Abstract
Objectives
Sexual selection typically centers on bodily and psychological traits. Non-bodily traits ranging from housing and vehicles through art to social media can, however, influence sexual selection even in absence of the phenotype proper. The theoretical framework of human sexual selection is updated in this article by unifying four theoretical approaches and conceptualizing non-bodily traits as extended phenotypic traits.
Methods
Existing research is synthesized with extended phenotype theory, life history theory, and behavioral ecology. To test population-level hypotheses arising from the review, ecological and demographic data on 122 countries are analyzed with multiple linear regression modelling.
Results
A four-factor model of intelligence, adolescent fertility, population density, and atmospheric cold demands predicts 64% of global variation in economic complexity in 1995 and 72% of the variation in 2016.
Conclusions
The evolutionary pathways of extended phenotypes frequently undergo a categorical broadening from providing functional benefits to carrying signalling value. Extended phenotypes require investments in skills and bioenergetic resources, but they can improve survival in high latitudes, facilitate the extraction of resources from the environment, and substantially influence sexual selection outcomes. Bioenergetic investments in extended phenotypes create individual- and population-level tradeoffs with competing life history processes, exemplified here as a global tradeoff between adolescent fertility and economic complexity. The merits of the present model include a more systematic classification of sexual traits, a clearer articulation of their evolutionary-developmental hierarchy, and an analysis of ecological, genetic, and psychological mechanisms that modulate the flow of energy into extended phenotypes and cultural innovations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Data gathered through observation and experimentation provide clues about the structure and function of the natural world, and theory organizes existing data and new ideas into a cohesive conceptual framework to both explain existing observations and make novel predictions. Theory reduces the apparent complexity of the natural world, because it captures essential features of a system, provides abstracted characterizations, and makes predictions for as-yet unobserved phenomena (Marquet et al. 2014).
A thought experiment: confiscate Shakespeare’s quill pens; eradicate the written record of his works—“bury it certain fathoms in the earth”. Dispossess Jimi Hendrix of his guitars and obliterate his extant musical recordings. Wipe out Hugh Hefner’s magazine oeuvre, dismantle the mansion, send the Bunnies home. Through these actions, each man’s pathway to global fame would have been thwarted. Lacking these culturally hallowed or notorious artefacts, each man’s capacity to signal his creativity and neurocognitive prowess would have been limited to bodily and behavioral traits. As such, each man would either have had to invent an alternative solution to the problem of signalling his creative and genetic quality (which are partially overlapping categories), or be resigned to cultural and/or reproductive anonymity.
Yet by using their instruments, creative tools, and surrounding social organizations in novel ways, each man has succeeded in creating artefacts that amplified his signalling capacity by orders of magnitude. Each man interacted with the external world in so successful a way that the artefacts he produced have infiltrated the minds of millions of people. This feat has been accomplished with remarkably little contact between the artefact creators and their audiences. These artefacts—from The Tempest (as cited above) through “All Along the Watchtower” to the first issue of Playboy featuring Marilyn Monroe—have enabled each man to make a luxurious living (e.g. Archer 2011a; Green 2017; Lawrence 2006), but they also bear inherent signalling value which informs others of the genetic and phenotypic quality of the individuals who produced them.
Following theory from evolutionary biology (Bailey 2012), such artefacts and non-bodily ornaments can be conceptualized as extended phenotypes—as parts of the phenotype expressed beyond the body boundary (Dawkins 1982). Extended phenotypes in humans have myriad predecessors in the animal kingdom (Schaedelin and Taborsky 2009). In this article, I highlight some of the many ways in which extended phenotypic “action at a distance” is of increasing relevance to contemporary sexual selection in humans, and is the missing link in a coherent theoretical framework for sexual traits.
Sexual selection comprises any variation in direct fitness among different phenotypes caused by their ability to gain sexual partners, produce fertilized eggs, and generate offspring (Cornwallis and Uller 2010). Sexual selection can occur via various mechanisms (Kordsmeyer et al. 2018; Puts 2016; Winegard et al. 2018), mate choice being among the most important in modern humans (Antfolk et al. 2015; Conroy-Beam 2018, and references therein). Effective mate choice consists of an evaluation of multiple sexual traits over a prolonged period of time (Barelds and Barelds-Dijkstra 2007; Candolin 2003; Groyecka et al. 2017; Harrison and Shortall 2011; Jonason et al. 2015; Miller and Todd 1998; Schaedelin and Taborsky 2009).
Sexual traits have been analyzed at several different levels in humans. In ascending order of abstraction, sexual traits can firstly be divided into specific observable cues, such as secondary sexual signals, material possessions, and acts of kindness (Miller and Todd 1998). Secondly, cues inform more abstract perceptions such as overall physical attractiveness, intelligence, and status (Csajbók and Berkics 2017; Miller and Todd 1998; Winegard et al. 2018). Thirdly, perceptions are proxies of evolutionarily relevant qualities which have an ultimate fitness impact (Miller and Todd 1998). A typical categorization at this level sequesters good genes, good provisioning ability, and good parental ability into different categories (Lu et al. 2015; Lu et al. 2017). These qualities can be associated with specific traits, which then influence perceptions about overall mate quality.
On the first level of observable cues, it is common to define sexual traits as falling roughly into three categories: physical, behavioral, and a third category which most notably consists of wealth, resources, and other status cues (Conroy-Beam et al. 2015; Conroy-Beam and Buss 2016; Sugiyama 2015; Winegard et al. 2018). The role of traits in this third non-bodily category has been extensively studied (e.g. Hughes and Aung 2017; Kruger and Kruger 2018), but it currently lacks a coherent theoretical framework to organize a range of disparate traits. Following theory from evolutionary biology, the current review conceptualizes this category as extended phenotypic traits (Dawkins 1982; Head et al. 2017; Schaedelin and Taborsky 2009).
The merits of the present model include a recognition of the evolutionary continuity and theoretical linkage between extended phenotypes in human and nonhuman animals; acknowledging that most extended phenotypes are not merely costly signals but that their signalling characteristics can evolve from a direct function; synthesizing extended phenotype theory with life history theory and behavioral ecology, thus illustrating how different environments shape selection pressures on extended phenotypes; a clearer articulation of the developmental hierarchy of sexual traits than currently exists in the literature; an empirical analysis of population differences in extended phenotypic trait expression based on global variation in economic complexity, including ecological and life history constraints; and an explicit emphasis given to the genetic underpinnings of extended phenotypic trait expression in humans.
These facets provide a number of theoretical and empirical advances on the current framework which conceptualizes wealth displays and cultural products merely as costly status signals (e.g. Kruger and Kruger 2018; Winegard et al. 2018), short-term mating aids (Sundie et al. 2011), and/or as indications of resources and provisioning capacity (e.g. Cloud and Taylor 2018; Hughes and Aung 2017). The extended phenotype approach also provides a novel theoretical framework for online dating behaviors and creative displays more generally, thus advancing synthesis between existing theory and empirical findings. To the best of my knowledge, this is the first article that integrates extended phenotype theory with life history theory—the most influential theory in ecology (Marquet et al. 2014) that has also presented its applied value in a range of other fields (Hurst and Kavanagh 2017; Krams et al. 2017a, 2017b, 2017c, 2019; Luoto et al. 2019; Maner et al. 2017; Sng et al. 2018; Wells et al. 2017; Woodley of Menie et al. 2017; Worthman and Trang 2018).
Extended phenotype is a concept that is relatively widely used in sexual selection research in nonhuman animals (Bailey 2012; Head et al. 2017; Jordan et al. 2016; Schaedelin and Taborsky 2009). An organism’s construction behaviors, for example, may sometimes modify the environment in fitness-enhancing (or fitness-damaging) ways. This creates a category of non-bodily traits—beaver dams, termite mounds, bird nests, spider webs, online dating profiles—that phenotypic or behavioral traits alone do not capture. Extended phenotypes (Dawkins 1982, 2004) result from the effects of an allele upon the world that influence that allele’s survival and/or proliferation. This includes any feature of the abiotic or biotic environment that is the result of manipulative action or construction behavior of an organism, but excludes any result of such manipulative behavior that does not affect the organism’s fitness negatively or positively (Dawkins 1982; Schaedelin and Taborsky 2009). This definition therefore includes the dam that a beaver builds, but not the footprints it makes when building it, because only the dam influences the fitness of the beaver (Dawkins 1982, p. 315).
The concept of extended phenotype has lately stimulated substantial research activity in nonhuman animal research as the tools of genomics and proteomics provide new evidence of its importance (Blamires et al. 2017, 2018; Geffre et al. 2017; Hoover et al. 2011; Hunter 2009, 2018). The main argument of the current article is that this theoretical framework should also be applied to research on human sexual selection and cultural evolution. Richard Dawkins did not originally apply the concept of extended phenotype to human sexual selection, and prior research on human sexual selection has almost completely overlooked its utility as an analytical tool. Geoffrey Miller’s work has benefited from conceptualizing certain sexual traits as extended phenotypes, but Miller (1999, 2000, 2001) only considered inanimate features of the external world as extended phenotypes and omitted the consideration of other organisms as such. As Miller (2000, p. 276) notes: “To explain our aesthetic preferences, we should be able to use the same sexual selection theories that biologists use to explain mating preferences.” This view is expanded in the current article to also evaluate the functional aspects of extended phenotypes, which informs analyses about the evolutionary pathways from direct functional utility to signalling.
Extended phenotypes are key functional traits (cf. Meunier et al. 2017). They can affect an individual’s fitness either functionally (Dawkins 1982), as a signal, or as a cue (Schaedelin and Taborsky 2009; see Glossary for full definitions of terms used in this article). For example, in addition to a nest’s function as a safe environment for rearing offspring, nests provide information about the constructor and have a sexual selection component in many bird species. Their original utilitarian function is complemented with signalling value. Potential mates assess these traits external to, or even in temporary absence of, the phenotype proper (Schaedelin and Taborsky 2009).
It is an evolutionary trend for extended phenotypes to undergo a gradual broadening from a purely utilitarian function to also carrying signalling value (Schaedelin and Taborsky 2009; cf. Winegard et al. 2018). Extended phenotypes with novel signalling value can therefore be classified as exaptations. An exaptation is a feature that enhances fitness in a way that differs from its “original” evolutionarily selected role (Buss et al. 1998; Gould 1991; Gould and Vrba 1982). In other words, an exaptation is a trait that currently has a positive fitness effect without being an adaptation specifically for that effect, having acquired a novel function in the course of evolution (Gangestad 2008; see also Maex et al. 2018 for a recent proximate-level discussion). This novel function may be further enhanced by secondary adaptations, i.e. features that improve a trait’s exapted rather than original function. Thus, any complex trait will be likely to include a sequential mixture of adaptations, exaptations, and secondary adaptations (Gould and Vrba 1982).
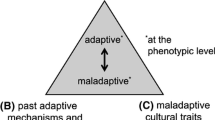
Another analytical dimension recognizes that extended phenotypes can impact an organism’s fitness either through viability selection (by improving survival, thus indirectly enhancing reproductive fitness) (Blamires et al. 2017, 2018; Hoover et al. 2011) and/or through sexual selection (Rubalcaba et al. 2016; Head et al. 2017; Miller 2001; Schaedelin and Taborsky 2009). It is important to recognize viability selection and sexual selection as different processes that generate different selection pressures on organisms to invest in extended phenotypes (Fig. 1) (cf. Jordan et al. 2016; Winegard et al. 2018).
Viability selection, sexual selection, and (to a lesser extent) kin selection impose different selection pressures on organisms to invest bioenergetic resources in the development of phenotypic, behavioral, and extended phenotypic traits. The bidirectional arrows between ultimate causes, proximate mechanisms, and phenotypic outcomes (Luoto and Rantala 2017; Rantala et al. 2018) indicate reciprocal causation—the idea that developmental processes and phenotypic outcomes can influence evolutionary change (Bateson and Gluckman 2011; Dickins and Barton 2013; Laland et al. 2011). Thus, proximate mechanisms, phenotypic plasticity, phenotypic variability, and behavioral variability can all feed back into shaping ultimate-level selection pressures. Phenotypic plasticity, for instance, can cause evolutionary innovations by inducing morphological novelty (Levis et al. 2018), while human cultural practices can precipitate complex evolutionary feedback loops between the three levels of analysis (Gienapp et al. 2008; Hoberg and Brooks 2015; Luoto et al. 2019; Reinisch et al. 2017).
Extreme trait elaboration in nonhuman animals has emerged via the mutually reinforcing synergism that takes place between viability selection and sexual selection, as well as the synergism that occurs between direct functionality and signalling. Examples of these synergistically evolved traits include 1) the powerful electric discharge of electric fishes: a behavioral trait used in predation and signalling, and 2) nest construction: a functional extended phenotype which also acts as a sexually selected signal in some bird and fish species (Head et al. 2017; Schaedelin and Taborsky 2009; West-Eberhard 2014, and references therein). The next section outlines how mutually reinforcing synergism between viability selection and sexual selection also exists in humans, and is particularly conspicuous with extended phenotypes.
Extended Phenotypes in Human Sexual Selection
Inanimate Extended Phenotypes
To exemplify the categorical broadening that some extended phenotypes have undergone from functional to signalling value, it is illustrative that the original function of human housing was to provide shelter, not to convey information. Paleolithic hunter-gatherers made constructions ranging from residential to special-purpose camps, which provided shelter and a stable locale for routines such as tool and clothing manufacture (Delson 2000, p. 636). These camps originally had a clear utility: to enhance the survival of the humans who inhabited them (e.g. Hoffecker and Hoffecker 2017b).
While housing has retained its viability selection component, it has also taken on a signalling function. To use the novel Pride and Prejudice as a historical example, the heroine’s resistance to a prominent male’s courtship undergoes a gradual transformation from initial resistance to finally accepting him as a mate when she is exposed to the grandeur and style of the territory that he occupies. Like a female bower bird, Elizabeth inspects the “nest” of a potential mate, Mr. Darcy, and at that very moment feels “that to be mistress of [that territory] might be something!” (Austen 2007/1813, p. 207). Likewise, a contemporary man presented in a luxury apartment is rated by women as significantly more attractive than when presented in an average apartment (Dunn and Hill 2014). Experimental research (Dunn and Hill 2014) on human housing as “augmented extended phenotypes” has equivalents in corresponding experimental interventions in fish, in which an augmented extended phenotype can lead to higher mating success in males (Jordan et al. 2016).
Cars provide another example of how extended phenotypes that were originally construed as functional innovations have taken on signalling characteristics. While seated in a Silver Bentley Continental GT, men were rated significantly more attractive by women than when seated in a Red Ford Fiesta ST (Dunn and Searle 2010). This finding has been replicated with various car models in other studies (Shuler and McCord 2010; Sundie et al. 2011), while still other car models were used in a study which found a positive correlation between the price of males’ cars and the likelihood of receiving phone numbers of young women in France (Guéguen and Lamy 2012).
Clothing provides yet another example of how functional extended phenotypes have been exapted for signalling. While the main function of clothing from the point of view of cultural evolution is to make humans adapted to different climates (d’Errico et al. 2018; Gilligan 2007, 2010), clothing also contains substantial signalling value in humans (Nelissen and Meijers 2011). Where women’s phenotypic features such as facial attractiveness (Roberts et al. 2004) and bodily scents (Singh and Bronstad 2001; Thornhill et al. 2003) have been found to undergo minor changes during ovulation, hypothetically to communicate fertility, evidence suggests that clothing functions as an additional extended phenotypic cue of ovulation (Durante et al. 2008; Eisenbruch et al. 2015; Haselton et al. 2007) and of courtship motivation (Grammer et al. 2004). Women tend to prefer clothing that is more revealing and more appealing to men close to the onset of ovulation (Durante et al. 2008; Eisenbruch et al. 2015; but see Blake et al. 2017 for mixed results and Arslan et al. 2018 for null findings). Clothing can therefore function as a malleable non-bodily ornament that signals its displayer’s mating motives (Elliot et al. 2013). Clothing can also augment evolved phenotypic features in humans, such as the use of high-heeled footwear which modifies women’s lumbar curvature and gait to be more appealing to men (Lewis et al. 2017b; Morris et al. 2013), while also exerting an effect on men’s prosocial behaviors towards women (Guéguen 2015).
Apartments, vehicles, and clothing are typical examples of extended phenotypes that have become cues of wealth, status, reproductive motivation, and reproductive value. Thus, they add cumulative fitness value above and beyond their functionality. Classifying their signalling value as an exaptation (Gould and Vrba 1982; Gould 1991) is consistent with evolutionary theory, since the original function of apartments, vehicles, and clothing has been “exapted” to convey information, thus taking on a new signalling role in human sexual selection. Whatever modifications they may have undergone for the novel signalling component to be enhanced can be seen to constitute secondary adaptations (cf. Gould and Vrba 1982).
Creativity is another important trait in mate choice: it was ranked as the 6th most desirable trait by women and 7th by men in a cross-cultural study on mate preferences (Buss et al. 1990; cf. Zietsch et al. 2012). Li and colleagues (Li et al. 2002) found that creativity is an important factor in mate choice once a baseline level of other traits had been satisfied. In an experiment that demonstrated the importance of extended phenotypic traits for assessing mate quality and creativity, an attractive male confederate solicited phone numbers from young women walking on the street in France (Guéguen et al. 2014). This act was performed under two experimental conditions and a control condition: the male was carrying either a large sports bag, a guitar case, or nothing at all. In the control condition, 14% of the young women provided the male with their phone numbers, compared with 9% in the sports bag condition and 31% in the guitar case condition (Guéguen et al. 2014). The guitar case seemed to have been a cue of the male’s personal qualities, yielding a significantly more positive courtship outcome. Notably, carrying the guitar case was a cue powerful enough to have an effect on his desirability, with no creative behavior as such needed to produce the desired effect. This highlights the difference between owning artefacts (a business magnate with an ostentatious Lamborghini) and creating them (an artist devoting immense effort and skill into their cultural oeuvre). Both outcomes may require different kinds of cognitive and personality traits, and may be differentially selected for by prospective mates (cf. de Manzano and Ullén 2018; Kruger and Kruger 2018; Nave et al. 2018; Sundie et al. 2011; Winegard et al. 2018).
Creativity can take several different forms, which are not all rated equally desirable by the opposite sex (Kaufman et al. 2014). Sculptures, paintings, musical recordings, and literature potentially constitute extended phenotypes that have a beneficial impact on the reproduction of the individual who produced (Clegg et al. 2011; Madison et al. 2018; Miller 2001; Winegard et al. 2018) or reproduced them. Written records of men’s compliments to women, for example, increase a man’s attractiveness when those compliments are creative rather than trite (Gao et al. 2017a). Indeed, artistic artefacts can even have an intergenerational effect (Boyd 2009) by acting as a cue of genetic quality which can improve the fitness of the descendants of a great artist. The intergenerational functional value of extended phenotypes has been noted by Lehmann (2008) and described as ecological inheritance by niche construction theorists (Laland et al. 2017; Odling-Smee et al. 2003). What I highlight is that extended phenotypes can also have signalling value in sexual selection across generations by communicating genetic rather than phenotypic quality. This can occur, for example, when an artist’s descendants continue to reap benefits from the enduring prestige of their progenitor’s art (Boyd 2009, p. 112; Winegard et al. 2018).
As a final example of inanimate extended phenotypes in human sexual selection, a central development in contemporary humans has been the emergence of online dating culture (Rosenfeld and Thomas 2012; Sumter et al. 2017). Where the beaver’s dam provides an enlarged catchment area to harvest energy from the environment (Dawkins 1982), a well-crafted profile on a dating site can provide vastly improved reproductive prospects to individuals who invest in such phenotypic extensions that can proliferate online without inducing a major cost for the signaller (cf. Shapiro et al. 2017; Sumter et al. 2017; Timmermans and Courtois 2018, though a rigorous empirical study comparing the mating success of users and non-users of online dating remains to be done).
Importantly, together with many other extended phenotypic traits in humans, online dating profiles share many of the potential advantages identified by Schaedelin and Taborsky (2009) as typical for extended phenotypic signals in nonhuman animals. These advantages are listed in Table 1. Despite potential advantages, however, a salient feature of online dating profiles as extended phenotypic cues is the ease with which they can be produced and modified; thus, they may occasionally be dishonest cues of underlying phenotypic and/or genetic quality (Donath 1998; Toma and Hancock 2010; Guadagno et al. 2012). This may be an advantage for the signaller but a disadvantage for the recipient.
