Abstract
Background
The success of minimally invasive parathyroidectomy (MIP) relies on accurate localization of the abnormal parathyroid glands. Concordant findings on ultrasound (US) and 99mTc-scintigraphy (sestamibi) are currently considered the ‘gold standard’. Computed tomography (CT) has also recently been used in preoperative planning. We sought to assess the accuracy of CT for localization of abnormal parathyroid glands in such patients.
Methods
An audit of 75 patients with primary hyperparathyroidism (PHPT) who underwent neck US and CT between 2017 and 2019 at our center as their first-line imaging.
Results
All 75 patients underwent US and CT and 54 (72.0%) also had sestamibi. CT alone identified a potential target in all patients, of which the location was correct in 63 (84.0%). The overall combined sensitivity of US and CT was 88% (95% CI 78–94) and was higher than the combined sensitivity of US and sestamibi (65% [95% CI 53–76]; p < 0.001). Twenty-one patients (28.0%) had an ectopic gland, and the sensitivity of US and CT was 86% (95% CI 64–96) versus US and sestamibi (57% [95% CI 34–77]; p = 0.016). For adenomas < 1.0 g (n = 36; 48%), the accuracy of CT was 81% (95% CI 64–91) compared with 62% (95% CI 44–77) for US and sestamibi (p = 0.04). The correct preoperative diagnosis of multiglandular disease (n = 9; 12%) seemed to be the most difficult, with similar accuracy for US and sestamibi (40% [95% CI 14–73]) and US and CT (50% [95% CI 20–80]) (p > 0.99).
Conclusion
The combination of US and CT was able to correctly identify the location of the abnormal parathyroid in 88% of patients and, in comparison with US and sestamibi, had better diagnostic accuracy, especially for smaller and ectopic adenomas. This finding suggests that US and CT could be considered as a first-line imaging modality in patients with PHPT considered for MIP.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Parathyroidectomy is the only definitive and cost-effective treatment for patients with primary hyperparathyroidism (PHPT) and is therefore recommended for all patients with symptomatic disease [1]. The first case of parathyroid surgery by means of a bilateral neck exploration (BNE) was described in 1925 [2]. Currently, minimally invasive parathyroidectomy (MIP) is increasingly employed in these patients.
The biggest challenge of this targeted surgical approach, however, is the wide variability in parathyroid gland anatomy and, therefore, this technique relies heavily on accurate preoperative anatomical localization of abnormal parathyroid glands. In most institutions, the diagnostic work-up consists of a combination of a nuclear medicine imaging scan (e.g., sestamibi (Technetium-99m-methoxyisobutyl isonitrile)) and ultrasonography (US) [3, 4].
More recently, a computed tomography scan (CT) has been used in the diagnostic work-up in patients with PHPT. Parathyroid glands are well vascularized and therefore possess a unique feature in that their uptake of contrast occurs very rapidly. This high vascularity aids the determination of their precise anatomical localization on CT. A meta-analysis including 34 studies reported a pooled sensitivity of CT for abnormal parathyroid localization ranging from 71 to 80%, depending on the exact scanning protocol used [5]. Apart from high sensitivity, CT is hypothesised to be less operator-dependent than real-time imaging and to enable a second opinion on distant location: it might therefore be a very valuable tool in the setting of forming an operative plan. Moreover, CT can provide a three-dimensional image of the parathyroid gland location.
Therefore, with the current study, we aimed to analyze the accuracy of a new diagnostic pathway of a parathyroid-specific CT with regard to the correct anatomical localization of the abnormal parathyroid glands in a four-quadrant classification.
Methods
The subjects of the current audit were 75 patients who presented with PHPT at the Centre for Endocrine Surgery (CES) (University College London Hospital, London, UK) between January 2017 and January 2019 and were followed up for at least 6 months. All patients had biochemically proven disease, based on increased levels of serum calcium and raised or inappropriately suppressed levels of parathyroid hormone (PTH) combined with high or normal urine calcium levels.
All patients had a neck US and a dedicated parathyroid CT as their first-line imaging at our center. Performance of a sestamibi was reserved for when there was diagnostic uncertainty, although most patients (n = 47) were referred to us with a sestamibi already performed at their referring hospital (i.e., without SPECT/CT (single-photon emission computerized tomography). Of these, 23 (48.9%) were negative.
