Abstract
In this paper, we develop a new mathematical model based on the Atangana Baleanu Caputo (ABC) derivative to investigate meningitis dynamics. We explain why fractional calculus is useful for modeling real-world problems. The model contains all of the possible interactions that cause disease to spread in the population. We start with classical differential equations and extended them into fractional-order using ABC. Both local and global asymptotic stability conditions for meningitis-free and endemic equilibria are determined. It is shown that the model undergoes backward bifurcation, where the locally stable disease-free equilibrium coexists with an endemic equilibrium. We also find conditions under which the model’s disease-free equilibrium is globally asymptotically stable. The approach of fractional order calculus is quite new for such a biological phenomenon. The effects of vaccination and treatment on transmission dynamics of meningitis are examined. These findings are based on various fractional parameter values and serve as a control parameter for identifying important disease-control techniques. Finally, the acquired results are graphically displayed to support our findings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Meningitis is one of the deadly infectious diseases that has caused an increase in the mortality rate of the populace. It is the inflammation of the meninges which are the protective membrane that covers the spinal cord and the brain [1, 2]. Meningitis can be caused by a variety of microorganisms, as well as non-infectious causes like cancer, injury in the head, and through the use of certain medications [3]. Bacterial meningitis has been identified to be the highest global burden among others, because of its widespread morbidity and mortality [2, 4]. Haemophilus influenzae, Streptococcus pneumoniae, Listeria monocytogenes, Group B Streptococcus, and Neisseria meningitidis are some of the bacteria that can cause it. In this study, we focus on Neisseria meningitidis which is responsible for causing meningococcal diseases [5, 6].
Although, meningococcal diseases are found globally; however, the highest-burden is in the sub-Saharan African meningitis belt. This region has the countries between Senegal and Ethiopia which include but is not limited to countries like Guinea, Gambia, Burkina Faso, Central African Republic, Togo, Ghana, Kenya, Mali, Niger, Uganda, and Nigeria which is the focus country in this study [6, 7]. In this region, an increase in meningitis incidence has been associated with the beginning of the dry season known as the harmattan season (from November to March). The season is characterized by a dry wind blowing south from the Sahara desert to the Gulf of Guinea [2, 5, 6].
Neisseria meningitidis is a human infection that can be passed from person to person via throat secretion or droplets of respiratory from a carrier of an infected individual. In addition, a close or prolonged personal contact with such individuals through kissing, sneezing, or sharing eating or drinking utensils with a susceptible person can facilitate the transmission of the disease. The incubation period of this disease varies between 2 and 10 days, thus, following contact with an infected individual, the symptoms characterized with meningitis are expected to start showing [2, 6]. These symptoms include high fever, severe headaches, the appearance of rashes, and some other flu-like symptoms. Complications from this disease can cause permanent disabilities such as deafness, brain damage, epilepsy, hearing loss, and individual can die in the absence of early diagnosis or proper treatment [1].
Treatment of infected individuals and the use of preventive measures for susceptible people are the primary ways to reduce the incidence of meningitis in the population. In areas with inadequate health infrastructure and resources, an infected individual can be treated with antibiotic medications after effective contact with the deadly disease [1]. Antibiotics and fluids can be given directly into a patient’s veins to prevent dehydration, and oxygen can be given through a face mask if there are complications or breathing difficulties. Another treatment option for meningitis is to use steroid medication to help reduce any swelling around the brain in any patient who has a brain complication [8]. Treatment care is mainly administered for meningitis-infected people, however, this disease can be mainly prevented by vaccination. Mass vaccination of the susceptible population and early detection of infected individuals have contributed to the mitigation of the meningitis epidemic [9]. Just like many other diseases, vaccination has been shown to reduce the outbreak of meningitis in many regions [10, 11]. Particularly, the administration of vaccines has been shown to prevent the disease in both children and adults. Conjugate and polysaccharide vaccines are the two types of vaccines that are currently used in Sub-Saharan Africa during routine immunization schedules, preventive campaigns, and response to the epidemic. These types of vaccines include monovalent A, C, and tetravalent A, C, Y, W. Conjugate vaccines provide long-time immunity and confer herd immunity. Polysaccharide vaccines can be bivalent (protecting against serogroups A and C), trivalent (protecting against serogroups A, C, and W), or tetravalent (protecting against serogroups A, C, Y, and W) [1, 12]. They have been available and in use, since the 1970s [13] with limited efficacy [9, 14], and they do not yield herd immunity [9, 15].
Mathematical models have become an effective useful tool that has been used by many researchers to understand the epidemiology of diseases in each population. Many models have been developed and analyzed using different methods to provide a better understanding of the transmission dynamics and control of the disease. Examples of these studies include [16,17,18,19,20,21,22,23,24,25]. More specifically, some studies have been carried out to provide a better understanding of the transmission of meningitis (see [1, 26,27,28]), and few of these studies focus on the effect of vaccination in controlling meningitis outbreaks in Nigeria [2, 6].
In this study, we employ the use of a fractional-order mathematical model to investigate the effect of vaccination and treatment on the control of meningitis in Nigeria. Numerous researchers have used fractional-order models to study the dynamics of different diseases (see [29,30,31,32,33,34,35] and references therein). Studies have shown that fractional-order models are more advantageous in characterizing memory of biological and epidemiological systems than the classical integral-order models [36, 37]. Other related studies via different modelling approach can be found in [38,39,40,41,42,43,44,45,46,47]. In addition, fractional-order models have been used in research to answer arising problems in different fields of study. Particularly, Atagana-Baleanu fractional operator has been used by many researchers to answer different questions. For examples of these studies, see [48,49,50,51] and the references therein. The memory properties of this operator allow the integration of more information from the past, to increase the accuracy of the model predictions. In the light of this advantage, we employ the use of a fractional-order model to study the effect of vaccination and treatment on the burden of meningitis in Nigeria. To the best of our knowledge, this is the first study to use a fractional-order model in investigating the dynamics of meningitis in the population. Another novelty of this study is the use of the explicit Euler fractional method to numerically simulate the effect of treatment and vaccination parameters on the dynamical behavior of a meningitis model. We first discuss the proposed model in detail in the integer case, then use the ABC derivative to extend the model into fractional-order and obtain the required findings. The rest of the article is organized as follows: Section two deals with the formulation of the model, section three deals with the analysis of the model in the integer-order and we extend it to the fractional-order model in section four. Section 5 discusses numerical simulations and a description of the fractional-order model. Finally, we concluded by summarizing our work in section six.