Genetic Architecture and Extended Phenotypic Plasticity
Besides the importance of cultural evolution and social learning (Blake et al. 2018; Creanza et al. 2017), it is reasonable to postulate the existence of a genetic basis in variation in extended phenotypes (cf. de Manzano and Ullén 2018). Dawkins (2004) elaborated on it as follows. Variations in replicators (i.e. alleles) need to be causally linked with variations in extended phenotypes so that, over generations, alleles associated with good extended phenotypes survive in the replicator pool at the expense of rival alleles associated with poorer extended phenotypes (Dawkins 2004, p. 379). Evidence for a genetic basis of an extended phenotype is not necessary to establish a feature of the external world as an extended phenotype, although in many instances there are strong grounds to believe that there is an important genetic component involved (Dawkins 2004; cf. de Manzano and Ullén 2018). In some cases, this genetic component has been identified (Hoover et al. 2011). In others, the ‘replicator-to-extended phenotype’ link may be more strained, but it is also challenging to delineate when there is no genetic component involved in between-individual variation in extended phenotypic trait expression.
Analyzing some of the examples of extended phenotypes given in the previous section through Dawkins’s (2004) strict allelocentric definition might suggest that an allele codes for, say, luxury car ownership and another allele in the same locus codes for more modest automotive display (likewise for any other example of extended phenotypes). This is not the intended meaning of the present argument, nor that of Dawkins. Instead, luxury car ownership, like any other complex trait, arises from a composite of polygenic traits (Charney 2012; Dawkins 1982) together with complex interactions with the animate and inanimate environment.
To exemplify this position, consider the following. Learning mechanisms are evolved adaptations to cope with changing environmental conditions (Ardiel and Rankin 2010; Dennett 1975; Muthukrishna and Henrich 2016); technical skills and constructive behaviors are adaptive behaviors that facilitate innovations and the colonization of harsh environments (Barton and Venditti 2014; Hoffecker and Hoffecker 2017b; Lane 2016; Sol et al. 2016). They are all polygenic traits (Barbot et al. 2013; Charney 2012; Davies et al. 2011; Hill et al. 2018a, 2018b; Reuter et al. 2006; Sniekers et al. 2017) that can lead to variation in functional and signalling extended phenotypes. Arriving at the genetic basis of variation in extended phenotypes may not be straightforward. That does not invalidate the hypothesis that there are genetic designs that produce better extended phenotypes (cf. Barbot et al. 2013; Dawkins 2004; de Manzano and Ullén 2018; Mosing et al. 2015). What it may require, however, is analyses extending to polygenic traits such as general learning capacity, intelligence, creativity, technical skills, and constructive predispositions (or their interactive effects) that underlie heritable variation in extended phenotypic trait expression.
Models of sexual selection which assume a direct relationship between genes and phenotypes have been rightly critiqued (Cornwallis and Uller 2010). Seeing phenotypic plasticity merely as a condition-dependent expression of sexual traits ignores the multi-dimensional nature of plasticity, which can also constitute an adaptive reaction to changing environmental conditions (Bateson and Gluckman 2011; Cornwallis and Uller 2010; Luoto et al. 2019). The resulting question therefore is: why should extended phenotypes be interpreted only as the effect of genes/alleles upon the world, when in fact conventional phenotypes are seen as the result of the interaction between genes and the environment?
Phenotypic plasticity refers to the ability of a single genotype to produce multiple phenotypes when exposed to different environments (Pigliucci 2005). The problem of coping with a cold environment, for example, can be solved a) phenotypically (e.g. by evolving subcutaneous adipose stores, which is an evolutionarily ancient solution: Ball et al. 2017; Speakman 2018; Steegmann et al. 2002; Wells 2012), b) behaviorally (e.g. by birds migrating ahead of cold spells: Briedis et al. 2017; Somveille et al. 2018), or c) by an efficient use of extended phenotypes (e.g. clothing and shelter in humans: Chu 2009; Gilligan 2007, 2010; Hoffecker and Hoffecker 2017b; MacDonald 2018; cf. burrow and shelter use in other mammals: Kowalczyk and Zalewski 2011; Reichman and Smith 1990).
Phenotypic plasticity has been so extensively documented that non-plastic phenotypes are an anomaly which almost requires more explanation than phenotypic plasticity itself (Cornwallis and Uller 2010, and references therein). For this reason, whilst investigations that identify the genetic component of extended phenotypes are valuable (e.g. Hoover et al. 2011), future research would greatly benefit from recognizing the inherent plasticity in the expression of extended phenotypes (cf. Blamires 2010; Blamires et al. 2017, 2018; Geffre et al. 2017). This approach empirically expands Dawkins’s original formulation of the extended phenotype theory and links it more explicitly with life history theory and behavioral ecology. That environmental influences are underdeveloped in Dawkins’s (1982, 2004) foundational work on extended phenotypes does not mean that the theory has to be abandoned—it means that it needs to be integrated with behavioral ecology and life history theory.
For the purposes of theoretical modelling, it is irrelevant whether an extended phenotype is an evolved adaptation that has arisen as a result of selection on the extended phenotype itself, or a byproduct of selection on some other general trait (such as intelligence or creativity)—provided that it shows heritable variation (Bailey 2012). Therefore, understanding the precise genetic mechanisms underlying extended phenotypic traits is not a prerequisite for meaningfully modelling the effects of those traits on sexual selection and/or viability selection. Just as behavioral plasticity and extended phenotypic plasticity can create novel traits for natural selection to act on, they may also generate novel interactions between various existing traits (Moczek et al. 2011) and their biological substrates. This idea is further explored in the next section by synthesizing the extended phenotype theory with life history theory and behavioral ecology.
Extended Phenotypes and Life History Evolution
Life history theory can be applied to the study of extended phenotypes both at the level of individuals (next section) and at the level of populations (see section “Integration with Behavioral Ecology”, below). Life history theory and extended phenotype theory have several fundamental points of contact, which provides a strong rationale for synthesizing them. Theory unification (Marquet et al. 2014) between sexual selection, extended phenotype theory, life history theory, and behavioral ecology is one of the major contributions of this article.
Widely used in evolutionary biology and increasingly in evolutionary psychology, life history (LH) theory is a mid-level theory derived from general evolutionary theory. The focus of LH theory is on how the entire life cycle of an organism is designed by natural selection to optimize reproductive success despite environmental challenges such as mortality and resource scarcity (Ellison 2017; Stearns et al. 2008). This design is set within a framework of constraints and tradeoffs that are shaped by past evolutionary pressures and current environmental contingencies (Krams et al. 2016, 2017a, 2017b, 2019; Meunier et al. 2017). Life history evolution works on the materials out of which organisms are built, as well as the developmental, physiological, and psychological traits that organisms have inherited from their ancestors and that are specifically calibrated to their environments (Bateson and Gluckman 2011; Ellison 2017; Figueredo et al. 2004, 2006; Flatt and Heyland 2011; Immonen et al. 2018; Krams et al. 2016; Luoto et al. 2019; Snell-Rood et al. 2015; Stearns et al. 2008).
LH theory presents a broad biobehavioral framework for understanding how organisms allocate bioenergetic resources between somatic effort and reproductive effort (Black et al. 2017; Worthman and Trang 2018) and how those allocations affect the developmental sequencing of human motivation (Kenrick et al. 2010). Extended phenotypic traits can (to varying degrees) be considered core life history traits: because LH theory is a theory about resource allocation, and because extended phenotypes are often energetically expensive, LH theory can and should be used to analyze the dynamic investments that organisms make between phenotypic, behavioral, and extended phenotypic traits. LH theory recognizes that because the time, energy, and resources at any organism’s disposal are finite (Ellison 2017; Kecko et al. 2017), bioenergetic investment in one type of LH trait is typically traded off with neglect of another trait (Krams et al. 2017c; Snell-Rood et al. 2015).
Extended Phenotypes in Individual-Level Life History Development
LH theory can be especially relevant to the recognition of a logical sequence underlying the development of phenotypic traits, behavioral traits, and extended phenotypic traits. Fetal growth and early childhood are characterized by high somatic investment as an embryo develops into adulthood. Organisms in early stages of development seldom make serious, protracted investments into producing extended phenotypes (the protective cases built by caddisfly larvae may be one counterexample; Dawkins 1982, p. 301). This is simply because an adequately developed phenotype—including perceptual, motor, cognitive, and brain development—is a prerequisite for the emergence of more elaborate behavioral traits. In a similar hierarchical sequence, it is almost impossible for an organism to produce or acquire extended phenotypes in absence of any behaviors.Footnote 1 In the majority of cases, behaviors are needed for an effective manipulation or acquisition of extended phenotypes. This is no trivial point, because it influences when in individual development extended phenotypic traits are likely to receive more bioenergetic investment from organisms.
For a developing organism, a unit of bioenergetic investment made into somatic traits pays higher dividends earlier in ontogeny (especially prenatally) than investment in behavioral or extended phenotypic traits (cf. Murphy et al. 2006). In childhood, allocation of bioenergetic resources to growth of somatic tissue is high and investment in reproductive tissue is very low or zero; in adolescence, there is simultaneous allocation to growth of somatic and reproductive tissue; in adulthood, somatic growth is mainly allocated into reproductive tissue (Ellison 2017; Gonzalez-Forero et al. 2017).Footnote 2 Age at maturity (Fig. 2) marks a central life history transition because energy previously allocated to somatic growth is then allocated into reproduction and other functions (Ellison 2017; Kuzawa 2007).
Diversity of sexual trait expression as a function of individual development. Category-specific examples are given in the right-hand panel. The lowest triangle (gray) represents phenotypic traits (e.g. physical attractiveness, secondary sexual signals, intelligence, and extraversion). The second triangle (blue) represents behavioral traits that arise from phenotypic traits (e.g. artistic behaviors, logodaedaly, research, construction, and working behaviors). The third triangle (purple) represents extended phenotypic traits (e.g. artistic products, scientific outputs, online dating profiles, apartments, and pets). These examples are not exhaustive. The trait categories overlap vertically because each successive stage starts developing before the earlier stage ceases to develop. The horizontal overlap between the triangles is imperfect because not all phenotypic traits lead to behavioral traits. Likewise, not all behavioral traits lead to extended phenotypic traits. Each successive trait category occupies wider space on the x-axis because diversity of expression (quantity of sexual traits) grows disproportionately with each successive stage. In other words, extended phenotypic traits are hypothetically broader in diversity than behavioral traits, which are more diverse than phenotypic traits. Behavioral stasis marks a point when an individual acquires or displays no novel behavioral traits. Nevertheless, the individual’s existing behavioral repertoire can still produce a wider array of extended phenotypic traits. An author, for instance, need not change their behavioral traits (writing) in order to produce novelty in extended phenotypic trait expression (books, poems, articles)
Gradually, as the organism becomes more developed, investments made into behavioral traits start yielding higher benefits because the organism’s interactions with the environment grow in frequency and importance (e.g. Hulteen et al. 2018; Stodden et al. 2008). That is when it will start having a variety of effects on its environment (Fig. 2). Earlier in ontogeny, these effects are unlikely to bear great fitness rewards. Instead, childhood play is a mechanism by which an organism prepares for future life scenarios with higher fitness payoffs (Boyd 2009; Nielsen 2012). Many childhood experiences hone not only an individual’s behavioral skills (Gonzalez-Forero et al. 2017), but they also lay a basis for an individual’s future capacity to manipulate the environment in fitness-enhancing ways (cf. Nielsen 2012; Remigereau et al. 2016; Ward and Kolomyts 2010).
Brain development occurring later in phylogeny (Barton and Venditti 2014; Iriki and Taoka 2012; Navarrete et al. 2016; Putt et al. 2017; Winegard et al. 2018) and later in ontogeny (Casey et al. 2008; Dickerson et al. 2018; Geary 1995) is likely a prerequisite for the behavioral skills, prolonged attention, and self-control needed for increasing sophistication in extended phenotypic trait expression (cf. Beck et al. 2012; Bukowski & Rudnicki, 2018; Chappell et al. 2013; Jordan 2015; Remigereau et al. 2016; Vaesen 2012). To act as efficient signals/cues in sexual selection, manipulations of the external world need to be skilful and elaborate enough to communicate the quality of the underlying neural architecture and/or mechanical skills of the individual who produced the extended phenotypes.
The impulse control and delay of gratification required for sustained goal-directed behavior is still only developing in childhood and adolescence (Casey et al. 2008; Steinberg et al. 2009, 2017), meaning that adolescents are unlikely to produce or acquire as elaborate extended phenotypes as adults are. What is more, formal education typically lasts until about 18 years of age in modern Western societies. Only gradually after that do individuals start accumulating the monetary resources needed, say, to owning a house. This marks an important LH transition (comparable in several nonhuman animal species: e.g. Head et al. 2017; Schaedelin and Taborsky 2009) because in most modern societies, house ownership typically signifies that an individual possesses adequate financial independence to produce viable offspring. The developmental time and bioenergetic resources needed for the full expression of one’s extended phenotypic repertoire may take decades of skill acquisition and resource accumulation (cf. Fig. 2). For this reason, humans are more likely to start displaying full-fledged elaboration and variation in extended phenotypes in their late twenties and beyond. Inherited wealth or fame, however, may advantage some people over others already in adolescence (Essock-Vitale 1984; Mace 1998; Pepper and Nettle 2017a).
Population-Level Life History Variation in Extended Phenotypic Trait Expression: Integration with Behavioral Ecology
An ecological analysis of extended phenotypes at the level of populations is likely to produce cross-culturally interesting results. In the course of an individual’s development, genes produce different phenotypic, behavioral, and extended phenotypic outcomes depending on informational inputs from the local ecology (Bateson and Gluckman 2011; Blamires 2010; Cornwallis and Uller 2010; Lewis et al. 2017a; Schaller 2015). Any effects that an individual might have on the environment can partly influence those informational inputs by creating a feedback loop between organismal behavior and environmental effects. Thus, phenotypic and behavioral traits influence the environment, which in turn influences phenotypic and behavioral traits (cf. González et al. 2017; Saltz and Foley 2011; Scott-Phillips et al. 2014; Varnum and Grossmann 2017). Mechanisms that yield greater functional benefits within that ecology become more fully developed, while less immediately relevant mechanisms may be developmentally neglected (Cornwallis and Uller 2010; Ellis et al. 2017; Schaller 2015).
As humans colonized locations farther and farther away from the equator, technological innovations were increasingly needed to provide shelter from the cold climate and to ensure subsistence regardless of within-year variation in temperature (Ash and Gallup 2007; Creanza and Feldman 2016; Hoffecker and Hoffecker 2017b; Jordan 2015; MacDonald 2018; cf. Fristoe et al. 2017). Do increased viability selection pressures on extended phenotypes translate also to greater emphasis given to extended phenotypic traits in sexual selection in human populations in high latitudes?
It is possible that innovative propensity has co-evolved with certain life history strategies in lineages exposed to environments with higher within-year variation in temperature. This co-evolution has hypothetically taken place through correlated selection and/or shared genetic/physiological mechanisms (Ash and Gallup 2007; Putt et al. 2017; Sol et al. 2016). Innovative propensity is likely magnified in areas with high population density owing to greater competition for resources (Bailey and Geary 2009; Flinn et al. 2005; Muthukrishna and Henrich 2016; Pianka 1970; Sng et al. 2017) and/or other historical factors (Carneiro 1970; Maloney and Valencia Caicedo 2016; Murray 2003, p. 376). Life history traits tend to evolve together, forming meaningful functional composites that are designated with the fast–slow continuum (Cain and Ketterson 2012; Figueredo et al. 2004, 2005; Luoto et al. 2019). The life history strategies of different species and different individuals within species can be arranged on a continuum from “fast” (early maturation and reproduction, short lifespan, and high investment in offspring quantity) to “slow” (late maturation and reproduction, long lifespan, and high investment in offspring quality) (Luoto et al. 2019; Sng et al. 2018; Woodley of Menie et al. 2017). Different life history strategies create adaptive variation between individuals and populations (Krams et al. 2019; Luoto et al. 2019; Sng et al. 2018; Wright et al. 2018), which is why LH theory is directly relevant to the evaluation of hypotheses on extended phenotypic trait expression not only at the developmental level of individuals but also at the ecological and cross-cultural level of populations.
Population-Level Hypotheses on Extended Phenotypic Trait Expression
The fast–slow continuum of LH variation could predictably correlate with traits underlying innovations (Sol et al. 2016) so that individuals with “slow” life histories are more likely to produce innovations in the domain of extended phenotypes than individuals with “fast” life histories. Ecological selection pressures generate further differences in the need to collect, assess, retain, and use environmental information, which can impact selection pressures for organismal longevity, intelligence, and future orientation (i.e. a preference for larger rewards in later time horizons over smaller rewards closer to the present time) (Baumard 2018; Lu et al. 2017; Minkov and Bond 2015; Minkov et al. 2016; Pepper and Nettle 2017a; Sih and Del Giudice 2012; Sol et al. 2016; Templer and Arikawa 2006; Van de Vliert and Murray 2018). Fundamental tradeoffs in energy allocations are likely to orchestrate these differences (Baumard 2018; Ellison 2017; Krams et al. 2019).
According to the cognitive buffer hypothesis (Sol 2009), plasticity in behavioral and extended phenotypic responses to challenging environments might directly affect the evolution of life histories by enhancing adult survival (Sol et al. 2016). Comparative research in primates further suggests that periods of environmental change have stimulated innovations (reviewed in Fogarty et al. 2015), a finding replicated in humans (De Dreu and van Dijk 2018). On the other hand, people inhabiting areas with high population density often need to compete for resources (Pianka 1970) and therefore may need to invest more heavily in individual development, future rewards, and slow LH strategies instead of early reproduction (Sng et al. 2017; Wright et al. 2018). Indeed, population density predicts variation in a range of LH measures such as sociosexuality, future orientation, and life expectancy so that high population density is likely to lead to LH strategies at the slow end of the spectrum (Sng et al. 2017; Wright et al. 2018). Population density may have also contributed to the evolution of greater hominid cranial capacity via social competition (Bailey and Geary 2009; Flinn et al. 2005). As we will see, this individual-level evolutionary finding bears relevance to population-level extended phenotype hypotheses.
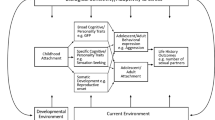
Leveraging these theoretical and empirical foundations from LH theory to testable hypotheses on extended phenotypic trait expression, it can be hypothesized that human populations inhabiting climates with greater within-year variation in temperature have (Hypothesis 1) higher future time orientation (Burke et al. 2017; Orosz et al. 2017; Van Lange et al. 2017) because of a recurring need to prepare for and survive cold seasons; (H2) higher innovative outputs because of adaptive responses to exogenous threats and selection pressures (De Dreu and van Dijk 2018; Fogarty 2018; Van de Vliert and Murray 2018); (H3a) greater and/or more diverse production of inanimate extended phenotypes (traded off against early reproduction) because they are essential for survival in such climatic conditions; and (H4) higher relative emphasis on inanimate extended phenotypic traits in sexual selection because of greater overall investment in cultural and technological innovations. (See Fig. 3 for a visualization of these hypotheses and Table 10 for a summary of existing evidence.) These effects are expected (H5) to become intensified in populations with higher population density because of greater competition for resources (Burger et al. 2017; Keeley 1988; Kline and Boyd 2010; Kokko and Rankin 2006; Munro et al. 2014; Sng et al. 2017; Tallavaara et al. 2018; Wright et al. 2018).