Details of imaging
All imaging reports were done according to a standardized template; specifically, the indication of one of the four quadrants indicating both the side, i.e., right or left, as well as upper or lower position of the suspected abnormal parathyroid gland, was reported. Also, it was noted if the pathological gland was suspected to be in an ectopic location (i.e., intrathyroidal, in the thyrothymic ligament in a dropped-superior position). Finally, a combined US and CT report was produced by the radiologist after independent performance and assessment of both imaging modalities. This final report was used for surgical planning.
For the neck US and CT, institutional protocols were used. The US was performed using a high-resolution linear ultrasound probe with the patient in neck extension. The neck was scanned in a neutral position and also with the head turned to each side in order to assess the peri-oesophageal/retrothyroid region. The neck was also scanned with a curvilinear probe to assess the deeper paratracheal tissues of the thoracic inlet. For the CT, patients were scanned using triple-phase imaging from the mandibular angle to just above the carina with 0.5 mm collimation. A precontrast scan was performed, followed by 100 mls Omnipaque 240 Mg iodine/ml at 5 mls/s scanning at 25 s (arterial phase) and 80 s (delayed phase).
Details of surgery
In general, most patients included in the current study were scheduled to undergo a MIP. This was performed through a 1.5–2-cm incision positioned over the enlarged parathyroid gland, as demonstrated on preoperative imaging. A lateral approach with access between the sternomastoid and strap muscles was employed. Identification and extirpation of the abnormal gland was followed by intraoperative PTH (IOPTH) measurements (Future Diagnostics STAT-IntraOperative-Intact-PTH (STAT-IO-I-PTH) Immunoassay Kit, Wijchen, the Netherlands). The modified Miami criterion was used to define intraoperative cure [6]. If there was insufficient drop in PTH concentration, an additional 20 min sample was analyzed before conversion to a BNE was undertaken through extension of the incision into a collar neck incision and additional midline access between the strap muscles. In this situation, all parathyroid glands were identified before any (additional) glands were removed. In select patients, BNE was the approach of first choice. No routine frozen sections were performed in our institution. Cure of the disease was defined as albumin-adjusted serum calcium of ≤ 2.6 mmol/L at 6 months following surgery.
Data collection
Standard demographic and clinical data such as sex and age at time of surgery were prospectively collected. Furthermore, the preoperative levels of serum calcium and PTH were noted.
The following details of surgery were noted: the type of operation (MIP or BNE), the exact location of the adenoma in one of the four quarters (i.e., right or left and superior or inferior position), ectopic location of the abnormal parathyroid gland (i.e., intrathyroidal, in the thyrothymic ligament in a dropped-superior position) and the presence of multiglandular disease. Moreover, all IOPTH values were recorded. The weight of the removed gland was also noted. The postoperative values of serum calcium and PTH were recorded at specific times (i.e., direct postoperative (< 24 h of surgery), 3 months after surgery and at the patients’ final follow-up appointment 6 months after surgery).
Statistical analysis
Summary statistics were obtained and presented as counts/percentages or median values (min, max). Sensitivity and 95% confidence intervals (95% CI) were calculated and compared for the different diagnostic tools in different settings using McNemar’s test. Overall, a p value of less than 0.05 was considered significant. All statistical analyses were performed using IBM SPSS Statistics for Macintosh, Version 25.0 (IBM Corp. IMB SPSS statistics, Armonk, NY, USA).
Results
In total, 75 patients with PHPT were included in this study and had a US and CT at our institution as a first-line imaging modality for their parathyroid disease in the preoperative work-up. Table 1 shows the demographic and preoperative laboratory data of our cohort. About three-quarters of patients were female (n = 58; 77.3%), with a median age of 57 years at time of the procedure. While all patients (n = 75; 100%) underwent an US and CT, 54 patients (72.0%) also had sestamibi.
Surgery
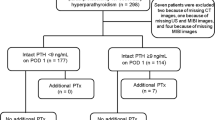
The details of surgery are presented in Table 2. Two-thirds of patients underwent a MIP (n = 50; 66.6%). Four patients (5.3%) could not undergo a MIP as they underwent concurrent thyroid surgery. In total, 86 parathyroid glands were removed. The majority of patients had single-gland disease (n = 66; 88.0%). In seven patients (9.3%), two glands were removed, while in two patients (2.7%), three glands were removed to achieve the desired drop in PTH. The locations of the parathyroid adenomas are depicted in Fig. 1. Notably, 21 parathyroid adenomas (28.0%) were in an ectopic position.
The median postoperative length of stay was 1 day (range 1–2) and none of the patients developed severe complications. Specifically, there were no patients who developed postoperative hypocalcemia.
