Abstract
Discharging of untreated or partially treated textile wastewater is common in Ethiopia, and this has detrimental effect to the environment. It is difficult to treat textile wastewater by conventional biological processes. In this study, real textile wastewater was taken and treated using sequencing batch reactor using a biomass taken from domestic wastewater treatment plant. Cycle period, air flowrate and sludge retention time (SRT) were initially optimized using the response surface methodology. The optimum ratio of cycle period/air flowrate/SRT which gives a 57% COD removal and 54% color removal was found to be 25 h/15 L/h/16 day. Using two types of wastewater substrate concentrations and various hydraulic retention times at optimized condition, COD removal, color removal, sludge volume index (SVI) and mixed liquor suspended solid were measured. The maximum of COD removal (73%) and color removal (65.8%) was obtained at an organic loading rate of 0.078 kg COD/m3 day. SVI at the optimized condition was found to be 90–92 mL/g. Finally, a first-order kinetic model was used to represent the degradation of textile wastewater.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The need of textile products is growing faster, and this leads to the increase in the quantity of textile effluent which discharges into the environment [1]. Currently, textile industry sector is growing faster in Ethiopia. In addition, the sector releases enormous amount of wastewater. It usually contains toxic and non-biodegradable pollutants. The effluent usually contains chemical oxygen demand (COD), biological oxygen demand (BOD), alkalinity and total dissolved solids. Moreover, textile effluent usually comprises synthetic dyes, dispersants, bases, acids, detergents, salts, inhibitory compounds and many other compounds. In addition to this, they are not easily degraded due to their complex structure and synthetic origin [1,2,3,4]. Some of the adverse effects of this effluent are reducing the amount of solar energy to penetrate the water bodies, lack of sufficient reoxygenation process and this cause detrimental effect to aquatic flora and fauna [5], and such type of wastewater can also cause cancer and extremely toxic to human beings [6].
By considering the detrimental effects of textile effluent on the environment, adequate and effective treatment technologies should be investigated [7]. They can be treated by physical and chemical process such as coagulation, flocculation, ozonation, oxidation, ion exchange, irradiation, precipitation and adsorption [8, 9]. However, these processes have some limitations such as excess amount of chemical usage, generation of excess sludge and additional cost to dispose such solid waste, requirement of high operational cost and sensitivity to variable wastewater influent [8, 10]. In this context, biological wastewater treatment is preferably selected due to its efficiency and inexpensive nature. The conventional activated sludge process was widely used for greater than 100 years for the treatment of wastewater. However, it has several operational limitations. Some of the limitations are: high biomass production, energy consumption and high operational cost [11]. On the other hand, recently, strict environmental protection legislation and emergence of better technology are the causes for the substitution of conventional activated sludge process by highly efficient and relatively cheap sequencing batch reactor (SBR) [11]. SBR is usually used for the removal of xenobiotic compounds in industrial and domestic wastewater. SBR has the following benefits at small scale: low construction and maintenance cost and flexibility in operation [12,13,14].
SBR usually uses aerobic bacteria similar to conventional activated sludge process. It can degrade and convert the biodegradable portion of the organic matter into new cells, gases and salts [15]. The SBR operates in five sequential stages (wastewater feeding, reaction, sedimentation, taking of the clarified effluent and idle stage) in discontinuous modes. This process has various advantages over conventional activated sludge process [11]. Some of the advantages are simplicity and flexibility, low cost, resistance to fluctuations in the influent and also process such as equalization, reaction and clarification that occur in the same reactor [11, 15].
The SBR wastewater treatment process is more efficient than conventional activated sludge process with respect to economy and time. Moreover, 60% treatment costs can be saved [16] and also above 90% removal efficiency of biodegradable organic matter can be obtained [17] as compared to conventional activated sludge process.
Previous research reported about the use of SBR for the treatment of both domestic and industrial wastewater [15]. It effectively removes nutrients from domestic wastewater [18]. In addition to this, it can remove various environmental pollutants from industrial influents, piggery [19], paper mill [14], land fill leachate [20, 21] and textile wastewater [22,23,24,25,26,27].
