. [31] showed combined AOP and biological treatment help out in reduction of COD and BOD. Several studies have been reported about successive oxidative treatment with chemicals and organisms [15]. These reports are precisely on the use of combination of AOPs and biological treatment for textile wastewater management.
In present study, we have focused on an evaluation of efficiency of combined biological (Aeromonas hydrophila SK16 bioremediation) and AOPs treatment using H2O2 for treating various textile dyes. This combined biological and AOPs approach is useful for removing contaminant from environment and reducing health associated problems. This combined process is easily adaptable in large scale for significant decontamination.
2 Materials and methods
2.1 Chemicals and dyes
The dyes Remazol Red (RR), Reactive Red 180 (RR 180) and Reactive Black 5 (RB 5) (Table 1) were obtained from Jamara Textile Industry SIPCOT Perundurai, Tamil Nadu, India. Decolorization studies were performed in Nutrient broth (Himedia, India). Hydrogen peroxide was procured from Merck, India. All reagents and chemicals were highly pure.
2.2 Culture condition and decolorization studies
Bacterial strain A. hydrophila SK16 was isolated earlier from the textile dye polluted soil [26]. The pure strain was maintained at 4 °C on nutrient agar. A. hydrophila SK16 was revived and used for the present study after examining its purity.
Decolorization studies of RR 180, RB 5 and RR were carried out in 100 ml nutrient broth (Beef extract—1.5 g, Yeast extract—1.5 g, Peptone—5 g and NaCl—5 g for 1 L). The concentration of 100 mg L−1 dyes was added in log phase culture after acquiring the bacterial count at 104–105 CFU. The experiments were executed in both static and shaking states at 37 °C. The H2 ion concentration of the pre-grown culture was optimized and adjusted to 8. Aliquot (5 ml) was withdrawn at every 2 h intervals, centrifuged (10,000×g, 15 min) and color removal was determined by computing λmax of the dyes. Abiotic control (without microorganisms) was kept along with every sets. All tests were executed in three sets and % decolorization was determined using following Eq. 1,
2.3 Advanced oxidation process
The advanced oxidation process was performed in 250 ml conical flask containing dye (100 mg L−1) and H2O2 (4%, based on the optimization from 1 to 10%). The flask was irradiated under solar light for 6 h. The same procedure was followed for dark conditions. Control was used without H2O2. The light intensity was measured by using Lux Meter (Lutron LX-101, Taiwan).
2.4 Combined biological and advanced oxidation process
The biomass was removed after biodegradation with the help of centrifugation. The remaining dye solution was exposed to AOPs treatment using 4% H2O2 (based on the optimization from 1 to 10%). The sample was exposed under sunlight for 3–6 h and then analyzed using UV–Vis Spectrophotometer. The intensity of light was measured using Lux Meter (Lutron LX-101, Taiwan).
2.5 BOD and COD determination of dye solution
BOD-Biological oxygen demand and COD-Chemical oxygen demand were determined using standard procedures according to APHA [3].
2.6 Sample preparation for enzyme analysis
Aeromonas hydrophila SK16 was grown in 100 ml nutrient medium for 24 h at 37 °C. The centrifuged (10,000 rpm, 15 min) pellet was resuspended in potassium phosphate buffer for sonication (40 A, 8 strokes of 40 s using sonics-vibracell). The prepared sample was used as source of enzyme. The same method of enzyme preparation was carried out for control as well as treated cell (dye exposed).
2.7 Enzyme studies
The assays of enzyme lignin peroxidase, tyrosinase, riboflavin reductase, NADH-DCIP reductase, azoreductase and laccase were carried out spectrophotometrically using crude source of enzyme. Lignin peroxidase assay was performed in a total 2.5 ml volume comprising tartaric acid (250 mM) and n-propanol (100 mM). The propanaldehyde production was estimated at 300 nm as reported by [24]. The assay of laccase was carried out in a reaction mixture of 2 mL containing 0.1 M acetate buffer (pH 4.9) with 10% ABTS and an increase in the absorbance was measured at 420 nm [19]. Tyrosinase activity was calculated in a reaction mixture of 2 mL, containing in 0.1 M phosphate buffer (pH 7.4) with 0.01% catechol at 495 nm [56]. Azoreductase assay was carried out using Methyl red as substrate as reported by Kurade et al. [27]. Riboflavin reductase activity was determined using riboflavin as substrate as mentioned in previously reported method [41]. Protein estimation was done by using Lowry et al. [30].