All patients had at least 6 months of follow-up after surgery (range 6–12). At the time of the last follow-up, overall median PTH was normal at 3.9 pmol/L (range 0.7–6.90), with an overall median serum calcium level of 2.39 mmol/L (range 2.30–2.60).
Imaging
The sensitivity of the different imaging tools is depicted in Table 3. CT alone established the correct location of the abnormal parathyroid gland in 63 patients (84.0%). Moreover, in 17 patients (22.7%), CT was the only modality that was correct. Unfortunately, in ten patients (13.3%), CT reported an incorrect location of the adenoma. Of these, in one (10.0%), sestamibi was the only modality that was correct. In another (10.0%), US was the only modality able to localize the intrathyroidal adenoma. In the remaining eight patients (80.0%), none of the modalities was correct.
In the 30 patients where US was not able to identify the adenoma, CT was able to localize the adenoma correctly in more than two-thirds (n = 21; 70.0%) and sestamibi was correct in four patients (13.3%). In patients with incorrect US localization, ten (33.3%) had ectopic pathology, and in eight patients (out of 19 with known weight; 42%), the noted weight of the adenoma was < 0.5 g.
Sestamibi alone was correct in only 18 patients (24.0%). In 17 of these patients (94.4%), CT was also correct in localizing the position of the adenoma. Moreover, sestamibi was negative in 27 patients (50.0%).
To assess if multimodality imaging would have resulted in overall higher sensitivity, the combined sensitivities were calculated. The standard combination or US and sestamibi, with which most patients were referred to our department, resulted in a sensitivity of 65% (95% CI 53–76). This was statistically less than the sensitivity for CT alone (p < 0.001). The imaging pathway, as was studied during this audit (i.e., a US and CT at our institution), resulted in a sensitivity of 88% (95% CI 78–94). Sestamibi was not helpful in any of the patients in whom US and CT combined failed to localize the adenoma.
Factors influencing sensitivity of imaging
Weight and, therefore, size of the abnormal parathyroid gland was the first important factor affecting the sensitivity of imaging modalities (Table 3). The sensitivity of CT for predicting the correct location of abnormal parathyroid glands weighing < 1.0 g was significantly better than that of US and sestamibi (p = 0.04). Combining the results of US and CT in patients with smaller glands did not lead to an increase in the sensitivity compared with CT alone (p > 0.99).
The second important factor was an ectopic location of abnormal parathyroid glands. The sensitivity for CT alone seemed better than for combined US and sestamibi (p = 0.07). However, combining US and CT led to a statistically significant increased accuracy of 86% (95% CI 64–96) (p = 0.016).
The correct preoperative diagnosis of multiglandular disease seemed to be the most challenging. With the standard combination of US and sestamibi, the accuracy rate was only 40% (95% CI 14–73). Among these patients, the sensitivity for combined US and CT was statistically similar (50%, p > 0.99).
Discussion
Currently, different protocols, driven by local expertise and resources available, are used for localization of abnormal parathyroid glands at various institutions, while there is an implicit agreement that there is no single imaging tool with perfect sensitivity. Some patients with negative imaging are even denied operations. It is therefore important for optimal ways of performing localization studies to be researched, while aiming at best accuracy, lowest costs, low radiation, and comprehensible logistics. This audit presents data on a newly implemented imaging paradigm, consisting of US and CT performed at CES as a first-line imaging modality in patients with PHPT. As the majority of patients had a sestamibi performed in their referring centers, the result of this nuclear imaging is also included in the final analysis. Our findings suggest that a combined US and CT protocol could be implemented as an initial imaging modality in patients with PHPT and that it could be superior to US and sestamibi alone or combined.
A total of 75 patients had a US and CT performed at our center. The sensitivity of CT for the precise localization of the abnormal parathyroid gland was 87% and comparable with that in other published series, although the rates reported vary significantly (range 66–89%) [7,8,9,10,11]. When combining US and CT, overall sensitivity was 88% (95% CI 78–94). While this study was not set up to assess the sensitivity of sestamibi within our cohort, both US (60%) and sestamibi (33%) as stand-alone imaging techniques had lower sensitivity than CT. Moreover, the combined sensitivity of sestamibi and US was also significantly lower among our patients. This most likely reflects the fact that CES is a tertiary referral centre and, therefore, many of our cases are not straightforward and possibly the negative sestamibi prompted their referral in the first place. When comparing the sensitivity of CT to that of US and sestamibi available in the literature, these seem comparable, with a pooled sensitivity for US of 80% and for scintigraphy of 83% in a recent large review article [12]. Other authors have demonstrated that the overall sensitivity is higher when a dual imaging diagnostic pathway consisting of sestamibi and US is employed, with sensitivity rates reported as up to 88% in different studies [13,14,15,16], comparable with the sensitivity of 88% for US and CT in our cohort. However, in contrast to our cohort, where only sestamibi scintigraphy was performed, most patients included in other current series undergo a sestamibi scan combined with CT (i.e., SPECT); thus, these patients are effectively undergoing a dual imaging protocol in one scan [11]. Several studies have shown that SPECT might be superior to sestamibi [17,18,19]. Specifically, Treglia et al. [20] report in their review article that the use SPECT increased the detection rate compared with sestamibi scintigraphy. While CT had a similar diagnostic performance compared with SPECT, this was at a higher radiation dose. The use of SPECT as a first-line imaging modality in preoperative planning may therefore be a suitable choice based on regional imaging capabilities.