Although a lot of researches have been carried out in the treatment of textile wastewater using SBR [22,23,24,25,26,27], to the best of authors’ knowledge, the process factors which were optimized in this study were not addressed by several previous researchers. Therefore, the main objective of this study was to investigate the optimum conditions for SBR process to remove COD and color from real textile wastewater using native bacteria taken from sludge of the municipal wastewater treatment plant and also to study the kinetics of the degradation of the textile wastewater.
Materials and methods
Wastewater collection
Raw textile wastewater sample was taken from KK acrylic fiber dyeing process textile factory located in Addis Ababa, capital city of Ethiopia. The samples were taken during dyeing process, particularly at a time of dark shading just after screening of course particles. Sampling bottles were cleaned and rinsed with distilled water before a new sample was taken. The characteristics of each sample were measured on site at the factory and were measured as soon as it reaches to the laboratory within that day to check any change of it during transportation process. It was refrigerated at 4 °C. The characteristics of KK textile wastewater are indicated in Table 1.
Sequential batch reactor
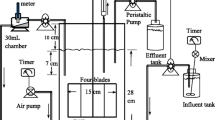
The setup of the laboratory-scale SBR is shown in Fig. 1. It is made of plexi glass with a total volume of 5.5L and working volume of 4L. Feeding of the wastewater and withdrawal of the decanted effluent were carried out using peristaltic pumps. Air is suppled with air diffuser. Mixing was taken place with mechanical stirrer at a speed of 150rph. The reactor was seeded with biomass collected from the Kalti domestic wastewater treatment plant in Addis Ababa, Ethiopia. In order to acclimatize the biomass, the SBR setup was left for 8 days with scheduled aeration at the rate of 8L/h. Consequently, the reactor was filled with textile wastewater. The pH of the wastewater was fixed to 6.8, and its temperature was also calibrated at 28 °C.
Experimental procedure of SBR
The potential process factors which were used in this study are: cycle period (12, 24 and 36 h), air flow rate (8, 13 and 18 L/h) and SRT (12, 16 and 20 day). The ranges and levels used for these process variables are shown in Table 2. The hydraulic retention time in the reactor was maintained as 4 days. These process factors were optimized with Box–Behnken design (BBD). The SBR was working with five consecutive batch processes, namely feeding, aeration, sedimentation, draw step (decant) and idle step. During 0.5 h, raw textile wastewater was fed into the reactor, aeration carried out for 10 h, sedimentation carried out for 1 h, the decanted effluent taken for 0.38 h and the system closed for 0.13 h. This process is recycled using fresh textile wastewater as a feed based on corresponding cycle periods. During the idle stage, excess sludge was removed at the bottom of the reactor.
Experiments were also conducted at optimized conditions at various organic loading rates (OLRs) and hydraulic retention times (HRTs) as shown in Table 3 using two distinct influent substrate concentrations obtained during dark shading and medium shading of the textile dyeing process (1.25 and 1.96 g COD/L). COD removal, color removal, mixed liquor suspended solid (MLSS) and sludge volume index (SVI) were measured using standard method of analysis.
Experimental design, analysis and statistical validation
Response surface methodology (RSM) was used for the optimization of experiments. In this research, BBD is used and RSM was employed for optimization of COD and color removal efficiencies. Here, a 23 factorial design was used to identify the influence of three parameters including cycle period (A), air flow rate (B) and sludge retention time (C). These factors were chosen based on the literature and preliminary experiments. From these preliminary experiments, the range of A, B and C was selected as shown in Table 2. A total of 17 experiments were conducted with five replicates at the central point, and the designation of values was according to Moghaddam et al. [28]. A second-order regression model was employed for analysis and proves to be a good estimation of response surface [29] and is expressed as shown in Eq. 1:
where y = response; xi and xj = input variables; β0 = intercept constant; βi = first-order regression coefficient; βii = second-order regression coefficient representing quadratic effect of factor i; and βij = coefficient of interaction between two factors i and j [30]. The analysis of variance (ANOVA) result was obtained using statistical software package Design-Expert® version 7.0.0 (Stat-Ease, Inc.) to study the results and to determine the implication of the fitted quadratic model. The fitted model was illustrated in the form of contour plots to know the interaction between the variables and responses. The quality of model was checked [31] using several coefficients such as the correlation coefficient R2, which evaluates the correlation between experimental data and predicted responses [32], the adjusted R2 which takes the number of factors into account [33] and the predicted R2 which indicates how much a regression model predicts responses for new observations.