2.8 Extraction and analytical studies of metabolites obtained after degradation
The decolorization study was evaluated by observing the change in spectra before and after biotransformation of dye with the help of spectrophotometer (Shimadzu 1800, Japan). The supernatant was obtained from culture by centrifugation after complete decolorization of RR 180, RB 5 and RR. The metabolites were extracted using ethyl acetate (1:1, v/v). The evaporation of extracts was done using Na2SO4 in a rotary evaporator. The residual solids were mixed in methanol (grade one) and subjected to different analytical systems. FTIR analysis was carried out to find out the functional groups of control and biodegraded sample. The analysis was carried out with 20 scan at infrared region (4000–400 cm−1). The samples of FTIR were prepared in high grade potassium bromide (5:95). The Shimadzu LC 40102010 instrument was used for HPLC study connected with C18 column. The solvent methanol was used in mobile phase with 1 ml min−1 flow rate and analysis was done at 470 nm.
Identification of degraded product after biotransformation was performed with the help of 45XGC-44 GC–MS (Bruker). The helium gas (mobile phase) was used for 26 min with 1 ml min−1 flow rate. The oven temperature conditions was primarily maintain for 120 s at 80 °C then progressively increased by 10 °C for each 60 s up to 250 °C and maintain for 26 min. The formed products after transformation were recognized with the help of mass spectra using the NIST library. The elucidation of intermediate product produced, and degradative pathway of dyes was proposed using a tool Chemsketch 2.0.
3 Results and discussion
3.1 Biological degradation of RR 180, RB 5 and RR by A. hydrophila SK16
The potential of A. hydrophila SK16 to decolorize various dyes such as RR 180, RB 5 and RR were studied in both static and shaking conditions. Present study represented the color removal capability of pure culture with respect to several textile dyes. The static condition showed noteworthy decolorization of RR 180, RB 5 and RR than shaking (91%; 50%), (90%; 72%) and (90%; 26%), respectively in 9 h (Fig. S1) (Table 2). Similarly, Pseudomonas sp. SUK1 also showed significant increased growth in shaking condition than static, but was not found decolorization even up to 48 h [23]. The dye Crystal violet was also removed by 100% at static condition [11]. The race held among dye and O2 aimed for reduced e− carrier could be possible reason for reduced decolorization in aerobic condition [22] or azo bond reduction activity was inhibited by the existence of O2 under shaking, because the aerobic respiration predominates usefulness of nicotinamide adenine dinucleotide (NADH), thus retard transferring of e− towards azo bonds [10]. As the decolorization efficiency was achieved at pH 8 and it is most appropriate for treatment of textile wastewater due to its alkaline condition [13]. Thus, these results concluded significant decolorization of RR 180, RB 5 and RR by A. hydrophila SK16 in static condition than shaking.
3.2 Decolorization of RR 180, RB 5 and RR by advanced oxidation process (AOPs)
The H2O2 concentration influences the efficiency of decolorization. AOPs showed 83, 37 and 68% decolorization of RR 180 (Fig. 1A, A.1.b), RB 5 (Fig. 1B, B.1.b) and RR (Fig. 1C, C.1.b), respectively, at 4% H2O2 and solar light exposure of 6 h (Table 2). Under AOPs, 30% color removal was observed by using TiO2 alone [1], thus, the single treatment of AOPs results incomplete degradation [36]. No color change was observed in the dark condition without using H2O2 [38].
The result of several researchers such as Almomani et al. [2], Foteinis et al. [14] and Rani and Shanker [37] reveals contribution of solar light is more in mineralization of wastewater and textile dyes. When the photon energy (hv from UV range of sunlight) of solar light is absorbed by H2O2 which leads to generation of hydroxyl radicals (OH·). The hydroxyl radicals destroys the organic pollutants. All the dyes are not degraded under AOPs especially structurally and chemically complex dyes. So, that combined treatment could overcome all the limitations which are occurred from other treatments.