The diagnostic accuracy of combined US and CT is at least comparable with a paradigm that includes sestamibi and US and also offers some advantages. Firstly, there is the possibility of offering US and CT in a one-stop fashion, and the performance of a CT is significantly shorter than that of sestamibi (reported times < 5 min versus > 300 min) [21]. Secondly, CT is hypothesized to be less operator-dependent than real-time imaging. Moreover, due to its permanent imaging records, it enables a second expert opinion at a distant location and can thus be of added value in the setting of preoperative planning. Permanent records could also facilitate the application of machine learning, a powerful technique for recognizing patterns in medical imaging, which can further improve localization of abnormal glands, making it less operator-dependent and more cost-effective. Thirdly, although the reported radiation dose for CT parathyroid (5.56–6.1 mSv) and sestamibi (3.33–5.6 mSv) are relatively comparable, the radiation exposure of a CT is lower than when sestamibi is combined with SPECT (12.4 mSv) [21,22,23]. And fourthly, while the pricing of different scans differs between countries, in general, US is by far the cheapest and CT is less costly than sestamibi, even without SPECT [24].
US, compared with CT, is of greater value in assessing the thyroid gland itself [25]. Thorough assessment of the thyroid prior to parathyroid surgery could reduce the number of patients needing thyroid surgery after removal of a parathyroid adenoma for disease that was already present. It could be hypothesized that US could be used in second-look modus directly after performance of a CT and, in this way, act as a confirmation tool of the location of the abnormal parathyroid gland. However, as US correctly localized the abnormal parathyroid in more than half of the patients, it could, in our view, be acceptable for the select Endocrine Centers which have IOPTH, to perform a MIP without any further investigations if the US finds a target [26]. Nevertheless, overall, we recommend the combined diagnostic pathway of US and CT as the first-line imaging modalities.
Furthermore, an important group of patients, who are increasingly scheduled for parathyroid surgery because of increased awareness of PHPT and an increase in screening, have relatively smaller adenomas and many of them in ectopic positions. Multiple authors have raised concerns that the sensitivity of US [27,28,29,30] and sestamibi [17, 31] is decreased in cases of small or ectopic adenomas as well as in multigland disease. Although a recent study on outcomes among different sizes of adenomas did not report a difference, these authors did note that smaller adenomas were more challenging to identify on preoperative imaging [32]. The average weight of an adenoma is 1 g [33, 34], with microadenomas defined as weighing less than 0.1 g and giant adenomas weighing over 2 g [35]. The median weight of the adenoma among our cohort was 0.72 g, and in more than one in four patients, it was found in an ectopic position. While our proportion of ectopic abnormal parathyroid glands is comparable with the percentage of ectopic glands reported by others [36, 37], it does underline the importance of being able to detect them preoperatively. This is especially important as these ectopic glands form a large risk factor for failure to cure the disease during primary surgery [38, 39]. Among our patients with smaller glands (weighing < 1.0 g), the sensitivity of CT alone (81%) was higher than that of US (54%) and US and sestamibi (62%). Moreover, while for adenomas in an ectopic location the sensitivity of CT alone (82%) was not statistically significantly higher than that of US and sestamibi (57%), in a dual-modality with US and CT, the accuracy increased to 86% and thus proved to be more accurate among these patients. Unfortunately, the sensitivity for localizing multiglandular disease was not increased among our patients by adding CT to their work-up, although this subgroup included only nine patients.