Formulation of Mathematical Model
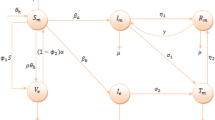
To understand the transmission dynamics of meningitis, we formulate a deterministic model with six compartments based on the epidemiological status of individuals. The compartments are susceptible S(t), Vaccinated V(t), Exposed E(t), Infected I(t) hospitalized Q(t) and Recovered R(t). We assume that the population of the susceptible class is increased either by immigration or by birth at a constant rate \(\theta \), the vaccinated population loses immunity at a rate \(\tau \). Movement from susceptible class to vaccinated class is at a rate \(\alpha \). Movement rate between exposed to infected class is at a rate \(\epsilon \). We assume a natural recovery rate for infected individuals at a rate \(\gamma \) or after receiving treatment from the hospital at a rate \(\phi \). Recovered individuals lose immunity and become susceptible upon recovery at a rate \(\sigma \), natural death occurs in all the classes at a rate \(\mu \) while meningitis induced death is at a rate \(\delta \). The population of the susceptible individuals is reduced through interaction with an individual in the infected class at a rate \(\lambda (t)\). Where \(\lambda (t) = \dfrac{\psi \{\rho E(t) + I(t)\}}{N(t)}\).
The effective transmission probability per contact with the infected individuals is denoted by \(\psi \) and the parameters \(\rho \le 1\) is a modification parameter that indicates the infectivity of individuals in the exposed class. The effectiveness of the vaccine is about 86-90% but not completely effective, therefore, we assume that vaccinated class decreases due to infection at the rates \((1 - \omega )\lambda (t)\) where \(0 \le \omega \le 1\). \(\omega \) determines the effectiveness of the vaccine. For instance, if \(\omega = 1\) in this case, the vaccine is assumed to be 100% effective. Thus, the above descriptions can be represented by a set of differential equations in (2.1), the compartmental diagram in Fig. 1 also illustrates the above descriptions.
Analysis of the Model
This section comprises the study of positivity and boundedness of the solutions, invariant region, condition of existence of equilibrium points, and basic reproduction number \((\mathcal {R}_0)\) of the meningitis model.
Positivity of the Solution
Considering the positivity solution of the meningitis in equation (2.1) with all positive initial conditions.
Theorem 1
If S(0), V(0), E(0), I(0), Q(0), R(0), be positive, then we can say that the solutions \(\left( S(t), V(t), E(t), I(t), Q(t), R(t), \right) \) in equation (2.1) are positive \(\forall \) \(t > 0\).
Proof
Let \(t^{*}= sup \{t> 0 : S(t)>0,V(t)> 0, E(t)>0, I(t)> 0, Q(t)> 0, R(t)> 0 \}\), so that \(t^{*} > 0\). From the equation (2.1), considering the first equation that is
Using integrating factor and multiplying it with equation (3.1) to obtain
Integrating both sides from \(t = 0\) to \(t = t^{*}\) and making \(S(t^{*})\) subject of the formula, we obtain
From the above result, it is clearly showed that \(S(t^{*})\) is \(\ge \) to the sum of positive terms which is positive. Similarly, following the same argument, we can show that \(E(t) \ge 0, I(t) \ge 0, Q(t) \ge 0, R(t) \ge 0, V(t) \ge 0\), \(\forall ~t^{*} > 0\). \(\square \)
Boundedness of the Solution
Theorem 2
All solutions S(0), V(0), E(0), I(0), Q(0), R(0) of the meningitis model(2.1) are bounded. This means that, if
then \(N(t) = S(t) + V(t)+E(t) + I(t) + Q(t) + R(t) \).
Proof
It is important to know that \(0 < I(t) \le N(t)\). Adding all the six equations of the meningitis model (2.1), we can say that
This can be seen that all solutions of meningitis model (2.1) are bounded. Therefore, equation (3.2) can be written as
\(\theta - \left( \mu + \delta \right) N(t) \le \dfrac{dN(t)}{dt} \le \theta - \mu N(t)\)
and we have
\(\square \)
Invariant Region of Meningitis Model
Theorem 3
The region \(\Theta \subset \mathbb {R}^6_+ \) is non-negatively invariant of the meningitis model (2.1) with positive initial conditions \(\mathbb {R}^6_+\).
Proof
To proof, Let \({\Theta }\) stand for feasible region of the meningitis model (2.1), this can now be written as
with
The conditions that are required to show the positive invariance of \(\Theta \) i.e. the solution in \(\Theta \) still remains in \(\Theta ~ \forall ~ t > 0\). This is obtained by adding equations of the meningitis model (2.1)
Using the standard comparison theorem [52], we can say that
Particularly, \(N(t) \le \dfrac{\theta }{\mu }\) if \(N(0) \le \dfrac{\theta }{\mu }\). Therefore, the region \(\Theta \) is non-negative invariant. \(\square \)
Existence of Equilibrium Point of Meningitis Model
In this section, it is necessary to discuss the existence of equilibrium points of the meningitis model. This can be divided into Meningitis free equilibrium (MFE) and Endemic equilibrium (EE) points. Considering the existence of meningitis free equilibrium point (MFEP), by equating the right-hand side of all the equations in (2.1) as well as setting E, I, Q and R to be zero, to obtain
Also, considering the existence of endemic equilibrium point (EEP) if I is non-zero and all the equations on the right hand side of model (2.1) are equating to be zero then by substituting \(S=S^{*}\), \(V=V^{*}\), \(E=E^{*}\), \(I=I^{*}\), \(Q=Q^{*}\) and \(R=R^{*}\) into model (2.1), we have
Simplifying further gives
where \(\lambda ^{*} (t) = \dfrac{\psi \{\rho E^{*} + I^{*}\}}{N^{*}}\), \(a_{1}=\alpha +\mu \), \(a_{2}=\tau +\mu \), \(a_{3}=\varepsilon +\mu \), \(a_{4}=(\delta +\beta +\gamma +\mu )\), \(a_{5}=(\phi +\delta +\mu )\), \(a_{6}=\sigma +\mu \), \(b_{1}= \left( 1-\omega \right) \left( a_{3}\,a_{4}\,a_{5}\,a_{6}-a_{5}\,\gamma \,\sigma \,\varepsilon -\beta \,\phi \,\sigma \,\varepsilon \right) \), \(b_{2}= \left( 1-\omega \right) \left( a_{1}\,a_{3}\,a_{4}\,a_{5}\,a_{6}-a_{5}\,\alpha \,\gamma \,\sigma \,\varepsilon -\alpha \,\beta \,\phi \,\sigma \,\varepsilon \right) \) and \(b_{3}=a_{3}\,a_{4}\,a_{5}\,a_{6} \left( a_{1}\,a_{2}-\alpha \,\tau \right) \)
From Eqs. (3.5) and (3.7), it has showed clearly that model (2.1) exists and has two equilibriums in the non-negative of \(\mathbb {R}_+^6\).