3.3 Decolorization of RR 180, RB 5 and RR by combined biological–AOPs treatment
The culture broth was centrifuged after biological treatment to separate biomass and used to optimize the concentration of H2O2. The supernatant was subjected for AOPs with various concentrations of H2O2 (1–10%) in 250 ml conical flask having 100 ml of supernatant. This mixture was subjected for treatment under sunlight. The decolorization of RR180 (Fig. 1A, A.1.d), RB 5 (Fig. 1B, B.1.d) and RR (Fig. 1C, C.1.d) was increased to 100% using 4% of H2O2 within 180 min (Table 2). This decoloization efficiency was better than the combined treatment (biological and AOPs) for RB5, RB13 and AO7 reported by Tantak and Chaudhari [[47]]. In another study, combined AOPs and biological treatment showed 99% color reduction [29]. The color removal was not effectual in the concentration range of 1–3% of H2O2. The decolorization efficiency was improved from 4% of H2O2. Combined treatment method was found to produce satisfactory results [4]. As per our finding, the combined Bio–AOPs treatment was more significant than single biological or AOPs processes for decolorization purposes.
3.4 Effect of combined Bio–AOPs treatment on BOD and COD of RR 180, RB 5 and RR after degradation
The sample was tested for BOD and COD reduction to assess the efficiency of Bio–AOPs system after decolorization. The BOD and COD of RR 180 were found to be reduced by 16; 10, 25; 24 and 78; 67% by the AOPs-H2O2, Biological-A. hydrophila SK 16 and Bio–AOPs, respectively (Fig. 2a). In case of RB 5, the BOD and COD were found to be reduced by 13; 35, 21; 52 and 55; 85% by the AOPs-H2O2, Biological-A. hydrophila SK 16 and Bio–AOPs, respectively (Fig. 2b). Also, BOD and COD of RR were found to be reduced by 11; 8, 22; 26 and 42; 45% by the AOPs-H2O2, Biological-A. hydrophila SK 16 and Bio–AOPs, respectively (Fig. 2b) (Table 2).
The above all results were showed that the individual AOPs and biological treatment is inadequate for textile dyes remediation, while combined Bio–AOPs treatment showed significant remediation. Both methods have some different limitation in waste treatment therefore, individually they unable to treat the dyes at maximum extent. In case of combined Bio–AOPs initially dye was degrade by A. hydrophila at some extent further AOPs was deal with the bio-recalcitrant compounds and showed maximum mineralization of all textile dyes. The significant reduction of BOD, COD and color in wastewater up to 98, 93 and 100%, respectively were observed during combined treatment process by Thiobaccilus ferrooxidans ATCC19859 [31]. The treatment with combined Fenton oxidation and biological treatment showed 98% COD removal of wastewater [48]. The COD (83%), BOD (88%), and total suspended solids (98%) were significantly reduced by combined treatment in non-biodegradable textile effluent [16]. The combined treatment not only achieve decolorization efficiency, but also contribute for the reduction of the treatment cost [9, 25].
3.5 Induction of enzyme activities after decolorization of RR 180, RB 5 and RR by A. hydrophila SK16
Enzyme studies exhibited the participation of number dye degrading biocatalyst like lignin peroxidase, tyrosinase, azo reductase, riboflavin reductase and laccase. The participation of these biocatalyst during color removal was documented by Telke et al. [49]. The levels of enzyme were meaningfully increased during dye transformation indicates complex mechanism of enzymes of dye degradation. In this study, activity of enzymes before and after decolorization of RR 180, RB 5 and RR were measured and found to be induced after decolorization. The decolorization of RR 180, RB 5 and RR showed induced activities of lignin peroxidase from 237.70, 121.84 and 82.59 to 536.99, 298.58 and 634.91 U ml−1min−1 (130, 145 and 669%)respectively. Laccase showed induction from 0.13, 0.03 and 0.06 to 0.22, 0.13 and 0.08 U ml−1 min−1 (69, 303 and 33%) after exposed to RR 180, RB 5 and RR, respectively. The activity of tyrosinase was elevated from 4.70, 1.33 and 0.30 to 28.86, 4.19 and 2.05 U ml−1 min−1 (514, 214 and 583%) by RR 180, RB 5 and RR exposure, respectively. The RR 180, RB 5 and RR dyes exposure was found to increase in the azo reductase from 91.12, 11.35 and 15.88 to 211.24, 19.47 and 78.05 µmol NADH reduced min−1 mg protein−1 (132, 72 and 391%) and riboflavin reductase from 38.34, 23.32 and 35.82 to 151.09, 98.24 and 40.28 µg of riboflavin reduced min−1 mg protein −1 (294, 321 and 12%), respectively (Table 3). Thus, our finding stated that, RR 180, RB 5 and RR significantly degraded by enhancing dye degrading enzymes activity in A. hydrophila SK16 at static condition.
