Abstract
Brown (leaf) rust of wheat (Puccinia triticina) was widely prevalent in all the wheat growing areas of India and neighbouring countries during 2013–16. To have an effective rust management strategy, monitoring of virulence patterns of P. triticina on wheat were undertaken. Brown rust samples were analyzed from the wheat growing areas in India, Bangladesh, Bhutan and Nepal using standard differential Indian System and named as per the binomial system of nomenclature. A new pathotype 77-12 was also identified during this period. Thirty seven pathotypes of P. triticina were identified in 1427 samples. Five predominant pathotypes 121R63-1(77-5 = THTTM), 121R60-1(77-9 = MHTKL), 21R55 (104-2 = PHTTL), 21R63 (104-3 = PHTTL) and 77-11(125R28 = MGTTL) accounted for 75.1% of the population. All of the prevalent pathotypes are virulent to Lr1, Lr3, Lr10, Lr11,Lr12 Lr13, Lr14a, Lr16, Lr17, Lr23, Lr26. Virulences for Lr9 and Lr19 though recorded from India long back, however, were not observed during present surveys whereas virulence on Lr28 was identified in 0.4% samples only. The proportion of pathotype 121R60-1 (77-9 = MHTKL) has increased during these years and was recorded in 23.7% samples against 12% in the previous report. Brown rust of wheat was not observed on Lr9, Lr24, Lr25, Lr32, Lr39 and Lr45 in the field population of Indian subcontinent during 2013–16. Diversity of pathotypes detected during this period could, to some extent, be related to cultivation of different varieties but was also appeared to be affected by different weather patterns in different agro-ecological areas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rusts are very damaging pathogens of wheat worldwide. Brown (leaf) rust of wheat caused by Puccinia triticina occurs in all the wheat growing areas and causes more yield losses than any other rust (Samborski 1985). It is a quick evolving pathogen and generally renders rust resistant varieties of wheat susceptible in a short span of about 4–5 years (Sawhney 1995). Most of the resistance genes present in bread wheat are susceptible to brown rust in India. Even genes Lr9, Lr19, Lr26, Lr28 transferred from alien sources have also become susceptible to new virulences of P. triticina. Virulence to these genes emerged much before the varieties based on these genes were commercialized (Bhardwaj et al. 2016; Nayar et al. 2003). In India, brown rust mostly appears in late December in Peninsular India whereas in most of the wheat growing areas of Northern and Central India initial infections occur by the end of January or sometimes may be later. Among the various options, cultivation of resistant wheat is cost effective, environmentally safe and effective way to manage wheat brown rust. However, frequent emergence of new virulent pathotypes can overcome the hitherto functional resistance, and therefore, breeding wheat for rust resistance is an ever going process.
Keeping in view quick emergence of virulent pathotypes of P. triticina, monitoring of rust incidence, identification of new pathotypes and mapping of pathotype distribution is undertaken regularly. Information on racial distribution of P. triticina provides the background data for the identification of novel resistance, resistance breeding of wheat and pathotype situation based strategic deployment of wheat genotypes in different wheat growing areas. Variability in brown rust of wheat is being monitored in India for the last about 90 years and previous results of pathotype distribution of P. triticina were published in 2016 (Bhardwaj et al. 2016). Patterns of physiologic race diversity observed during 2013–16 in the Indian subcontinent are described in this publication.
Materials and methods
Brown rust samples of wheat were collected/received from different growing areas of India and neighbouring nations during 2013–16. The leaf scrapings of uredospore infected samples were inoculated on 7-day old plants of Agra Local (susceptible local bread wheat). Fresh uredospores were collected from the inoculated plants after a fortnight. Uredosporic dust of each sample was inoculated on three sets of differentials. Naming of pathotypes (Pts., singular Pt.) was done by following Indian system of binomial nomenclature (Nagarajan et al. 1983), with some modifications (Table 1) necessitated by the emergence of new pathotypes of P. triticina (Bhardwaj et al. 2012). All the optimal greenhouse procedures of inoculation, infection, post-inoculation care and recording of observations were followed (Stakman et al. 1962; Nayar et al. 1997). For the understanding of Wheat Pathologists of other countries, the Indian designations were also coded according to the North American system of racial nomenclature (Kolmer et al. 2009) as illustrated in Table 2.