The adequate precision was used to describe the signal-to-noise ratio [34]. This value should be higher than 4. Finally, the coefficient of variation factor describes the repeatability and reproducibility of the models generated [35].
Results and discussion
Regression model and statistical testing
The experimental results originating from the BBD are summarized in Table 4, and the resulting model values obtained by using the RSM model are summarized in Tables 5, 6 and 7. Optimal values for color removal COD and measurement of sludge volume index in the experimental runs could be determined by a second-order polynomial as expressed by Eqs. 2, 3 and 4:
where R1 stands for color removal, R2 stands for COD removal and R3 accounts for SVI, showing the regression coefficients and model performance indicators for the RSM model. In Eqs. 2, 3 and 4, positive effect of a factor implies the response is improved when the factor level increases and a negative effect of the factor means that the response is not improved when the factor level increases [36]. Values of probability < 0.05 show that model terms are significant, and values > 0.1000 point out that model terms are not significant [35].
In the case of both COD and color removal, it can be seen that A, B, C, BC, A2, B2 and C2 were significant terms, and the rest of the terms were not significant and therefore not included in the equation for both COD and color removal while for SVI it was concluded that A, B, C, AC, BC, A2, B2 and C2 were proved to be significant. Thus, statistical analysis showed that not all variables had a significant effect.
The relationship between each of the factors and the role of each process factor can be described using Fisher’s test. The smaller P > f value and the greater F value usually indicate the adequacy of the model [37]. It was further observed that the corresponding p-values of the Fisher’s test for COD removal, color removal and SVI were < 0.0001 which indicated the regression itself was significant and adequate [35].
The value of the predicted R2 for all the three responses was found to be > 0.97. This implies the prediction of experimental data was satisfactory. Low values of coefficient of variation (C.V) indicated high accuracy and dependability of experiments as the values are < 10%. In this case, all the three responses have a C.V value of < 10%.
The contour figures were drawn to indicate the interaction between two independent factors. Moreover, the value of one variable is kept constant to clearly see the interaction among two independent process factors. The contour plots clearly illustrate the behavior of the system in the experimental design. These plots are indicated in Figs. 2 and 4. The behavior of the response surface plot usually indicates the relationship between the process factors. The nature of the response surface curves shows the interaction between the variables. The elliptical shape of the curve indicates good interaction between the two variables, and circular shape indicates no interaction between the variables. From the figures, it is observed that the elliptical nature of the contour in all the graphs depicts the mutual interactions of all the variables. There was a relatively significant interaction between every two variables, and predicted decolorization and COD reduction were maximum as indicated by the surface confined in the smallest ellipse in the contour diagrams. Hence, in these plots, there was a significant interaction between process factors and predicted COD and color removal were highest which is shown by the surface confined in the smallest ellipse in the counter plots.
Effect of process variables on treatment of textile wastewater in SBR
Figure 2 shows the effect of air flow rate and SRT on COD removal. From this figure, it can be seen that an initial increase in air flow rate causes the increase in the removal of COD. However, further increase in air flow rate above 13.8 L/h causes the decrease in COD removal efficiency. The possible reason for the increase in COD removal at low value of air flowrate was due to better interaction between air and microorganisms. On the contrary, the decrease in COD removal at high air flow rate is due to the decrease in the retention time of air. Moreover, the decrease in the retention time of air can lead to the decrease in the interaction between microorganisms and air molecules.
Figure 2 also shows that the increase in SRT up to 16 days leads to the increase in COD removal efficiency. The effect of air flow rate and SRT on decolorization (Fig. 3) was in similar pattern with that of COD removal observed in Fig. 2, while cycle period has no effect for both COD and color removal. Similar trend of results was reported by previous researcher [38].