3.6 Effect of combined Bio–AOPs treatment on RR 180, RB 5 and RR
The spectral scrutiny (350–750 nm) was done to confirm transformation of RB 5, RR 180 and RR. The absorption spectra after decolorization of RR 180, RB 5 and RR by AOPs (H2O2), biological and combined Bio–AOPs along with control showed complete reduction of RR 180 (Fig. 3A), RB 5 (Fig. 3B) and RR (Fig. 3C) due to combined Bio–AOPs. These UV–Vis spectral results stated that combined Bio–AOPs method is more noteworthy than individual one for the removal of dye contaminant from wastewater.
3.7 Spectroscopic and chromatographic analysis of decolorized product after combined Bio–AOPs treatment
3.7.1 FTIR analysis
The transformation of textile dye into different degraded product was evaluated by Fourier-transform infrared spectroscopy (FTIR) spectral analysis via comparison between RR 180, RB 5 and RR and its degraded products produced by A. hydrophila SK16 at static condition within 24 h. The FTIR spectra of RR 180 before biodegradation and after biodegradation differed with number of peaks and their position. The FTIR spectrum of untreated RR 180 indicated number peak at 3413.22 cm−1 representing N–H stretching (str) of secondary amides and 1758.41 cm−1 representing C=O str. of amide. The peak at 1621.24 cm−1 confirmed the presence of azo (N=N) bond by a while, a peak at 1553.09 cm−1 represent N–H deformation (def). The peak at 1462.22 cm−1 represented N=O str. of nitrosamine, and peak at 1131.86 cm−1 C–H def. in tri-substituted benzenes. The presence of sulfur group is represented by a peak at 1043.36 cm−1 for S=O str. of sulfonic acid, while a peak at 836.71 cm−1 representing C–H def. of benzene ring possessing 2 adjacent H2 atoms (Fig. S2A). Whereas, the products of the dye formed after decolorization by A. hydrophila SK16 showed a peak at 3244.65 cm−1 revealed O–H str. and 2417.27 cm−1 representing NH+ str. of charged amines. Although a peak at 1595.42 cm−1 representing NH3+ def. of charged amine derivatives, while a peak at 1410.66 cm−1 representing S=O str. of sulfites. The peak at 1115.42 cm−1 revealed C–OH str. of 2° alcohols and at 989.28 cm−1 revealed C–H def. of mono-substituted alkanes (Fig. S2B). The arrival several new peaks in biotransformed sample and disappearing of peaks in untreated spectra confirmed the biodegradation of dye RR 180 by A. hydrophila SK1.
Similarly, FTIR spectral analysis of RB 5 before biodegradation showed peaks at 3435.71, 2418.55, 1813.31, 1591.10, 1492.15 and 1418.68 cm−1 which represents the presence of N–H str., O–H def., C=O symmetric str., –N=N– azo bond, aromatic compound, C–H def. of alkanes, respectively. Peaks at 1050.27 and 631.90 cm−1 showed –C=O str. and –C=S str. indicates the presence of sulfur containing groups. Peaks at 1130.59 and 839.92 cm−1 represents C–H def., a peak at 895.28 cm−1 for aromatic and a peak at 631.90 cm−1 showed C–S sulfur group presents in control dye (Fig. S3A). After biodegraded product showed various peaks at 3273.37 cm−1 for O–H str., C=O str. at 1658.46 cm−1, a peak at 1110.14 cm−1 for C–OH secondary alcohol. The disappearance of 1591.10 cm−1 in degraded metabolites confirms azo bond cleavage. Peaks at 983.15, 866.13 and 619.09 cm−1 represent C–OS stretching, C–HS stretching and C–H deformation (Fig. S3B). Finally, disappearance of peaks and formation of new peaks in metabolites confirms biodegradation of RB 5.