Together with genetic differences, species-typical plastic responses to environmental cues (including social cues from conspecifics) can lead to cross-cultural differences in human psychology. Model based on existing theory and empirical findings (Azam 2017; Bukowski & Rudnicki, 2018; De Dreu and van Dijk 2018; Fincher et al. 2008; Fincher and Thornhill 2012; Gurven 2018; Honegger and de Bivort 2018; Minkov and Bond 2015; Obschonka et al. 2018; Thornhill et al. 2010; Van de Vliert 2009; Van de Vliert and Murray 2018; Van Lange et al. 2017; Wei et al. 2017; Wright et al. 2018; figure adapted from Lewis et al. 2017a)
There is existing empirical evidence to support some of these hypotheses. It has been argued that an increase in patience and future orientation increases research in humans (Romer 1990) and innovation in nonhuman animals (Sol et al. 2016). The genetic factors underlying time orientation and LH strategy are increasingly well understood, with polymorphisms in an androgen receptor gene, dopamine receptor gene, and serotonin transporter gene being associated with life history strategies and time orientation (reviewed in Minkov and Bond 2015; see also Gettler et al. 2017; Luoto et al. 2019; Pine et al. 2010). Minkov and Bond (2015) provided evidence that a national index of these three genetic loci predicts future time orientation and life history strategy at the national level.
The data collected by Minkov and Bond (2015) and by Wang et al. (2016) suggest that people from nations with colder climates have a higher future time orientation and slower LH strategies, although that is a research question that neither study directly addressed. These data are concordant with a study on 31 countries, which found that warmer countries located near the equator had a more present-oriented “pace of life” than countries with colder climates (Levine and Norenzayan 1999). Other studies have reported preliminary evidence on self-control being positively correlated with distance from the equator, so that populations farther from the equator have higher levels of self-control than populations closer to the equator (Botchkavar et al. 2015; see also IJzerman et al. 2017; Van Lange et al. 2017).
Sternberg (2017) has suggested that it is intelligence that underlies climatic population-level variation in LH strategies, future time orientation, aggression, and self-control. Preliminary evidence lends some support to that argument: people from colder climates show higher intelligence than people from hotter climates (Kanazawa 2008; Lynn and Vanhanen 2002). As Sternberg (2017) suggested, extreme heat may in fact be a proximate mechanism that lowers intelligence as a state (rather than as a trait), which then precipitates to other cognitive mechanisms that underlie LH strategy, time orientation, self-control, and aggression (cf. Minkov and Bond 2015; Minkov et al. 2016). Recent empirical findings on the effects of heat on cognitive function provide some support for this hypothesis (Cedeño Laurent et al. 2018). Additional (though relatively circumstantial) evidence is provided by the association between temperature, crime events (Schinasi and Hamra 2017), and intimate partner violence (Sanz-Barbero et al. 2018) as well as between temperature and conflicts in Classic Maya (Carleton et al. 2017).
Future orientation, life history speed, and intelligence have an impact on a country’s level of innovation. Wang and colleagues (Wang et al. 2016) investigated whether a country’s innovation capacity (derived from the Global Competitive Report 2008–2009) can be predicted by the future time orientation of its citizens. Even after controlling for a country’s level of wealth, the population’s future time orientation still significantly predicted a country’s innovation factor (Wang et al. 2016). Bukowski and Rudnicki’s (2018) multiple regression analyses between eight independent variables showed that future orientation was the most powerful predictor of country-level differences in innovation (intelligence was not included in the analyses). A country’s innovation factor is, furthermore, strongly predicted by its absolute latitude (r = .61, N = 142 countries: Rindermann et al. 2015, using data from the Global Innovation Index, 2013, available in Dutta and Lanvin 2013).Footnote 3 Similar results have been reported for creativity, with cold temperatures promoting creativity especially in richer populations (Van de Vliert and Murray 2018). Using the Economic Complexity Index (ECI), Azam (2017) showed that intelligence is an important predictor (r = .76) of innovation at the level of nations. ECI ranks countries (Fig. 4) according to the level of diversification and complexity of their export baskets, thus reflecting the existing productive knowledge and industrial composition of a society (Azam 2017; Hartmann et al. 2017; Hausmann et al. 2014). Based on these findings, it is reasonable to hypothesize (H6) that intelligence mediates the influence of climate on innovation and economic complexity.
In addition to the evidence cited above, Hypothesis 2 (human populations inhabiting climates with greater within-year variation in temperature have higher innovative outputs) is also supported by findings on hunter-gatherer technological complexity. Latitude (positively) and effective temperature (negatively) predict the complexity of food-getting technology used by hunter-gatherer societies (Collard et al. 2005; Hoffecker and Hoffecker 2017a, 2017b; Torrence 1983, 2000). An analysis of hominid cranial capacity further suggests that variation in temperature has had a substantial effect on brain expansion (Bailey and Geary 2009), with hominid cranial volume growing as distance from the equator increases (Ash and Gallup 2007; Meisenberg and Woodley 2013).Footnote 4 These converging lines of evidence give preliminary empirical support to the hypotheses derived above on population-level extended phenotypic trait expression using LH theory and behavioral ecology.
Testing Population-Level Hypotheses with the Economic Complexity Index: Materials
The Economic Complexity Index (ECI) was selected as a dependent variable with which to test the above hypotheses. A holistic measure of the knowledge, industrial composition, and production characteristics of a country, ECI is used here as a population-level proxy for the expression of inanimate extended phenotypes. Data on ECI were collected for two time periods, 2016 and 1995, to control for effects of time. Country-level data on adolescent fertility rates (births per 1000 women ages 15–19) were used as one of the independent variables in the analyses to more explicitly study the interrelationships between economic complexity, sexual selection, and life history variability. Sources for all data are shown in Table 2. Because of high skew, data on adolescent fertility rates, population density, population size, distance from Brussels, and GDP per capita were natural logarithm transformed.
Results
Hypothesis 2 (that “human populations inhabiting climates with higher within-year variation in temperature have higher innovative outputs”) is supported by an analysis of a country’s Economic Complexity Index and distance from the equator. The farther a country is from the equator, the higher is its ECI (r = .68, p < .00001, N = 122 nations). Similarly, atmospheric cold demands is another significant predictor of ECI (r = .58, p < .00001, N = 122). The more severe the cold demands, the more diverse is a country’s industrial composition (Fig. 5). These data also support Hypothesis 3a, that “human populations inhabiting climates with higher within-year variation have greater and/or more diverse production of inanimate extended phenotypes (traded off against early reproduction)”.
Crucially, the life history tradeoff between early reproduction and bioenergetic investment in extended phenotypes becomes apparent when comparing global adolescent fertility rates with global variation in economic complexity (Fig. 6). The significant negative correlation between adolescent fertility and ECI (r = −.75, p < .00001, N = 122) suggests that there is a fundamental population-level LH tradeoff between the flow of energy into early reproduction and extended phenotypic trait expression.
Existing findings in hunter-gatherer societies (Keeley 1988) support Hypothesis 5: that population density increases economic complexity. A linear regression analysis between population density (loge) and ECI was performed to test whether population density increases economic complexity also in modern societies. The results show a small but significant correlation in the hypothesized direction (r = .27, p = 0.0026, N = 122 nations).
The finding that parasite stress predicts a country’s economic complexity (r = −.70, p < .00001, N = 122 nations; Table 3) is in line with general predictions arising from behavioral ecology (Gurven 2018). Countries with a higher parasite stress have a less diverse economic composition. One explanation for this finding is that high parasite stress ties bioenergetic and immunological resources, creating an immunological LH tradeoff at the level of individuals and populations (Krams et al. 2017a, 2017b, 2019; Said-Mohamed et al. 2018). Thus, populations with high parasite stress may be unable to invest equally high amounts of bioenergetic resources in innovation and technology as populations with lower parasite stress. Another explanation takes into account the high correlation between adolescent fertility and parasite stress (r = .77, p < .00001, N = 122 nations; Table 3), which indicates that ecological conditions with high pathogen prevalence favor fast LH strategies. One outcome of these selection pressures could thus be low investment in innovation and economic development.
Multiple Linear Regression Results, Control Analyses, and Discussion
Correlations between all variables are shown in Tables 3 and 4 (ECI values for 2016 and 1995). Multiple linear regression analyses were performed to analyze the relationships between these variables in detail. They revealed redundancy between predictor variables. Distance from the equator and atmospheric cold demands were highly correlated (r = .88) and showed multicollinearity (VIF = 3.78). This was also the case with parasite stress and cold demands (r = −.67, VIF = 4.71). Thus, the multiple regression model with the best fit excluded parasite stress and distance from the equator and included four variables (R2 = .72, F(4,117) = 76.82, p < .0001), which all significantly and independently predicted ECI. These remaining four variables were intelligence, adolescent fertility rate (loge), population density (loge), and cold demands. This four-factor model accounted for 72% of the variation in global economic complexity in 2016 (Table 5).
To eliminate possible confounds, a further analysis was conducted controlling for other factors. Since the global economic centre has roughly hovered around Europe during the past century (Dobbs et al. 2012, p. 17), it could be that trade distance from Europe has driven global country-level differences in ECI so that countries farther away from Europe are less able to develop their economic complexity. Thus, the distance between each of the countries’ capital and Brussels was measured using the European Commission’s (2018) distance calculator.Footnote 5 It is also possible that population size confounds the results reported above (cf. Fogarty and Creanza 2017), which is why population size was added as a second control variable. GDP per capita was added as a third control variable to control for the influence of wealth on economic complexity (cf. Baumard 2018; Murray 2003, pp. 349–351). As values for all three of these control variables were highly skewed, they were log-transformed prior to fitting them in the model.
After introducing these three control variables, the results were still highly significant for all four predictor variables except for adolescent fertility rate. The high correlation between adolescent fertility rate and per capita GDP (r = −.74, p < .00001) rendered adolescent fertility non-significant when the three control variables were introduced. No major multicollinearity was detected between adolescent fertility rate and the other six variables (all VIFs <3.6). The final model with four predictor variables and three control variables is shown in Table 7.
An additional possibility is that these results may have arisen as a chance occurrence for economic complexity in the year 2016 alone. Thus, additional analyses were performed with ECI values from 1995. Data on predictor variables and control variables were also collected from 1995 to analyze correlations between the variables for the appropriate time period. These control analyses largely confirmed the findings reported above on ECI in 2016. A stepwise multiple linear regression analysis showed that the same four variables that predicted 72% of the variation in global economic complexity in 2016 predicted 64% of the variation in 1995 (Table 6). The correlation matrix in Table 4 shows significant correlations between the variables in 1995, further indicating that the findings for 2016 did not arise by chance. Finally, a multiple regression analysis on the same variables as for 2016 showed that the effects of population density and cold demands on ECI remained significant after the three control variables were introduced (Table 8). However, the effects of intelligence and adolescent fertility were no longer significant, indicating that their influence on ECI was significantly mediated by the control variables in 1995 (Table 8). No major multicollinearity was detected between adolescent fertility rate, intelligence, and the other five variables (all VIFs <3.4).
Since the data on cold demands were extracted from a relatively long time period of 1960–1990 (Van de Vliert, personal communication), they may not adequately represent climatic conditions for the study years of 1995 and 2016. Therefore, to check the validity of cold demands (Van de Vliert 2013) as a measure of climate that reliably predicts ECI, additional climate data were collected from Climate Change Knowledge Portal (2018). A variable coldest month was calculated for each country as the average of the mean temperature (in Celsius degrees) of the coldest month for each year 1993–1995, which were then averaged across the 3 years. Linear regression analyses between coldest month (1993–1995) and cold demands (1960–1990) showed a high correlation (r = −.87). Further bivariate correlational analyses between the new variable coldest month, ECI 1995 (rs = −.68, p < .00001), and ECI 2016 (rs = −.59, p < .00001) showed equally good predictive power as the variable cold demands.Footnote 6 The latter was used in the overall analyses because it is based on data from 1960 to 1990, thus reflecting a longer period that will have, in part, influenced the complex causal process between climate and ECI.
The mediating effect of intelligence on the relationship between atmospheric cold demands and ECI was explored to test Hypothesis 6 (Table 10). Intelligence was not entered as a control variable in the multiple regression analyses because atmospheric cold demands are hypothesized to be among the main driving forces behind increases in intelligence (Kanazawa 2008; Templer and Arikawa 2006), innovation (De Dreu and van Dijk 2018; Van de Vliert and Murray 2018), GDP per capita (Rindermann and Becker 2018), and thus behind the development of economic complexity. A mediation analysis with 5,000 bootstrap samples was conducted in SPSS version 25 using PROCESS macro 3.2. The results showed significant correlations between cold demands and intelligence as well as between intelligence and ECI (Fig. 7). The bivariate correlation between cold demands and intelligence was exactly the same (r = .66; Table 3) as reported by Templer and Arikawa (2006) between mean low temperature in winter and intelligence. The residual predictive power of cold demands on economic complexity (c’ path) was not significant when the influence of intelligence was factored into the model (Fig. 7 and Table 9). The change from the direct c path (b = .023, t(120) = 7.72, p < .001) to the mediated c’ path (b = .003, t(119) = 1.15, p = .25) was significant (Δb = .020, BootSE = .003, 95% BootCIs [.015, .025], Sobel’s z = 6.87, p < .01). The percentage of variance accounted for (%VAF) between c path and c’ path by introducing intelligence as a mediator was 85.22% (VAF = indirect effect / total effect * 100 = .0196 / .023 * 100 = 85.22). With VAF > 80% and c’ path being non-significant, these results show that the effect of cold demands on economic complexity is fully mediated by intelligence. This finding supports the hypothesis that cold ecological conditions select for certain phenotypic traits (higher intelligence) which in various ways enable organisms to produce cultural innovations (here: ECI) that improve their fitness.
To control for non-independence of data (e.g. Pollet et al. 2014), correlations were analyzed by dividing the global data into six world regions. The predictive power of intelligence, adolescent fertility, population density, and cold demands on ECI generally held within these six world regions: 22/24 correlations were in the expected direction. Population density in North America (r = −.62, p = .14, n = 7 nations) and in Africa (r = 0, n = 29 nations) were two exceptions. Though reduced sample sizes meant that p-values were less significant than in the global analyses, nine out of 24 within-region correlations were statistically significant (Figs. 8, 9, 10 and 11).
Correlation between intelligence and ECI value in six world regions. North America (r = .73, p = .06, n = 7 nations); South America (r = .31, p = .26, n = 15 nations); West Eurasia (r = .85, p < .00001, n = 49 nations)***; Africa (r = .48, p < .009, n = 29 nations)**; East Eurasia (r = .69, p = .003, n = 16 nations)**; Insular Pacific (r = .36, p = .48, n = 6 nations). * p < .05. ** p < .01. *** p < .001
Correlation between adolescent fertility (loge) and ECI value in six world regions. North America (r = −.59, p = .16, n = 7 nations); South America (r = −.52, p = .047, n = 15 nations)*; West Eurasia (r = −.63, p < .00001, n = 49 nations)***; Africa (r = −.50, p < .006, n = 29 nations)**; East Eurasia (r = −.78, p < .0004, n = 16 nations)***; Insular Pacific (r = −.48, p = .34, n = 6 nations). * p < .05. ** p < .01. *** p < .001
Correlation between population density (loge) and ECI value in six world regions. North America (r = −.62, p = .14, n = 7 nations); South America (r = .24, p = .39, n = 15 nations); West Eurasia (r = .22, p = .13, n = 49 nations); Africa (r = 0, n = 29 nations); East Eurasia (r = .28, p = .29, n = 16 nations); Insular Pacific (r = .38, p = .46, n = 6 nations). * p < .05. ** p < .01. *** p < .001
Correlation between atmospheric cold demands and ECI value in six world regions. North America (r = .72, p = .07, n = 7 nations); South America (r = .05, p = .86, n = 15 nations); West Eurasia (r = .53, p < .0001, n = 49 nations)***; Africa (r = .61, p < .0005, n = 29 nations)***; East Eurasia (r = .04, p = .88, n = 16 nations); Insular Pacific (r = .14, p = .79, n = 6 nations). * p < .05. ** p < .01. *** p < .001
These analyses do not address all possible confounding factors. They do, however, further strengthen the hypothesis that cold temperatures and slow LH strategies—driven here by low adolescent fertility, high intelligence, and high population density (cf. Meisenberg and Woodley 2013; Minkov et al. 2016; Sng et al. 2017)—are important predictors of population-level innovative output and extended phenotypic trait expression as indexed by ECI.
The next section extends the current theoretical synthesis to analysing sex differences in sexual selection, with a particular focus on extended phenotypic traits. This is an important conceptual extension to the current theoretical synthesis because of the wealth of research conducted on sex differences in mate choice.
Sex Differences in the Importance of Extended Phenotypic Traits
Extended phenotypes are expected to be better indicators of male rather than female genetic quality and/or parental ability (Head et al. 2017) for logical biological reasons: females tend to make higher somatic investments in reproduction than males (Clutton-Brock 2007, 2017; Jasienska et al. 2017), which is typically (though not always) reciprocated by resource provisioning inputs from males (Bribiescas et al. 2012; Stanyon and Bigoni 2014; Wang et al. 2018). Just as ancestral women who displayed attractive secondary sexual signals (e.g. breasts, gluteofemoral fat deposits) were favored by sexual selection because they gained direct male-delivered material benefits (Thornhill and Gangestad 2008), male-delivered material benefits had a signalling value in and of themselves due to their value for offspring development.
Features associated with high mate value in men differ from those associated with high mate value in women (Conroy-Beam et al. 2015; Sugiyama 2015). One of the most robust findings in sexual selection research pertains to sex differences in mate preferences, as originally reported by Buss (1989) and replicated in countless studies. Although men and women agree on the importance of several traits (a fact that tends to be sometimes forgotten when research findings on sexual selection are oversimplified), men place consistently more value on physical attractiveness than women do, while women value wealth, resources, and status more than men (Buss 1989; Hughes and Aung 2017; Oda 2001; Schwarz and Hassebrauck 2012; Shackelford et al. 2005; Souza et al. 2016; Wang et al. 2018). This results in significant sexual dimorphism in human mate preferences, yielding a multivariate effect size (Mahalanobis D) of 2.41 (Conroy-Beam et al. 2015).
Sex differences in mate preferences have been replicated in studies in which subjects rate the attractiveness of potential mates when presented with an extended phenotypic cue. As discussed above, males seated in a luxury car were rated as significantly more attractive by women than men seated in a standard car, yet the luxury car manipulation had no effect on how attractive men rated women to be (Dunn and Searle 2010). This finding has been replicated by Sundie and colleagues (Sundie et al. 2011) using a verbal description, rather than a photograph, of a potential mate and their car. Therefore, despite noticeable increases in female ownership of luxury cars (Dunn and Searle 2010), the signalling function that cars provide in sexual selection is negligible when displayed by women. Likewise, men are rated more attractive when presented in a luxury apartment, but women’s attractiveness ratings remain unchanged regardless of residential context (Dunn and Hill 2014).
In another line of research, a correlational study found that men’s artistic success with visual art was a significant predictor of the number of their mating partners—a pattern not found in female artists (Clegg et al. 2011). Similar results have been recorded with literary art and other creative products; the ‘art as sexual display’ hypothesis is supported by the finding that men are overwhelmingly in the majority when it comes to the quantity of creative products produced for public consumption (Kanazawa 2000; Lange and Euler 2014; Miller 1999). Men also report being more motivated to write books than women (Lange 2011) whilst women are more avid readers of fiction than men and consume more high-brow culture in general than men (reviewed in Christin 2012). These findings support the hypothesis of male supply and female evaluation of creative products that arises from sexual selection theory (Crocchiola 2014; Gao et al. 2017b; Lange and Euler 2014; see also Winegard et al. 2018 for an alternative hypothesis: the status competition model).
Animate Extended Phenotypes
Interestingly, heterospecifics can also constitute extended phenotypic cues. Pet ownership, for instance, increases a male’s attractiveness to a woman (Tifferet et al. 2013). In a simple but clever experimental set-up, a highly attractive male confederate set about soliciting phone numbers from young women on the street (Guéguen and Ciccotti 2008). In the experimental condition, the confederate had a dog with him (kept on a lead) when he was asking for women’s phone numbers, whereas in the control condition the dog was absent. In the control condition, 9.2% of the women complied with the confederate’s request to receive the woman’s phone number; when the dog was present, 28.3% of the women provided their phone numbers (Guéguen and Ciccotti 2008). The dog therefore may have acted as an extended phenotypic cue of the man’s underlying qualities, leading to a significantly more positive courtship outcome because of the woman’s inferences of what dog ownership communicates about the man. Alternatively, the dog could have become a direct motivational incentive (Toates 2009) to the women, arousing their attention irrespective of any inferences that the dog’s presence can elicit about the man’s qualities.