The effect of air flow rate on SVI is shown in Fig. 4; initially, it was observed that the value of air flowrate increases from 8 to 16 L/h while the value of SVI was decreased. However, further increase in air flowrate leads to the increase in the value of SVI. This phenomenon can be occurred due to the breakdown of sludge’s at higher air flow rate. Furthermore, the figures also illustrated that air flowrate beyond 16L/h improves the value of SVI. In addition, the effect of SRT on SVI is shown in Fig. 4. Accordingly, the increase in SRT from 10 to 16 days causes the decrease in the value of SVI. However, at the maximum level of SRT (20d) SVI values were proportionally increased.
In Fig. 4, cycle period has no effect on the value of SVI. SVI was the best indicator of sludge settling properties. The value of SVI obtained experimentally in this research was 80–115 mL/g. The value of SVI can be in the range of 30–400 mL/g. If the value of SVI is less than 150 mL/g, it usually shows better settling properties of the sludge. On the other hand, if the value of SVI is greater than 150 mL/g, it usually indicates bulking of the sludge [39]. In this sense, the value of SVI which was experimentally measured in this research was 80–115 mL/g. This result was relatively lower than the results which were reported by previous researchers [40, 41].
Optimization of process factors of SBR-treated textile waste water for color and COD removal
Optimization was carried out using statistical software package Design-Expert® version 7.0.0 (Stat-Ease, Inc.) with BBD. Optimal values of the three responses were based on Eqs. 2, 3 and 4. Furthermore, the main purpose of optimization was to achieve the maximum efficiency of the SBR wastewater treatment process. In addition, optimization was carried out based on preferred selected criteria. In this sense, COD and color removal efficiency were selected to be maximized, cycle period, air flow rate and SRT were selected in range, while SVI was selected to be minimized. Based on these selection criteria, optimum working conditions and percent removal efficiencies were obtained. The optimum values obtained from BBD based on the selected criteria were: cycle period 25 h, air flow rate 15 L/h and SRT 16 d with COD and color removal efficiency of 57% and 54%, respectively.
Optimum condition performance of SBR at various OLRs
Further experiments were conducted based on optimum conditions of the process factors by varying influent substrate concentrations (1.25 and 1.96 g COD/L) and HRT (4, 3 and 2d). Moreover, the value of OLR varies based on the duration of these HRTs and quantities of influent substrate concentrations. The detailed working conditions are given in Table 3. The total number of days of operation was seventy 7 days. In this reactor, continuous working days, COD removal, decolorization and value of SVI were investigated based on standard methods of water and wastewater treatment analysis [42].
In the beginning of the reactor performance, the influent substrate concentration was 1.25 g COD/L and HRT was 4 days while the OLR was 0.078 kg COD/m3 day. In this process condition, the removal of both COD and color was minimum due to the microorganism required longer time for adaptation of the reactor environment. However, gradually within 17 days (1–17 days) of the reactor continuous operation the system reached steady-state condition. At this stage, a maximum of 73% COD and 65.8% color removal efficiency was achieved. Likewise, on the 18th day the OLR was increased to 0.104 kg COD/m3 day by decreasing the HRT to 3 days while the influent wastewater concentration was kept constant. In these process conditions, in the rector within 6 days (18–23 days) another steady-state condition was achieved. At this stage, a maximum of 71.6% COD and 63.7% color removal was obtained. Similarly, on the 24th day the OLR was increased to 0.156 kg COD/m3 day by decreasing the HRT to 2 days while keeping the influent concentration constant. In this process condition, the rector reached steady state within 14 days of continuous operation (24–37 days). At this stage, a maximum of 64.5% COD and 58% color removal was obtained.