Growing of seedlings, inoculation and recording observations
Brown rust of wheat was established on Agra Local grown in small plastic pots (4 inch size). Sets of differentials were grown in aluminum bread pans (29 cm long × 12 cm wide × 7 cm deep size) containing fresh loam soil and farmyard manure (3:1). A week old plants were inoculated through gentle application of uredospores with a lancet needle. Seedlings were then sprayed with a fine mist of water and incubated in dew chambers at 22 ± 2 °C for 24 h. Saturated relative humidity and 12 h of daylight were maintained in these chambers. Inoculated materials were then transferred on to the greenhouse benches having temperature of 22 ± 2 °C, relative humidity of 40–60% and light of about 15,000 lx for 12 h. Wheat-rust infection response (low or high) were recorded on differentials after a fortnight of inoculations using the scale of Stakman et al. (1962) with some modifications. Response 0 to 2 (micro flecks to small-moderate uredial pustules with or without chlorosis) were categorized as resistant whereas 3 to 4 (large uredial pustules with or without chlorosis) were designated susceptible. Response 33 + was categorized where both 3 and 3 + pustules occurred simultaneously. When the infection type data was different to the known/published pathotypes, new pathotype was designated and added to the culture collection.
Results and discussion
Pathotype distribution
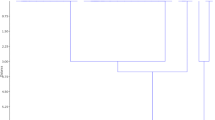
In 1427 wheat brown rust samples, 35 pathotypes were identified from India and three neighboring countries during 2013–16. Three pathotypes 121R63-1(77-5 = THTTM), 121R60-1(77-9 = MHTKL), 21R55 (104-2 = PHTTL) were found to be present in most of the samples ranging from 12 to 38% and were also widespread across different states of the country. Other pathotypes namely 109R63 (77-1 = THTTQ), 125R55 (77-3 = THTTS), 121R55-1 (77-6 = THTTQ), 77-11(125R28 = MGTTL), 21R63 (104-3 = PHTTL) and 162-2 (93R39 = KHTTL) were identified in 5–7% samples across more than five states of the country. The avirulence/virulence formulae of these pathotypes are depicted in Table 3. Pathotypes of 77 group comprised 58% of the population against 22% of 104 group. Pathotype (pt.) 77-5 (26.6%), has been predominant for about 20 years and now is being replaced gradually by 77-9 (23.8%) identified in 2008, indicating a clear shift in pathotype predominance observed in the population of 77-9 and decreasing trend was observed for the other pathotypes 77-5,104-2 and 104-3 (Fig. 1). Lr9, Lr24, Lr25, Lr32, Lr39 and Lr45 remained resistant to the field population of wheat brown rust pathogen in India and neighboring countries during 2013–16.
Among the 793 samples of brown rust of wheat analyzed from 15 states of India and three neighbouring countries, 30 pathotypes were observed during 2013–14. Pathotype 77-5, virulent to Lr23 and Lr26 genes which occur in many of the wheat varieties, was the most widespread and occurred in 14 Indian states, Nepal, Bhutan and Bangladesh. Similarly, pathotype 104-2, also virulent to Lr23 and Lr26 was the second most frequent and was identified from 12 states of India, Nepal and Bangladesh. The proportion of pathotype 77-9 has increased in Nilgiris, as compared to the previous years, as it was sampled in 56% of the samples from Nilgiris. In Karnataka, three predominant pathotypes were 77-9, 12 and 77-11, and other pathotypes 77-5, 12-2 and 104-2 were also identified in substantial number of samples. In the remaining samples, 18 other pathotypes were identified.
Surprisingly, the dominant trend was in favour of pt.12 in Maharashtra followed by pathotype 77-5 whereas remaining pathotypes were observed in few samples only. It hints at cultivation of some indigenous or local wheat germplasm in Maharashtra. Pathotype 104-2 was the most frequent in the states of Gujarat, Rajasthan and Madhya Pradesh. A total of 25 samples were analyzed from Bihar and West Bengal and pathotype 77-5 was the most common. In Uttar Pradesh, both pts. 77-5 and 104-2 were identified in most of the samples and twelve other pathotypes were identified in few samples only. Pathotype 104-2 was most frequent in Uttarakhand whereas in Himachal Pradesh both pts 77-5 and 104-2 were more common and had equal frequency. Pathotype 77-3 along with other five pathotypes were picked up in samples from Punjab. In Jammu and Kashmir, pt. 12-9 was identified in two samples whereas 104-2 occurred in one sample only. In Nepal, Bhutan and Bangladesh, both pts. 77-5 and 104-2 were identified in most of the samples analyzed whereas fifteen other pathotypes occurred in few samples only (Table 4). During this year, a new pathotype designated as 77-12 was identified in few samples analyzed from the Nilgiris hills. The new pathotype is close to the pathotype 77-9 but has avirulence to Lr20.