Pet ownership provides another example of how men and women differ in attentional bias between phenotypic and extended phenotypic traits. Significantly more men (21.6% of 476 pet owners) than women (6.3% of 727 pet owners) confessed to having used a pet to attract a mate (Gray et al. 2015). Gray and colleagues (2015) also found that significantly more women than men reported greater opposite-sex attraction caused by pet ownership of a potential partner. This effect was particularly noticeable for women in their 20s. Older women were less likely to report being attracted to men’s pet ownership. Men who reported increased attraction caused by women’s pet ownership were significantly outnumbered in all age categories by men who did not report greater attraction caused by women’s pet ownership (Gray et al. 2015). Notably, all the subjects studied by Gray et al. (2015) were pet owners themselves. The irrelevance of women’s pet ownership to men might be even more pronounced in the population at large, given the general male emphasis on reproductive rather than parenting effort (Bribiescas et al. 2012), and assuming that pet-keeping is an alternative to, or a distraction from, parenting effort (Archer 2011b).
In the aggregate, these findings highlight the centrality of somatic traits when men evaluate women’s mate value and the relative importance of extended phenotypic traits in women’s assessments of men’s mate value (Fig. 12).
Typical trajectories of sexual trait development and preferences in men and women partitioned into phenotypic, behavioral, and extended phenotypic traits. This model provides a schematic approximation (based on e.g. Dunn and Hill 2014; Dunn and Searle 2010; Gregor 1985; Hughes and Aung 2017; Israel and Strassberg 2009; Jones et al. 1995; Madison et al. 2018; Marcinkowska et al. 2017; Motta-Mena and Puts 2017; Munro et al. 2014; Schwarz and Hassebrauck 2012; Thornhill and Gangestad 2008; Wells 2007). Development of extended phenotypic traits (bottom panel) lags behind female preferences for those traits (top panel), which may partially contribute to women’s well-documented preference for older men (Antfolk et al. 2015; Schwarz and Hassebrauck 2012). Individual variation within sexes in trait development and preferences may, however, result in considerable individual-level deviations from these patterns (Widemo and Sæther 1999). Trait preferences also fluctuate across the ovulatory cycle and/or according to long-term or short-term relationship contexts (e.g. Confer et al. 2010; Havlíček et al. 2015; Motta-Mena and Puts 2017). Cross-cultural variation is largely absent from the model as it is mainly based on research conducted in Western, educated, industrialized, rich and democratic (WEIRD) societies
What Causes Sex Differences in Preferences for Extended Phenotypic Traits?
Physical attractiveness is the most honest signal of women’s reproductive value (Andrews et al. 2017; Thornhill and Gangestad 2008). Just as men have evolved to fixate on women’s physical attractiveness (Maner et al. 2007), women have evolved a preference to assess a male’s reproductive value not only through phenotypic and behavioral traits but also by the extended phenotypic traits that men exhibit (Austen 2007/1813; Dunn and Hill 2014; Guéguen and Lamy 2012; Hughes and Aung 2017; Sundie et al. 2011; Fig. 12). Any bioenergetic and material investments made in existing offspring compromise men’s ability to invest in extended phenotypes, such as luxury apartments, vehicles, or art. This underlines a tangible LH tradeoff between mating effort and parental effort (Figueredo et al. 2006).
One of the most important constituents of mate value is residual reproductive value—an individual’s future reproductive potential or total expected reproductive success from the present time forward (Fisher 1930; Williams 1966). Just as the phenotypic ornaments that function to display women’s quality also reflect their age-based and parity-based residual reproductive value (Thornhill and Gangestad 2008), for males, existing resources and/or a capacity to acquire them are major constituents of reproductive value. Between-sex interaction in sex-specific markers of reproductive value is exemplified by the finding that the greater the physical attractiveness a woman had, the greater financial and occupational status she demanded from potential mates. Similarly, the more resources men had, the greater were their demands for women’s physical attractiveness (Bereczkei et al. 1997). A small-scale study in American, Chinese, and European populations found that annual income had between 4.3 times (Chinese) to 7.5 times (American) higher impact on men’s attractiveness to women than vice versa (Wang et al. 2018). These findings highlight the different contributions that phenotypic traits and extended phenotypic traits have on reproductive value in men and women (yet see Buston and Emlen 2003 for a more refined set of findings supporting assortative mating).
Reproductive value signalled by extended phenotypic traits can have fitness consequences either materially, bioenergetically, or genetically. A man who occupies desirable territory will provide a propitious developmental niche for offspring (cf. Krams et al. 2019; Stotz 2017). This has both direct benefits and indirect benefits to offspring, if one makes the assumption that the male’s capacity to win over that territory has a heritable component. In modern humans, the existence of this heritable component is suggested by studies reporting that genetic differences account for variation in income, educational attainment, and intelligence (Hill et al. 2016, 2018a, 2018b; Lee et al. 2018; Minkov et al. 2016; Okbay et al. 2016; Sniekers et al. 2017). Women’s sexual ornaments and extended sexuality (i.e. the desire to mate despite fertility being at or near zero e.g. outside the fertile phase of the menstrual cycle or during pregnancy) evolved partly because they enhanced acquisition of male-delivered material benefits and services (Thornhill and Gangestad 2008; see also Cloud and Taylor 2018). Much like the beaver that constructs a dam to have an enlarged catchment area to harvest energy from the environment (Dawkins 1982), women select males not only to provide good genes for their offspring but also to act as “an enlarged catchment aid” that harvests energy from the environment to the benefit of the woman’s offspring. In some cases, however, it might be in the man’s benefit to invest those bioenergetic resources in the production of new offspring.
It is in this sense that males who provide material benefits to women and their offspring act as functional extended phenotypes that boost the fitness of those other genotypes. In a large German sample, most men (75.3%) could imagine marrying someone who does not have regular employment, while relatively few women (28.0%) were interested in such a partner (Schwarz and Hassebrauck 2012). In the Tsimane of Bolivia, successful male hunters have more attractive wives, higher levels of fertility, and are rated as more influential than those who are unsuccessful (Gurven et al. 2009; cf. similar results in an Amazonian society: Escasa et al. 2010). Interestingly, following predictions from assortative mating in Western societies (Buston and Emlen 2003; Robinson et al. 2017), productive Tsimane men are paired with productive women, with these pairs having higher in-pair fertility and their children showing higher survivorship than children in other families (Gurven et al. 2009). The view that a male provides the sole functional extended phenotype for the offspring, however, is misleading in a number of ways (e.g. Bribiescas et al. 2012). Both parents typically act as functional extended phenotypes for offspring in humans (the obvious exception being single parenthood). What is important to note is the diverging fitness landscapes of each individual—those of the father, those of the mother, those of each offspring—and how each individual manipulates one another in an attempt to maximize one’s own fitness (Liu et al. 2017; Said-Mohamed et al. 2018; Schlomer et al. 2011).
Theoretical Considerations
Niche Construction
Because applying the extended phenotype concept in research on human sexual selection has not received substantive attention, it is necessary to predict some of the most obvious criticisms that may be levelled against it. Since the publication of The Extended Phenotype in 1982, a theory significantly overlapping with Dawkins’s theory has been proposed under the name of niche construction (Odling-Smee et al. 2003). Dawkins (2004) has subsequently criticized the emergence of niche construction, concluding that it “is a phrase that should be abandoned forthwith”, though others have pointed out that the niche construction approach is not in conflict with the extended phenotype approach (Hunter 2009). Recent critical discussions of niche construction and the extended phenotype theory (Hunter 2009; Scott-Phillips et al. 2014; Wells 2015) surpass the scope of the present article.
It is more logical from the point of view of sexual selection research to use the extended phenotype theory rather than niche construction. This is because the central focus of the extended phenotype theory is on particular end-products of an individual’s manipulative and construction behaviors, whilst niche construction is more concerned with macroevolutionary processes (see e.g. Laland and O’Brien 2011; Scott-Phillips et al. 2014). Furthermore, niche construction has given substantially more attention to viability selection than sexual selection (Odling-Smee et al. 2003). A recent review on niche construction, for example, makes only a passing reference to sexual selection (Laland et al. 2017), while research on sexual selection (in nonhuman animals) frequently utilizes the extended phenotype approach (Head et al. 2017; Jordan et al. 2016; Rubalcaba et al. 2016; Schaedelin and Taborsky 2009).
Initial Hypotheses and Future Research
I have found that the viewpoint represented by the label ‘extended phenotype’ has made me see animals and their behaviour differently, and I think I understand them better for it. The extended phenotype may not constitute a testable hypothesis in itself, but it so far changes the way we see animals and plants that it may cause us to think of testable hypotheses that we would otherwise never have dreamed of. (Dawkins 1982, p. 2).
Evolutionary explanations are sometimes criticized for not generating testable predictions and hypotheses (Boutwell et al. 2015). Owing to the scope of the present theoretical framework (cf. Dawkins 1982, pp. 1–4), there is a range of hypotheses and predictions that can be derived from it. Some of these (H1–H6) have already been discussed and tested above. Others are summarized in Table 10, though the list is by no means exhaustive.
Limitations
The present review has sketched a broad argument that unites several theoretical approaches, coupled with an analysis of cross-cultural biodemographic, ecological, and economic data. Within the scope of this article, it is impossible to provide a detailed discussion of all hypothesized covariates and proximate mechanisms underlying these large-scale patterns of human behavioral ecology. Thus, the current article is not exempt from the axiomatic tradeoff between breadth of appeal and specificity of prediction (Bailey 2012; Pepper and Nettle 2017b) despite the concentrated attempt to spell out specific hypotheses in the previous sections. Nor is it always unproblematic to extrapolate individual-level hypotheses to population-level data: thus, caution is recommended when interpreting population-level findings without complementary longitudinal or individual-level studies (Minkov et al. 2016; Pollet et al. 2014).
The current findings on economic complexity describe global trends: the strong negative correlation (Figs. 6 and 9) between ECI and adolescent fertility, for example, indicates a substantial tradeoff between economic complexity and reproductive timing that is of significant interest to life history theorists. Nevertheless, despite the inclusion of adolescent fertility as an independent variable, the relationship between economic complexity and sexual rather than viability selection remains to be explored in greater detail in future research (cf. Hypothesis 4). It is also noteworthy that ECI measures economic activity rather than cultural activity, which has been studied elsewhere using other measures (Murray 2003; Van de Vliert and Murray 2018; Winegard et al. 2018). Though the ECI data span 122 countries across the globe, the examples of extended phenotypes reviewed in the “Inanimate Extended Phenotypes” section are mainly from modern Western cultures: it would thus be valuable to explore the role of extended phenotypic traits in non-Western and traditional cultures. The present framework is therefore intended to encourage inputs from a range of scholars to further test its utility.
Discussion
This article has fostered theory unification by synthesizing the four approaches named in the title: extended phenotype theory, life history theory, behavioral ecology, and human sexual selection. What the framework sets out to do is to initiate a systematizing of bodies of research that could otherwise seem disjointed, but are in fact unified by operationalizing evolutionary theory in the interactions that organisms have with their environments. The formalization of extended phenotypic traits as a component in human sexual selection is the missing link in a larger conceptualization which divides sexual traits into three categories: phenotypic, behavioral, and extended phenotypic traits. Extended phenotypic traits include not only ostentatious displays of wealth (Dunn and Hill 2014; Miller 2010; Sundie et al. 2011), but a myriad of other artefacts and social cues that can convey the cultural competencies, neurocognitive qualities, or provisioning abilities of their producer or displayer (Gao et al. 2017a, 2017b; Hughes and Aung 2017; Lange and Euler 2014; Luoto and Spriggs 2018; Miller 2001; Miller and Todd 1998; Miller 2000; Rosenfeld and Thomas 2012; Winegard et al. 2018).
The conceptual overlap between the extended phenotypes produced and/or manipulated by humans and nonhuman animals is substantial (Table 1). This fundamental realization can set in motion research programmes on the evolutionary continuity and/or divergences between extended phenotypic traits in humans and nonhuman animals. Another fundamental realization—that the signalling value of many human-made extended phenotypes is an exaptation (Gould and Vrba 1982) of their original functional value, as in nonhuman animals (Schaedelin and Taborsky 2009)—can further augment the depth and impact of research on extended phenotypes and sexual selection. A third central point is a consideration of the genetic basis that creates individual differences in extended phenotypic trait expression, a point that is too often neglected in research on non-bodily traits in human sexual selection.
Human life histories comprise behaviors that transfer energy from the environment into phenotypes and further into extended phenotypes. A life history perspective recognizes that extended phenotypic traits can constitute core LH traits: they impose substantial bioenergetic costs but have profound fitness implications for humans. Bioenergetic resources invested in extended phenotypes are traded off against other LH traits. As shown in Figs. 6 and 9, for example, adolescent fertility negatively correlates with economic complexity. This suggests that there is a population-level LH tradeoff between early reproduction and investment in extended phenotypes.
Adopting a life history perspective to analysing sexual traits does not ipso facto mean that viability selection and sexual selection need to be treated as antagonistic processes (Fig. 1). Granted, sometimes there are grounds to do so (Jordan et al. 2016), but sometimes a unit of investment into functional extended phenotypes can also pay dividends as extended phenotypic cues that can influence sexual selection (e.g. Rubalcaba et al. 2016). That way, extended phenotypic cues can demonstrate the synergistic relationship between viability selection and sexual selection (Rubalcaba et al. 2016; Schaedelin and Taborsky 2009)—as several phenotypic signals do (Lassek and Gaulin 2008; Sell et al. 2017; Thornhill and Gangestad 2008)—while occasionally drawing attention to tradeoffs between viability and sexual selection.
LH theory further predicts that there are different fitness benefits that organisms are likely to receive for any unit of bioenergetic investment made into sexual traits in each of the three categories at any given point in organismal development (Fig. 12; cf. Ellison 2017). Traits in these categories have a hierarchical onset of development (Fig. 2). It is important to note that not only the biochemical constitution of the phenotype per se is the target of selection. Viability selection and sexual selection also exert selection pressures (Fig. 1) on the way in which organisms channel energy flows from phenotypes into extended phenotypes (e.g. Blamires et al. 2018; González et al. 2017; Luhring et al. 2017; Meunier et al. 2017). A possible evolutionary outcome of these selection pressures may be the high correlation between atmospheric cold demands and economic complexity (Fig. 5), which suggests that greater rates of innovation and reliance on extended phenotypic traits are required for human thriving in ecological conditions that impose greater cold demands on survival (cf. De Dreu and van Dijk 2018). These ecological conditions may also select for slower LH strategies, as suggested by the strong negative correlation between cold demands and adolescent fertility (r = −.51 in 1995 and r = −.56 in 2016: Tables 3 and 4).
That sex-specific mate preferences also follow the predictions that arise from the current theoretical framework constitutes important psychobehavioral evidence for the utility of the framework. These biodemographic and psychobehavioral findings reflect the basic tenet of life history evolution: that the essence of life can be defined as variant forms competing to harvest energy from the environment in the service of reproduction (Del Giudice et al. 2015; Ellison 2017; Snell-Rood et al. 2015). The production and manipulation of extended phenotypes can be a proximate mechanism underlying that ultimate function (Fig. 1), which provides an additional rationale for synthesizing extended phenotype theory with life history theory.
A high quality phenotype runs the risk of being evolutionarily futile if its quality is not effectively signalled to the opposite sex. Extended phenotypic traits amplify the scope of organismal signalling vastly beyond the limitations of phenotypic and behavioral traits by utilizing many aspects of the innovative capacity that biological evolution has endowed humans with (Lane 2016; Schaedelin and Taborsky 2009). During the past decades, sexual traits that are independent of the body have risen in importance due to increasing globalization and the advent of the Internet (Oishi 2010; Rosenfeld and Thomas 2012; Sumter et al. 2017; Timmermans and De Caluwé 2017), providing magnified opportunities and payoffs for extended phenotypic signalling. Insight into the evolutionary dynamics between each of the trait categories—phenotypic, behavioral, extended phenotypic—is crucial for enhancing our understanding of the complexity of human sexual selection at the level of individuals, cultures, and populations (cf. Wilson et al. 2017). This is a particularly salient research question in the rapidly changing modern world in which technological innovations and globalization (Blake et al. 2018; Lane 2016; Oishi 2010; Rosenfeld and Thomas 2012) revolutionize the ways in which people harvest energy from the environment, interact with one another, and fall—or don’t fall—in love.
Conclusion
Evolution causes biological diversity through adaptation to environmental conditions. Human cultural evolution has enabled the near-global spread of our species, unprecedented population growth, and an order-of-magnitude increase in cultural innovations. All of these outcomes shape the way in which viability selection and sexual selection operate in modern humans.
Yet not all populations are equally innovative, not all economies equally complex. With a dataset comprising 122 nations, I explored the ways in which ecological and demographic variables predict global variation in economic complexity. The results of these analyses are in line with predictions arising from evolutionary theory: economic complexity is higher in countries with colder winters, an effect significantly mediated by country-level differences in intelligence (Tables 5 and 6; Fig. 7). Because of a significant heritable component in intelligence and creativity (de Manzano and Ullén 2018; Hill et al. 2016, 2018a, 2018b; Lee et al. 2018; Minkov et al. 2016; Okbay et al. 2016; Sniekers et al. 2017), this result suggests the existence of genetic population-level underpinnings in extended phenotypic trait expression as originally postulated at the level of individuals by Dawkins (1982; cf. Hoover et al., 2011). Economic complexity is also significantly constrained by population-level adolescent fertility rates (Fig. 6), showing the expected life history tradeoff between early reproduction, innovation, and economic development. These results shed light on the ways in which evolutionary processes shape human adaptation to local environments. The results further suggest that these adaptive processes occur both at the level of psychological traits (intelligence, innovative capacity) and de facto behaviors, indexed by global variation in reproductive timing, innovation, and economic complexity.
Echoing the influential work of Gould and Vrba (1982), whether this is a trivial article on terminology or a deeply meaningful statement about evolution and human development ultimately hinges upon the importance of extended phenotypes to human behavior and sexual selection. Adopting the synergistic viewpoint offered by behavioral ecology, life history theory, sexual selection, and extended phenotype theory is beneficial only to the extent that a multilevel, evolutionary analysis contributes to an improved understanding of human behavior (cf. Luoto et al. 2019; Rantala et al. 2018). I argue that it does, since the present theory unification offers an explicitly articulated synthetic approach to organize research on sexual selection, life history theory, behavioral ecology, and cultural evolution. The absence of this synthesis has restricted the range and theoretical cohesion of existing evolutionary research in hitherto unrecognized ways.
Notes
Inherited wealth may be an exception. In a limited number of cases, the causal succession from a phenotype to behavior to extended phenotypes can potentially skip the intermediate step behavior.
Equatorial distance is a proxy of climate: it correlates significantly (r = .90) with daily minimum temperature (IJzerman et al. 2017) and cold demands (r = .88, as described below).
See Roth and Pravosudov (2009), Vincze (2016), and especially Fristoe et al. (2017), for similar results in bird species, as well as a discussion on direction of causality, which suggests that, in birds, it was brain expansion that led to the colonization of variable habitats rather than colonization of variable habitats leading to brain expansion.
Brussels was chosen as a proxy for Europe’s central point for the present purposes since it is located between Europe’s geographical centre (Suchowola, Poland) and financial centre (London, UK) and constitutes the administrative centre of the European Union.
Spearman’s rank correlation coefficient was used because of non-normal distribution of data for the variable coldest month.
References
Andrews, T. M., Lukaszewski, A. W., Simmons, Z. L., & Bleske-Rechek, A. (2017). Cue-based estimates of reproductive value explain women's body attractiveness. Evolution and Human Behavior, 38(4), 461–467. https://doi.org/10.1016/j.evolhumbehav.2017.04.002.
Antfolk, J., Salo, B., Alanko, K., Bergen, E., Corander, J., Sandnabba, N. K., & Santtila, P. (2015). Women's and men's sexual preferences and activities with respect to the partner's age: Evidence for female choice. Evolution and Human Behavior, 36(1), 73–79. https://doi.org/10.1016/j.evolhumbehav.2014.09.003.