After 37 days of reactor operation (on the 38th day), the inlet substrate concentration was increased to 1.96 g COD/L, the HRT was 4 days, and the corresponding OLR was 0.122 kg COD/m3 day. In this process condition, within 15 days of the reactor continuous operations (38–52 days), the system reached steady-state condition. At this stage, a maximum of 56.1% COD reduction and 51.6% color removal was achieved. In addition, on the 53rd day the OLR was increased to 0.163 kg COD/m3 day by decreasing the HRT to 3 days while keeping the influent substrate concentration constant. In these process conditions, within 9 days (53–61 days) of reactor operation steady-state conditions were achieved. At this stage, a maximum of 55.6% COD and 50.5% color removal was obtained. Finally, on the 62nd day the OLR was increased to 0.245 kg COD/m3 day by decreasing the HRT to 2 days while keeping the influent concentration constant. In this process condition, the reactor reached steady state within 16 days of continuous operation (62–77 days). At this stage, a maximum of 48.7% COD and 42.5% color removal was obtained.
The percent of COD removal and decolorization using various OLRs and HRTs for a total of 77 continuous days at steady-state conditions are summarized in Table 8. In addition, the trends of COD and color removal using various HRTs and OLRs are also clearly illustrated in Fig. 5 . In this sense, whenever HRT decreased, the OLR increased, and usually, such condition always results in the decrease in percent removal efficiency of both COD and color. This outcome also indicated how SBR resists the variation in OLR. Similar trend was also observed by previous researchers by increasing the OLR and using various inlet concentrations. Kapdan and Oztekin did experiment on SBR performance on simulated dye wastewater using different HRT, and they reported that whenever HRT decreased, COD removal and decolorization also decreased [43].
The trend of measured SVI in SBR is clearly illustrated in Fig. 6. Initially, the value of SVI in the reactor was 180 mg/L. However, after 17 days of reactor operation the value of SVI was dramatically changed. Gradually, the SVI starts to decrease; at the end of the reactor operation, the value of SVI decreased to 90 mL/g. The measured SVI at optimum condition was mainly in the range of 90–92 mL/g. The value of SVI obtained in this study was small and in agreement with previous studies. Previous researchers obtained the value of SVI in the range of 30–60 mL/g [41] and also in the range of 65–105 mL/g [40].
During the initial stage of SBR operation, MLSS was decreased from 1495 to 1290 mg/L. The possible reason for the decrease in MLSS at this stage could be due to discharge of smaller sludge. On the other hand, after the SBR runs continuously for several days, the MLSS starts to increase from 1780 to 5993 mg/L. However, gradually the amount of sludge starts to slightly decrease due to gradual formation of large-sized granules. It is known that the formation of large-sized granules inhibits the availability of dissolved oxygen and nutrients to the microorganisms. In addition, this phenomenon also inhibits the growth of microorganisms in the system. These trends of similar results were also addressed by previous researchers [44,45,46,47,48].
Kinetic study in SBR
The kinetic study was carried out for the treatment of textile wastewater at various levels of substrate concentration and hydraulic retention time. The microorganisms usually used the available substrate concentrations in wastewater. The biodegradation process is usually expressed by the first-order kinetic model. By integrating in between defined limit, such a first-order model can be expressed as:
where CSo is the initial substrate concentrations (g COD/L), CS is the substrate concentration (g COD/L), t is the degradation time (day), K1 is the first-order rate constant (day−1). Figure 7 shows how the experimental data fitted with the first-order model at different substrate concentrations and various hydraulic retention times. The first-order rate constant (K1) was calculated from the slope of the line by least square fit in the figure.
The value of R2 and K is shown in Table 9. The values of R2 indicated the ability of the model to represent the first order used in this kinetic model. K values for 4 day and 3 day were nearer to each other, the smallest K values in the SBR were observed at the highest organic loading rate (0.245 KgCOD/m3 day), and the possible reason for this was due to inhibitory effect of the substrate on the microbial activity.
