Abstract
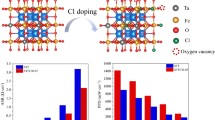
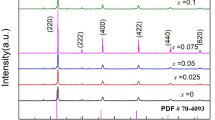
Sr-doped LaMnO3 (LSM) which is the first-generation cathode for solid oxide fuel cells (SOFCs) has been tailored with Zn ions, aiming to achieve improved protonation ability for proton-conducting SOFCs (H-SOFCs). The new Sr and Zn co-doped LaMnO3 (LSMZ) can be successfully synthesized. The first-principle studies indicate that the LSMZ improves the protonation of LSM and decreases the barriers for oxygen vacancy formation, leading to high performance of the LSMZ cathode-based cells. The proposed LSMZ cell shows the highest fuel cell performance among ever reported LSM-based H-SOFCs. In addition, the superior fuel cell performance does not impair its stability. LSMZ is stable against CO2, as demonstrated by both in-situ CO2 corrosion tests and the first-principles calculations, leading to good long-term stability of the cell. The Zn-doping strategy for the traditional LSM cathode with high performance and good stability brings back the LSM cathode to intermediate temperatures and paves a new way for the research on the LSM-based materials as cathodes for SOFCs.

摘要
Sr掺杂的LaMnO3 (LSM)是固体氧化物燃料电池(SOFC)的第一 代阴极. 为了提高其质子化能力从而应用于质子导体SOFC(H-SOFC), 本研究成功合成了Zn和Sr共掺杂的LaMnO3材料(LSMZ), 即通过Zn离 子调节LSM的性能. 第一性原理研究表明, LSMZ提高了材料的质子化 能力, 而且降低了材料中氧空位的形成能, 使LSMZ在电池应用中展现 出较高的性能, 是文献报道同类阴极在H-SOFC中的最大值. 此外, 良好 的燃料电池性能并没有影响材料的稳定性. 原位CO2腐蚀测试和第一性 原理计算表明LSMZ材料对于CO2具有高稳定性, 使LSMZ电池在工作 状态下具有较好的长期稳定性. Zn掺杂策略将传统LSM阴极的高稳定 性与高性能相结合, 将LSM阴极带回到中温工作区间.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Duan C, Kee RJ, Zhu H, et al. Highly durable, coking and sulfur tolerant, fuel-flexible protonic ceramic fuel cells. Nature, 2018, 557: 217–222
Medvedev DA, Lyagaeva JG, Gorbova EV, et al. Advanced materials for SOFC application: Strategies for the development of highly conductive and stable solid oxide proton electrolytes. Prog Mater Sci, 2016, 75: 38–79
Ding H, Wu W, Jiang C, et al. Self-sustainable protonic ceramic electrochemical cells using a triple conducting electrode for hydrogen and power production. Nat Commun, 2020, 11: 1907
Song Y, Chen Y, Wang W, et al. Self-assembled triple-conducting nanocomposite as a superior protonic ceramic fuel cell cathode. Joule, 2019, 3: 2842–2853
Chen Y, Yoo S, Pei K, et al. An in situ formed, dual-phase cathode with a highly active catalyst coating for protonic ceramic fuel cells. Adv Funct Mater, 2018, 28: 1704907
Li Y, Singh M, Zhuang Z, et al. Efficient reversible CO/CO2 conversion in solid oxide cells with a phase-transformed fuel electrode. Sci China Mater, 2021, 64: 1114–1126
Jiang SP. Nanoscale and nano-structured electrodes of solid oxide fuel cells by infiltration: Advances and challenges. Int J Hydrogen Energy, 2012, 37: 449–470
Wachsman ED, Lee KT. Lowering the temperature of solid oxide fuel cells. Science, 2011, 334: 935–939
Zhang Y, Chen B, Guan D, et al. Thermal-expansion offset for highperformance fuel cell cathodes. Nature, 2021, 591: 246–251
Tarutin AP, Lyagaeva JG, Medvedev DA, et al. Recent advances in layered Ln2NiO4+δ nickelates: Fundamentals and prospects of their applications in protonic ceramic fuel and electrolysis cells. J Mater Chem A, 2021, 9: 154–195
Li J, Wang C, Wang X, et al. Sintering aids for proton-conducting oxides—a double-edged sword? A mini review. Electrochem Commun, 2020, 112: 106672
Cao D, Zhou M, Yan X, et al. High performance low-temperature tubular protonic ceramic fuel cells based on barium cerate-zirconate electrolyte. Electrochem Commun, 2021, 125: 106986
Duan C, Huang J, Sullivan N, et al. Proton-conducting oxides for energy conversion and storage. Appl Phys Rev, 2020, 7: 011314
Kilner JA, Burriel M. Materials for intermediate-temperature solidoxide fuel cells. Annu Rev Mater Res, 2014, 44: 365–393
Saranya AM, Pla D, Morata A, et al. Engineering mixed ionic electronic conduction in La0.8Sr0.2MnO3+δ nanostructures through fast grain boundary oxygen diffusivity. Adv Energy Mater, 2015, 5: 1500377
Graves C, Ebbesen SD, Jensen SH, et al. Eliminating degradation in solid oxide electrochemical cells by reversible operation Nat Mater, 2015, 14: 239–244
Zhang X, Liu L, Zhao Z, et al. Enhanced oxygen reduction activity and solid oxide fuel cell performance with a nanoparticles-loaded cathode Nano Lett, 2015, 15: 1703–1709
Painter AS, Huang YL, Wachsman ED. Durability of (La0.8Sr0.2)0.95-MnO3−δ-(Er0.2Bi0.8)2O3 composite cathodes for low temperature SOFCs. J Power Sources, 2017, 360: 391–398