Difference between parent dye and metabolite produced after 9 h of RR revealed biodegradation. Parent dye represents NH group at 3415.31 cm−1, a peak at 2925.76 cm−1 for C–H alkane stretching, C–H stretching at 1756.05 cm−1, a peak at 1597.78 cm−1 for –N=N– azo bond, a peak at 1489.79 cm−1 for C=C stretching (aromatic compound). Peaks at 1407.47, 1319.41, 1291.04, 1126.64, 1042.82, 843.49 and 622.21 cm−1 represents O–H str. vibration (vib), C–N vib., O–NO2 vib., C–O str., C–S sulfonated (aromatic compound), and C–H def., respectively. Peaks at 793.08 and 743.51 cm−1 represented for C–H bending (Fig. S4A). biotransformed RR showed vanishing of major peaks and appearance of new peaks 3294.46 cm−1 for O–H str., a peak at 1648.22 cm−1 for C=N str., a peak at 1458.19 cm−1 for C–H def. (alkanes), respectively (Fig. S4B).
3.7.2 HPLC analysis
HPLC profile of the parent dye represented major peak with at retention time (Rt) of 2.101 min (Fig. 4a), while biotransformed dye by A. hydrophila SK16 represent 4 new peaks with the Rt of 1.499, 1.820, 3.564 and 3.805 min (Fig. 4b). It is the confirmation of degradation of RR 180 by A. hydrophila SK16 by appearing of some new peaks at different retention time of some new metabolites. Although, analysis of RB 5 before degradation represent main peak at 2.141 min and a slight peak at 6.113 min Rt (Fig. 4c), however in biodegraded sample represent 4 new peaks at 1.766, 1.918, 2.752 and 3.192 min Rt (Fig. 4d) confirmed biodegradation of RB 5. Similarly, analysis of RR represents a main peak at 2.192 min and a slight peak at 6.202 min Rt (Fig. 4e). Biodegraded sample represents 4 different peaks at 1.487, 1.835, 3.236 and 3.697 min Rt suggested biodegradation of RR (Fig. 4f).
Profile of HPLC elution at 470 nm of dyes a Reactive Red 180 and b extracted metabolic products of Reactive Red 180 after decolorization by A. hydrophila SK16; c Reactive Black 5 and d extracted metabolites of Reactive Black 5 after decolorization by A. hydrophila SK16; e Remazol Red RR and f extracted metabolites of Remazol Red RR after decolorization by A. hydrophila SK16
3.8 The proposed pathways of dyes transformation
Fate of metabolism of RR 180 by A. hydrophila SK16 is proposed on the basis of increased level of enzyme A. hydrophila SK16 and the GC–MS profiling. Initially, by the action of azo reductase on azo bond, it leads to the production of transitional compound [A] and [B]. Desulfonation of compound [A] provided an unidentified product. Asymmetric cleavage of laccase on Intermediate [B] gave compound [C] and [D] which was identified as benzamide [Rt 21.093 min; Mol.wt-121; m/z-123]. Further, desulphonation and deamination of Intermediate [C] led to formation of naphthalene-2-thiol [Rt 20.673 min; Mol.wt-160; m/z-157] (Fig. 5). The asymmetrical cleavage of azo dyes is well studied action of laccase [46].
In case of RB 5, initially azo reductase activity breaks azo linkage leads the production of Intermediate [I] and [II]. Deionization of Intermediate [I] resulted in the formation of 2-[(4-aminophenyl)sulfonyl]ethyl sulfate. Cleavage of Intermediate [II] by laccase formed Intermediate [III] and [IV]. The Intermediate [III] delaminated to the formation of 4-dihydronaphthalene-2-sulfonate. Further, breakdown of Intermediate [IV] in the formation of 3,5,6-triamino-3,4-dihydronaphthalene-2-sulfonate was through desulfonation and deionization. Sequential deamination produced 5-amino-3,4-dihydronaphthalene-2-sulfonate and 3,4-dihydronaphthalene-2-sulfonate as final product (Fig. 6). Similarly, GCMS profiling was done to analyze the biotransformed product during degradation of RR and identified the metabolites as 3-diazenyl-4-methoxyphenol, (2-methoxyphenyl)diazene, methoxybenzene, phenol, naphthalene-1-sulfonic acid and naphthalene (Fig. 7).
4 Conclusion
This combined Bio–AOPs treatment not only achieves significant decolorization, but also contributes towards reduction in BOD and COD of wastewater. It is more efficient than an individual treatment. It is reliable, efficient, passive, economical, feasible solar energy dependent and requires less maintenance for treatment of textile dyes and wastewater as compared to individual biological and advanced oxidation process.