Population of wheat brown rust during 2014–15 comprised twenty five pathotypes intercepted in 386 samples received from nine states of India and three neighbouring countries. The frequency of 77-9 was found to increase in Tamil Nadu, Karnatka, Maharashtra, MP and Punjab. Three predominant pathotypes i.e., 77-9 (38%), 77-5 (32%) and 104-2 (14.5%) comprised 85% of the flora. Among these, both pathotypes 77-5 and 104-2 occurred in eight states of India and three neighboring countries. In Madhya Pradesh, pt. 77-5 was most frequent, followed by 77-9 whereas in Gujarat and Haryana, pt.104-2 was the most prevalent. Pathotype 77-9 was observed only in seven states of India but not in the neighboring countries. Remaining 22 pathotypes occurred in few samples only. The brown rust population in Bangladesh, Bhutan and Nepal was similarly dominated by three pathotypes. Among these, pt. 77-5 was most frequent followed by 104-2 and 12-4 (Table 5).
Diversity of pathotypes was lower during 2015–16. Among the 248 samples analyzed from 13 states of India, Nepal and Bhutan, 19 pathotypes of P. triticina were identified. Increased frequency in favour of pathotype 77-9 (121R60-1) continued as it was identified from 37.9% samples, succeeded by pts.77-5 (121R63-1) and 104-2 (21R55), which occurred in more than 15% samples each (Table 6). Pathotype 77-9 was the most frequent in Tamil Nadu, Chhatisgarh, Karnataka and Himachal Pradesh whereas 77-5 was frequent in Haryana, Maharashtra and Madhya Pradesh. The most dominant pt. was 104-2 in Uttarakhand. P. triticina population was quite diverse in Karnataka and Himachal Pradesh where eight pts. each were observed in these states. In Karnataka pathotype 77-9 was predominant followed by pathotypes 77-5 and 104-2. Remaining pathotypes were identified in few samples only. In Himachal Pradesh, both pts. 77-9 and 104-2 were equally common. Pathotype 77-9 was found in maximum number of samples analyzed from Chhattisgarh indicating its spread to other states. Both pts. 77-5 and 104-2 were identified in one and three samples, respectively. Some of the historical virulences like 11, 12-1, 106, 107-1, 162, 162A and other recent ones namely 12-6, 77-8, and 77-10, were observed in one sample each. Eighteen samples of brown rust were analyzed from Nepal and Bhutan. Six pathotypes with pt.77-5 as more frequent followed by 104-2 were observed in these countries.
Large number of pathotypes detected from Nilgiri Hills (Tamil Nadu), Karnatka and Himachal Pradesh, different agro-ecological areas provided further evidence that these areas continue to remain active source area for the brown rust spread in the country.
Pathotypes and wheat material in farmer’s fields
Information was also generated on the pathotypes in relation to the cultivated wheat varieties in the farmers’ fields. Fourteen pathotypes were identified from ten states of India and neighboring countries Nepal and Bangladesh. Mostly, pts. 77-5, 77-9, 104-2 and 104-3 occurred on the popular wheat varieties (Table 7). It means that present day varieties are providing enough surviving space for these pathotypes. Our observations could not validate based on host resistance, existence of different virulences in the farmers field. However, it does hint at weather conditions existing in different agro ecological areas which most likely have impacted the prevalence of different pathotypes independent of their virulences.