Archer, I. W. (2011a). Economy. In A. F. Kinney (Ed.), The Oxford Handbook of Shakespeare (pp. 165–181). Oxford: Oxford University Press. https://doi.org/10.1093/oxfordhb/9780199566105.013.0010.
Archer, J. (2011b). Pet keeping: A case study in maladaptive behavior. In C. Salmon & T. K. Shackelford (Eds.), The Oxford handbook of evolutionary family psychology (pp. 281–296). Oxford: Oxford University Press.
Ardiel, E. L., & Rankin, C. H. (2010). An elegant mind: Learning and memory in Caenorhabditis elegans. Learning & Memory, 17(4), 191–201. https://doi.org/10.1101/lm.960510.
Arslan, R. C., Schilling, K. M., Gerlach, T. M., & Penke, L. (2018). Using 26,000 diary entries to show ovulatory changes in sexual desire and behavior. Journal of Personality and Social Psychology. https://doi.org/10.1037/pspp0000208.
Ash, J., & Gallup, G. G. (2007). Paleoclimatic variation and brain expansion during human evolution. Human Nature, 18(2), 109–124.
Austen, J. (2007/1813). Pride and Prejudice. In Ebrary, Chandni Chowk, Delhi: Global Media.
Azam, S. (2017). A cross-country empirical test of cognitive abilities and innovation nexus. International Journal of Educational Development, 53, 128–136.
Bailey, N. W. (2012). Evolutionary models of extended phenotypes. Trends in Ecology & Evolution, 27(10), 561–569.
Bailey, D. H., & Geary, D. C. (2009). Hominid brain evolution. Human Nature, 20(1), 67–79. https://doi.org/10.1007/s12110-008-9054-0.
Ball, H. C., Londraville, R. L., Prokop, J. W., George, J. C., Suydam, R. S., Vinyard, C., Thewissen, J. G. M., & Duff, R. J. (2017). Beyond thermoregulation: Metabolic function of cetacean blubber in migrating bowhead and beluga whales. Journal of Comparative Physiology B, 187(1), 235–252. https://doi.org/10.1007/s0036.
Barbot, B., Tan, M., & Grigorenko, E. L. (2013). The genetics of creativity: The generative and receptive sides of the creativity equation. In O. Vartanian, A. S. Bristol, & J. C. Kaufman (Eds.), The neuroscience of creativity (pp. 71–93). Cambridge: MIT Press.
Barelds, D. P., & Barelds-Dijkstra, P. (2007). Love at first sight or friends first? Ties among partner personality trait similarity, relationship onset, relationship quality, and love. Journal of Social and Personal Relationships, 24(4), 479–496.
Barton, R. A., & Venditti, C. (2014). Rapid evolution of the cerebellum in humans and other great apes. Current Biology, 24(20), 2440–2444.
Bateson, P., & Gluckman, P. (2011). Plasticity, robustness, development and evolution. Cambridge: Cambridge University Press.
Baumard, N. (2018). Psychological origins of the industrial revolution. Behavioral and Brain Sciences, 1–47. https://doi.org/10.1017/S0140525X1800211X.
Beck, S. R., Chappell, J., Apperly, I. A., & Cutting, N. (2012). Tool innovation may be a critical limiting step for the establishment of a rich tool-using culture: A perspective from child development. Behavioral and Brain Sciences, 35(4), 220–221.
Bereczkei, T., Voros, S., Gal, A., & Bernath, L. (1997). Resources, attractiveness, family commitment; reproductive decisions in human mate choice. Ethology, 103(8), 681–699.
Black, C. J., Figueredo, A. J., & Jacobs, W. J. (2017). Substance, history, and politics: An examination of the conceptual underpinnings of alternative approaches to the life history narrative. Evolutionary Psychology, 15(1), 1474704916670402.
Blake, K. R., Dixson, B. J., O'Dean, S. M., & Denson, T. F. (2017). No compelling positive association between ovarian hormones and wearing red clothing when using multinomial analyses. Hormones and Behavior, 90, 129–135.
Blake, K. R., Bastian, B., Denson, T. F., Grosjean, P., & Brooks, R. C. (2018). Income inequality not gender inequality positively covaries with female sexualization on social media. Proceedings of the National Academy of Sciences, 115(35), 8722–8727. https://doi.org/10.1073/pnas.1717959115.
Blamires, S. J. (2010). Plasticity in extended phenotypes: Orb web architectural responses to variations in prey parameters. The Journal of Experimental Biology, 213(18), 3207–3212. https://doi.org/10.1242/jeb.045583.
Blamires, S. J., Hasemore, M., Martens, P. J., & Kasumovic, M. M. (2017). Diet-induced co-variation between architectural and physicochemical plasticity in an extended phenotype. The Journal of Experimental Biology, 220(Pt 5), 876–884. https://doi.org/10.1242/jeb.150029.
Blamires, S. J., Martens, P. J., & Kasumovic, M. M. (2018). Fitness consequences of plasticity in an extended phenotype. Journal of Experimental Biology, 221(4), jeb167288. https://doi.org/10.1242/jeb.167288.
Bolondi, A., Caldarelli, F., Di Felice, F., Durano, D., Germani, G., Michetti, L., et al. (2017). What is a gene? A two sided view. Evolutionary Biology, 44(1), 1–4. https://doi.org/10.1007/s11692-016-9392-5.
Botchkavar, E., Marshell, I. H., Rocque, M., & Posick, C. (2015). The importance of parenting in the development of self-control in boys and girls: Results from a multinational study of youth. Journal of Criminal Justice, 34, 133–141. https://doi.org/10.1016/j.jcrimjus.2015.02.001.
Boutwell, B. B., Barnes, J. C., Beaver, K. M., Haynes, R. D., Nedelec, J. L., & Gibson, C. L. (2015). A unified crime theory: The evolutionary taxonomy. Aggression and Violent Behavior, 25, 343–353.
Boyd, B. (2009). On the origin of stories: Evolution, cognition, and fiction. Cambridge: Harvard University Press.
Bribiescas, R. G., Ellison, P. T., & Gray, P. B. (2012). Male life history, reproductive effort, and the evolution of the genus homo. Current Anthropology, 53(S6), S424–S435. https://doi.org/10.1086/667538.
Briedis, M., Hahn, S., & Adamík, P. (2017). Cold spell en route delays spring arrival and decreases apparent survival in a long-distance migratory songbird. BMC Ecology, 17(1), 11. https://doi.org/10.1186/s12898-017-0121-4.
Bukowski, A., & Rudnicki, S. (2018). Not Only Individualism: The Effects of Long-Term Orientation and Other Cultural Variables on National Innovation Success. Cross-Cultural Research, 1-44, 1069397118785546. https://doi.org/10.1177/1069397118785546.
Burger, J. R., Weinberger, V. P., & Marquet, P. A. (2017). Extra-metabolic energy use and the rise in human hyper-density. Scientific Reports, 7. https://doi.org/10.1038/srep43869.
Burke, D., Sulikowski, D., Stephen, I., & Brooks, R. (2017). Inconsistent with the data: Support for the CLASH model depends on the wrong kind of latitude. Behavioral and Brain Sciences, 40, e80. https://doi.org/10.1017/S0140525X16001242,e80.
Buss, D. M. (1989). Sex differences in human mate preferences: Evolutionary hypotheses tested in 37 cultures. Behavioral and Brain Sciences, 12(01), 1–14.
Buss, D. M., Abbott, M., Angleitner, A., Asherian, A., Biaggio, A., Blanco-Villasenor, A., ... Deraad, B. (1990). International preferences in selecting mates: A study of 37 cultures. Journal of Cross-Cultural Psychology, 21(1), 5–47.
Buss, D. M., Haselton, M. G., Shackelford, T. K., Bleske, A. L., & Wakefield, J. C. (1998). Adaptations, exaptations, and spandrels. American Psychologist, 53(5), 533–548. https://doi.org/10.1037/0003-066X.53.5.533.
Buston, P. M., & Emlen, S. T. (2003). Cognitive processes underlying human mate choice: The relationship between self-perception and mate preference in western society. Proceedings of the National Academy of Sciences of the United States of America, 100(15), 8805–8810. https://doi.org/10.1073/pnas.1533220100.
Cain, K. E., & Ketterson, E. D. (2012). Competitive females are successful females; phenotype, mechanism, and selection in a common songbird. Behavioral Ecology and Sociobiology, 66, 241–252.
Camera, D. M., Smiles, W. J., & Hawley, J. A. (2016). Exercise-induced skeletal muscle signaling pathways and human athletic performance. Free Radical Biology and Medicine, 98, 131–143. https://doi.org/10.1016/j.freeradbiomed.2016.02.007.
Candolin, U. (2003). The use of multiple cues in mate choice. Biological Reviews, 78(4), 575–595.
Carleton, W. C., Campbell, D., & Collard, M. (2017). Increasing temperature exacerbated classic Maya conflict over the long term. Quaternary Science Reviews, 163, 209–218. https://doi.org/10.1016/j.quascirev.2017.02.022.
Carneiro, R. L. (1970). A theory of the origin of the state: Traditional theories of state origins are considered and rejected in favor of a new ecological hypothesis. Science, 169(3947), 733–738.
Casey, B., Jones, R. M., & Hare, T. A. (2008). The adolescent brain. Annals of the New York Academy of Sciences, 1124(1), 111–126.
Cedeño Laurent, J. G., Williams, A., Oulhote, Y., Zanobetti, A., Allen, J. G., & Spengler, D. A. (2018). Reduced cognitive function during a heat wave among residents of non-air-conditioned buildings: An observational study of young adults in the summer of 2016. PLoS Medicine, 15(7), e1002605. https://doi.org/10.1371/journal.pmed.1002605.
Chappell, J., Cutting, N., Apperly, I. A., & Beck, S. R. (2013). The development of tool manufacture in humans: What helps young children make innovative tools? Philosophical Transactions of the Royal Society, B: Biological Sciences, 368(1630), 20120409. https://doi.org/10.1098/rstb.2012.0409.
Charney, E. (2012). Behavior genetics and postgenomics. Behavioral and Brain Sciences, 35(5), 331–358. https://doi.org/10.1017/S0140525X11002226.
Christin, A. (2012). Gender and highbrow cultural participation in the United States. Poetics, 40(5), 423–443.
Chu, W. (2009). A functional approach to Paleolithic open-air habitation structures. World Archaeology, 41(3), 348–362.
Clegg, H., Nettle, D., & Miell, D. (2011). Status and mating success amongst visual artists. Frontiers in Psychology, 2(310). https://doi.org/10.3389/fpsyg.2011.00310.
Climate Change Knowledge Portal (2018). Historical climate data. Online Database Available at: http://sdwebx.worldbank.org/climateportal/index.cfm Retrieved on September 3, 2018.
Cloud, J. M., & Taylor, M. H. (2018). The effect of mate value discrepancy on hypothetical engagement ring purchases. Evolutionary Psychological Science. https://doi.org/10.1007/s40806-018-0156-6.
Clutton-Brock, T. (2007). Sexual selection in males and females. Science, 318(5858), 1882–1885.
Clutton-Brock, T. (2017). Reproductive competition and sexual selection. Phil. Trans. R. Soc. B, 372(1729), 20160310. https://doi.org/10.1098/rstb.2016.0310.
Collard, M., Kemery, M., & Banks, S. (2005). Causes of toolkit variation among hunter-gatherers: A test of four competing hypotheses. Canadian Journal of Archaeology/Journal Canadien D'Archéologie, 29(1), 1–19. https://www.jstor.org/stable/41103514.
Confer, J. C., Perilloux, C., & Buss, D. M. (2010). More than just a pretty face: men's priority shifts toward bodily attractiveness in short-term versus long-term mating contexts. Evolution and Human Behavior, 31(5), 348–353.
Conroy-Beam, D. (2018). Euclidean mate value and power of choice on the mating market. Personality and Social Psychology Bulletin, 44(2), 252–264. https://doi.org/10.1177/0146167217739262.
Conroy-Beam, D., & Buss, D. M. (2016). Mate preferences. In T. K. Shackelford, & V. A. Weekes-Shackelford (Eds.), Encyclopedia of evolutionary psychological science (pp. 1–11) Springer. https://doi.org/10.1007/978-3-319-16999-6_1-1.
Conroy-Beam, D., Buss, D. M., Pham, M. N., & Shackelford, T. K. (2015). How sexually dimorphic are human mate preferences? Personality and Social Psychology Bulletin, 41(8), 1082–1093.
Cornwallis, C. K., & Uller, T. (2010). Towards an evolutionary ecology of sexual traits. Trends in Ecology & Evolution, 25(3), 145–152.
Creanza, N., & Feldman, M. W. (2016). Worldwide genetic and cultural change in human evolution. Current Opinion in Genetics & Development, 41, 85–92.
Creanza, N., Kolodny, O., & Feldman, M. W. (2017). Cultural evolutionary theory: How culture evolves and why it matters. Proceedings of the National Academy of Sciences, 114(30), 7782–7789. https://doi.org/10.1073/pnas.1620732114.
Crocchiola, D. (2014). Art as an indicator of male fitness: Does prenatal testosterone influence artistic ability? Evolutionary Psychology, 12(3), 521–533. https://doi.org/10.1177/147470491401200303.
Csajbók, Z., & Berkics, M. (2017). Factor, factor, on the whole, who's the best fitting of all?: Factors of mate preferences in a large sample. Personality and Individual Differences, 114, 92–102.
d’Errico, F., Doyon, L., Zhang, S., Baumann, M., Lázničková-Galetová, M., Gao, X., Chen, F., & Zhang, Y. (2018). The origin and evolution of sewing technologies in Eurasia and North America. Journal of Human Evolution, 125, 71–86. https://doi.org/10.1016/j.jhevol.2018.10.004.
Davies, G., Tenesa, A., Payton, A., Yang, J., Harris, S. E., Liewald, D., ... Luciano, M. (2011). Genome-wide association studies establish that human intelligence is highly heritable and polygenic. Molecular Psychiatry, 16(10), 996–1005.
Dawkins, R. (1982). The extended phenotype: The long reach of the gene. Oxford: Oxford University Press.
Dawkins, R. (2004). Extended phenotype–but not too extended. A reply to Laland, turner and Jablonka. Biology and Philosophy, 19(3), 377–396.
De Dreu, C. K., & van Dijk, M. A. (2018). Climatic shocks associate with innovation in science and technology. PLoS One, 13(1), e0190122. https://doi.org/10.1371/journal.pone.0190122.
de Manzano, Ö., & Ullén, F. (2018). Genetic and environmental influences on the phenotypic associations between intelligence, personality, and creative achievement in the arts and sciences. Intelligence, 69, 123–133. https://doi.org/10.1016/j.intell.2018.05.004.
Del Giudice, M., Gangestad, S. W., & Kaplan, H. S. (2015). Life history theory and evolutionary psychology. In D. M. Buss (Ed.), The handbook of evolutionary psychology (2nd ed., pp. 88–114). New York: Wiley Online Library. https://doi.org/10.1002/9781119125563.evpsych102.
Delson, E. (2000). Encyclopedia of human evolution and prehistory (2nd ed.). New York: Taylor & Francis.
Dennett, D. C. (1975). Why the law of effect will not go away. Journal for the Theory of Social Behaviour, 5(2), 169–188.
Dickerson, K. L., Ainge, J. A., & Seed, A. M. (2018). The role of Association in pre-schoolers’ solutions to “spoon tests” of future planning. Current Biology, 28(14), 2309–2313. https://doi.org/10.1016/j.cub.2018.05.052.
Dickins, T. E., & Barton, R. A. (2013). Reciprocal causation and the proximate–ultimate distinction. Biology and Philosophy, 28(5), 747–756. https://doi.org/10.1007/s10539-012-9345-z.
Dobbs, R., Remes, J., Manyika, J., Roxburgh, C., Smit, S., & Schaer, F. (2012). Urban world: Cities and the rise of the consuming class. McKinsey global institute. Available at: https://www.mckinsey.com/global-themes/urbanization/urban-world-cities-and-the-rise-of-the-consuming-class. Retrieved on January 1, 2018.
Donath, J. S. (1998). Identity and deception in the virtual community. In M. A. Smith & P. Kollack (Eds.), Communities in cyberspace (pp. 29–59). London: Routledge.
Dunn, M. J., & Hill, A. (2014). Manipulated luxury-apartment ownership enhances opposite-sex attraction in females but not males. Journal of Evolutionary Psychology, 12(1), 1–17.
Dunn, M. J., & Searle, R. (2010). Effect of manipulated prestige-car ownership on both sex attractiveness ratings. British Journal of Psychology, 101(1), 69–80.
Durante, K. M., Li, N. P., & Haselton, M. G. (2008). Changes in women's choice of dress across the ovulatory cycle: Naturalistic and laboratory task-based evidence. Personality and Social Psychology Bulletin, 34(11), 1451–1460. https://doi.org/10.1177/0146167208323103.
Dutta, S., & Lanvin, B. (2013). The global innovation index 2013. The local dynamics of innovation. Ithaca: Cornell University, World Intellectual Property Organization (WIPO). (Retrieved from www.globalinnovationindex.org/content.aspx?page=gii-fullreport-2013).
Eisenbruch, A. B., Simmons, Z. L., & Roney, J. R. (2015). Lady in red: Hormonal predictors of women's clothing choices. Psychological Science, 26(8), 1332–1338. https://doi.org/10.1177/0956797615586403.
Elliot, A. J., Greitemeyer, T., & Pazda, A. D. (2013). Women's use of red clothing as a sexual signal in intersexual interaction. Journal of Experimental Social Psychology, 49(3), 599–602.
Ellis, B. J., Bianchi, J., Griskevicius, V., & Frankenhuis, W. E. (2017). Beyond risk and protective factors: An adaptation-based approach to resilience. Perspectives on Psychological Science, 12(4), 561–587. https://doi.org/10.1177/1745691617693054.
Ellison, P. T. (2017). Endocrinology, energetics, and human life history: A synthetic model. Hormones and Behavior, 91, 97–106.
Escasa, M., Gray, P. B., & Patton, J. Q. (2010). Male traits associated with attractiveness in Conambo, Ecuador. Evolution and Human Behavior, 31(3), 193–200.
Essock-Vitale, S. M. (1984). The reproductive success of wealthy Americans. Ethology and Sociobiology, 5(1), 45–49.
European Commission (2018). Distance calculator. Available at: http://ec.europa.eu/dgs/education_culture/tools/distance_en.htm. Retrieved on January 1, 2018.
Figueredo, A. J., Vásquez, G., Brumbach, B. H., & Schneider, S. M. (2004). The heritability of life history strategy: The k-factor, covitality, and personality. Social Biology, 51(3–4), 121–143.
Figueredo, A. J., Vásquez, G., Brumbach, B. H., Sefcek, J. A., Kirsner, B. R., & Jacobs, W. J. (2005). The K-factor: Individual differences in life history strategy. Personality and Individual Differences, 39, 1349–1360.
Figueredo, A. J., Vásquez, G., Brumbach, B. H., Schneider, S. M., Sefcek, J. A., Tal, I. R., ... Jacobs, W. J. (2006). Consilience and life history theory: From genes to brain to reproductive strategy. Developmental Review, 26(2), 243–275.
Fincher, C. L., & Thornhill, R. (2012). Parasite-stress promotes in-group assortative sociality: The cases of strong family ties and heightened religiosity. Behavioral and Brain Sciences, 35(02), 61–79.
Fincher, C. L., Thornhill, R., Murray, D. R., & Schaller, M. (2008). Pathogen prevalence predicts human cross-cultural variability in individualism/collectivism. Proceedings of the Royal Society of London B: Biological Sciences, 275(1640), 1279–1285. https://doi.org/10.1098/rspb.2008.0094.
Fisher, R. A. (1930). The genetical theory of natural selection. Oxford: Oxford University Press.
Flatt, T., & Heyland, A. (Eds.). (2011). Mechanisms of life history evolution: The genetics and physiology of life history traits and trade-offs. Oxford: Oxford University Press.
Flinn, M. V., Geary, D. C., & Ward, C. V. (2005). Ecological dominance, social competition, and coalitionary arms races: Why humans evolved extraordinary intelligence. Evolution and Human Behavior, 26(1), 10–46. https://doi.org/10.1016/j.evolhumbehav.2004.08.005.
