Abstract
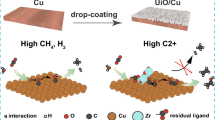
The electrocatalytic CO2 reduction technology is expected to simultaneously alleviate increasing CO2 emissions and the depletion of fossil resources. However, it is still a big challenge to improve the selectivity toward valuable multi-carbon products in electrocatalytic CO2 reduction reaction. In this study, the synergistic role of Cu+ and Cu0 species in Cu2O-Cu interfaces is unravelled through density functional theory (DFT) calculations, in which the electrode surface exhibits low free energy of *COCO intermediate formation and H2O dissociation, which are beneficial to the high selectivity towards multi-carbon products, especially C2H4. Guided by these DFT results, an oxide-derived copper electrode activation strategy that builds the synergistic Cu+ and Cu0 on Cu2O-Cu interfaces is designed to boost the selectivity toward multi-carbon products. Interestingly, Cu2O cubes chosen as the pristine catalyst are activated via a square-wave (SW) potential treatment to form SW-Cu2O cubes that bear Cu+ and Cu0 species. The as-prepared SW-Cu2O cubes exhibit superior Faradaic efficiencies for C2H4 (60%) and C2+ products (75%) in an H-type cell, which are about 1.5 times that of the Cu2O cubes. This study demonstrates the synergistic Cu0 and Cu+ on Cu2O-Cu interfaces for improving the selectivity of a specific valuable multi-carbon product in electrocatalytic CO2 conversion.

摘要
电催化CO2还原技术有望同时缓解化石燃料濒临枯竭及大气中CO2浓度不断攀升等问题. 然而, 对于高附加值的电催化CO2还原多碳产物的选择性提升, 仍然面临巨大挑战. 密度泛函理论(DFT)计算表明,Cu2O−Cu界面上Cu+和Cu0的协同耦合效应使其表面上*COCO中间体的生成能降低, 同时H2O的解离自由能也降低, 从而有利于电催化CO2还原高选择性生成多碳产物特别是C2H4. 受DFT计算结果的启发, 本文设计了一种氧化物衍生铜电极的活化策略, 构建Cu2O−Cu界面, 以Cu+和Cu0协同促进电催化CO2还原高效高选择性生成C2+产物. 其中, Cu2O立方体被用作初始催化剂, 经方波电位处理后, 在Cu2O−Cu界面上形成了具有Cu+和Cu0协同作用的SW-Cu2O/Cu立方体. 在H型电解池中, SW-Cu2O/Cu电催化CO2还原生成C2H4和C2+产物的法拉第效率分别为60%和75%, 约为前驱体Cu2O立方体的1.5倍, 证明Cu0和Cu+在Cu2O−Cu界面上的协同作用的确可提高电催化CO2还原过程中特定高附加值多碳产物的选择性.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Lee MY, Park KT, Lee W, et al. Current achievements and the future direction of electrochemical CO2 reduction: A short review. Crit Rev Environ Sci Tech, 2019, 50: 769–815
De Luna P, Hahn C, Higgins D, et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes?. Science, 2019, 364: aav3506
Peter SC. Reduction of CO2 to chemicals and fuels: A solution to global warming and energy crisis. ACS Energy Lett, 2018, 3: 1557–1561
Qiao J, Liu Y, Hong F, et al. A review of catalysts for the electroreduction of carbon dioxide to produce low-carbon fuels. Chem Soc Rev, 2014, 43: 631–675
Li X, Wu X, Lv X, et al. Recent advances in metal-based electrocatalysts with hetero-interfaces for CO2 reduction reaction. Chem Catal, 2021, 2: 262–291
Wang D, Chang K, Zhang Y, et al. Unravelling the electrocatalytic activity of bismuth nanosheets towards carbon dioxide reduction: Edge plane versus basal plane. Appl Catal B-Environ, 2021, 299: 120693
Wang D, Liu C, Zhang Y, et al. CO2 electroreduction to formate at a partial current density up to 590 mA mg−1via micrometer-scale lateral structuring of bismuth nanosheets. Small, 2021, 17: 2100602
Liang S, Huang L, Gao Y, et al. Electrochemical reduction of CO2 to CO over transition metal/N-doped carbon catalysts: The active sites and reaction mechanism. Adv Sci, 2021, 8: 2102886
Wang X, Feng S, Lu W, et al. A new strategy for accelerating dynamic proton transfer of electrochemical CO2 reduction at high current densities. Adv Funct Mater, 2021, 31: 2104243
Wakerley D, Lamaison S, Ozanam F, et al. Bio-inspired hydrophobicity promotes CO2 reduction on a Cu surface. Nat Mater, 2019, 18: 1222–1227
Nitopi S, Bertheussen E, Scott SB, et al. Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte. Chem Rev, 2019, 119: 7610–7672
Popović S, Smiljanić M, Jovanovič P, et al. Stability and degradation mechanisms of copper-based catalysts for electrochemical CO2 reduction. Angew Chem Int Ed, 2020, 59: 14736–14746
Gao D, Arán-Ais RM, Jeon HS, et al. Rational catalyst and electrolyte design for CO2 electroreduction towards multicarbon products. Nat Catal, 2019, 2: 198–210