Per cent effectiveness of Lr genes
Effectiveness of different rust resistance genes in Indian wheat material to brown rust population is presented in Table 8. Lr9, Lr19, Lr24 and Lr28 were highly effective against field population of P. triticina in India and adjoining countries whereas Lr1, Lr10, Lr23 and Lr26 were not effective. In addition, Lr3, Lr13, Lr14a were susceptible to all the prevalent pathotypes of P. triticina in India, Bangladesh, Bhutan and Nepal. Evaluation of resistance genes/lines against field isolates revealed that Lr9, Lr24, Lr25, Lr32, Lr39 and Lr45 were resistant to the brown rust population of the Indian sub-continent during 2013–16 (Table 8).
During 2008–2013, a diverse population of 37 races of P. triticina were identified (Bhardwaj et al. 2016) in 2424 samples, likewise during 2013–16 also, 37 pathotypes were identified in 1407 samples analyzed from India, Bangladesh, Bhutan and Nepal. Pathotypes 121R63-1 (77-5 = THTTS) in 26.6% samples, 121R60-1(77-9 = MHTTS) in 23.8%, 21R55 (104-2 = PHTTL) in 17.7%, 21R63 (104-3(PHTKL) in 4.2% and 77-11(125R28 = MGTTL) in 2.7% samples were the five most widespread virulences during 2013–16. In Indian subcontinent the proportion of five predominant pathotypes has increased from 68 to 75% with a shift in virulence pattern in favour of a new pathotype 77-9(121R60-1), which was identified in 23.8% of the samples during these years against 12% in the last survey. Based on the recent studies, it has been observed that pathotype 121R60-1 is more competitive and has high fitness potential in comparison to 121R63-1 (Bhardwaj et al. 2014). In Maharashtra pt. 12 was predominant, which hints at cultivation of some old/indigenous wheat varieties. Simple pathotypes are promoted by varieties having no effective leaf rust resistance (Bhardwaj et al. 2014).In China PHT (23.7%), THT (14.7%), PHJ (11.4%) and THJ (4.2%) were the four most predominant races. Among Lr genes; Lr9, Lr24, Lr25, Lr28, Lr32, Lr39 and Lr45 were resistant (Liu and Chen 2012). The racial pattern is comparable to that of India, however, in China predominant pathotype resembles to 104-2 (PHTTL) whereas in India, predominant pts. are 77-5(THTTS) or 77-9(MHTTS). The races of P. triticina from China are coded on old system; however, if the first three letters are seen, Indian flora matches partially.
Virulence to Lr9, 19, 28 did not exist in Pakistan (Fayyaz et al. 2008), though in India, we have virulence for all these genes, however, virulence to Lr9 was not observed in the field population during these years. On the other hand predominant races in Slovakia (Hanzalova et al. 2008) are virulent to Lr9, Lr24 and Lr28 to which Indian pathotypes identified during this period were avirulent except for the few samples virulent to Lr28. When compared with the pathotypic flora of United States of America, Indian pathotypes are strikingly different as pathotypes MBTNB, TBBGS and TCRKG were widespread (Kolmer and Hughes 2016). Prevalent population of wheat rust in United States of America is virulent to Lr9, 24, 28 and 39 whereas avirulent in India. Population of P. triticina in Middle East and central Asia (Kolmer et al. 2011) is also strikingly different to that of India. The wheat leaf rust population of Zimbabwe, Zambia and Malawi was quite different to that of India as races MCDS (74.6%), TCPS (12.7%), FBPT (16.3%) and SCDS(6.3%) were predominant (Pretorius et al. 2014). In Egypt, most frequent races of P triticina observed during 2012–14 were PTTTT, TTTST, TTTTT and PKTST (El-Orabey et al. 2015), which are different to that of the Indian sub-continent.
Since rust resistant variety lasts for about 5 years (Sawhney 1995), therefore, identification of new pathotypes and pathotype distribution becomes a guiding force to re-orient the wheat breeding program. Like previous surveys (Bhardwaj et al. 2016) brown rust population remained avirulent to Lr9, 24, 25, 32, 39 and 45. With the shift in brown rust population, resistance of seedling, adult and slow rusting types are being identified in upcoming wheat material by screening against most virulent and predominant pathotypes including 77-9 also. Likewise the pathotype distribution information of brown rust is being used for wheat varietal deployment. The success of the programme can be envisaged from the fact that there was no wheat rust epidemic during the last 46 years.