Basic Reproduction Number \((\mathcal {R}_0)\) of Meningitis Model
In this section, we shall consider basic reproduction number (\(\mathcal {R}_0\)) in order to determine the nature of the meningitis disease. To this end, we will use next generation matrix method to find basic reproduction number (\(\mathcal {R}_0\)). Therefore, meningitis model (2.1) can be written as
and
Using Jacobian Matrix to solve equations (3.8) and (3.9), we respectively obtain equations (3.10) and (3.11).
and
According to [53] basic reproduction number is the obtained as the spectral radius of \(\mathbf {F} \mathbf {V}^{-1}\). Therefore,
Local stability of Disease-Free Equilibrium (DFE)
The following theorem establishes the local stability of disease-free equilibrium.
Theorem 4
When \(R_0 < 1,\) the disease-free equilibrium \(E_0\) is locally asymptotically stable, otherwise it is unstable.
Proof
The Jacobian matrix of the system 2.1 at disease-free equilibrium point \(E_0\) is obtained as follows:

Now, four of the six eigenvalues of the Jacobian matrix \(J_0\) can be easily obtained as \(-\mu , -(\mu +\sigma ), -(\alpha +\mu +\tau )\) and \( -(\delta +\phi +\mu )\). The other two eigenvalues can be given as:
and
where
Now Consider \(R_0<1 \), which implies
which further implies:
which implies \(\zeta < 0\). Now it can be easily verified that all the eigenvalues of \(J_0\) have a negative real part. Hence Meningitis free equilibrium Point (MFEP) is locally asymptotically stable provided \(R_0<1\), and unstable when \(R_0 > 1\). \(\square \)
Global Stability of Disease-Free Equilibrium (DFE)
In order to derive the conditions for the global stability of \(E_0\), we will use the method provided in [54]. Here is the brief overview of the technique. If we have a model system in the below form.
where \(U\in {\varvec{R}}^{\varvec{m}}\) represents the uninfected individuals whereas \(V\in {\varvec{R}}^{\varvec{n}}\) is the infected population. Now, if \(R_0 <1\) then the two criteria given below establish the global stability of the disease-free equilibria. (N1) For \(\frac{dU}{dt}=F(U,0)\), \(U_0\) is globally asymptotically stable. (N2) \(G\left( U,V\right) =BV-\hat{G}\left( U,V\right) \), where \(\hat{G}\left( U,V\right) \ge 0\) for \((U,V)\in \Theta \).
Here, \(B=D_VG\left( U_0,0\right) \) represents an M-matrix (M-matrix is a matrix whose non-diagonal elements are non-negative) and \(\Theta \) is the region where the model makes biological sense. Now, using this result, we can provide the following theorem:
Theorem 5
If \(R_0<1\), the disease-free equilibrium is globally asymptotically stable and unstable for \(R_0>1\).
Proof
With respect to the notations of the method described above, the meningitis free equilibrium (MFE) can be given by \(Q_0=(U_0,0)\). In order to prove this theorem we need to prove conditions (N1) and (N2). We begin by showing (N1). Consider
Here,
and the corresponding Jacobian matrix can be given as:
The characteristics polynomial of the above system is
We can clearly see that, one eigenvalue is \(-(\mu +\sigma )\) and other two eigenvalues are either -ve or have -ve real part. Hence \(U_0\) is globally asymptotically stable. Hence (N1) is satisfied. Now we will show (N2) as follows: We have,
Clearly, B is M-Matrix and \(\hat{G}\left( U,V\right) \ge 0\) which implies (N2) holds for the system. Hence, we have shown that both (N1) and (N2) holds for the system, which also completes the proof of the theorem. \(\square \)
Stability of Endemic Equilibrium
We will use the Routh-Hurwitz stability criterion to derive the conditions under which the endemic equilibrium point (EEP) is locally stable. The Jacobian matrix of the model system 2.1 at endemic equilibrium point (3.7) can be given as:

In order to avoid further complication, we will write the matrix \(J_0\) equivalent to
Here, \(a_{i,J}\) where \( i = 1,2...6 \) and \( j = 1,2...6\) are the corresponding entries. Now, the characteristic equation of the matrix \(J^*_0\) can be given as:
Now, the above equation [3.17] is further written in form of a polynomial equation as
The values of \(A_{i^s}: i= 1,2...6\) are provided in Appendix (See: Section 7).
Now, the endemic equilibrium point (8) will be locally stable provided the following four conditions hold:
-
(i)
\( A_i > 0 \forall i = 1,2,...6.\)
-
(ii)
\(A_1A_2> A_3, A_1 A_2 A_3 +A_1A_5 >A^2_1 A_4 + A^2_3\).
-
(iii)
\( A_1A_4(A_2A_3+2A_5)+A_2(A^2_1 A_6+A_3A_6)> A_1(A_1A^2_4+A^2_2A_5+A_3A_6)+A^2_3A_4+A^2_5\).