Fogarty, L. (2018). Cultural complexity and evolution in fluctuating environments. Philosophical Transactions of the Royal Society B, 373(1743), 20170063. https://doi.org/10.1098/rstb.2017.0063.
Fogarty, L., & Creanza, N. (2017). The niche construction of cultural complexity: Interactions between innovations, population size and the environment. Philosophical Transactions of the Royal Society B, 372(1735), 20160428. https://doi.org/10.1098/rstb.2016.0428.
Fogarty, L., Creanza, N., & Feldman, M. W. (2015). Cultural evolutionary perspectives on creativity and human innovation. Trends in Ecology & Evolution, 30(12), 736–754. https://doi.org/10.1016/j.tree.2015.10.004.
Fristoe, T. S., Iwaniuk, A. N., & Botero, C. A. (2017). Big brains stabilize populations and facilitate colonization of variable habitats in birds. Nature Ecology & Evolution, 1, 1706–1715. https://doi.org/10.1038/s41559-017-0316-2.
Gangestad, S. W. (2008). Biological adaptations and human behavior. In C. Crawford & D. Krebs (Eds.), Foundations of evolutionary psychology (pp. 153–172). New York: Lawrence Erlbaum Associates.
Gao, Z., Gao, S., Xu, L., Zheng, X., Ma, X., Luo, L., & Kendrick, K. M. (2017a). Women prefer men who use metaphorical language when paying compliments in a romantic context. Scientific Reports, 7, 40871. https://doi.org/10.1038/srep40871.
Gao, Z., Yang, Q., Ma, X., Becker, B., Li, K., Zhou, F., & Kendrick, K. M. (2017b). Men who compliment a Woman's appearance using metaphorical language: Associations with creativity, masculinity, intelligence and attractiveness. Frontiers in Psychology, 8. https://doi.org/10.3389/fpsyg.2017.02185.
Geary, D. C. (1995). Reflections of evolution and culture in children's cognition: Implications for mathematical development and instruction. American Psychologist, 50(1), 24–37.
Geffre, A. C., Liu, R., Manfredini, F., Beani, L., Kathirithamby, J., Grozinger, C. M., & Toth, A. L. (2017). Transcriptomics of an extended phenotype: Parasite manipulation of wasp social behaviour shifts expression of caste-related genes. Proceedings of the Royal Society of London B: Biological Sciences, 284(1852), 20170029. https://doi.org/10.1098/rspb.2017.0029.
Gettler, L. T., Ryan, C. P., Eisenberg, D. T., Rzhetskaya, M., Hayes, M. G., Feranil, A. B., . . . Kuzawa, C. W. (2017). The role of testosterone in coordinating male life history strategies: The moderating effects of the androgen receptor CAG repeat polymorphism. Hormones and Behavior, 87, 164–175. https://doi.org/10.1016/j.yhbeh.2016.10.012.
Gienapp, P., Teplitsky, C., Alho, J. S., Mills, J. A., & Merilä, J. (2008). Climate change and evolution: Disentangling environmental and genetic responses. Molecular Ecology, 17(1), 167–178. https://doi.org/10.1111/j.1365-294X.2007.03413.x.
Gilligan, I. (2007). Neanderthal extinction and modern human behaviour: The role of climate change and clothing. World Archaeology, 39(4), 499–514. https://doi.org/10.1080/00438240701680492.
Gilligan, I. (2010). The prehistoric development of clothing: Archaeological implications of a thermal model. Journal of Archaeological Method and Theory, 17(1), 15–80. https://doi.org/10.1007/s10816-009-9076-x.
González, A. L., Dézerald, O., Marquet, P. A., Romero, G. Q., & Srivastava, D. S. (2017). The multidimensional stoichiometric niche. Frontiers in Ecology and Evolution, 5, 110. https://doi.org/10.3389/fevo.2017.00110.
Gonzalez-Forero, M., Faulwasser, T., & Lehmann, L. (2017). A model for brain life history evolution. PLoS Computational Biology, 13(3), e1005380. https://doi.org/10.1371/journal.pcbi.1005380.
Gould, S. J. (1991). Exaptation: A crucial tool for an evolutionary psychology. Journal of Social Issues, 47(3), 43–65.
Gould, S. J., & Vrba, E. S. (1982). Exaptation—A missing term in the science of form. Paleobiology, 8(01), 4–15.
Grammer, K., Renninger, L., & Fischer, B. (2004). Disco clothing, female sexual motivation, and relationship status: Is she dressed to impress? Journal of Sex Research, 41(1), 66–74.
Gray, P. B., Volsche, S. L., Garcia, J. R., & Fisher, H. E. (2015). The roles of pet dogs and cats in human courtship and dating. Anthrozoös, 28(4), 673–683.
Green, R. (2017). Hugh Hefner, the international academy of sex research, and its founding president. Archives of Sexual Behavior, 46, 2211–2212. https://doi.org/10.1007/s10508-017-1098-y.
Gregor, T. (1985). Anxious pleasures: The sexual lives of an Amazonian people. Chicago: University of Chicago Press.
Groyecka, A., Pisanski, K., Sorokowska, A., Havlíček, J., Karwowski, M., Puts, D., ... & Sorokowski, P. (2017). Attractiveness is multimodal: Beauty is also in the nose and ear of the beholder. Frontiers in Psychology, 8, 778. https://doi.org/10.3389/fpsyg.2017.00778.
Guadagno, R. E., Okdie, B. M., & Kruse, S. A. (2012). Dating deception: Gender, online dating, and exaggerated self-presentation. Computers in Human Behavior, 28(2), 642–647.
Guéguen, N. (2015). High heels increase women’s attractiveness. Archives of Sexual Behavior, 44(8), 2227–2235. https://doi.org/10.1007/s10508-014-0422-z.
Guéguen, N., & Ciccotti, S. (2008). Domestic dogs as facilitators in social interaction: An evaluation of helping and courtship behaviors. Anthrozoös, 21(4), 339–349.
Guéguen, N., & Lamy, L. (2012). Men’s social status and attractiveness. Swiss Journal of Psychology, 71, 157–160. https://doi.org/10.1024/1421-0185/a000083.
Guéguen, N., Meineri, S., & Fischer-Lokou, J. (2014). Men’s music ability and attractiveness to women in a real-life courtship context. Psychology of Music, 42(4), 545–549.
Gurven, M. (2018). Broadening horizons: Sample diversity and socioecological theory are essential to the future of psychological science. Proceedings of the National Academy of Sciences of the United States of America, 115(45), 11420–11427. https://doi.org/10.1073/pnas.1720433115.
Gurven, M., Winking, J., Kaplan, H., Von Rueden, C., & McAllister, L. (2009). A bioeconomic approach to marriage and the sexual division of labor. Human Nature, 20(2), 151–183.
Harrison, M. A., & Shortall, J. C. (2011). Women and men in love: Who really feels it and says it first? The Journal of Social Psychology, 151(6), 727–736.
Hartmann, D., Guevara, M. R., Jara-Figueroa, C., Aristarán, M., & Hidalgo, C. A. (2017). Linking economic complexity, institutions, and Income Inequality. World Development, 93, 75–93. https://doi.org/10.1016/j.worlddev.2016.12.020.
Haselton, M. G., Mortezaie, M., Pillsworth, E. G., Bleske-Rechek, A., & Frederick, D. A. (2007). Ovulatory shifts in human female ornamentation: Near ovulation, women dress to impress. Hormones and Behavior, 51(1), 40–45.
Hausmann, R., Hidalgo, C. A., Bustos, S., Coscia, M., Simoes, A., & Yildirim, M. A. (2014). The atlas of economic complexity: Mapping paths to prosperity. Cambridge: MIT Press.
Havlíček, J., Cobey, K. D., Barrett, L., Klapilová, K., & Roberts, S. C. (2015). The spandrels of Santa Barbara? A new perspective on the peri-ovulation paradigm. Behavioral Ecology, 26(5), 1249–1260. https://doi.org/10.1093/beheco/arv064.
Head, M. L., Fox, R. J., & Barber, I. (2017). Environmental change mediates mate choice for an extended phenotype, but not for mate quality. Evolution, 71(1), 135–144. https://doi.org/10.1111/evo.13091.
Hill, W. D., Hagenaars, S. P., Marioni, R. E., Harris, S. E., Liewald, D. C., Davies, G., et al. (2016). Molecular genetic contributions to social deprivation and household income in UK biobank. Current Biology, 26(22), 3083–3089. https://doi.org/10.1016/j.cub.2016.09.035.
Hill, W. D., Arslan, R. C., Xia, C., Luciano, M., Amador, C., Navarro, P., ... & Deary, I. J. (2018a). Genomic analysis of family data reveals additional genetic effects on intelligence and personality. Molecular Psychiatry, 1. https://doi.org/10.1038/s41380-017-0005-1.
Hill, W. D., Marioni, R. E., Maghzian, O., Ritchie, S. J., Hagenaars, S. P., McIntosh, A. M., Gale, C. R., Davies, G., & Deary, I. J. (2018b). A combined analysis of genetically correlated traits identifies 187 loci and a role for neurogenesis and myelination in intelligence. Molecular Psychiatry. https://doi.org/10.1038/s41380-017-0001-5.
Hoberg, E. P., & Brooks, D. R. (2015). Evolution in action: Climate change, biodiversity dynamics and emerging infectious disease. Phil. Trans. R. Soc. B, 370(1665), 20130553. https://doi.org/10.1098/rstb.2013.0553.
Hoffecker, J. F., & Hoffecker, I. T. (2017a). The structural and functional complexity of hunter-gatherer technology. Journal of Archaeological Method and Theory, 1–24. https://doi.org/10.1007/s10816-017-9332-4.
Hoffecker, J. F., & Hoffecker, I. T. (2017b). Technological complexity and the global dispersal of modern humans. Evolutionary Anthropology, 26(6), 285–299. https://doi.org/10.1002/evan.21553.
Holmberg, A. R. (1969). Nomads of the long Bow: The Siriono of eastern Bolivia. Garden City: Natural History Press.
Honegger, K., & de Bivort, B. (2018). Stochasticity, individuality and behavior. Current Biology, 28, R1–R16. https://doi.org/10.1016/j.cub.2017.11.058.
Hoover, K., Grove, M., Gardner, M., Hughes, D. P., McNeil, J., & Slavicek, J. (2011). A gene for an extended phenotype. Science, 333(6048), 1401. https://doi.org/10.1126/science.1209199.
Hughes, S. M., & Aung, T. (2017). Modern-day female preferences for resources and provisioning by long-term mates. Evolutionary Behavioral Sciences, 11(3), 242–261. https://doi.org/10.1037/ebs0000084.
Hulteen, R. M., Morgan, P. J., Barnett, L. M., Stodden, D. F., & Lubans, D. R. (2018). Development of foundational movement skills: A conceptual model for physical activity across the lifespan. Sports Medicine, 48(7), 1533–1540. https://doi.org/10.1007/s40279-018-0892-6.
Hunter, P. (2009). Extended phenotype redux. EMBO Reports, 10(3), 212–215. https://doi.org/10.1038/embor.2009.18.
Hunter, P. (2018). The revival of the extended phenotype: After more than 30 years, Dawkins’ extended phenotype hypothesis is enriching evolutionary biology and inspiring potential applications. EMBO Reports, 19, e46477. https://doi.org/10.15252/embr.201846477.
Hurst, J. E., & Kavanagh, P. S. (2017). Life history strategies and psychopathology: The faster the life strategies, the more symptoms of psychopathology. Evolution and Human Behavior, 38(1), 1–8. https://doi.org/10.1016/j.evolhumbehav.2016.06.001.
IJzerman, H., Čolić, M. V., Hennecke, M., Hong, Y., Hu, C. P., Joy-Gaba, J., ... & Schubert, T. (2017). Does distance from the equator predict self-control? Lessons from the human penguin project. Behavioral and Brain Sciences, 40. https://doi.org/10.1017/S0140525X16000406.
Immonen, E., Hämäläinen, A., Schuett, W., & Tarka, M. (2018). Evolution of sex-speciic pace-of-life syndromes: Genetic architecture and physiological mechanisms. Behavioral Ecology and Sociobiology, 72(3), 60. https://doi.org/10.1007/s00265-018-2462-1.
Iriki, A., & Taoka, M. (2012). Triadic (ecological, neural, cognitive) niche construction: A scenario of human brain evolution extrapolating tool use and language from the control of reaching actions. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 367(1585), 10–23. https://doi.org/10.1098/rstb.2011.0190.
Israel, E., & Strassberg, D. S. (2009). Viewing time as an objective measure of sexual interest in heterosexual men and women. Archives of Sexual Behavior, 38(4), 551–558.
Jasienska, G., Bribiescas, R. G., Furberg, A. S., Helle, S., & Núñez-de la Mora, A. (2017). Human reproduction and health: An evolutionary perspective. The Lancet, 390(10093), 510–520. https://doi.org/10.1016/S0140-6736(17)30573-1.
Jonason, P. K., Garcia, J. R., Webster, G. D., Li, N. P., & Fisher, H. E. (2015). Relationship dealbreakers: Traits people avoid in potential mates. Personality and Social Psychology Bulletin, 41(12), 1697–1711. https://doi.org/10.1177/0146167215609064.
Jones, D., Brace, C. L., Jankowiak, W., Laland, K. N., Musselman, L. E., Langlois, J. H., Roggman, L. A., Pérusse, D., Schweder, B., & Symons, D. (1995). Sexual selection, physical attractiveness, and facial neoteny: Cross-cultural evidence and implications. Current Anthropology, 36(5), 723–748.
Jordan, P. (2015). Technology as human social tradition: Cultural transmission among hunter-gatherers. Oakland: University of California Press.
Jordan, L. A., Maguire, S. M., Hofmann, H. A., & Kohda, M. (2016). The social and ecological costs of an ‘over-extended’ phenotype. Proceedings of the Royal Society of London B: Biological Sciences, 283, 20152359. https://doi.org/10.1098/rspb.2015.2359.
Kanazawa, S. (2000). Scientific discoveries as cultural displays: A further test of Miller's courtship model. Evolution and Human Behavior, 21(5), 317–321. https://doi.org/10.1016/S1090-5138(00)00051-9.
Kanazawa, S. (2008). Temperature and evolutionary novelty as forces behind the evolution of general intelligence. Intelligence, 36(2), 99–108. https://doi.org/10.1016/j.intell.2007.04.001.
Kaufman, S. B., Kozbelt, A., Silvia, P., Kaufman, J. C., Ramesh, S., & Feist, G. J. (2014). Who finds bill gates sexy? Creative mate preferences as a function of cognitive ability, personality, and creative achievement. The Journal of Creative Behavior, 50(4), 294–307.
Kecko, S., Mihailova, A., Kangassalo, K., Elferts, D., Krama, T., Krams, R., Luoto, S., Rantala, M. J., & Krams, I. A. (2017). Sex-specific compensatory growth in the larvae of the greater wax moth Galleria mellonella. Journal of Evolutionary Biology, 30(10), 1910–1918. https://doi.org/10.1111/jeb.13150.
Keeley, L. H. (1988). Hunter-gatherer economic complexity and “population pressure”: A cross-cultural analysis. Journal of Anthropological Archaeology, 7(4), 373–411. https://doi.org/10.1016/0278-4165(88)90003-7.
Kenrick, D. T., Griskevicius, V., Neuberg, S. L., & Schaller, M. (2010). Renovating the pyramid of needs: Contemporary extensions built upon ancient foundations. Perspectives on Psychological Science, 5(3), 292–314.
Kline, M. A., & Boyd, R. (2010). Population size predicts technological complexity in Oceania. Proceedings of the Royal Society of London B: Biological Sciences, 277(1693), 2559–2564. https://doi.org/10.1098/rspb.2010.0452.
Kokko, H., & Rankin, D. J. (2006). Lonely hearts or sex in the city? Density-dependent effects in mating systems. Philosophical Transactions of the Royal Society of London B: Biological Sciences, 361(1466), 319–334.
Kordsmeyer, T. L., Hunt, J., Puts, D. A., Ostner, J., & Penke, L. (2018). The relative importance of intra-and intersexual selection on human male sexually dimorphic traits. Evolution and Human Behavior, 39(4), 424–436. https://doi.org/10.1016/j.evolhumbehav.2018.03.008.
Kowalczyk, R., & Zalewski, A. (2011). Adaptation to cold and predation—Shelter use by invasive raccoon dogs Nyctereutes procyonoides in Białowieża primeval Forest (Poland). European Journal of Wildlife Research, 57(1), 133–142. https://doi.org/10.1007/s10344-010-0406-9.
Kramer, R. S., & Mulgrew, J. (2018). Displaying red and black on a first date: A field study using the “first dates” television series. Evolutionary Psychology, 16(2), 1474704918769417. https://doi.org/10.1177/1474704918769417.
Krams, I., Inwood, S. E., Trakimas, G., Krams, R., Burghardt, G. M., Butler, D. M., ... & Krama, T. (2016). Short-term exposure to predation affects body elemental composition, climbing speed and survival ability in Drosophila melanogaster. PeerJ, 4, e2314. https://doi.org/10.7717/peerj.2314.
Krams, I., Kecko, S., Inashkina, I., Trakimas, G., Krams, R., Elferts, D., ... & Contreras-Garduño, J. (2017a). Food quality affects the expression of antimicrobial peptide genes upon simulated parasite attack in the larvae of greater wax moth. Entomologia Experimentalis et Applicata, 165(2–3), 129–137. https://doi.org/10.1111/eea.12629.
Krams, I., Kecko, S., Jõers, P., Trakimas, G., Elferts, D., Krams, R., et al. (2017b). Microbiome symbionts and diet diversity incur costs on the immune system of insect larvae. Journal of Experimental Biology, 220(22), 4204–4212. https://doi.org/10.1242/jeb.169227.
Krams, I., Rumvolt, K., Saks, L., Krams, R., Elferts, D., Vrublevska, J., et al. (2017c). Reproduction compromises adaptive immunity in a cyprinid fish. Ecological Research, 32, 559–566. https://doi.org/10.1007/s11284-017-1467-y.
Krams, I., Luoto, S., Rubika, A., Krama, T., Elferts, D., Kecko, S., Skrinda, I., Moore, F. R., Krams, R., & Rantala, M. J. (2018). A head start for life history development? Family income causes associations between body height and immune response in men. American Journal of Physical Anthropology. https://doi.org/10.1002/ajpa.23754.
Kruger, D., & Kruger, J. S. (2018). What do economically costly signals signal?: A life history framework for interpreting conspicuous consumption. Evolutionary Psychological Science, 4(4), 420–427. https://doi.org/10.1007/s40806-018-0151-y.
Kuzawa, C. W. (2007). Developmental origins of life history: Growth, productivity, and reproduction. American Journal of Human Biology, 19(5), 654–661.
Laidre, M. E., & Johnstone, R. A. (2013). Animal signals. Current Biology, 23(18), R829–R833.
Laland, K. N., & O’Brien, M. J. (2011). Cultural niche construction: An introduction. Biological Theory, 6(3), 191–202.
Laland, K. N., Sterelny, K., Odling-Smee, J., Hoppitt, W., & Uller, T. (2011). Cause and effect in biology revisited: Is Mayr’s proximate-ultimate dichotomy still useful? Science, 334(6062), 1512–1516.
Laland, K., Odling-Smee, J., & Endler, J. (2017). Niche construction, sources of selection and trait coevolution. Interface Focus, 7(5), 20160147.
Lane, D. A. (2016). Innovation cascades: Artefacts, organization and attributions. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 371, 20150194. https://doi.org/10.1098/rstb.2015.0194.
Lange, B. P. (2011). Male proneness to verbal display production. Acta Linguistica, 5, 97–104.
Lange, B. P., & Euler, H. A. (2014). Writers have groupies, too: High quality literature production and mating success. Evolutionary Behavioral Sciences, 8(1), 20–30. https://doi.org/10.1037/h0097246.
Lassek, W. D., & Gaulin, S. J. (2008). Waist-hip ratio and cognitive ability: Is gluteofemoral fat a privileged store of neurodevelopmental resources? Evolution and Human Behavior, 29(1), 26–34.