Conclusion
In this study, real textile wastewater was taken and treated using SBR using a biomass taken from domestic wastewater treatment plant. Process factors such as cycle period, air flowrate and sludge retention time (SRT) were optimized using RSM. The optimum ratio of cycle period/air flowrate/HRT which gives a 57% COD removal and 54% color removal was found to be 25 h/15L/h/16 d. Using two ranges of influent substrate concentrations and HRT at optimized condition COD removal, color removal, SVI and MLSS were measured. The maximum removal of 73% COD and 65.8% color was obtained at an organic loading rate of 0.078 kg COD/m3 day. It is also possible to conclude that as HRT decreases, there is a decrease in the percent COD and color removal. Significant reduction in percent COD and color removal is observed between HRT 3 and 2 days. Small value of SVI usually indicated good performance of SBR. The first-order model was best to represent degradation of textile wastewater using SBR. It is possible to conclude that SBR can be applied in the treatment of textile wastewater.
References
Daud NK, Akpan UG, Hameed BH (2011) Decolorization of sunzol black DN conc. in aqueous solution by Fenton oxidation process, effect of system parameters and kinetic study. Desalin Water Treat 3994:1–7. https://doi.org/10.5004/dwt.2012.1213
Hayat H, Mahmood Q, Pervez A et al (2015) Comparative decolorization of dyes in textile wastewater using biological and chemical treatment. Sep Purif Technol 154:149–153. https://doi.org/10.1016/j.seppur.2015.09.025
Ellouze E, Tahri N, Ben Amar R (2012) Enhancement of textile wastewater treatment process using Nanofiltration. Desalination 286:16–23. https://doi.org/10.1016/j.desal.2011.09.025
Fersi C, Dhahbi M (2008) Treatment of textile plant effluent by ultrafiltration and/or nanofiltration for water reuse. Desalination 222:263–271. https://doi.org/10.1016/j.desal.2007.01.171
Clarke EA, Anliker R (1980) Organic dyes and pigments. Springer, New York
Puvaneswari N, Muthukrishnan J, Gunasekaran P (2006) Toxicity assessment and microbial degradation of azo dyes. IJEB 1009:618–626
Chung K, Fulk GE, Egan M (1978) Reduction of Azo dyes by intestinal anaerobes. Appl Environ Microbiol 35:558–562
Shi B, Li G, Wang D et al (2007) Removal of direct dyes by coagulation: the performance of preformed polymeric aluminum species. J Hazard Mater 143:567–574. https://doi.org/10.1016/j.jhazmat.2006.09.076
Rajeshkannan R, Rajasimman M, Rajamohan N (2010) Optimization, equilibrium and kinetics studies on sorption of Acid Blue 9 using brown marine algae Turbinaria conoides. Biodegradation 21:713–727. https://doi.org/10.1007/s10532-010-9337-0
Verma P, Madamwar D (2003) Decolourization of synthetic dyes by a newly isolated strain of Serratia marcescens. World J Microbiol Biotechnol 19:615–618. https://doi.org/10.1023/A:1025115801331
Singh M, Srivastava RK (2011) Sequencing batch reactor technology for biological wastewater treatment: a review. Asia Pac J Chem Eng 199–203. https://doi.org/10.1002/apj
Oliveira RP, Ghilardi JA, Ratusznei SM et al (2008) Anaerobic sequencing batch biofilm reactor applied to automobile industry wastewater treatment: volumetric loading rate and feed strategy effects. Chem Eng Process Process Intensif 47:1374–1383. https://doi.org/10.1016/j.cep.2007.06.014
Mohan SV, Rao NC, Prasad KK et al (2005) Treatment of complex chemical wastewater in a sequencing batch reactor (SBR) with an aerobic suspended growth configuration. Process Biochem 40:1501–1508. https://doi.org/10.1016/j.procbio.2003.02.001
Tsang YF, Hua FL, Chua H et al (2007) Optimization of biological treatment of paper mill effluent in a sequencing batch reactor. Biochem Eng J 34:193–199. https://doi.org/10.1016/j.bej.2006.12.004