-
(iv)
\( A_2A_3A^2_5+A^3_3A_6>A^2_3A_4A_5+A^3_5+A^3_1A^2_6+A^2_1(A^2_4A_5-A_3A_4A_6-2A_2A_5A_6)+ A_1(A^2_2A^2_5+A_2A_3(-A_4A_5+A_3A_6)+A_5(-2A_4A_5+3A_3A_6))\).
Bifurcation Analysis
The presence of endemic equilibrium suggests that backward bifurcation is exist. We will use the “Center Manifold based theorem" present in [55], to validate the existence of backward bifurcation.
The estimate expression for \(R_0\) in 3.12 is
which gives \(\psi :\) effective transmission probability as the bifurcation parameter and \(\psi ^*\) as the critical point of bifurcation. Setting \(R_0 = 1\), we obtain
At \(\psi = \psi ^*\), the jacobian of the model system at Meningitis free equilibrium point (MFEP) is

It can be easily verified that zero is one eigenvalue of \(J_0\). Now the associated right eigenvector \(\mathbb {W} = (w_1,w_2,w_3,w_4,w_5,w_6)\) corresponding to eigenvalue zero and matrix \(J_0\) can be obtained by solving the following system of equations:
Here \( P = \dfrac{\psi \rho }{\alpha +\tau +\mu }\).
On solving system of equations 3.19, we get
Also the left eigenvector \(\mathbb {V} = (v_1,v_2,v_3,v_4,v_5,v_6)\) and satisfying \( \mathbb {W}.\mathbb {V} = 1\) can be obtained by solving:
Further calculations leads us to
Using Theorem 2 of article [55] the coefficients a and b can be computed as:
Further calculations lead to:
Substituting these values in expressions of a and b i.e. expression 3.23, we obtain:
Now from the derived expressions of a and b i.e. equations 3.24 and 3.25 it is clear that our proposed model will exhibit backward bifurcation only if
Analysis of Fractional Order Meningitis Model
In this section, we present some properties of Atangana-Baleanu Operator. [29, 56,57,58,59] which will be used in the analysis of the fractional order meningitis model 4.7.
Definition 1
Let a function \(\phi _1\in F^1(a,b)\) such that \(a<b\) and \(p\in [0,1]\) represent the fractional order, then the Atangana-Baleanu (AB) operator can be expressed as
Definition 2
If \(\phi _1\in F^1(a,b)\) such that \(b>a\) and \(p\in [0,1]\), then we have the form ABC in the Riemann- Liouville sense (ABR) expressed as
Definition 3
The AB fractional integral operator can be expressed as
where M(p) represent normalized function.
Theorem 6
If \(\phi _1\) is continuous on [a, b], the inequality holds.
Theorem 7
The ABR and ABC derivatives satisfy the Lipschitz criteria below
Fractional Order Meningitis Model
The fractional order version of the meningitis model 2.1 in AB derivative can be written as
with the initials for each of the variables given as \(S(0)=n_1, V(0)=n_2, E(0)=n_3, I(0)=n_4, Q(0)=n_5, R(0)=n_6\).
Existence and Uniqueness of the Fractional Order Solution
In this section, we show that the existence and uniqueness of the fractional ABC model in 4.7 using fixed point theory. We express 4.7 in equivalent form as
The fractional integral derivative of ABC on 4.7 is given as
Using the definition of integral fractional derivative in AB sense, we have that
Theorem 8
The kernel \(G_1\) satisfy the Lipschitz condition and contaction if the inequality in holds
Proof
To establish the Lipschitz condition, we consider two functions, S and \(S_1\)
\(\square \)
Where
This implies that
The rest of the equations followed the same procedure
Where \(\sigma _1\),,,,\(\sigma _6\) represent the corresponding Lipschitz constants. As a result, the Lipschitz conditions are met. The model in 4.7 can be written in a recursive terms as
Furthermore, the difference of successive terms is expressed as
Consider that,
Hence, we have
Furthermore, we derive the relation
Hence, S(t), V(t), E(t), I(t), Q(t), R(t) are bounded functions.
Theorem 9
The solution of the meningitis fractional order model 4.7 exist and is unique if there exist \(t_0\) such that;
Proof
It is established that S(t), V(t), E(t), I(t), Q(t), R(t) are bounded functions and satisfy Lipschitz conditions. From 4.20, we obtain the succeeding expression below
\(\square \)
The existence of the solutions are proved. We define the following functions in order to show that 4.22 represent the solution of the fraction order model 4.7
Further, we get
By repeating the same procedure at \(t_0\) we have
Taking the limit of 4.25 as \(n \longrightarrow \infty \) clearly, \(\parallel C_{1n}(t)\parallel \longrightarrow \)0. By the same procedure, \(\parallel C_{2n}(t)\parallel \), \(\parallel C_{3n}(t)\parallel \), \(\parallel C_{4n}(t)\parallel \), \(\parallel C_{5n}(t)\parallel \) and \(\parallel C_{6n}(t)\parallel \) \(\longrightarrow \)0.
Theorem 10
The solution of the fractional order model 4.7 is unique if the assumption below holds
Proof
In other to proof the uniqueness of the solution of 4.7, we consider another solution such as, \(S_{1}(t),V_{1}(t),E_{1}(t),I_{1}(t),Q_{1}(t),R_{1}(t)\) then,
\(\square \)
Hence, \(S(t)=S_{1}(t)\), if the condition 4.26 holds. Similarly, \( V(t)=V_{1}(t), E(t)=E_{1}(t), I(t)=I_{1}(t), Q(t)=Q_{1}(t)\) and \(R(t)=R_{1}(t)\). Hence, the solution is unique.
Numerical Simulation
Some of the parameter values were obtained from the literature, as shown in Table 2, and the demographic parameter values were estimated. The natural death rate is estimated to be \(\mu =\frac{1}{60.87 \times 365}\), with Nigerians having an average life expectancy of 60.87 years [60]. Furthermore, the recruitment rate is estimated by \(\mu \times N\), where the total population N is reported as 219,463,862 [60].