References
Ahmadi M, Amiri P, Amiri N (2015) Combination of TiO2-photocatalytic process and biological oxidation for the treatment of textile wastewater. Korean J Chem Eng 32(7):1327–1332
Almomani FA, Bhosale RR, Khraisheh MAMM, Kumar A, Kennes C (2018) Mineralization of dichloromethane using solar-oxidation and activated TiO2: pilot scale study. Sol Energy 172(2):116–127
APHA (1998) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association, Washington, D.C.
Arslan-Alaton I, Cokgor EU, Koban B (2007) Integrated photochemical and biological treatment of a commercial textile surfactant: process optimization, process kinetics and COD fractionation. J Hazard Mater 146(3):453–458
Bedekar PA, Saratale RG, Saratale GD, Govindwar SP (2014) Development of low cost upflow column bioreactor for degradation and detoxification of Blue HERD and textile effluent by Lysinibacillus sp. RGS immobilized on Loofa. Int Biodeterior Biodegradation 96:112–120
Bharagava RN, Mani S, Mulla SI, Saratale GD (2018) Degradation and decolourization potential of an ligninolytic enzyme producing Aeromonas hydrophila for crystal violet dye and its phytotoxicity evaluation. Ecotoxicol Environ Saf 156(7):166–175
Bhargava DA (2016) Physico-chemical waste water treatment technologies: an overview. Int J Sci Res Educ 4(5):5308–5319
Bila DM, Filipe Montalvão A, Silva AC, Dezotti M (2005) Ozonation of a landfill leachate: evaluation of toxicity removal and biodegradability improvement. J Hazard Mater 117(3):235–242
Brosillon S, Djelal H, Merienne N, Amrane A (2008) Innovative integrated process for the treatment of azo dyes: coupling of photocatalysis and biological treatment. Desalination 222(3):331–339
Chang JS, Lin CY (2001) Decolorization kinetics of a recombinant Escherichia coli strain harboring azo-dye-decolorizing determinants from Rhodococcus sp. Biotech Lett 23(8):631–636
Chen CH, Chang CF, Ho CH, Tsai TL, Liu SM (2008) Biodegradation of crystal violet by a Shewanella sp. NTOU1. Chemosphere 72(11):1712–1720
Chequer FMD, Lizier TM, de Felício R, Zanoni MVB, Debonsi HM, Lopes NP et al (2011) Analyses of the genotoxic and mutagenic potential of the products formed after the biotransformation of the azo dye Disperse Red 1. Toxicol Vitro 25(8):2054–2063
Dawkar VV, Jadhav UU, Jadhav SU, Govindwar SP (2008) Biodegradation of disperse textile dye Brown 3REL by newly isolated Bacillus sp. VUS. J Appl Microbiol 105(1):14–24
Foteinis S, Borthwick AGL, Frontistis Z, Mantzavinos D, Chatzisymeon E (2018) Environmental sustainability of light-driven processes for wastewater treatment applications. J Clean Prod 182:8–15
Gaya UI, Abdullah AH (2008) Heterogeneous photocatalytic degradation of organic contaminants over titanium dioxide: a review of fundamentals, progress and problems. J Photochem Photobiol C 9(1):1–12
Ghoreishi SM, Haghighi R (2003) Chemical catalytic reaction and biological oxidation for treatment of non-biodegradable textile effluent. Chem Eng J 95(3):163–169
Guadie A, Gessesse A, Xia S (2018) Halomonas sp. strain A55, a novel dye decolorizing bacterium from dye-uncontaminated Rift Valley Soda lake. Chemosphere 206(5):59–69
Haq I, Raj A (2018) Biodegradation of Azure-B dye by Serratia liquefaciens and its validation by phytotoxicity, genotoxicity and cytotoxicity studies. Chemosphere 196(7):58–68
Hatvani N, Mécs I (2001) Production of laccase and manganese peroxidase by Lentinus edodes on malt-containing by-product of the brewing process. Process Biochem 37(5):491–496
Jadhav JP, Phugare SS, Dhanve RS, Jadhav SB (2010) Rapid biodegradation and decolorization of Direct Orange 39 (Orange TGLL) by an isolated bacterium Pseudomonas aeruginosa strain BCH. Biodegradation 21(3):453–463
Jonstrup M, Punzi M, Mattiasson B (2011) Comparison of anaerobic pre-treatment and aerobic post-treatment coupled to photo-Fenton oxidation for degradation of azo dyes. J Photochem Photobiol A 224(1):55–61