Peng C, Melnik J, Li J, et al. ZnO-doped BaZr0.85Y0.15O3−δ proton-conducting electrolytes: Characterization and fabrication of thin films. J Power Sources, 2009, 190: 447–452
Da’as EH, Bi L, Boulfrad S, et al. Nanostructuring the electronic conducting La0.8Sr0.2MnO3−δ cathode for high-performance in proton-conducting solid oxide fuel cells below 600°C. Sci China Mater, 2018, 61: 57–64
Peng R, Wu T, Liu W, et al. Cathode processes and materials for solid oxide fuel cells with proton conductors as electrolytes J Mater Chem, 2010, 20: 6218–6225
Muñoz-García AB, Pavone M. First-principles design of new electrodes for proton-conducting solid-oxide electrochemical cells: A-site doped Sr2Fe1.5Mo0.5O6−δ perovskite. Chem Mater, 2016, 28: 490–500
Xu X, Wang H, Fronzi M, et al. Tailoring cations in a perovskite cathode for proton-conducting solid oxide fuel cells with high performance. J Mater Chem A, 2019, 7: 20624–20632
Zohourian R, Merkle R, Raimondi G, et al. Mixed-conducting perovskites as cathode materials for protonic ceramic fuel cells: Understanding the trends in proton uptake. Adv Funct Mater, 2018, 28: 1801241
Xu X, Xu Y, Ma J, et al. Tailoring electronic structure of perovskite cathode for proton-conducting solid oxide fuel cells with high performance. J Power Sources, 2021, 489: 229486
Xu X, Bi L, Zhao XS. Highly-conductive proton-conducting electrolyte membranes with a low sintering temperature for solid oxide fuel cells. J Membrane Sci, 2018, 558: 17–25
Bi L, Shafi SP, Da’as EH, et al. Tailoring the cathode-electrolyte interface with nanoparticles for boosting the solid oxide fuel cell performance of chemically stable proton-conducting electrolytes. Small, 2018, 14: 1801231
Wang B, Liu X, Bi L, et al. Fabrication of high-performance proton-conducting electrolytes from microwave prepared ultrafine powders for solid oxide fuel cells. J Power Sources, 2019, 412: 664–669
Kresse G, Furthmüller J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys Rev B, 1996, 54: 11169–11186
Blöchl PE, Jepsen O, Andersen OK. Improved tetrahedron method for brillouin-zone integrations. Phys Rev B, 1994, 49: 16223–16233
Xu Y, Xu X, Cao N, et al. Perovskite ceramic oxide as an efficient electrocatalyst for nitrogen fixation. Int J Hydrogen Energy, 2021, 46: 10293–10302
Tao Z, Xu X, Bi L. Density functional theory calculations for cathode materials of proton-conducting solid oxide fuel cells: A mini-review. Electrochem Commun, 2021, 129: 107072
Ji Q, Xu X, Liu X, et al. Improvement of the catalytic properties of porous lanthanum manganite for the oxygen reduction reaction by partial substitution of strontium for lanthanum. Electrochem Commun, 2021, 124: 106964
Muñoz-García AB, Tuccillo M, Pavone M. Computational design of cobalt-free mixed proton-electron conductors for solid oxide electrochemical cells. J Mater Chem A, 2017, 5: 11825–11833
Zhang X, Pei C, Chang X, et al. FeO6 octahedral distortion activates lattice oxygen in perovskite ferrite for methane partial oxidation coupled with CO2 splitting. J Am Chem Soc, 2020, 142: 11540–11549
Ren R, Wang Z, Meng X, et al. Tailoring the oxygen vacancy to achieve fast intrinsic proton transport in a perovskite cathode for protonic ceramic fuel cells. ACS Appl Energy Mater, 2020, 3: 4914–4922
Kreuer KD. Proton-conducting oxides. Annu Rev Mater Res, 2003, 33: 333–359
Parbey J, Wang Q, Lei J, et al. High-performance solid oxide fuel cells with fiber-based cathodes for low-temperature operation. Int J Hydrogen Energy, 2020, 45: 6949–6957
Dai H, Xu X, Liu C, et al. Tailoring a LaMnO3 cathode for proton-conducting solid oxide fuel cells: Integration of high performance and excellent stability. J Mater Chem A, 2021, 9: 12553–12559
Wang Q, Hou J, Fan Y, et al. Pr2BaNiMnO7−δ double-layered Rud-dlesden-Popper perovskite oxides as efficient cathode electrocatalysts for low temperature proton conducting solid oxide fuel cells. J Mater Chem A, 2020, 8: 7704–7712
Li J, Hou J, Lu Y, et al. Ca-containing Ba0.95Ca0.05Co0.4Fe0.4Zr0.1O3−δ cathode with high CO2-poisoning tolerance for proton-conducting solid oxide fuel cells. J Power Sources, 2020, 453: 227909
Bu Y, Joo S, Zhang Y, et al. A highly efficient composite cathode for proton-conducting solid oxide fuel cells. J Power Sources, 2020, 451: 227812
Zhang Y, Zhu A, Guo Y, et al. Electrochemical performance and effect of moisture on Ba0.5Sr0.5Sc0.175Nb0.025Co0.8O3−δ oxide as a promising electrode for proton-conducting solid oxide fuel cells. Appl Energy, 2019, 238: 344–350