The numerical dynamics of the meningitis outbreak as modeled with the ABC operator are examined in this section, while the remaining parameters are taken from Table 2. In this paper, an explicit Euler fractional type numerical approach is used to simulate fractional ABC type ordinary differential equations. The approach itself, as well as its complete analysis based on convergence and error boundaries, may be found in [61,62,63]. Because of its simplicity and versatility, the numerical approach devised for this purpose has garnered a lot of praise in the current literature. The use of such readily available routines on MathWorks made simulations far more useful for the current research study for the ABC meningitis model. It should be noted that we utilize the MATLAB program version (R2019a).
We ran simulations for each state variable using the ABC operator model. This simulation consists of two charts for each class, one comparing the classical and fractional order derivatives Figs. 2, 3 and the other varying the fractional-order Figs. (4), (5) values to study the dynamics of the compartments. Susceptible people appear to diminish with fractional value variation, however, the vaccinated and exposed groups exhibit symmetrical behavior with the bell-shaped curve representing, once again, the true behavior of the disease. Concerning the infectious and quarantine classes, they fall dramatically at first but thereafter follow the bell-shaped structure. Last but not least, the recovered class’s behavior is depicted in Fig. (5(C)). There aren’t many deaths in this class since the infectious population declines after a while, and as a result, people begin to recover, as seen. We also test the effect of the vaccine by varying the values of the vaccine rate on the susceptible population as can be seen in Fig. (6). The treatment rate shows sensitive behavior as the infectious class got decreased with increasing values of the treatment rate (7). Based on this information, it is possible to conclude that the meningitis outbreak is manageable if it can be prevented from spreading by increasing the rate of vaccination. Vaccinating against meningitis is the most effective strategy to lessen the disease’s incidence and effects by providing long-term protection. Treatments are also given to people who are at a high risk of contracting meningococcal illness. Controlling meningitis epidemics, on the other hand, necessitates both vaccine and therapy.
Conclusion
A new mathematical model for the transmission dynamics of meningitis in fractional ABC derivatives has been investigated. The meningitis outbreak is particularly fast-acting and has shockingly dangerous implications. It is vital to investigate the dynamics of this epidemic in greater depth. The fractional operator utilized in this study has been shown in the literature to be effective for analyzing the transmission dynamics of various epidemic models. p is the fractionalized order. As a result, the model formulation, existence, and uniqueness of the solution via the fixed point theorem, bifurcation analysis, global and local stability analysis, and, most importantly, the basic number of reproductions for the suggested fractional version of the model have all been demonstrated. Furthermore, the model’s parameters are derived from actual cases of the infection. Data from the 2017 Nigerian meningitis outbreak was used to parameterize the model [6]. It is worth mentioning that the fractional type illness model under consideration understands disease behavior better than the integer-order variation. The treatment and vaccine’s effect has been demonstrated by altering the vaccine rate values. Furthermore, several numerical simulations were run using an efficient numerical scheme to provide more light on the model’s behavior. Moreover, in order to predict meningitis eradication, we investigated the effects of multiple control techniques in reducing the disease’s prevalence in the population. The findings of the study are expected to provide a necessary framework for assessing meningitis management in any part of the world. Because the prevalence of meningitis varies by region, our analytical results can show the genuine picture of meningitis dynamics in a given region.
Appendix
The values of \(A_{i^s}: i= 1,2...6\) used in Section 3.8 are given as:
-
\( A_1 = (- a_{11} - a_{22} - a_{33} - a_{44} - a_{55} - a_{66}) \).
-
\( A_2 = -\alpha \tau - \lambda a_{13} - a_{23}a_{32} - \epsilon a_{34} + a_{11} (a_{22}+a_{33}+a_{44}+a_{55}+a_{66}) + a_{22}(a_{33} +a_{44}+a_{55}+a_{66})\ +\) \(a_{33}(a_{44}+a_{55}+a_{66}) + a_{44}(a_{55}+a_{66})+a_{55}a_{66} \)
-
\( A_3 = -\epsilon a_{24} a_{32}+\alpha \tau (a_{33}+a_{44})+\epsilon a_{22} a_{34}+a_{23} a_{32} a_{44}-a_{22} a_{33}( a_{44}-a_{55} - a_{66}) + \big ( \alpha \tau +a_{23} a_{32} +\epsilon a_{34} -(a_{22} -a_{33}) a_{44} \big ) a_{55}\ +\) \(\big (\alpha \tau +a_{23} a_{32} + \epsilon a_{34} -(a_{22} -a_{33}) a_{44} -a_{55}(a_{22} -a_{33} -a_{44}) \big ) a_{66} +a_{11} \big (a_{23} a_{32}+\epsilon a_{34}-a_{33} a_{44}-a_{33} a_{55}-a_{44} a_{55}-(a_{33} -a_{44} -a_{55}) a_{66}\ -\) \(a_{22} \big (a_{33} +a_{44}+a_{55}+a_{66}\big )\big )-\epsilon a_{14} \lambda ^*-\tau a_{23} \lambda ^*+a_{13} (-\alpha a_{32}+(a_{22}+a_{44}+a_{55}+a_{66}) \lambda ^*) \)
-
\( A_4 = \alpha \epsilon \tau a_{34}+\alpha a_{13} a_{32} (a_{44}+a_{55}+a_{66})-\alpha \tau a_{33}( a_{44}+a_{55} +a_{66})+\epsilon a_{24} a_{32} a_{55}- \epsilon a_{22} a_{34} a_{55}\ -\) \((\alpha \tau -a_{23} a_{32} +a_{22} a_{33}) a_{44} a_{55}+\epsilon a_{24} a_{32} a_{66} -\epsilon a_{22} a_{34} a_{66}- \big ( \alpha \tau -a_{23} a_{32} +a_{22} a_{33}\big )a_{44} a_{66}\ +\) \((-\alpha \tau - a_{23} a_{32} +a_{22} a_{33} -\epsilon a_{34} +a_{22} a_{44} +a_{33} a_{44})a_{55} a_{66} +a_{11} ( (\epsilon a_{24} -a_{23} a_{44}-a_{23} a_{55} ) a_{32}-\epsilon a_{34} a_{55}+a_{33} a_{44} a_{55}- \big (a_{23} a_{32} -\epsilon a_{34} + a_{33} a_{44} +a_{33} a_{55} +a_{44} a_{55}\big ) a_{66} +a_{22} (-\epsilon a_{34}+a_{44} a_{55}+a_{44} a_{66}+a_{55} a_{66}\ +\) \(a_{33} (a_{44}+a_{55}+a_{66})))- \lambda ^* \Big (\gamma \epsilon \sigma -\epsilon \tau a_{24} -a_{13} a_{22} a_{44} +\tau a_{23} a_{44} - (a_{13} a_{22} +\tau a_{23} a_{13} a_{44} )a_{55} - a_{66}(a_{13} a_{22} +\tau a_{23} -a_{13} a_{44} -a_{13} a_{55}) +\epsilon a_{14} \left( -\alpha a_{32}+\left( a_{22}+a_{55}+a_{66}\right) \right) \Big )\).
