Abstract
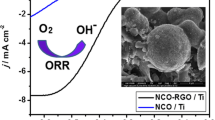
Graphene oxide (GO) is an important derivative of graphene, fascinating the entire world with its dazzling properties and versatile performance. However, the synthesis of GO via chemical routes often results in limited control of the density of functionalities and their distribution, presenting a barrier to the spread of GO applications. We modified Hummers’ method and aimed at controlling the oxygen functionality of GO. The highest oxygen content of the modified synthetic GO (MdGO) occurs at edge regions, and the large proportion of carboxyl groups can be easily removed upon annealing. The excellent conductivity of intrinsic graphene can thus be recovered after the removal of the main functional groups. The resulting MdGO was reduced and doped with NH3, and the reduced MdGO (rMdGO) was determined to be an excellent support for oxygen reduction reaction (ORR) electrocatalysts. As a demonstration, a composite of CoO and N-rMdGO was fabricated, which exhibited highly comparable ORR performance in alkaline relative to 20 wt.% Pt/C.
中文摘要
本文采用液相化学法对氧化石墨烯氧化官能团的分布进行控制合成, 使较多含氧官能团在氧化石墨烯的边缘区域. X射线 光电子能谱显示其具有高的含氧官能度, 且C=O/C–O比值在2左右, 远高于原有Hummers方法制备的氧化石墨烯的0.17. 其深黑色的外 观与其他文献对比, 可以证明其主要的含氧官能团为羧基且羰基主要存在于边缘区域. 具有该结构的氧化石墨烯可以经过简单还原后, 极大地恢复其高导电性, 层面上存有的少量官能团可以用来有效地抓住催化剂颗粒, 使得这种氧化石墨烯所制得的CoO复合物具有非 常优秀的催化氧气还原反应性能.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Guo S, Zhang S, Wu L, Sun S. Co/CoO nanoparticles assembled on graphene for electrochemical reduction of oxygen. Angew Chem, 2012, 124: 11940–11943
He Q, Li Q, Khene S, et al. High-loading cobalt oxide coupled with nitrogen-doped graphene for oxygen reduction in anion-exchange-membrane alkaline fuel cells. J Phys Chem C, 2013, 117: 8697–8707
Jiang S, Zhu C, Dong S. Cobalt and nitrogen-cofunctionalized graphene as a durable non-precious metal catalyst with enhanced ORR activity. J Mater Chem A, 2013, 1: 3593–3599
Liang Y, Li Y, Wang H, et al. Co3O4 nanocrystals on graphene as a synergistic catalyst for oxygen reduction reaction. Nat Mater, 2011, 10: 780–786
Liang Y, Wang H, Zhou J, et al. Covalent hybrid of spinel manganese–cobalt oxide and graphene as advanced oxygen reduction electrocatalysts. J Am Chem Soc, 2012, 134: 3517–3523
Wang H, Liang Y, Li Y, Dai H. Co1–x S-graphene hybrid: a high-performance metal chalcogenide electrocatalyst for oxygen reduction. Angew Chem Int Ed, 2011, 50: 10969–10972
Wang G, Yang J, Park J, et al. Facile synthesis and characterization of graphene nanosheets. J Phys Chem C, 2008, 112: 8192–8195
Zhou X, Qiao J, Yang L, Zhang J. A review of graphene-based nanostructural materials for both catalyst supports and metal-free catalysts in pem fuel cell oxygen reduction reactions. Adv Energ Mater, 2014, 4: 1289–1295
Luo J, Kim J, Huang J. Material processing of chemically modified graphene: some challenges and solutions. Acc Chem Res, 2013, 46: 2225–2234
Wang H, Dai H. Strongly coupled inorganic–nano-carbon hybrid materials for energy storage. Chem Soc Rev, 2013, 42: 3088–3113
Marcano DC, Kosynkin DV, Berlin JM, et al. Improved synthesis of graphene oxide. ACS Nano, 2010, 4: 4806–4814
Luo D, Zhang G, Liu J, Sun X. Evaluation criteria for reduced graphene oxide. J Phys Chem C, 2012, 115: 11327–11335.
Pei S, Cheng HM. The reduction of graphene oxide. Carbon, 2012, 50: 3210–3228.
Chua CK, Pumera M. Chemical reduction of graphene oxide: a synthetic chemistry viewpoint. Chem Soc Rev, 2014, 43: 291–312
Gao W, Alemany LB, Ci L, Ajayan PM. New insights into the structure and reduction of graphite oxide. Nat Chem, 2009, 1: 403–408
Jeon IY, Shin YR, Sohn GJ, et al. Edge-carboxylated graphene nanosheets via ball milling. Proc Natl Acad Sci, 2012, 109: 5588–5593
Huang NM, Lim HN, Chia CH, Yarmo MA, Muhamad MR. Simple room-temperature preparation of high-yield large-area graphene oxide. Int J Nanomed, 2010, 6: 3443–3448
Sun X, Zhuang L, Welsher K, et al. Nano-graphene oxide for cellular imaging and drug delivery. Nano Res, 2008, 1: 203–212.
Dimiev AM, Tour JM. Mechanism of graphene oxide formation. ACS Nano, 2014, 8: 3060–3068
Silvestre-Albero J. Ultrahigh CO2 adsorption capacity on carbon molecular sieves at room temperature. Chem Commun, 2011, 47: 6840–6842
Xia Y, Mokaya R, Walker GS, Zhu Y. Superior CO2 adsorption capacity on N-doped, high-surface-area, microporous carbons templated from zeolite. Adv Energ Mater, 2011, 1: 678–683
Yang Z, Lisiecki I, Walls M, Pileni MP. Nanocrystallinity and the ordering of nanoparticles in two-dimensional superlattices: controlled formation of either core/shell (Co/CoO) or hollow CoO nanocrystals. ACS Nano, 2013, 7: 1342–1350
Author information
Authors and Affiliations
Corresponding author
Additional information
Xiaoming Sun was born in Pingdu, Shandong Province, China. He received his BSc and PhD degrees from the Department of Chemistry, Tsinghua University in 2000 and 2005, respectively. After post-doctoral work in Prof. Hongjie Dai’s group at Stanford University, he joined the faculty of Beijing University of Chemical Technology at 2008. His research interests focus on controlled synthesis, the separation and assembly of functional inorganic nanomaterials and carbon materials, and the structure and property regulation of metal-oxide arrays.
Rights and permissions
About this article
Cite this article
Zhang, G., Xu, Y., Wang, L. et al. Rational design of graphene oxide and its hollow CoO composite for superior oxygen reduction reaction. Sci. China Mater. 58, 534–542 (2015). https://doi.org/10.1007/s40843-015-0064-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-015-0064-z