Rodrigues CSD, Madeira LM, Boaventura RAR (2014) Synthetic textile dyeing wastewater treatment by integration of advanced oxidation and biological processes—performance analysis with costs reduction. 2:1027–1039
Ng WJ, Sim TS, Ong SL et al (1993) Efficiency of sequencing batch reactor (SBR) in the removal of selected microorganisms from domestic sewage. Water Res 27:1591–1600. https://doi.org/10.1016/0043-1354(93)90105-Q
Metcalf & Eddy (2003) Wastewater engineering: treatment and reuse, 4th edn. McGraw Hill, New York
Ersu CB, Arslankaya E (2006) Biological nutrient removal in a sequencing batch reactor. Water Sci Technol 33:29–38
Su J, Kung C, Lin J et al (1997) Utilization of sequencing batch reactor for in situ piggery wastewater treatment. J Environ Sci Heal Part A 32:391–405
Morling S (2010) Nitrogen removal and heavy metals in leachate treatment using SBR technology. J Hazard Mater 174:679–686. https://doi.org/10.1016/j.jhazmat.2009.09.104
Klimiuk E, Kulikowska D (2004) Effectiveness of organics and nitrogen removal from municipal landfill leachate in single- and two-stage SBR systems. Polish J Environ Stud 13:525–532
Sirianuntapiboon S, Sadahiro O, Salee P (2007) Some properties of a granular activated carbon-sequencing batch reactor (GAC-SBR) system for treatment of textile wastewater containing direct dyes. J Environ Manage 85:162–170. https://doi.org/10.1016/j.jenvman.2006.09.001
Sirianuntapiboon S, Chairattanawan K, Jungphungsukpanich S (2006) Some properties of a sequencing batch reactor system for removal of vat dyes. Bioresour Technol 97:1243–1252. https://doi.org/10.1016/j.biortech.2005.02.052
Abu-ghunmi LN, Jamrah AI (2006) Biological treatment of textile wastewater using sequencing batch reactor technology. Environ Model Assess 11:333–343. https://doi.org/10.1007/s10666-005-9025-3
Fu LY, Wen XH, Yi Qian QLL (2001) Treatment of dyeing wastewater in two SBR systems. Process Biochem 36:1111–1118. https://doi.org/10.1016/S0032-9592(01)00143-1
Lourenco ND, Novais JM, Pinheiro HM (2000) Reactive textile dye colour removal in a sequencing batch reactor. Water Sci Technol 42:321–328
Khouni I, Marrot B, Ben R (2012) Treatment of reconstituted textile wastewater containing a reactive dye in an aerobic sequencing batch reactor using a novel bacterial consortium. Sep Purif Technol 87:110–119. https://doi.org/10.1016/j.seppur.2011.11.030
Moghaddam SS, Moghaddam MRA, Arami M (2010) Coagulation/flocculation process for dye removal using sludge from water treatment plant: optimization through response surface methodology. J Hazard Mater 175:651–657. https://doi.org/10.1016/j.jhazmat.2009.10.058
Zhu X, Tian J, Liu R, Chen L (2011) Optimization of Fenton and electro-Fenton oxidation of biologically treated coking wastewater using response surface methodology. Sep Purif Technol 81:444–450. https://doi.org/10.1016/j.seppur.2011.08.023
Xu H, Qi S, Li Y, Zhao Y (2013) Heterogeneous Fenton-like discoloration of Rhodamine B using natural schorl as catalyst: optimization by response surface methodology. Env Sci Pollut Res 20:5764–5772. https://doi.org/10.1007/s11356-013-1578-0
Gilpavas E, Dobroz-Gomoz I G-GM (2012) Decolorization and mineralization of Diarylide Yellow 12 (PY12) by photo-Fenton process : the Response Surface Methodology as the optimization tool Edison GilPavas, Izabela Dobrosz-Gómez and Miguel Ángel Gómez-García. Water Sci Technol 65:1795–1801. https://doi.org/10.2166/wst.2012.078
Fathinia M, Khataee AR, Zarei M, Aber S (2010) A: chemical comparative photocatalytic degradation of two dyes on immobilized TiO2 nanoparticles: Effect of dye molecular structure and response surface approach. J Mol Catal AChemical 333:73–84. https://doi.org/10.1016/j.molcata.2010.09.018
Sharma S, Kapoor S, Sharma RAC (2017) Effect of Fenton process on treatment of simulated textile wastewater: optimization using response surface methodology. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-017-1253-y