-
\( A_5 = ((\alpha \tau -a_{11} a_{22} ) a_{55}+(\alpha \tau a_{11} a_{22}) a_{66}) a_{33} a_{44} +(\alpha \tau a_{33} - a_{11} a_{22} a_{33} +\alpha \tau a_{44} - a_{11} a_{22} a_{44} - (a_{11} -a_{22} )a_{33} a_{44} ) a_{55} a_{66} \ +\) \(\epsilon a_{34} ((-\alpha \tau +a_{11} a_{22}) a_{66}+a_{55} (-\alpha \tau +a_{22} a_{66}+a_{11} (a_{22}+a_{66})))-a_{32} (\alpha \gamma \epsilon \sigma +\alpha a_{13} a_{44} a_{55}+\alpha a_{13} a_{44} a_{66}+(\alpha a_{13} +\epsilon a_{24} -a_{23} a_{44}) a_{55} a_{66}-\alpha \epsilon a_{14} (a_{55}+a_{66})+a_{11} (\epsilon a_{24} (a_{55}+a_{66})-a_{23} (a_{55} a_{66}+a_{44} (a_{55}+a_{66}))))\ -\) \(\lambda ^* \big (\beta \epsilon \sigma \phi +\gamma \epsilon \sigma a_{22} +(\gamma \epsilon \sigma - \epsilon a_{14} a_{22} +\epsilon \tau a_{24} +a_{13} a_{22} a_{44} -\tau a_{23} a_{44}) a_{55} -(\epsilon a_{14} a_{22} +\epsilon \tau a_{24} +a_{13} a_{22} a_{44} -\tau a_{23} a_{44} )a_{66} -(\epsilon a_{14} +a_{13} a_{22} -\tau a_{23} +a_{13} a_{44} )a_{55}a_{66} \big ) \).
-
\(A_6 = \big (\epsilon \left( \alpha \tau -a_{11} a_{22}\right) a_{34} -(\alpha \tau +a_{11} a_{22} )a_{33} a_{44} \big ) a_{55} a_{66}+a_{32} (-\alpha \beta \epsilon \sigma \phi +a_{55} (-\alpha \epsilon a_{14} a_{66}+a_{11} (\epsilon a_{24}-a_{23} a_{44}) a_{66}+\alpha (\gamma \epsilon \sigma +a_{13} a_{44} a_{66})))\ +\) \(\lambda ^*\Big [\beta \epsilon \sigma \phi a_{22} -\gamma \epsilon \sigma a_{22} a_{55} + a_{55} a_{66}\big (\epsilon a_{14} a_{22} -\epsilon \tau a_{24} -a_{13} a_{22} a_{44} + \tau a_{23} a_{44} \big ) \Big ] \)
Data Availability Statement
Data used to support the findings of this study are included within the article. Authors used a set of parameter values whose sources are from the literature as shown in table 1.
References
Asamoah J.K.K., Nyabadza F., Seidu B., Chand M., Dutta H., Mathematical modelling of bacterial meningitis transmission dynamics with control measures, Computational and mathematical methods in medicine (2018)
Ojo M.M.: Mathematical modeling of neisseria meningitidis: a case study of nigeria, Ph.D. thesis, University of Kansas (2019)
Ginsberg, L.: Difficult and recurrent meningitis. J. Neurol. Neurosurg. Psychiatry 75(suppl 1), 116–121 (2004)
Oordt-Speets, A.M., Bolijn, R., van Hoorn, R.C., Bhavsar, A., Kyaw, M.H.: Global etiology of bacterial meningitis: a systematic review and meta-analysis. PloS One 13(6), e0198772 (2018)
Rouphael N.G., Stephens D.S., Neisseria meningitidis: biology, microbiology, and epidemiology, Neisseria meningitidis (2012) 1–20
Agusto, F., Leite, M.: Optimal control and cost-effective analysis of the: Meningitis outbreak in Nigeria. Infect. Disease Model. 4(2019), 161–187 (2017)
Agier, L., Martiny, N., Thiongane, O., Mueller, J.E., Paireau, J., Watkins, E.R., Irving, T.J., Koutangni, T., Broutin, H.: Towards understanding the epidemiology of neisseria meningitidis in the african meningitis belt: a multi-disciplinary overview. Int. J. Infect. Diseases 54, 103–112 (2017)
National Health Service, Treatment of meningitis, Available from: https://www.nhs.uk/conditions/meningitis/treatment/
LaForce F., Konde S.K., Viviani, M.-P. Préziosi, The meningitis vaccine project, Vaccine 25 (2007) A97–A100
Kuznik, A., Iliyasu, G., Lamorde, M., Mahmud, M., Musa, B.M., Nashabaru, I., Obaro, S., Mohammed, I., Habib, A.G.: Cost-effectiveness of expanding childhood routine immunization against neisseria meningitidis serogroups c, w and y with a quadrivalent conjugate vaccine in the African meningitis belt. PloS One 12(11), e0188595 (2017)
Lingani C., Bergeron-Caron C., Stuart J.M., Fernandez K., Djingarey M.H., Ronveaux O., Schnitzler J.C., Perea W.A., Meningococcal meningitis surveillance in the African meningitis belt, 2004–2013, Clinical infectious diseases 61 (suppl_5) (2015) S410–S415
Trotter, C.L., Ramsay, M.E.: Vaccination against meningococcal disease in Europe: review and recommendations for the use of conjugate vaccines. FEMS Microbiol. Rev. 31(1), 101–107 (2007)
C. for Disease Control, Prevention, et al., Meningococcal disease: epidemiology and prevention of vaccine-preventable diseases, The Pink Book: Course Textbook. 201212th ed. Atlanta, GA Centers for Disease Control and Prevention Available at: http://www.cdc.gov/vaccines/pubs/pinkbook/downloads/mening.pdf. Accessed April 26 (2012)
Reingold, A., Hightower, A., Bolan, G., Jones, E., Tiendrebeogo, H., Broome, C., Ajello, G., Adamsbaum, C., Phillips, C., Yada, A.: Age-specific differences in duration of clinical protection after vaccination with meningococcal polysaccharide a vaccine. The Lancet 326(8447), 114–118 (1985)
Hassan-King, M., Wall, R., Greenwood, B.: Meningococcal carriage, meningococcal disease and vaccination. J. Infect. 16(1), 55–59 (1988)