Kalme S, Ghodake G, Govindwar S (2007) Red HE7B degradation using desulfonation by Pseudomonas desmolyticum NCIM 2112. Int Biodeterior Biodegrad 60(4):327–333
Kalyani DC, Telke AA, Dhanve RS, Jadhav JP (2009) Ecofriendly biodegradation and detoxification of Reactive Red 2 textile dye by newly isolated Pseudomonas sp. SUK1. J Hazard Mater 163(3):735–742
Kalyani DC, Phugare SS, Shedbalkar UU, Jadhav JP (2011) Purification and characterization of a bacterial peroxidase from the isolated strain Pseudomonas sp. SUK1 and its application for textile dye decolorization. Ann Microbiol 61(3):483–491
Kiran S, Ali S, Asgher M (2013) Degradation and mineralization of azo dye reactive blue 222 by sequential photo-fenton’s oxidation followed by aerobic biological treatment using white rot fungi. Bull Environ Contam Toxicol 90(2):208–215
Kumar SS, Shantkriti S, Muruganandham T, Murugesh E, Rane N, Govindwar SP (2016) Bioinformatics aided microbial approach for bioremediation of wastewater containing textile dyes. Ecol Inform 31:112–121
Kurade MB, Waghmode TR, Tamboli DP, Govindwar SP (2013) Differential catalytic action of Brevibacillus laterosporus on two dissimilar azo dyes Remazol red and Rubine GFL. J Basic Microbiol 53(2):136–146
Lade H, Kadam A, Paul D, Govindwar S (2015) Biodegradation and detoxification of textile azo dyes by bacterial consortium under sequential microaerophilic/aerobic processes. EXCLI J 14(10):158–174
Lodha B, Chaudhari S (2007) Optimization of Fenton-biological treatment scheme for the treatment of aqueous dye solutions. J Hazard Mater 148(1–2):459–466
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin-phenol reagent. J Biol Chem 193:265–275
Mandal T, Maity S, Dasgupta D, Datta S (2010) Advanced oxidation process and biotreatment: their roles in combined industrial wastewater treatment. Desalination 250(1):87–94
Mantzavinos D, Psillakis E (2004) Enhancement of biodegradability of industrial wastewaters by chemical oxidation pre-treatment. J Chem Technol Biotechnol 79(5):431–454
Oller I, Malato S, Sánchez-Pérez JA (2011) Combination of advanced oxidation processes and biological treatments for wastewater decontamination: a review. Sci Total Environ 409(20):4141–4166
Phugare S, Patil P, Govindwar S, Jadhav J (2010) Exploitation of yeast biomass generated as a waste product of distillery industry for remediation of textile industry effluent. Int Biodeterior Biodegrad 64(8):716–726
Punzi M, Mattiasson B, Jonstrup M (2012) Treatment of synthetic textile wastewater by homogeneous and heterogeneous photo-Fenton oxidation. J Photochem Photobiol A 248:30–35
Ramteke LP, Gogate PR (2015) Removal of ethylbenzene and p-nitrophenol using combined approach of advanced oxidation with biological oxidation based on the use of novel modified prepared activated sludge. Process Saf Environ Prot 95:146–158
Rani M, Shanker U (2018) Sun-light driven rapid photocatalytic degradation of methylene blue by poly(methyl methacrylate)/metal oxide nanocomposites. Colloids Surf A 559:136–147
Riga A, Soutsas K, Ntampegliotis K, Karayannis V, Papapolymerou G (2007) Effect of system parameters and of inorganic salts on the decolorization and degradation of Procion H-exl dyes. Comparison of H2O2/UV, Fenton, UV/Fenton, TiO2/UV and TiO2/UV/H2O2 processes. Desalination 211(1–3):72–86
Rizzo L (2011) Erratum to “Bioassays as a tool for evaluating advanced oxidation processes in water and wastewater treatment” [Water Research 45 (2011) 4311–4340]. Water Res 45(17):5805
Rueda-Márquez JJ, Sillanpää M, Pocostales P, Acevedo A, Manzano MA (2015) Post-treatment of biologically treated wastewater containing organic contaminants using a sequence of H2O2 based advanced oxidation processes: photolysis and catalytic wet oxidation. Water Res 71:85–96
Russ R, Rau J, Stolz A (2000) The function of cytoplasmic flavin reductases in the reduction of azo dyes by bacteria. Appl Environ Microbiol 66(4):1429–1434