Other newer functional imaging modalities should also be considered in the imaging of patients with PHPT. One promising modality for visualizing parathyroid adenomas is 18F-fluorocholine (FCH) PET/CT [40,41,42,43]. Specifically, a recent large retrospective study showed a high rate of correct detection at 96% (per patient-based) and 90% (per lesion-based) [44]. However, this technique is not widely available and insights into its cost-effectiveness are currently lacking. It could thus be best considered for those patients with diagnostic uncertainty or perhaps recurrence of their PHPT. Another emerging technique is 11C-methionine (Met) PET/CT [45]. While this scan seems to have a high localization rate (79–91%), its main disadvantage is the restricted availability and the relatively high costs of Met-PET/CT. It seems that this scan is therefore better reserved for selective rather than routine use. Furthermore, the usefulness of 18F-FDG PET/CT in detecting the adenoma in patients with PHPT is still under debate, as current data do not support the utilization of this technique [20, 46].
The main strength of this study is that it describes a fairly typical population of patients with PHPT. About three-quarters of our patients were female, with a median age of 57 years at the time of the procedure: the majority had single-gland disease (88.0%), with the proportion of patients with multigland disease being just under one-eighth (12.0%). This is comparable with most cohorts on PHPT which report the percentage of patients in whom more than one gland is affected as ranging from 8 to 33% [47,48,49,50,51,52,53,54,55]. These similarities suggest that our data could be applied to most patients with PHPT and that our experience should be transferable to other units. The other advantage is that the current study is one of the largest specifically set up to address adding CT to the preoperative work-up for patients with PHPT. Although we were able to demonstrate a statistically significant difference when doing whole group analysis, certain subgroup analyses could not be performed. Traditionally, most published imaging studies comment on the correct lateralization of abnormal parathyroids. As the success of MIP depends on precise localization, we have opted for four-quadrant-localization rather than just indicating the side of pathology. Therefore, this was the primary outcome measure in the current study. Sensitivity of preoperative localization techniques may be affected by numerous factors. With regard to US, one factor known to influence sensitivity is experience of the operator [11]. Therefore, at our center we have a dedicated team of radiologists performing all our parathyroid US scans.
Weaknesses of this study include a specific referral pattern resulting in the different nature of the included patients, as supported by the relatively high proportion of negative results on sestamibi and, hence, a skewed result. As sestamibi had already been performed in a substantial subgroup of patients, these results could have influenced the outcome of the US and CT reports from CES. Furthermore, the US and CT scans were not routinely performed at the same time and there was no formal blinding of the radiologist. A combined report was drawn up after performance of both imaging studies, therefore confounding could have occurred.
In conclusion, CT alone is a sensitive imaging modality able to identify abnormal parathyroid glands in patients with PHPT better than US and sestamibi alone or combined. CT is also superior in identifying smaller and ectopic glands. When combined with US, correct localization was achieved in 88% of patients and, therefore, we recommend that the dual-modality imaging of US and CT should be considered as a first-line imaging modality in patients with PHPT.
References
Wilhelm SM, Wang TS, Ruan DT, Lee JA, Asa SL, Duh QY et al (2016) The American Association of Endocrine Surgeons Guidelines for definitive management of primary hyperparathyroidism. JAMA Surg 151(10):959–968
Delbridge LW, Palazzo FF (2007) First parathyroid surgeon: Sir John Bland-Sutton and the parathyroids. ANZ J Surg 77(12):1058–1061
Kunstman JW, Kirsch JD, Mahajan A, Udelsman R (2013) Clinical review: parathyroid localization and implications for clinical management. J Clin Endocrinol Metab 98(3):902–912