Lawrence, S. (2006). Jimi Hendrix: The man, the magic, the truth. New York: Harper Entertainment.
Lee, J. J., Wedow, R., Okbay, A., Kong, E., Maghzian, O., Zacher, M., ... & Fontana, M. A. (2018). Gene discovery and polygenic prediction from a genome-wide association study of educational attainment in 1.1 million individuals. Nature Genetics, 50(8), 1112–1121. https://doi.org/10.1038/s41588-018-0147-3.
Lehmann, L. (2008). The adaptive dynamics of niche constructing traits in spatially subdivided populations: Evolving posthumous extended phenotypes. Evolution, 62(3), 549–566.
Levine, R. V., & Norenzayan, A. (1999). The pace of life in 31 countries. Journal of Cross-Cultural Psychology, 30(2), 178–205.
Levis, N. A., Isdaner, A. J., & Pfennig, D. W. (2018). Morphological novelty emerges from pre-existing phenotypic plasticity. Nature Ecology & Evolution, 2(8), 1289–1297. https://doi.org/10.1038/s41559-018-0601-8.
Lewis, D. M., Al-Shawaf, L., Conroy-Beam, D., Asao, K., & Buss, D. M. (2017a). Evolutionary psychology: A how-to guide. American Psychologist, 72(4), 353–373. https://doi.org/10.1037/a0040409.
Lewis, D. M., Russell, E. M., Al-Shawaf, L., Ta, V., Senveli, Z., Ickes, W., & Buss, D. M. (2017b). Why women wear high heels: Evolution, lumbar curvature, and attractiveness. Frontiers in Psychology, 8, 1875. https://doi.org/10.3389/fpsyg.2017.01875.
Li, N. P., Balley, J., Kenrick, D. T., & Linsenmeier, J. A. (2002). The necessities and luxuries of mate preferences: Testing the tradeoffs. Journal of Personality and Social Psychology, 82(6), 947–955.
Liu, J., Duan, C., & Lummaa, V. (2017). Parent-offspring conflict over family size in current China. American Journal of Human Biology, 29(3), e22946. https://doi.org/10.1002/ajhb.22946.
Lu, H. J., Zhu, X. Q., & Chang, L. (2015). Good genes, good providers, and good fathers: Economic development involved in how women select a mate. Evolutionary Behavioral Sciences, 9(4), 215–228.
Lu, H. J., Wong, K., & Chang, L. (2017). The association between life history strategy and mate preference in men. Personality and Individual Differences, 116, 157–163. https://doi.org/10.1016/j.paid.2017.04.047.
Luhring, T. M., DeLong, J. P., & Semlitsch, R. D. (2017). Stoichiometry and life-history interact to determine the magnitude of cross-ecosystem element and biomass fluxes. Frontiers in Microbiology, 8(814). https://doi.org/10.3389/fmicb.2017.00814.
Luoto, S., & Rantala, M. J. (2017). Specificity of women’s sexual response: Proximate mechanisms and ultimate causes. Archives of Sexual Behavior, 46, 1195–1198. https://doi.org/10.1007/s10508-017-0961-1.
Luoto, S., & Spriggs, M. J. (2018). Commentary: The neural basis of human female mate copying: An empathy-based social learning process. Frontiers in Psychology, 9, 397. https://doi.org/10.3389/fpsyg.2018.00397.
Luoto, S., Krams, I., & Rantala, M. J. (2019). A life history approach to the female sexual orientation spectrum: Evolution, development, causal mechanisms, and health. Archives of Sexual Behavior, 1–36. [Target Article]. https://doi.org/10.1007/s10508-018-1261-0.
Lynn, R. (2012). IQs predict differences in the technological development of nations from 1000 BC through 2000 AD. Intelligence, 40(5), 439–444.
Lynn, R. & Vanhanen, T. (2002). IQ and the wealth of nations. Westport, Conn.: Praeger.
MacDonald, K. (2018). Fire-free hominin strategies for coping with cool winter temperatures in North-Western Europe from before 800,000 to circa 400,000 years ago. PaleoAnthropology, 7–26. https://doi.org/10.4207/PA.2018.ART109.
Mace, R. (1998). The coevolution of human fertility and wealth inheritance strategies. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 353(1367), 389–397. https://doi.org/10.1098/rstb.1998.0217.
Madison, G., Holmquist, J., & Vestin, M. (2018). Musical improvisation skill in a prospective partner is associated with mate value and preferences, consistent with sexual selection and parental investment theory: Implications for the origin of music. Evolution and Human Behavior, 39(1), 120–129. https://doi.org/10.1016/j.evolhumbehav.2017.10.005.
Maex, M., Treer, D., de Greve, H., Proost, P., van Bocxlaer, I., & Bossuyt, F. (2018). Exaptation as a mechanism for functional reinforcement of an animal pheromone system. Current Biology, 28(18), 2955–2960. https://doi.org/10.1016/j.cub.2018.06.074.
Maloney, W. F., & Valencia Caicedo, F. (2016). The persistence of (subnational) fortune. The Economic Journal, 126(598), 2363–2401. https://doi.org/10.1111/ecoj.12276.
Maner, J. K., Gailliot, M. T., & DeWall, C. N. (2007). Adaptive attentional attunement: Evidence for mating-related perceptual bias. Evolution and Human Behavior, 28(1), 28–36.
Maner, J. K., Dittmann, A., Meltzer, A. L., & McNulty, J. K. (2017). Implications of life-history strategies for obesity. Proceedings of the National Academy of Sciences, 114(32), 8517–8522.
Marcinkowska, U. M., Dixson, B. J., Kozlov, M. V., Prasai, K., & Rantala, M. J. (2017). Men’s preferences for female facial femininity decline with age. The Journals of Gerontology: Series B, 72(1), 180–186.
Marquet, P. A., Allen, A. P., Brown, J. H., Dunne, J. A., Enquist, B. J., Gillooly, J. F., ... & O’Dwyer, J. (2014). On theory in ecology. BioScience, 64(8), 701–710.
Meisenberg, G., & Woodley, M. A. (2013). Global behavioral variation: A test of differential-K. Personality and Individual Differences, 55(3), 273–278. https://doi.org/10.1016/j.paid.2012.04.016.
Meunier, C. L., Boersma, M., El-Sabaawi, R., Halvorson, H. M., Herstoff, E. M., Van de Waal, D. B., ... & Litchman, E. (2017). From elements to function: Toward unifying ecological stoichiometry and trait-based ecology. Frontiers in Environmental Science, 5, 18. https://doi.org/10.3389/fenvs.2017.00018.
Miller, G. F. (1999). Sexual selection for cultural displays. In R. Dunbar, C. Knight, & C. Power (Eds.), The evolution of culture (pp. 71–91). New Brunswick: Rutgers University Press.
Miller, G. F. (2000). The mating mind: How sexual choice shaped the evolution of human nature. New York: Doubleday.
Miller, G. F. (2001). Aesthetic fitness: How sexual selection shaped artistic virtuosity as a fitness indicator and aesthetic preferences as mate choice criteria. Bulletin of Psychology and the Arts, 2(1), 20–25.
Miller, G. F. (2010). Spent: Sex, evolution, and consumer behavior. New York: Penguin.
Miller, G. F., & Todd, P. M. (1998). Mate choice turns cognitive. Trends in Cognitive Sciences, 2(5), 190–198.
Minkov, M., & Bond, M. H. (2015). Genetic polymorphisms predict national differences in life history strategy and time orientation. Personality and Individual Differences, 76, 204–215.
Minkov, M., Welzel, C., & Bond, M. H. (2016). The impact of genes, geography, and educational opportunities on national cognitive achievement. Learning and Individual Differences, 47, 236–243. https://doi.org/10.1016/j.lindif.2016.03.004.
Moczek, A. P., Sultan, S., Foster, S., Ledón-Rettig, C., Dworkin, I., Nijhout, H. F., ... & Pfennig, D. W. (2011). The role of developmental plasticity in evolutionary innovation. Proceedings of the Royal Society of London B: Biological Sciences, 278(1719), 2705–2713.
Morris, P. H., White, J., Morrison, E. R., & Fisher, K. (2013). High heels as supernormal stimuli: How wearing high heels affects judgements of female attractiveness. Evolution and Human Behavior, 34(3), 176–181. https://doi.org/10.1016/j.evolhumbehav.2012.11.006.
Mosing, M. A., Verweij, K. J., Madison, G., Pedersen, N. L., Zietsch, B. P., & Ullén, F. (2015). Did sexual selection shape human music? Testing predictions from the sexual selection hypothesis of music evolution using a large genetically informative sample of over 10,000 twins. Evolution and Human Behavior, 36(5), 359–366. https://doi.org/10.1016/j.evolhumbehav.2015.02.004.
Motta-Mena, N. V., & Puts, D. A. (2017). Endocrinology of human female sexuality, mating, and reproductive behavior. Hormones and Behavior, 91, 19–35. https://doi.org/10.1016/j.yhbeh.2016.11.012.
Munro, K. R., Flood, N. J., McKellar, A. E., & Reudink, M. W. (2014). Female mate preference varies with age and environmental conditions. Behaviour, 151(14), 2059–2081. https://doi.org/10.1163/1568539X-00003231.
Murphy, V. E., Smith, R., Giles, W. B., & Clifton, V. L. (2006). Endocrine regulation of human fetal growth: The role of the mother, placenta, and fetus. Endocrine Reviews, 27(2), 141–169.
Murray, C. (2003). Human accomplishment: The pursuit of excellence in the arts and sciences, 800 BC to 1950. New York: Harper Collins.
Muthukrishna, M., & Henrich, J. (2016). Innovation in the collective brain. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 371(1690), 20150192. https://doi.org/10.1098/rstb.2015.0192.
Navarrete, A. F., Reader, S. M., Street, S. E., Whalen, A., & Laland, K. N. (2016). The coevolution of innovation and technical intelligence in primates. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 371(1690), 20150186. https://doi.org/10.1098/rstb.2015.0186.
Nave, G., Nadler, A., Dubois, D., Zava, D., Camerer, C., & Plassmann, H. (2018). Single-dose testosterone administration increases men’s preference for status goods. Nature Communications, 9(1), 2433. https://doi.org/10.1038/s41467-018-04923-0.
Nelissen, R. M., & Meijers, M. H. (2011). Social benefits of luxury brands as costly signals of wealth and status. Evolution and Human Behavior, 32(5), 343–355.
Nielsen, M. (2012). Childhood and advances in human tool use. Behavioral and Brain Sciences, 35(4), 232–233.
Obschonka, M., Stuetzer, M., Rentfrow, P. J., Shaw-Taylor, L., Satchell, M., Silbereisen, R. K., ... & Gosling, S. D. (2018). In the shadow of coal: How large-scale industries contributed to present-day regional differences in personality and well-being. Journal of Personality and Social Psychology, 115(5), 903–927. https://doi.org/10.1037/pspp0000175.
Oda, R. (2001). Sexually dimorphic mate preference in Japan. Human Nature, 12(3), 191–206.
Odling-Smee, F. J., Laland, K. N., & Feldman, M. W. (2003). Niche construction: The neglected process in evolution. Princeton: Princeton University Press.
Oishi, S. (2010). The psychology of residential mobility: Implications for the self, social relationships, and well-being. Perspectives on Psychological Science, 5(1), 5–21. https://doi.org/10.1177/1745691609356781.
Okbay, A., Beauchamp, J. P., Fontana, M. A., Lee, J. J., Pers, T. H., Rietveld, C. A., ... & Oskarsson, S. (2016). Genome-wide association study identifies 74 loci associated with educational attainment. Nature, 533(7604), 539–542. https://doi.org/10.1038/nature17671.
Orosz, G., Zimbardo, P. G., Boőthe, B., & Tóth-Király, I. (2017). The paradoxical effect of climate on time perspective considering resource accumulation. Behavioral and Brain Sciences, 40, e92.
Pepper, G. V., & Nettle, D. (2017a). The behavioural constellation of deprivation: Causes and consequences. Behavioral and Brain Sciences, 1–72. https://doi.org/10.1017/S0140525X1600234X, e314.
Pepper, G. V., & Nettle, D. (2017b). Strengths, altered investment, risk management, and other elaborations on the behavioural constellation of deprivation. Behavioral and Brain Sciences, 40, e346. https://doi.org/10.1017/S0140525X1700190X.
Pianka, E. R. (1970). On r-and K-selection. The American Naturalist, 104(940), 592–597.
Pigliucci, M. (2005). Evolution of phenotypic plasticity: Where are we going now? Trends in Ecology & Evolution, 20(9), 481–486.
Pine, A., Shiner, T., Seymour, B., & Dolan, R. J. (2010). Dopamine, time, and impulsivity in humans. The Journal of Neuroscience, 30(26), 8888–8896. https://doi.org/10.1523/JNEUROSCI.6028-09.2010.
Pollet, T. V., Tybur, J. M., Frankenhuis, W. E., & Rickard, I. J. (2014). What can cross-cultural correlations teach us about human nature? Human Nature, 25(3), 410–429. https://doi.org/10.1007/s12110-014-9206-3.
Pollock, D. (2002). Partible paternity and multiple maternity among the Kulina. In S. Beckerman & P. Valentine (Eds.), Cultures of multiple fathers: The theory of practice of partible paternity in lowland South America (pp. 42–61). Gainesville: University Press of Florida.
Puts, D. (2016). Human sexual selection. Current Opinion in Psychology, 7, 28–32. https://doi.org/10.1016/j.copsyc.2015.07.011.
Putt, S. S., Wijeakumar, S., Franciscus, R. G., & Spencer, J. P. (2017). The functional brain networks that underlie early stone age tool manufacture. Nature Human Behaviour, 1. https://doi.org/10.1038/s41562-017-0102.
Rantala, M. J., Luoto, S., Krams, I., & Karlsson, H. (2018). Depression subtyping based on evolutionary psychiatry: Proximate mechanisms and ultimate functions. Brain, Behavior, and Immunity, 69, 603–617. https://doi.org/10.1016/j.bbi.2017.10.012.
Reichman, O. J., & Smith, S. C. (1990). Burrows and burrowing behavior by mammals. Current Mammalogy, 2, 197–244.
Reinisch, J. M., Mortensen, E. L., & Sanders, S. A. (2017). Prenatal exposure to progesterone affects sexual orientation in humans. Archives of Sexual Behavior, 46(5), 1239–1249. https://doi.org/10.1007/s10508-016-0923-z.
Remigereau, C., Roy, A., Costini, O., Osiurak, F., Jarry, C., & Le Gall, D. (2016). Involvement of technical reasoning more than functional knowledge in development of tool use in childhood. Frontiers in Psychology, 7, 1625. https://doi.org/10.3389/fpsyg.2016.01625.
Reuter, M., Roth, S., Holve, K., & Hennig, J. (2006). Identification of first candidate genes for creativity: A pilot study. Brain Research, 1069(1), 190–197.
Rindermann, H., & Becker, D. (2018). Flynn-effect and economic growth: Do national increases in intelligence lead to increases in GDP? Intelligence, 69, 87–93. https://doi.org/10.1016/j.intell.2018.05.001.
Rindermann, H., Kodila-Tedika, O., & Christainsen, G. (2015). Cognitive capital, good governance, and the wealth of nations. Intelligence, 51, 98–108. https://doi.org/10.1016/j.intell.2015.06.002.
Roberts, S. C., Havlíček, J., Flegr, J., Hruskova, M., Little, A. C., Jones, B. C., ... Petrie, M. (2004). Female facial attractiveness increases during the fertile phase of the menstrual cycle. Proceedings of the Royal Society of London B: Biological Sciences, 271 Suppl 5, S270–S272. https://doi.org/10.1098/rsbl.2004.0174.
Robinson, M. R., Kleinman, A., Graff, M., Vinkhuyzen, A. A., Couper, D., Miller, M. B., ... Nolte, I. M. (2017). Genetic evidence of assortative mating in humans. Nature Human Behaviour, 1, 0016. https://doi.org/10.1038/s41562-016-0016.
Romer, P. M. (1990). Endogenous technological change. Journal of Political Economy, 98(5, Part 2), S71–S102.
Rosenfeld, M. J., & Thomas, R. J. (2012). Searching for a mate: The rise of the internet as a social intermediary. American Sociological Review, 77(4), 523–547.
Roth, T. C., & Pravosudov, V. V. (2009). Hippocampal volumes and neuron numbers increase along a gradient of environmental harshness: A large-scale comparison. Proceedings of the Royal Society of London B: Biological Sciences, 276(1656), 401–405. https://doi.org/10.1098/rspb.2008.1184.
Rubalcaba, J. G., Polo, V., Maia, R., Rubenstein, D. R., & Veiga, J. P. (2016). Sexual and natural selection in the evolution of extended phenotypes: The use of green nesting material in starlings. Journal of Evolutionary Biology, 29(8), 1585–1592. https://doi.org/10.1111/jeb.12893.
Said-Mohamed, R., Pettifor, J. M., & Norris, S. A. (2018). Life history theory hypotheses on child growth: Potential implications for short and long-term child growth, development and health. American Journal of Physical Anthropology, 165(1), 4–19. https://doi.org/10.1002/ajpa.23340.
Saltz, J. B., & Foley, B. R. (2011). Natural genetic variation in social niche construction: Social effects of aggression drive disruptive sexual selection in Drosophila melanogaster. The American Naturalist, 177(5), 645–654.
Sanz-Barbero, B., Linares, C., Vives-Cases, C., González, J. L., López-Ossorio, J. J., & Díaz, J. (2018). Heat wave and the risk of intimate partner violence. Science of the Total Environment, 644, 413–419. https://doi.org/10.1016/j.scitotenv.2018.06.368.
Schaedelin, F. C., & Taborsky, M. (2009). Extended phenotypes as signals. Biological Reviews, 84(2), 293–313. https://doi.org/10.1111/j.1469-185X.2008.00075.x.
Schaller, M. (2015). The behavioral immune system. In D. M. Buss (Ed.), The handbook of evolutionary psychology (2nd ed., pp. 1–17). Hoboken: Wiley Online Library.
Schinasi, L. H., & Hamra, G. B. (2017). A time series analysis of associations between daily temperature and crime events in Philadelphia, Pennsylvania. Journal of Urban Health, 94(6), 892–900. https://doi.org/10.1007/s11524-017-0181-y.
Schlomer, G. L., Del Giudice, M., & Ellis, B. J. (2011). Parent–offspring conflict theory: An evolutionary framework for understanding conflict within human families. Psychological Review, 118(3), 496–521. https://doi.org/10.1037/a0024043.
Schwarz, S., & Hassebrauck, M. (2012). Sex and age differences in mate-selection preferences. Human Nature, 23(4), 447–466. https://doi.org/10.1007/s12110-012-9152-x.
Sell, A., Lukazsweski, A. W., & Townsley, M. (2017). Cues of upper body strength account for most of the variance in men's bodily attractiveness. Proceedings of the Royal Society of London B: Biological Sciences, 284(1869), 20171819. https://doi.org/10.1098/rspb.2017.1819.
Shackelford, T. K., Schmitt, D. P., & Buss, D. M. (2005). Universal dimensions of human mate preferences. Personality and Individual Differences, 39(2), 447–458.
Shapiro, G. K., Tatar, O., Sutton, A., Fisher, W., Naz, A., Perez, S., & Rosberger, Z. (2017). Correlates of tinder use and risky sexual behaviors in young adults. Cyberpsychology, Behavior and Social Networking, 20(12), 727–734.
Shuler, G. A., & McCord, D. M. (2010). Determinants of male attractiveness: “Hotness” ratings as a function of perceived resources. American Journal of Psychological Research, 6(1), 10–23.
Sih, A., & Del Giudice, M. (2012). Linking behavioural syndromes and cognition: A behavioural ecology perspective. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 367(1603), 2762–2772. https://doi.org/10.1098/rstb.2012.0216.
Singh, D., & Bronstad, P. M. (2001). Female body odour is a potential cue to ovulation. Proceedings of the Royal Society of London B: Biological Sciences, 268(1469), 797–801. https://doi.org/10.1098/rspb.2001.1589.