Peter, O.J., Viriyapong, R., Oguntolu, F.A., Yosyingyong, P., Edogbanya, H.O., Ajisope, M.O.: Stability and optimal control analysis of an scir epidemic model. J. Math. Comput. Sci. 10(6), 2722–2753 (2020)
M. Ojo, F. Akinpelu, Lyapunov functions and global properties of seir epidemic model, International journal of Chemistry, Mathematics and Physics 1 (1) (2017)
Ojo, M.M., Gbadamosi, B., Benson, T.O., Adebimpe, O., Georgina, A.: Modeling the dynamics of lassa fever in Nigeria. J. Egyptian Math. Soc. 29(1), 1–19 (2021)
Peter O.J., Kumar S., Kumari N., Oguntolu F.A., Oshinubi K., Musa R., Transmission dynamics of monkeypox virus: a mathematical modelling approach, Model. Earth Syst. Environ., (2021) 1–12
Peter, O.J., Qureshi, S., Yusuf, A., Al-Shomrani, M., Idowu, A.A.: A new mathematical model of covid-19 using real data from pakistan. Results Phys. 24, 104098 (2021)
Ojo, M., Gbadamosi, B., Olukayode, A., Oluwaseun, O.R.: Sensitivity analysis of dengue model with saturated incidence rate. Open Access Library J 5(03), 1 (2018)
Abioye, A., Ibrahim, M., Peter, O., Amadiegwu, S., Oguntolu, F.: Differential transform method for solving mathematical model of seir and sei spread of malaria 40(1), 197–219 (2018)
Abioye, A.I., Peter, O.J., Ogunseye, H.A., Oguntolu, F.A., Oshinubi, K., Ibrahim, A.A., Khan, I.: Mathematical model of covid-19 in Nigeria with optimal control. Results Phys. 28, 104598 (2021)
Ayoola, T.A., Edogbanya, H.O., Peter, O.J., Oguntolu, F.A., Oshinubi, K., Olaosebikan, M.L.: Modelling and optimal control analysis of typhoid fever. J. Math. Comput. Sci. 11(6), 6666–6682 (2021)
Peter O., Ibrahim M., Oguntolu F., Akinduko O., Akinyemi S., Direct and indirect transmission dynamics of typhoid fever model by differential transform method (2018)
Musa, S.S., Zhao, S., Hussaini, N., Habib, A.G., He, D.: Mathematical modeling and analysis of meningococcal meningitis transmission dynamics. Int. J. Biomath. 13(01), 2050006 (2020)
Karachaliou A., Conlan A.J., Preziosi M.-P., Trotter C.L., Modeling long-term vaccination strategies with menafrivac in the african meningitis belt, Clinical Infectious Diseases 61 (suppl_5) (2015) S594–S600
Tilahun, G.T.: Modeling co-dynamics of pneumonia and meningitis diseases. Adv. Differ. Equ. 2019(1), 1–18 (2019)
Peter, O.J., Yusuf, A., Oshinubi, K., Oguntolu, F.A., Lawal, J.O., Abioye, A.I., Ayoola, T.A.: Fractional order of pneumococcal pneumonia infection model with caputo fabrizio operator. Results Phys. 29, 104581 (2021)
Peter O.J., Shaikh A.S., Ibrahim M.O., Nisar K.S., Baleanu D., Khan I., Abioye A.I., Analysis and dynamics of fractional order mathematical model of covid-19 in nigeria using atangana-baleanu operator, Comput. Mater. Continua (2021) 1823–1848
Khan, H., Gómez-Aguilar, J., Alkhazzan, A., Khan, A.: A fractional order hiv-tb coinfection model with nonsingular mittag-leffler law. Math. Methods Appl. Sci. 43(6), 3786–3806 (2020)
Ahmad, S., Ullah, A., Al-Mdallal, Q.M., Khan, H., Shah, K., Khan, A.: Fractional order mathematical modeling of covid-19 transmission. Chaos, Solitons Fractals 139, 110256 (2020)
Peter O.J., Transmission dynamics of fractional order brucellosis model using caputo–fabrizio operator, Int. J. Differ. Equ. (2020)
Shaikh, A.A., Qureshi, S.: Comparative analysis of riemann-liouville, caputo-fabrizio, and atangana-baleanu integrals. J. Appl. Math. Comput. Mech. 21(1), 91–101 (2022)
Yusuf, A., Qureshi, S., Mustapha, U.T., Musa, S.S., Sulaiman, T.A.: Fractional modeling for improving scholastic performance of students with optimal control. Int. J. Appl. Comput. Math. 8(1), 1–20 (2022)
Arafa A., Khalil M., Sayed A., A non-integer variable order mathematical model of human immunodeficiency virus and malaria coinfection with time delay, Complexity (2019)
Carvalho, A.R., Pinto, C.M., Baleanu, D.: Hiv/hcv coinfection model: a fractional-order perspective for the effect of the hiv viral load. Adv. Differ. Equ. 2018(1), 1–22 (2018)
Nazeer, M., Hussain, F., Khan, M.I., El-Zahar, E.R., Chu, Y.-M., Malik, M., et al.: Theoretical study of mhd electro-osmotically flow of third-grade fluid in micro channel. Appl. Math. Comput. 420, 126868 (2022)
Chu, Y.-M., Shankaralingappa, B., Gireesha, B., Alzahrani, F., Khan, M.I., Khan, S.U.: Combined impact of cattaneo-christov double diffusion and radiative heat flux on bio-convective flow of maxwell liquid configured by a stretched nano-material surface. Appl. Math. Comput. 419, 126883 (2022)
Zhao T.-H., Khan M.I., Chu Y.-M., Artificial neural networking (ann) analysis for heat and entropy generation in flow of non-newtonian fluid between two rotating disks, Mathematical Methods Appl. Sci. (2021)
Chu, Y.-M., Nazir, U., Sohail, M., Selim, M.M., Lee, J.-R.: Enhancement in thermal energy and solute particles using hybrid nanoparticles by engaging activation energy and chemical reaction over a parabolic surface via finite element approach. Fractal Fractional 5(3), 119 (2021)