Elaraj DM, Sippel RS, Lindsay S, Sansano I, Duh QY, Clark OH et al (2010) Are additional localization studies and referral indicated for patients with primary hyperparathyroidism who have negative sestamibi scan results? Arch Surg 145(6):578–581
Kluijfhout WP, Pasternak JD, Beninato T, Drake FT, Gosnell JE, Shen WT et al (2017) Diagnostic performance of computed tomography for parathyroid adenoma localization; a systematic review and meta-analysis. Eur J Radiol 88:117–128
Barczynski M, Konturek A, Hubalewska-Dydejczyk A, Cichon S, Nowak W (2009) Evaluation of Halle, Miami, Rome, and Vienna intraoperative iPTH assay criteria in guiding minimally invasive parathyroidectomy. Langenbeck's Arch Surg 394(5):843–849
Rodgers SE, Hunter GJ, Hamberg LM, Schellingerhout D, Doherty DB, Ayers GD et al (2006) Improved preoperative planning for directed parathyroidectomy with 4-dimensional computed tomography. Surgery. 140(6):932–940 discussion 40-1
Rameau A, Eng S, Vu J, Saket R, Jun P, Friduss M (2017) Four-dimensional computed tomography scan utility in parathyroidectomy for primary hyperparathyroidism with low baseline intact parathyroid hormone. Laryngoscope. 127(6):1476–1482
Day KM, Elsayed M, Beland MD, Monchik JM (2015) The utility of 4-dimensional computed tomography for preoperative localization of primary hyperparathyroidism in patients not localized by sestamibi or ultrasonography. Surgery. 157(3):534–539
Suh YJ, Choi JY, Kim SJ, Chun IK, Yun TJ, Lee KE et al (2015) Comparison of 4D CT, ultrasonography, and 99mTc sestamibi SPECT/CT in localizing single-gland primary hyperparathyroidism. Otolaryngol Head Neck Surg 152(3):438–443
Cheung K, Wang TS, Farrokhyar F, Roman SA, Sosa JA (2012) A meta-analysis of preoperative localization techniques for patients with primary hyperparathyroidism. Ann Surg Oncol 19(2):577–583
Nafisi Moghadam R, Amlelshahbaz AP, Namiranian N, Sobhan-Ardekani M, Emami-Meybodi M, Dehghan A et al (2017) Comparative diagnostic performance of ultrasonography and 99mTc-sestamibi scintigraphy for parathyroid adenoma in primary hyperparathyroidism; systematic review and meta-analysis. Asian Pac J Cancer Prev 18(12):3195–3200
Scattergood S, Marsden M, Kyrimi E, Ishii H, Doddi S, Sinha P (2019) Combined ultrasound and sestamibi scintigraphy provides accurate preoperative localisation for patients with primary hyperparathyroidism. Ann R Coll Surg Engl 101(2):97–102
Haber RS, Kim CK, Inabnet WB (2002) Ultrasonography for preoperative localization of enlarged parathyroid glands in primary hyperparathyroidism: comparison with (99m)technetium sestamibi scintigraphy. Clin Endocrinol 57(2):241–249
Perie S, Fessi H, Tassart M, Younsi N, Poli I, St Guily JL et al (2005) Usefulness of combination of high-resolution ultrasonography and dual-phase dual-isotope iodine 123/technetium Tc 99m sestamibi scintigraphy for the preoperative localization of hyperplastic parathyroid glands in renal hyperparathyroidism. Am J Kidney Dis 45(2):344–352
Lee JB, Kim WY, Lee YM (2015) The role of preoperative ultrasonography, computed tomography, and sestamibi scintigraphy localization in secondary hyperparathyroidism. Ann Surg Treat Res 89(6):300–305
Lavely WC, Goetze S, Friedman KP, Leal JP, Zhang Z, Garret-Mayer E et al (2007) Comparison of SPECT/CT, SPECT, and planar imaging with single- and dual-phase (99m)Tc-sestamibi parathyroid scintigraphy. J Nucl Med 48(7):1084–1089
Shafiei B, Hoseinzadeh S, Fotouhi F, Malek H, Azizi F, Jahed A et al (2012) Preoperative (9)(9)mTc-sestamibi scintigraphy in patients with primary hyperparathyroidism and concomitant nodular goiter: comparison of SPECT-CT, SPECT, and planar imaging. Nucl Med Commun 33(10):1070–1076
Eslamy HK, Ziessman HA (2008) Parathyroid scintigraphy in patients with primary hyperparathyroidism: 99mTc sestamibi SPECT and SPECT/CT. Radiographics. 28(5):1461–1476
Treglia G, Trimboli P, Huellner M, Giovanella L (2018) Imaging in primary hyperparathyroidism: focus on the evidence-based diagnostic performance of different methods. Minerva Endocrinol 43(2):133–143
Madorin CA, Owen R, Coakley B, Lowe H, Nam KH, Weber K et al (2013) Comparison of radiation exposure and cost between dynamic computed tomography and sestamibi scintigraphy for preoperative localization of parathyroid lesions. JAMA Surg. 148(6):500–503