References
Bhardwaj SC, Gangwar OP, Singh SB, Saharan MS, Sangeeta Sharma (2012) Rust situation and pathotypes of Puccinia species in Leh Ladakh in relation to recurrence of wheat rusts in India. Indian Phytopathol 65(3):230–232
Bhardwaj SC, Gupta N, Sharma TR, Pal Dharam, Prasad P (2014) Competitive ability and fitness potential among the pathotypes of Puccinia triticina on wheat in India. Indian Phytopathol 67(1):33–37
Bhardwaj SC, Prashar M, Kumar S, Gangwar OP, Neha Gupta, Prasad P, Khan H (2016) Patterns of physiologic diversity of Puccinia triticina on wheat in Indian subcontinent during 2008–2013. Indian J Agric Sci 86(1):55–64
El-Orabey WM, Sallam ME, Omara R, Abd El-Malik I (2015) Geographical distribution of Puccinia triticina physiologic races in Egypt during 2012–2014 growing seasons. Afr J Agric Res 10(45):4193–4203
Fayyaz M, Rattu AR, Ahmad J, Akhtar MA, Hakro AA, Kazi M (2008) Current status in the occurrence and distribution of Puccinia triticina, wheat leaf rust virulences in Pakistan. Pak J Bot 40(2):887–895
Hanzalova A, Bartos P, Sumikova T (2008) Virulence of wheat leaf rust (Puccinia triticina Eriks.) in the years 2013–15 and resistance of wheat cultivars in Slovakia. Cereal Res Commun 44(4):585–593
Kolmer JA, Hughes ME (2016) Physiologic specialization of Puccinia triticina on wheat in the United states in 2014. Plant Dis 102:1066–1071
Kolmer JA, Long DL, Hughes ME (2009) Physiologic specialization of Puccinia triticina on wheat in the United States in 2007. Plant Dis 95:538–544
Kolmer JA, Ordonez ME, Manisterski J, Anikster Y (2011) Genetic differentiation of Puccinia triticina populations in the middle east and genetic similarity with population in Central Asia. Phytopathology 101:870–877
Liu TG, Chen WQ (2012) Race and virulence dynamics of Puccinia triticina in China during 2002-2006. Plant Dis 96:1601–1607
Nagarajan S, Nayar SK, Bahadur P (1983) The proposed brown rust of wheat (Puccinia recondia f. sp tritici virulence monitoring system. Curr Sci 52(9):413–416
Nayar SK, Prashar M, Bhardwaj SC (1997) Manual of current techniques in wheat rusts. Res Bull 2:32 (Regional Station, Directorate of Wheat Research Flowerdale, Shimla HP)
Nayar SK, Jain SK, Prashar M, Kumar S, Menon MK (2003) Appearance of new pathotype of Puccinia recondita tritici virulent on Lr9 in India. Indian Phytopathol 56(2):196–198
Pretorius ZA, Terefe TV, Herselman PR, Soko T, Siwale J, Mutari B, Selinga TI, Hodson DP (2014) Races of Puccinia triticina detected on wheat in Zimbabwe, Zambia and Malawi and regional germplasm responses. Australas Plant Pathol 44(2):217–224
Samborski DJ (1985) Wheat leaf rust. In: Roelfs AP, Bushnell WR (eds) The cereal rusts. Academic Press, New York, pp 39–59
Sawhney RN (1995) Genetics of wheat rust interaction. Plant Breed Rev 13:293–343
Stakman EC, Stewart DM, Loegering WQ (1962) Identification of physiologic races of Puccinia graminis tritici. US Agric Res Serv 617:53
Acknowledgements
The authors are grateful to the Director, ICAR-IIWBR, Karnal, Haryana for providing liberal resources and first author also acknowledges the financial assistance received from Department of Biotechnology, Govt. of India through the Project ‘Puccinia triticina genomics network on De novo genome sequencing, fitness, variation and pathogenicity’. Help of all the partners in monitoring wheat rusts and sampling is thankfully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants or animals
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Bhardwaj, S.C., Gangwar, O.P., Prasad, P. et al. Physiologic specialization and shift in Puccinia triticina pathotypes on wheat in Indian subcontinent during 2013–2016. Indian Phytopathology 72, 23–34 (2019). https://doi.org/10.1007/s42360-018-00110-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42360-018-00110-9