Lei Q, Zhu H, Song K, et al. Investigating the origin of enhanced C2+ selectivity in oxide-/hydroxide-derived copper electrodes during CO2 electroreduction. J Am Chem Soc, 2020, 142: 4213–4222
Kuhl KP, Cave ER, Abram DN, et al. New insights into the electrochemical reduction of carbon dioxide on metallic copper surfaces. Energy Environ Sci, 2012, 5: 7050–7059
Zheng Y, Vasileff A, Zhou X, et al. Understanding the roadmap for electrochemical reduction of CO2 to multi-carbon oxygenates and hydrocarbons on copper-based catalysts. J Am Chem Soc, 2019, 141: 7646–7659
Velasco-Vélez JJ, Jones T, Gao D, et al. The role of the copper oxidation state in the electrocatalytic reduction of CO2 into valuable hydrocarbons. ACS Sustain Chem Eng, 2019, 7: 1485–1492
Mistry H, Varela AS, Bonifacio CS, et al. Highly selective plasma-activated copper catalysts for carbon dioxide reduction to ethylene. Nat Commun, 2016, 7: 12123
Wang J, Tan HY, Zhu Y, et al. Linking the dynamic chemical state of catalysts with the product profile of electrocatalytic CO2 reduction. Angew Chem Int Ed, 2021, 60: 17254–17267
Kim D, Lee S, Ocon JD, et al. Insights into an autonomously formed oxygen-evacuated Cu2O electrode for the selective production of C2H4 from CO2. Phys Chem Chem Phys, 2014, 17: 824–830
Yang PP, Zhang XL, Gao FY, et al. Protecting copper oxidation state via intermediate confinement for selective CO2 electroreduction to C2+ fuels. J Am Chem Soc, 2020, 142: 6400–6408
Arán-Ais RM, Scholten F, Kunze S, et al. The role of in situ generated morphological motifs and Cu(I) species in C2+ product selectivity during CO2 pulsed electroreduction. Nat Energy, 2020, 5: 317–325
Tang HF, Huang ZY, Xiao M. Effects of particle size and temperature on surface thermodynamic functions of cubic nano-Cu2O. Acta Physico-Chim Sin, 2016, 32: 2678–2684
Kresse G, Furthmüller J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput Mater Sci, 1996, 6: 15–50
Kresse G, Furthmüller J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys Rev B, 1996, 54: 11169–11186
Kresse G, Joubert D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys Rev B, 1999, 59: 1758–1775
Perdew JP, Burke K, Ernzerhof M. Generalized gradient approximation made simple. Phys Rev Lett, 1998, 77: 3865–3868
Gao Y, Chen Z, Zhu Y, et al. New insights into the generation of singlet oxygen in the metal-free peroxymonosulfate activation process: Important role of electron-deficient carbon atoms. Environ Sci Technol, 2020, 54: 1232–1241
De Luna P, Quintero-Bermudez R, Dinh CT, et al. Catalyst electroredeposition controls morphology and oxidation state for selective carbon dioxide reduction. Nat Catal, 2018, 1: 103–110
Todorova TK, Schreiber MW, Fontecave M. Mechanistic understanding of CO2 reduction reaction (CO2RR) toward multicarbon products by heterogeneous copper-based catalysts. ACS Catal, 2019, 10: 1754–1768
Kortlever R, Shen J, Schouten KJP, et al. Catalysts and reaction pathways for the electrochemical reduction of carbon dioxide. J Phys Chem Lett, 2015, 6: 4073–4082
Mandal SC, Rawat KS, Garg P, et al. Hexagonal Cu(111) monolayers for selective CO2 hydrogenation to CH3OH: Insights from density functional theory. ACS Appl Nano Mater, 2019, 2: 7686–7695
Guo X, Liu S, Huang S. Single Ru atom supported on defective graphene for water splitting: DFT and microkinetic investigation. Int J Hydrogen Energy, 2018, 43: 4880–4892
Wu T, López N, Vegge T, et al. Facet-dependent electrocatalytic water splitting reaction on CeO2: A DFT + U study. J Catal, 2020, 388: 1–10
Li P, Xie Q, Zheng L, et al. Topotactic reduction of layered double hydroxides for atomically thick two-dimensional non-noble-metal alloy. Nano Res, 2017, 10: 2988–2997
Yang F, Deng PL, Wang Q, et al. Metal-organic framework-derived cupric oxide polycrystalline nanowires for selective carbon dioxide electroreduction to C2 valuables. J Mater Chem A, 2020, 8: 12418–12423
Martin L, Martinez H, Poinot D, et al. Comprehensive X-ray photoelectron spectroscopy study of the conversion reaction mechanism of CuO in lithiated thin film electrodes. J Phys Chem C, 2013, 117: 4421–4430
Guo PP, He ZH, Yang SY, et al. Electrocatalytic CO2 reduction to ethylene over ZrO2/Cu−Cu2O catalysts in aqueous electrolytes. Green Chem, 2022, 24: 1527–1533