Czarnecki CA, Einsiedel PF, Phal PM, Miller JA, Lichtenstein M, Stella DL (2018) Dynamic CT for parathyroid adenoma detection: how does radiation dose compare with nuclear medicine? AJR Am J Roentgenol 210(5):1118–1122
Vaiman M, Cohenpur M, Halevy A, Volkov O, Gold R, Peer M et al (2019) Low-radiation of technetium-99m-sestamibi and single-photon emission computed tomography/computed tomography to diagnose parathyroid lesions. World J Nucl Med 18(1):52–57
Wang TS, Cheung K, Farrokhyar F, Roman SA, Sosa JA (2011) Would scan, but which scan? A cost-utility analysis to optimize preoperative imaging for primary hyperparathyroidism. Surgery. 150(6):1286–1294
Yoo J, Ahn HS, Kim SJ, Park SH, Seo M, Chong S (2018) Evaluation of diagnostic performance of screening thyroid ultrasonography and imaging findings of screening-detected thyroid cancer. Cancer Res Treat 50(1):11–18
Shawky M, Abdel Aziz T, Morley S, Beale T, Bomanji J, Soromani C et al (2019) Impact of intraoperative parathyroid hormone monitoring on the management of patients with primary hyperparathyroidism. Clin Endocrinol 90(2):277–284
Gilat H, Cohen M, Feinmesser R, Benzion J, Shvero J, Segal K et al (2005) Minimally invasive procedure for resection of a parathyroid adenoma: the role of preoperative high-resolution ultrasonography. J Clin Ultrasound 33(6):283–287
Grosso I, Sargiotto A, D'Amelio P, Tamone C, Gasparri G, De Filippi PG et al (2007) Preoperative localization of parathyroid adenoma with sonography and 99mTc-sestamibi scintigraphy in primary hyperparathyroidism. J Clin Ultrasound 35(4):186–190
Lo CY, Lang BH, Chan WF, Kung AW, Lam KS (2007) A prospective evaluation of preoperative localization by technetium-99m sestamibi scintigraphy and ultrasonography in primary hyperparathyroidism. Am J Surg 193(2):155–159
Ozkaya M, Elboga U, Sahin E, Kalender E, Korkmaz H, Demir HD et al (2015) Evaluation of conventional imaging techniques on preoperative localization in primary hyperparathyroidism. Bosn J Basic Med Sci 15(1):61–66
Nichols KJ, Tomas MB, Tronco GG, Rini JN, Kunjummen BD, Heller KS et al (2008) Preoperative parathyroid scintigraphic lesion localization: accuracy of various types of readings. Radiology. 248(1):221–232
Abdel-Aziz TE, Gleeson F, Sadler G, Mihai R (2019) Dwarfs and giants of parathyroid adenomas-no difference in outcome after parathyroidectomy. J Surg Res 237:56–60
Gupta M, Singhal L, Kumar A (2018) Hyperparathyroidism mimicking metastatic bone disease: a case report and review of literature. J Adolesc Young Adult Oncol 7(3):400–403
Allen E, Fingeret A (2019) Anatomy, head and neck, Thyroid. StatPearls, Treasure Island
Wolfe SA, Sharma S (2019) Parathyroid adenoma. StatPearls, Treasure Island
Roy M, Mazeh H, Chen H, Sippel RS (2013) Incidence and localization of ectopic parathyroid adenomas in previously unexplored patients. World J Surg 37(1):102–106
Kim J, Cubangbang M, Adkins L, Chia S, DeKlotz TR, Boyle L et al (2017) Ectopic parathyroid adenoma in the pyriform sinus. Head Neck 39(10):E110–E1E3
Patrinos A, Zarokosta M, Piperos T, Tsiaoussis J, Noussios G, Mariolis-Sapsakos T (2019) An anatomic aberration and a surgical challenge: mediastinal parathyroid adenoma anterior the pericardium. A case report. Int J Surg Case Rep 58:153–156
Gouveia S, Rodrigues D, Barros L, Ribeiro C, Albuquerque A, Costa G et al (2012) Persistent primary hyperparathyroidism: an uncommon location for an ectopic gland--case report and review. Arq Bras Endocrinol Metabol 56(6):393–403
Michaud L, Burgess A, Huchet V, Lefevre M, Tassart M, Ohnona J et al (2014) Is 18F-fluorocholine-positron emission tomography/computerized tomography a new imaging tool for detecting hyperfunctioning parathyroid glands in primary or secondary hyperparathyroidism? J Clin Endocrinol Metab 99(12):4531–4536
Michaud L, Balogova S, Burgess A, Ohnona J, Huchet V, Kerrou K et al (2015) A pilot comparison of 18F-fluorocholine PET/CT, ultrasonography and 123I/99mTc-sestaMIBI dual-phase dual-isotope scintigraphy in the preoperative localization of hyperfunctioning parathyroid glands in primary or secondary hyperparathyroidism: influence of thyroid anomalies. Medicine (Baltimore) 94(41):e1701
Treglia G, Piccardo A, Imperiale A, Strobel K, Kaufmann PA, Prior JO et al (2019) Diagnostic performance of choline PET for detection of hyperfunctioning parathyroid glands in hyperparathyroidism: a systematic review and meta-analysis. Eur J Nucl Med Mol Imaging 46(3):751–765