Saratale RG, Saratale GD, Chang JS, Govindwar SP (2011) Bacterial decolorization and degradation of azo dyes: a review. J Taiwan Inst Chem Eng 42(1):138–157
Suryaman D, Hasegawa K, Kagaya S (2006) Combined biological and photocatalytic treatment for the mineralization of phenol in water. Chemosphere 65(11):2502–2506
Sutar H, Das C (2015) International journal of research in chemistry and environment phytoremediation: a review focusing on phytoremediation mechanisms. Int J Res Chem Environ 5(3):1–9
Tabrizi GB, Mehrvar M (2004) Integration of advanced oxidation technologies and biological processes: recent developments, trends, and advances. J Environ Sci Health A Toxic Hazard Subst Environ Eng 39(11–12):3029–3081
Tamboli DP, Kurade MB, Waghmode TR, Joshi SM, Govindwar SP (2010) Exploring the ability of Sphingobacterium sp. ATM to degrade textile dye Direct Blue GLL, mixture of dyes and textile effluent and production of polyhydroxyhexadecanoic acid using waste biomass generated after dye degradation. J Hazard Mater 182(1–3):169–176
Tantak NP, Chaudhari S (2006) Degradation of azo dyes by sequential Fenton’s oxidation and aerobic biological treatment. J Hazard Mater 136(3):698–705
Tekin H, Bilkay O, Ataberk SS, Balta TH, Ceribasi IH, Sanin FD et al (2006) Use of Fenton oxidation to improve the biodegradability of a pharmaceutical wastewater. J Hazard Mater 136(2):258–265
Telke AA, Kalyani DC, Jadhav UU, Parshetti GK, Govindwar SP (2009) Purification and characterization of an extracellular laccase from a Pseudomonas sp. LBC1 and its application for the removal of bisphenol A. J Mol Catal B Enzymatic 61(3–4):252–260
Torrades F, García-Montaño J, García-Hortal JA, Domènech X, Peral J (2004) Decolorization and mineralization of commercial reactive dyes under solar light assisted photo-Fenton conditions. Sol Energy 77(5):573–581
Vilar VJP, Pinho LX, Pintor AMA, Boaventura RAR (2011) Treatment of textile wastewaters by solar-driven advanced oxidation processes. Sol Energy 85(9):1927–1934
Vincenzo Naddeo AC (2013) Wastewater treatment by combination of advanced oxidation processes and conventional biological systems. J Bioremediat Biodegrad 04(08):222–230
Waghmode TR, Kurade MB, Govindwar SP (2011) Time dependent degradation of mixture of structurally different azo and non azo dyes by using Galactomyces geotrichum MTCC 1360. Int Biodeterior Biodegrad 65(3):479–486
Wanyonyi WC, Onyari JM, Shiundu PM, Mulaa FJ (2017) Biodegradation and detoxification of malachite Green Dye using novel enzymes from Bacillus cereus Strain KM201428: kinetic and metabolite analysis. Energy Proc 119:38–51
Zapata A, Malato S, Sánchez-Pérez JA, Oller I, Maldonado MI (2010) Scale-up strategy for a combined solar photo-Fenton/biological system for remediation of pesticide-contaminated water. Catal Today 151(1–2):100–106
Zhang X, Flurkey WH (1997) Phenoloxidases in Portabella mushrooms. J Food Sci 62(1):97–100
Acknowledgements
The authors are thankful and would like to acknowledge the Science and Engineering Research Board (SERB), Department of Science and Technology (DST), Govt. of India, New Delhi for funding the research project under Fast Track Scheme for Young Scientist. Project Ref. No: SR/FT/LS/-121/2011 Dt. 29.05.2012. The authors are grateful to the Principal Dr. R. Sundararaman, Director Dr. K. Anbarasu and Shri. K. Ragunathan Secretary of National College for providing facilities. Suhas Kadam and Shivtej Biradar are thankful to UGC, New Delhi for providing fellowship (DRS-SAP II, Grant No. F.4-8/2015/DRS II (SAP-II)).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declares that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Thanavel, M., Kadam, S.K., Biradar, S.P. et al. Combined biological and advanced oxidation process for decolorization of textile dyes. SN Appl. Sci. 1, 97 (2019). https://doi.org/10.1007/s42452-018-0111-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-018-0111-y