Mason R, Gunst R, Hess J (2003) Statistical design and analysis of experiments with applications to engineering and science, 2nd edn. Wiley, New York
Rosales E, Sanromán MA, Pazos M (2012) Application of central composite face-centered design and response surface methodology for the optimization of electro-Fenton decolorization of Azure B dye. Env Sci Pollut Res 19:1738–1746. https://doi.org/10.1007/s11356-011-0668-0
Saldana-Robels A, Gerra-Sanchez R, Maldonado-Rubio MP-HJ (2014) Optimization of the operating parameters using RSM for the Fenton oxidation process and adsorption on vegetal carbon of MO solutions. J Ind Eng Chem 20:848–857. https://doi.org/10.1016/j.jiec.2013.06.015
Montogomery D (2010) Design and analysis of experimenters, 7th edn. Wiley India Pvt Ltd, New Delhi
Novais JM, Pinheiro HM (2001) Effect of some operational parameters on textile dye biodegradation in a sequential batch reactor. J Biotechnol 89:163–174
Palm JC, Jenkins D, Parker DS (1980) Relationship between organic oxygen sludge dissolved loading, and concentration in the completely settleability mixed activated process sludge. J WPCF 52:2484–2506
Zinatizadeh AAL, Mansouri Y, Akhbari A, Pashaei S (2011) Biological treatment of a synthetic dairy wastewater in a sequencing batch biofilm reactor: statistical modeling using optimization using response surface methodology. Chem Ind Chem Eng Q 17:485–495. https://doi.org/10.2298/CICEQ110524034Z
Janczukowicz W, Szewczyk M, Krzemieniewski M, Pesta J (2001) Settling properties of activated sludge from a sequencing batch reactor (SBR)z. Polish J Environ Stud 10:15–20
Clesceri LS, Greenbaerg AE, Eaton AD (1998) Standard methods for examination of water and wastewater (standard methods for the examination of water and wastewater), 20th edn. American Public Health Association (APHA), Washington
Kapdan IK, Oztekin R (2006) The effect of hydraulic residence time and initial COD concentration on color and COD removal performance of the anaerobic-aerobic SBR system. J Hazard Mater 136:896–901. https://doi.org/10.1016/j.jhazmat.2006.01.034
Ong SA, Toorisaka E, Hirata M, Hano T (2005) Treatment of azo dye Orange II in aerobic and anaerobic-SBR systems. Process Biochem 40:2907–2914. https://doi.org/10.1016/j.procbio.2005.01.009
Zheng YM, Yu HQ, Sheng GP (2005) Physical and chemical characteristics of granular activated sludge from a sequencing batch airlift reactor. Process Biochem 40:645–650. https://doi.org/10.1016/j.procbio.2004.01.056
Arrojo B, Mosquera-Corral A, Garrido JM, Méndez R (2004) Aerobic granulation with industrial wastewater in sequencing batch reactors. Water Res 38:3389–3399. https://doi.org/10.1016/j.watres.2004.05.002
Tay J-H, Liu Q-S, Liu Y (2001) Microscopic observation of aerobic granulation in sequential aerobic sludge blanket reactor. J Appl Microbiol 91:168–175. https://doi.org/10.1046/j.1365-2672.2001.01374.x
Tay J-H, Liu QS, Liu Y (2002) The effects of shear force on the formation, structure and metabolism of aerobic granules. Appl Microbiol Biotechnol 57:227–233. https://doi.org/10.1007/s002530100766
Acknowledgements
First, we would like to thank NORAD project of Hawaasa University for the financial support. Finally, the authors would like to acknowledge laboratory workers of Addis Ababa Institute of Technology, Addis Ababa University, for providing the necessary laboratory facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Solomon, D., Kiflie, Z. & Van Hulle, S. Kinetic investigation and optimization of a sequencing batch reactor for the treatment of textile wastewater. Nanotechnol. Environ. Eng. 4, 15 (2019). https://doi.org/10.1007/s41204-019-0062-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41204-019-0062-6