Gao Y, Yu S, Zhou P, et al. Promoting electrocatalytic reduction of CO2 to C2H4 production by inhibiting C2H5OH desorption from Cu2O/C composite. Small, 2022, 18: 2105212
Wang J, Cheng T, Fenwick AQ, et al. Selective CO2 electrochemical reduction enabled by a tricomponent copolymer modifier on a copper surface. J Am Chem Soc, 2021, 143: 2857–2865
Zhang J, Guo Y, Shang B, et al. Unveiling the synergistic effect between graphitic carbon nitride and Cu2O toward CO2 electroreduction to C2H4. ChemSusChem, 2021, 14: 929–937
Meng DL, Zhang MD, Si DH, et al. Highly selective tandem electroreduction of CO2 to ethylene over atomically isolated nickel-nitrogen site/copper nanoparticle catalysts. Angew Chem Int Ed, 2021, 60: 25485–25492
Qiu XF, Zhu HL, Huang JR, et al. Highly selective CO2 electroreduction to C2H4 using a metal-organic framework with dual active sites. J Am Chem Soc, 2021, 143: 7242–7246
Jiang Y, Choi C, Hong S, et al. Enhanced electrochemical CO2 reduction to ethylene over CuO by synergistically tuning oxygen vacancies and metal doping. Cell Rep Phys Sci, 2021, 2: 100356
Yuan X, Chen S, Cheng D, et al. Controllable Cu0−Cu+ sites for electrocatalytic reduction of carbon dioxide. Angew Chem Int Ed, 2021, 60: 15344–15347
Yin Z, Yu C, Zhao Z, et al. Cu3N nanocubes for selective electrochemical reduction of CO2 to ethylene. Nano Lett, 2019, 19: 8658–8663
Gao Y, Wu Q, Liang X, et al. Cu2O nanoparticles with both {100} and {111} facets for enhancing the selectivity and activity of CO2 electroreduction to ethylene. Adv Sci, 2020, 7: 1902820
Choi C, Kwon S, Cheng T, et al. Highly active and stable stepped Cu surface for enhanced electrochemical CO2 reduction to C2H4. Nat Catal, 2020, 3: 804–812
Wei X, Yin Z, Lyu K, et al. Highly selective reduction of CO2 to C2+ hydrocarbons at copper/polyaniline interfaces. ACS Catal, 2020, 10: 4103–4111
Acknowledgements
This work was supported by the National Key Research and Development Project (2018YFB1502401 and 2018YFA0702002), the Program for Changjiang Scholars and Innovation Research Team in the University (IRT1205), the Fundamental Research Funds for the Central Universities, and the long-term subsidy mechanism from the Ministry of Finance and the Ministry of Education of China. We thank Lu Bai for her help in electron microscopy characterization.
Author information
Authors and Affiliations
Contributions
Wang S carried out the experiments and analyzed the data. Wang D participated in the data analysis, and writing and editing of the manuscript. Tian B plotted figures and assisted in the experiments and data analysis. Gao X carried out the DFT calculations. Han L, Zhong Y, Song S and Wang Z assisted to carry out electrochemical experiments. Li Y directed the DFT calculations. Gui J took part in the format editing of the manuscript. Zhang Y supervized the project and participated in editing and review of the manuscript. Kuang Y and Sun X provided resources, and directed the research.
Corresponding authors
Additional information
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary information
Experimental details and supporting data are available in the online version of the paper.
Ying Zhang received her PhD degree in 2019 from Monash University, Australia, under the supervision of Prof. Jie Zhang and Prof. Alan. M. Bond. Currently, she is an associate professor at Jiangnan University, China. Her research focuses on electrochemical reduction of CO2 including developing efficient catalysts and fundamental study of mechanisms.
Yun Kuang obtained his PhD degree in 2015 and joined the College of Chemistry, Beijing University of Chemical Technology in the same year. His recent research focuses on the design of material interface for active and stable electrocatalysis in energy and environment-related processes.
Rights and permissions
About this article
Cite this article
Wang, S., Wang, D., Tian, B. et al. Synergistic Cu+/Cu0 on Cu2O-Cu interfaces for efficient and selective C2+ production in electrocatalytic CO2 conversion. Sci. China Mater. 66, 1801–1809 (2023). https://doi.org/10.1007/s40843-022-2344-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-022-2344-2