Piccardo A, Trimboli P, Rutigliani M, Puntoni M, Foppiani L, Bacigalupo L et al (2019) Additional value of integrated (18)F-choline PET/4D contrast-enhanced CT in the localization of hyperfunctioning parathyroid glands and correlation with molecular profile. Eur J Nucl Med Mol Imaging 46(3):766–775
Broos WAM, Wondergem M, Knol RJJ, van der Zant FM (2019) Parathyroid imaging with (18)F-fluorocholine PET/CT as a first-line imaging modality in primary hyperparathyroidism: a retrospective cohort study. EJNMMI Res 9(1):72
Weber T, Luster M (2014) Localization of parathyroid adenomas with C11-methionine PET-CT. Chirurg. 85(7):601–606
Kluijfhout WP, Pasternak JD, Drake FT, Beninato T, Gosnell JE, Shen WT et al (2016) Use of PET tracers for parathyroid localization: a systematic review and meta-analysis. Langenbeck's Arch Surg 401(7):925–935
Weber CJ, Sewell CW, McGarity WC (1994) Persistent and recurrent sporadic primary hyperparathyroidism: histopathology, complications, and results of reoperation. Surgery. 116(6):991–998
Tezelman S, Rodriguez JM, Shen W, Siperstein AE, Duh QY, Clark OH (1995) Primary hyperparathyroidism in patients who have received radiation therapy and in patients who have not received radiation therapy. J Am Coll Surg 180(1):81–87
Bonjer HJ, Bruining HA, Birkenhager JC, Nishiyama RH, Jones MA, Bagwell CB (1992) Single and multigland disease in primary hyperparathyroidism: clinical follow-up, histopathology, and flow cytometric DNA analysis. World J Surg 16(4):737–743 discussion 43-4
Attie JN, Bock G, Auguste LJ (1990) Multiple parathyroid adenomas: report of thirty-three cases. Surgery. 108(6):1014–1019 discussion 9-20
Molinari AS, Irvin GL 3rd, Deriso GT, Bott L (1996) Incidence of multiglandular disease in primary hyperparathyroidism determined by parathyroid hormone secretion. Surgery. 120(6):934–936 discussion 6-7
Rose DM, Wood TF, Van Herle AJ, Cohan P, Singer FR, Giuliano AE (2001) Long-term management and outcome of parathyroidectomy for sporadic primary multiple-gland disease. Arch Surg 136(6):621–626
Shane E (2001) Clinical review 122: parathyroid carcinoma. J Clin Endocrinol Metab 86(2):485–493
Adam MA, Untch BR, Olson JA Jr (2010) Parathyroid carcinoma: current understanding and new insights into gene expression and intraoperative parathyroid hormone kinetics. Oncologist. 15(1):61–72
Obara T, Fujimoto Y (1991) Diagnosis and treatment of patients with parathyroid carcinoma: an update and review. World J Surg 15(6):738–744
Author information
Authors and Affiliations
Contributions
MC de Jong: Study conception and design; acquisition, analysis, and interpretation of data; drafting of manuscript.
K Jamal: Study conception and design; acquisition of data; critical revision.
S Morley: Acquisition of data; analysis and interpretation of data.
T Beale: Acquisition of data; analysis and interpretation of data.
T Chung: Study conception and design; acquisition of data; analysis and interpretation of data.
S Jawad: Acquisition of data; analysis and interpretation of data.
S Hurel: Acquisition of data; analysis and interpretation of data.
H Simpson: Acquisition of data; analysis of data.
U Srirangalingam: Acquisition of data; analysis of data.
SE Baldeweg: Critical revision of revised manuscript.
V Rozalén García: Acquisition of data; analysis and interpretation of data.
S Otero: Acquisition of data; analysis of data.
M Shawky: Acquisition of data; analysis and interpretation of data.
TE Abdel-Aziz: Study conception and design; critical revision.
TR Kurzawinski: Analysis and interpretation of data; drafting of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of our institution and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. This retrospective study was registered as an audit of our current practice with our IRB and was exempt from review.
Informed consent
Not applicable; anonymized retrospective data analysis, consent requirement waived.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Jong, M.C., Jamal, K., Morley, S. et al. The use of computed tomography as a first-line imaging modality in patients with primary hyperparathyroidism. Hormones 20, 499–506 (2021). https://doi.org/10.1007/s42000-020-00205-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42000-020-00205-x