Xia Y, Jin Z, Wang H, et al. A novel cobalt-free cathode with triple-conduction for proton-conducting solid oxide fuel cells with unprecedented performance. J Mater Chem A, 2019, 7: 16136–16148
Pikalova E, Kolchugin A, Koroleva M, et al. Functionality of an oxygen Ca3Co4O9+δ electrode for reversible solid oxide electrochemical cells based on proton-conducting electrolytes. J Power Sources, 2019, 438: 226996
Xie Y, Shi N, Huan D, et al. A stable and efficient cathode for fluorine-containing proton-conducting solid oxide fuel cells. ChemSusChem, 2018, 11: 3423–3430
Huan D, Shi N, Zhang L, et al. New, efficient, and reliable air electrode material for proton-conducting reversible solid oxide cells. ACS Appl Mater Interfaces, 2018, 10: 1761–1770
Choi S, Kucharczyk CJ, Liang Y, et al. Exceptional power density and stability at intermediate temperatures in protonic ceramic fuel cells. Nat Energy, 2018, 3: 202–210
Fabbri E, Bi L, Pergolesi D, et al. High-performance composite cathodes with tailored mixed conductivity for intermediate temperature solid oxide fuel cells using proton conducting electrolytes. Energy Environ Sci, 2011, 4: 4984–4993
He F, Wu T, Peng R, et al. Cathode reaction models and performance analysis of Sm0.5Sr0.5CoO3−δ-BaCe0.8Sm0.2O3−δ composite cathode for solid oxide fuel cells with proton conducting electrolyte. J Power Sources, 2009, 194: 263–268
Kournoutis VC, Tietz F, Bebelis S. AC impedance characterisation of a La0.8Sr0.2Co0.2Fe0.8O3 electrode. Fuel Cells, 2009, 9: 852–860
Lu X, Yang X, Jia L, et al. First principles study on the oxygen reduction reaction of the La1−xSrxMnO3 coated Ba1−xSrxCo1−yFeyO3 cathode for solid oxide fuel cells. Int J Hydrogen Energy, 2019, 44: 16359–16367
Acknowledgements
This work was supported by the National Natural Science Foundation of China (51972183 and 51972128) and the Startup Funding for Talents at the University of South China.
Author information
Authors and Affiliations
Contributions
Wu S and Bi L designed this study. Wu S and Li X performed the experiments. Xu X performed the DFT calculations and analyzed the data. Bi L wrote the manuscript with other co-authors and all authors discussed the results and provided their approval to the final version.
Corresponding author
Additional information
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary information
Supporting data are available in the online version of the paper.
Shuai Wu is a postgraduate student, studying in the group of Professor Lei Bi. His research interest is the utilization of the doping strategy to tailor cathode materials for H-SOFCs with enhanced performance. Designing new cathode materials with protonation ability is now his major research topic.
Xi Xu was a master student in the group of Professor Lei Bi and now she is a PhD student at Imperial College London and doing her research in the laboratory of Professor Stephen Skinner from October 2020. Her current research topic is the development of advanced cathode materials for H-SOFCs, which covers both the experimental part and the theoretical simulation part using density functional theory (DFT).
Xiaomei Li is a postgraduate student in the group of Professor Lei Bi. After joining the group, she started the work of developing high-performing H-SOFCs, by microstructure optimizations and design of new materials. She is also interested in understanding the electrode working mechanisms for H-SOFCs.
Lei Bi is now a full professor at the University of South China, leading a group working on H-SOFCs. After obtaining his PhD degree from the University of Science and Technology of China, he spent six years working at the National Institute for Materials Science in Japan as a postdoc and King Abdullah University of Science and Technology in Saudi Arabia as a research scientist. His research interests are the development of H-SOFCs, using both the first-principles calculation method and experimental approaches.
Supporting Information
40843_2021_1821_MOESM1_ESM.pdf
High performance proton-conducting solid oxide fuel cells using the first-generation Sr-doped LaMnO3 cathode tailored with Zn ions
Rights and permissions
About this article
Cite this article
Wu, S., Xu, X., Li, X. et al. High-performance proton-conducting solid oxide fuel cells using the first-generation Sr-doped LaMnO3 cathode tailored with Zn ions. Sci. China Mater. 65, 675–682 (2022). https://doi.org/10.1007/s40843-021-1821-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-021-1821-4