Madhukesh, J., Kumar, R.N., Gowda, R.P., Prasannakumara, B., Ramesh, G., Khan, M.I., Khan, S.U., Chu, Y.-M.: Numerical simulation of aa7072-aa7075/water-based hybrid nanofluid flow over a curved stretching sheet with newtonian heating: a non-fourier heat flux model approach. J. Mole. Liquids 335, 116103 (2021)
Wang, J., Khan, M.I., Khan, W.A., Abbas, S.Z., Khan, M.I.: Transportation of heat generation/absorption and radiative heat flux in homogeneous-heterogeneous catalytic reactions of non-newtonian fluid (oldroyd-b model). Computer Methods Programs Biomed. 189, 105310 (2020)
Khan, M.I., Hayat, T., Waqas, M., Alsaedi, A.: Outcome for chemically reactive aspect in flow of tangent hyperbolic material. J. Mole. Liquids 230, 143–151 (2017)
Hayat, T., Tamoor, M., Khan, M.I., Alsaedi, A.: Numerical simulation for nonlinear radiative flow by convective cylinder. Results Phys. 6, 1031–1035 (2016)
Qayyum, S., Khan, M.I., Hayat, T., Alsaedi, A.: Comparative investigation of five nanoparticles in flow of viscous fluid with joule heating and slip due to rotating disk. Physica B: Condensed Matter 534, 173–183 (2018)
Khan, M.I., Qayyum, S., Kadry, S., Khan, W., Abbas, S.: Irreversibility analysis and heat transport in squeezing nanoliquid flow of non-newtonian (second-grade) fluid between infinite plates with activation energy. Arabian J. Sci. Eng. 45(6), 4939–4947 (2020)
Arqub, O.A., Maayah, B.: Numerical solutions of integrodifferential equations of fredholm operator type in the sense of the Atangana-baleanu fractional operator. Chaos Solitons Fractals 117, 117–124 (2018)
Arqub, O.A., Al-Smadi, M.: Atangana-baleanu fractional approach to the solutions of bagley-torvik and painlevé equations in hilbert space. Chaos Solitons Fractals 117, 161–167 (2018)
Momani S., Abu Arqub O., Maayah B., Piecewise optimal fractional reproducing kernel solution and convergence analysis for the atangana–baleanu–caputo model of the lienard’s equation, Fractals 28 (08) (2020) 2040007
Momani, S., Maayah, B., Arqub, O.A.: The reproducing kernel algorithm for numerical solution of van der pol damping model in view of the atangana-baleanu fractional approach. Fractals 28(08), 2040010 (2020)
Choi, S., Kang, B., Koo, N.: Stability for caputo fractional differential systems. Abstract Appl. Anal. 2014, 631419 (2014)
Geng X., Katul G.G., Gerges F., Bou-Zeid E., Nassif H., Boufadel M.C., A kernel-modulated sir model for covid-19 contagious spread from county to continent 118 (21) (2021). https://doi.org/10.1073/pnas.2023321118.
Castillo-Chavez C., Blower S., Driessche P., Kirschner D., Yakubu A., Mathematical approaches for emerging and reemerging infectious diseases: models method and theory (2002) 84
Castillo-Chavez, C., Song, B.: Dynamical models of tuberculosis and their applications. Math. Biosci. Eng. 1(2), 361–404 (2004)
Atangana, A., Baleanu, D.: New fractional derivatives with non-local and non-singular kernel: Theory and application to heat transfer model. Thermal Sci. 20(2), 763–769 (2016)
Qureshi, S., Yusuf, A.: Modeling chickenpox disease with fractional derivatives: from caputo to atangana-baleanu. Chaos Solitons Fractals 122, 111–118 (2019)
Khan, M., Ullah, S., Farooq, M.: A new fractional model for tuberculosis with relapse via atangana baleanu derivative. Chaos Solitons Fractals 116, 227–238 (2018)
Qureshi, S., Atangana, A.: Mathematical analysis of dengue fever outbreak by novel fractional operators with field data. Physica A Stat. Mech. Appl. 526, 121127 (2019)
Central Intelligence Agency, The world factbook, Available from: https://www.cia.gov/the-world-factbook/countries/nigeria/
Changpin L., Fanhai Z., Numerical methods for fractional calculus 24 (2015)
Jajarmi A., Baleanu D., A new fractional analysis on the interaction of hiv with cd4+ t-cells, Chaos, Solitons and Fractals 113 (2018) 221–229. https://doi.org/10.1016/j.chaos.2018.06.009.
Baleanu D., Jajarmi A., Hajipour M., On the nonlinear dynamical systems within the generalized fractional derivatives with mittag-leffler kernel. Nonlinear dynamics, Nonlinear dynamics 94 (1) (2018) 397–414. https://doi.org/10.1016/j.chaos.2018.06.009.
Funding
No funding received.
Author information
Authors and Affiliations
Contributions
OJP conceived the idea, model formulation, OJP AF, MMO,SK,NK and FAO Writing, qualitative analysis, editing, proof reading
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Peter, O.J., Yusuf, A., Ojo, M.M. et al. A Mathematical Model Analysis of Meningitis with Treatment and Vaccination in Fractional Derivatives. Int. J. Appl. Comput. Math 8, 117 (2022). https://doi.org/10.1007/s40819-022-01317-1
Accepted:
Published:
DOI: https://doi.org/10.1007/s40819-022-01317-1