Snell-Rood, E., Cothran, R., Espeset, A., Jeyasingh, P., Hobbie, S., & Morehouse, N. I. (2015). Life-history evolution in the anthropocene: Effects of increasing nutrients on traits and trade-offs. Evolutionary Applications, 8(7), 635–649. https://doi.org/10.1111/eva.12272.
Sng, O., Neuberg, S. L., Varnum, M. E. W., & Kenrick, D. T. (2017). The crowded life is a slow life: Population density and life history strategy. Journal of Personality and Social Psychology, 112(5), 736–754. https://doi.org/10.1037/pspi0000086.
Sng, O., Neuberg, S. L., Varnum, M. E., & Kenrick, D. T. (2018). The behavioral ecology of cultural psychological variation. Psychological Review, 125(5), 714–743. https://doi.org/10.1037/rev0000104.
Sniekers, S., Stringer, S., Watanabe, K., Jansen, P. R., Coleman, J. R., Krapohl, E., ... & Amin, N. (2017). Genome-wide association meta-analysis of 78,308 individuals identifies new loci and genes influencing human intelligence. Nature Genetics, 49, 1107–1112. https://doi.org/10.1038/ng.3869.
Socrata. (2017). Country list ISO 3166 codes latitude longitude. Internet database. Available at: https://opendata.socrata.com/dataset/Country-List-ISO-3166-Codes-Latitude-Longitude/mnkm-8ram/data. Retrieved on December 6, 2017.
Sol, D. (2009). Revisiting the cognitive buffer hypothesis for the evolution of large brains. Biology Letters, 5(1), 130–133. https://doi.org/10.1098/rsbl.2008.0621.
Sol, D., Sayol, F., Ducatez, S., & Lefebvre, L. (2016). The life-history basis of behavioural innovations. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 371(1690), 20150187. https://doi.org/10.1098/rstb.2015.0187.
Somveille, M., Rodrigues, A. S. L., & Manica, A. (2018). Energy efficiency drives the global seasonal distribution of birds. Nature Ecology & Evolution, 2, 962–969. https://doi.org/10.1038/s41559-018-0556-9.
Souza, A. L., Conroy-Beam, D., & Buss, D. M. (2016). Mate preferences in Brazil: Evolved desires and cultural evolution over three decades. Personality and Individual Differences, 95, 45–49. https://doi.org/10.1016/j.paid.2016.01.053.
Speakman, J. R. (2018). Response to ‘fat is not just an energy store’. Journal of Experimental Biology, 221(12), jeb184499. https://doi.org/10.1242/jeb.184499.
Stanyon, R., & Bigoni, F. (2014). Sexual selection and the evolution of behavior, morphology, neuroanatomy and genes in humans and other primates. Neuroscience & Biobehavioral Reviews, 46, 579–590. https://doi.org/10.1016/j.neubiorev.2014.10.001.
Stearns, S. C., Allal, N., & Mace, R. (2008). Life history theory and human development. In C. Crawford & D. Krebs (Eds.), Foundations of evolutionary psychology (pp. 47–69). New York: Lawrence Erlbaum Associates.
Steegmann, A. T., Cerny, F. J., & Holliday, T. W. (2002). Neandertal cold adaptation: Physiological and energetic factors. American Journal of Human Biology, 14(5), 566–583. https://doi.org/10.1002/ajhb.10070.
Steinberg, L., Graham, S., O’Brien, L., Woolard, J., Cauffman, E., & Banich, M. (2009). Age differences in future orientation and delay discounting. Child Development, 80(1), 28–44. https://doi.org/10.1111/j.1467-8624.2008.01244.x.
Steinberg, L., Icenogle, G., Shulman, E. P., Breiner, K., Chein, J., Bacchini, D., ... & Fanti, K. A. (2017). Around the world, adolescence is a time of heightened sensation seeking and immature self-regulation. Developmental Science. 00; e12532. https://doi.org/10.1111/desc.12532.
Sternberg, R. J. (2017). An alternative interpretation of climate data: Intelligence. Behavioral and Brain Sciences, 40, e96.
Stodden, D. F., Goodway, J. D., Langendorfer, S. J., Roberton, M. A., Rudisill, M. E., Garcia, C., & Garcia, L. E. (2008). A developmental perspective on the role of motor skill competence in physical activity: An emergent relationship. Quest, 60(2), 290–306. https://doi.org/10.1080/00336297.2008.10483582.
Stotz, K. (2017). Why developmental niche construction is not selective niche construction: And why it matters. Interface Focus, 7(5), 20160157. https://doi.org/10.1098/rsfs.2016.0157.
Sugiyama, L. S. (2015). Physical attractiveness: An adaptationist perspective. In D. M. Buss (Ed.), The handbook of evolutionary psychology (2nd ed., pp. 317–384). Wiley Online Library. https://doi.org/10.1002/9781119125563.evpsych112.
Sumter, S. R., Vandenbosch, L., & Ligtenberg, L. (2017). Love me tinder: Untangling emerging adults’ motivations for using the dating application tinder. Telematics and Informatics, 34(1), 67–78.
Sundie, J. M., Kenrick, D. T., Griskevicius, V., Tybur, J. M., Vohs, K. D., & Beal, D. J. (2011). Peacocks, Porsches, and Thorstein Veblen: Conspicuous consumption as a sexual signaling system. Journal of Personality and Social Psychology, 100(4), 664–680.
Tallavaara, M., Eronen, J. T., & Luoto, M. (2018). Productivity, biodiversity, and pathogens influence the global hunter-gatherer population density. Proceedings of the National Academy of Sciences, 115(6), 1232–1237. https://doi.org/10.1073/pnas.1715638115.
Templer, D. I., & Arikawa, H. (2006). Temperature, skin color, per capita income, and IQ: An international perspective. Intelligence, 34(2), 121–139. https://doi.org/10.1016/j.intell.2005.04.002.
The Atlas of Economic Complexity (2017). Country Complexity Rankings. Internet Database Available at: http://atlas.cid.harvard.edu/rankings/ Retrieved on December 6, 2017 and September 3, 2018.
Thornhill, R., & Gangestad, S. W. (2008). The evolutionary biology of human female sexuality. Oxford: Oxford University Press.
Thornhill, R., Gangestad, S. W., Miller, R., Scheyd, G., McCollough, J. K., & Franklin, M. (2003). Major histocompatibility complex genes, symmetry, and body scent attractiveness in men and women. Behavioral Ecology, 14(5), 668–678.
Thornhill, R., Fincher, C. L., Murray, D. R., & Schaller, M. (2010). Zoonotic and non-zoonotic diseases in relation to human personality and societal values: Support for the parasite-stress model. Evolutionary Psychology, 8(2), 147470491000800201.
Tifferet, S., Kruger, D. J., Bar-Lev, O., & Zeller, S. (2013). Dog ownership increases attractiveness and attenuates perceptions of short-term mating strategy in cad-like men. Journal of Evolutionary Psychology, 11(3), 121–129.
Timmermans, E., & Courtois, C. (2018). From swiping to casual sex and/or committed relationships: Exploring the experiences of tinder users. The Information Society, 34(2), 59–70.
Timmermans, E., & De Caluwé, E. (2017). To tinder or not to tinder, that's the question: An individual differences perspective to tinder use and motives. Personality and Individual Differences, 110, 74–79.
Toates, F. (2009). An integrative theoretical framework for understanding sexual motivation, arousal, and behavior. Journal of Sex Research, 46(2–3), 168–193.
Toma, C. L., & Hancock, J. T. (2010). Looks and lies: The role of physical attractiveness in online dating self-presentation and deception. Communication Research, 37(3), 335–351.
Torrence, R. (1983). Time budgeting and hunter-gatherer technology. In G. Bailey (Ed.), Hunter-gatherer economy in prehistory: A European perspective (pp. 11–22). Cambridge: Cambridge University Press.
Torrence, R. (2000). Hunter-gatherer technology: Macro- and microscale approaches. In C. Panter-Brick, R. H. Layton, & P. Rowley-Conwy (Eds.), Hunter-gatherers: An interdisciplinary perspective (pp. 99–143). Cambridge: Cambridge University Press.
Tskhay, K. O., Clout, J. M., & Rule, N. O. (2017). The impact of health, wealth, and attractiveness on romantic evaluation from photographs of faces. Archives of Sexual Behavior, 46, 2365–2376. https://doi.org/10.1007/s10508-017-0963-z.
UN Population Division (2018). World Population Prospects. Internet database. Available at: https://data.worldbank.org/indicator/sp.ado.tfrt. Retrieved on September 3, 2018.
UN Statistics Division (2017). World Development Indicators 2014. Internet database. Available at: http://data.un.org/Default.aspx. Retrieved on December 20, 2017.
Vaesen, K. (2012). The cognitive bases of human tool use. Behavioral and Brain Sciences, 35(4), 203–218.
Van de Vliert, E. (2009). Climate, affluence, and culture. New York: Cambridge University Press.
Van de Vliert, E. (2013). White, gray, and black domains of cultural adaptations to climato-economic conditions. Behavioral and Brain Sciences, 36(5), 503–521. https://doi.org/10.1017/S0140525X13000277.
Van de Vliert, E., & Murray, D. R. (2018). Climate and creativity: Cold and heat trigger invention and innovation in richer populations. Creativity Research Journal, 30(1), 17–28. https://doi.org/10.1080/10400419.2018.1411571.
Van Lange, P. A., Rinderu, M. I., & Bushman, B. J. (2017). Aggression and violence around the world: A model of CLimate, aggression, and self-control in humans (CLASH). Behavioral and Brain Sciences, 40. https://doi.org/10.1017/S0140525X16000406,e75.
Varnum, M. E. W., & Grossmann, I. (2017). Cultural change: The how and the why. Perspectives on Psychological Science, 12(6), 956–972. https://doi.org/10.1177/1745691617699971.
Vincze, O. (2016). Light enough to travel or wise enough to stay? Brain size evolution and migratory behavior in birds. Evolution, 70(9), 2123–2133. https://doi.org/10.1111/evo.13012.
Walasek, L., & Brown, G. D. (2015). Income inequality and status seeking: Searching for positional goods in unequal US states. Psychological Science, 26(4), 527–533. https://doi.org/10.1177/0956797614567511.
Walasek, L., Bhatia, S., & Brown, G. D. (2017). Positional goods and the social rank hypothesis: Income inequality affects online chatter about high and low status brands on twitter. Journal of Consumer Psychology, 28, 138–148. https://doi.org/10.1002/jcpy.1012.
Wang, M., Rieger, M. O., & Hens, T. (2016). How time preferences differ: Evidence from 53 countries. Journal of Economic Psychology, 52, 115–135.
Wang, G., Cao, M., Sauciuvenaite, J., Bissland, R., Hacker, M., Hambly, C., Vaanholt, L. M., Niu, C., Faries, M. D., & Speakman, J. R. (2018). Different impacts of resources on opposite sex ratings of physical attractiveness by males and females. Evolution and Human Behavior, 39(2), 220–225. https://doi.org/10.1016/j.evolhumbehav.2017.12.008.
Ward, T. B., & Kolomyts, Y. (2010). Cognition and creativity. In J. C. Kaufman & R. J. Sternberg (Eds.), The Cambridge handbook of creativity (pp. 93–112). New York: Cambridge University Press.
Wei, W., Lu, J. K., Galinsky, A. D., ... & Wang, L. (2017). Regional ambient temperature is associated with human personality. Nature Human Behaviour, 1, 890–895. https://doi.org/10.1038/s41562-017-0240-0.
Wells, J. C. (2006). The evolution of human fatness and susceptibility to obesity: An ethological approach. Biological Reviews, 81(2), 183–205. https://doi.org/10.1017/S1464793105006974.
Wells, J. C. (2007). Sexual dimorphism of body composition. Best Practice & Research Clinical Endocrinology & Metabolism, 21(3), 415–430. https://doi.org/10.1016/j.beem.2007.04.007.
Wells, J. C. (2012). Sexual dimorphism in body composition across human populations: Associations with climate and proxies for short-and long-term energy supply. American Journal of Human Biology, 24(4), 411–419. https://doi.org/10.1002/ajhb.22223.
Wells, D. A. (2015). The extended phenotype(s): A comparison with niche construction theory. Biology and Philosophy, 30(4), 547–567.
Wells, J. C., Nesse, R. M., Sear, R., Johnstone, R. A., & Stearns, S. C. (2017). Evolutionary public health: Introducing the concept. The Lancet, 390(10093), 500–509. https://doi.org/10.1016/S0140-6736(17)30572-X.
West-Eberhard, M. J. (2014). Darwin's forgotten idea: The social essence of sexual selection. Neuroscience & Biobehavioral Reviews, 46, 501–508.
Widemo, F., & Sæther, S. A. (1999). Beauty is in the eye of the beholder: Causes and consequences of variation in mating preferences. Trends in Ecology & Evolution, 14(1), 26–31.
Williams, G. C. (1966). Adaptation and natural selection: A critique of some current evolutionary thought. Princeton: Princeton University Press.
Wilson, M. L., Miller, C. M., & Crouse, K. N. (2017). Humans as a model species for sexual selection research. Proceedings of the Royal Society of London B: Biological Sciences, 284, 20171320. https://doi.org/10.1098/rspb.2017.1320.
Winegard, B., Winegard, B., & Geary, D. C. (2018). The status competition model of cultural production. Evolutionary Psychological Science, 4, 1–21. https://doi.org/10.1007/s40806-018-0147-7.
Woodley of Menie, M. A., Cabeca de Baca, T., Fernandes, H. B., Madison, G., Figueredo, A. J., & Aguirre, M. P. (2017). Slow and steady wins the race: K positively predicts fertility in the USA and Sweden. Evolutionary Psychological Science, 3(2), 109–117. https://doi.org/10.1007/s40806-016-0077-1.
Worthman, C. M., & Trang, K. (2018). Dynamics of body time, social time and life history at adolescence. Nature, 554(7693), 451–457. https://doi.org/10.1038/nature25750.
Wright, J., Bolstad, G. H., Araya-Ajoy, Y. G., & Dingemanse, N. J. (2018). Life-history evolution under fluctuating density-dependent selection and the adaptive alignment of pace-of-life syndromes. Biological Reviews. https://doi.org/10.1111/brv.12451.
Zietsch, B. P., Verweij, K. J., & Burri, A. V. (2012). Heritability of preferences for multiple cues of mate quality in humans. Evolution, 66(6), 1762–1772.
Acknowledgements
This work was supported by the Otto A. Malm foundation, The University of Auckland Doctoral Scholarship, and the Emil Aaltonen Young Researcher Grant. The author is grateful for Brian Boyd’s and Quentin Atkinson’s meticulous criticism, Markus J. Rantala’s guidance, and for Oliver Sheehan, Ania Grant, and Patrick Neilands for their valuable comments on earlier drafts. The author wishes to thank the University of Auckland Language, Culture and Cognition group for several thoughtful discussions on the topic. The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Allele
-
each of two or more alternative forms of a gene found in the same genetic locus.
- Atmospheric cold demands
-
a climatic variable that is coded as the sum of the absolute downward deviations from 22 °C for the average lowest temperature in the coldest month, the average highest temperature in the coldest month, the average lowest temperature in the hottest month, and the average highest temperature in the hottest month (Van de Vliert 2013).
- Behavioral ecology
-
studies the evolutionary basis for behavior shaped by ecological pressures such as climate, habitat characteristics, and conspecific interactions.
- Cognitive buffer hypothesis
-
suggests that the primary adaptive function of a large brain is to buffer individuals against environmental challenges by facilitating the construction of behavioral responses (Sol 2009).
- Conspecifics
-
are members of the same species; heterospecifics are members of a different species.
- Cues
-
can provide information to other organisms, but they were not evolutionarily selected for the specific purpose of conveying information. Many signals have nevertheless evolved from cues through the evolutionary process of signaller precursor route: signals can evolve from behaviors or structures that originally were informative aspects of the signaller and then became specialized over evolutionary time to convey information more effectively (Laidre and Johnstone 2013). It can be challenging to establish what is a cue and what is a signal (Sugiyama 2015) especially in contemporary human sexual selection on extended phenotypic traits. This is why the current work (mostly) refrains from discussing extended phenotypic traits as signals and instead refers to them simply as cues or traits.
- Ecological inheritance
-
refers to ecological legacies passed on to future generations that are generated by niche construction activities of past generations.
- Economic Complexity Index (ECI)
-
a holistic measure of the knowledge, industrial composition, and production characteristics of a country.
- Exaptation
-
a feature that enhances fitness in a way that differs from its “original” evolutionarily selected role; i.e. it is a trait that currently has a positive fitness effect without being an adaptation specifically for that effect (Buss et al. 1998; Gould and Vrba 1982).
- Extended phenotypes
-
are a part of the phenotype expressed beyond the body. In simplified terms, and not taking into account the polygenic architecture underlying the creation or manipulation of extended phenotypes, they result from the effects of an allele upon the world that have a bearing on that allele’s survival or proliferation (Dawkins 2004). This includes any feature of the abiotic or biotic environment that is the result of manipulative action or construction behavior of an individual, but excludes any result of such manipulative behavior that does not affect the organism’s fitness negatively or positively (Dawkins 1982; Schaedelin and Taborsky 2009). This definition therefore includes the dam that a beaver builds, but not the footprints it makes when building it (Dawkins 1982, p. 315). Both result from the action of an organism upon the world, but only the dam has a fitness implication to the beaver. Extended phenotypes can be divided into inanimate and animate ones.
- Extended phenotypic plasticity
-
refers to the process of extended phenotypes adapting to the demands of the environment, interacting with it, and gaining feedback from it, thereby creating phenotype–environment interactions and extended phenotype–environment interactions that flow through generations (Blamires 2010; Lehmann 2008).
- Functional traits
-
are the morphological, physiological, phenological, and behavioral characteristics of an organism that have an influence on performance or fitness (Meunier et al. 2017).
- Kin selection
-
refers to any variation in indirect fitness among different phenotypes caused by their ability to influence the reproductive success and survival of genetically similar individuals.
- Natural selection
-
is the sum total of viability selection, sexual selection, and kin selection (Cornwallis and Uller 2010).
- Niche construction
-
is a theoretical framework that studies the ways in which organisms choose and modify components of their local environments.
- Phenotype
-
is the composite of an organism’s observable physical or biochemical features (as determined by genetic makeup and environmental influences) (Bolondi et al. 2017). Cognitive traits such as intelligence are classified as phenotypic traits in this article. Thus, phenotypic traits can be further divided into somatic (physical) and cognitive (psychological) traits (e.g. Miller and Todd 1998). Much literature exists on behavioral phenotypes; for reasons of theoretical accuracy and empirical distinction, however, behavioral traits are here distinguished from phenotypic traits. Likewise, though extended phenotypic traits could be seen as a subcategory of phenotypic traits, they are treated as a separate category because they do not fulfil the orthodox definition of a phenotype.
- Phenotypic plasticity
-
refers to the ability of a single genotype to produce multiple phenotypes when exposed to different environments.
- Secondary adaptations
-
(sometimes called post-adaptations) are adaptive changes that an exaptation undergoes to become better suited to its evolutionarily novel function.
- Secondary sexual signals
-
are sexually selected traits that give an organism an advantage over its rivals in mate choice without being directly involved in reproduction. Examples in humans include enlarged breasts and muscles.
- Sexual selection
-
is defined as any variation in direct fitness among different phenotypes caused by their ability to gain sexual partners, produce fertilized eggs, and generate offspring.
- Signals
-
are traits that were evolutionarily selected for because they conveyed information about the signaller, affecting the behavior of other organisms and ultimately impacting the fitness of both the signaller and the recipient (Laidre and Johnstone 2013).
- Trait-based ecology
-
provides a functional approach to understanding ecological interactions by focusing on the expression of functional traits under distinct environmental conditions (Meunier et al. 2017).
- Viability selection
-
refers to any variation in direct fitness among different phenotypes caused by their ability to survive.
Rights and permissions
About this article
Cite this article
Luoto, S. An Updated Theoretical Framework for Human Sexual Selection: from Ecology, Genetics, and Life History to Extended Phenotypes. Adaptive Human Behavior and Physiology 5, 48–102 (2019). https://doi.org/10.1007/s40750-018-0103-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40750-018-0103-6