Abstract
The objective of this study was to evaluate the corrosion kinetics and microstructural change of a Ti–6Al–4V alloy with machined and treated surfaces (sandblasted with aluminum oxide or double acid-etched) at three salivary pH levels. For an electrochemical test (n = 5), open-circuit potential and electrochemical impedance spectroscopy analyses were conducted in artificial saliva at pH 3, 6.5, and 9 in the three types of Ti–6Al–4V surfaces. The capacitance (CPE) and the polarization resistance (R p) of the titanium oxide layer were determined. Disk surfaces were characterized using scanning electron microscopy, atomic force microscopy, and profilometry, which provided the surface roughness (Ra) of the disks before (baseline) and after the electrochemical test. The corrosion parameters were compared by two-way ANOVA. One-way ANOVA was applied for analyzing Ra values of the specimens before and after the electrochemical process. For results that were statistically significant, the Tukey HSD test was used. The level of significance used for all tests was 0.05. The highest value of R p was observed on disks with machined surfaces and treated with double acid-etching (p = 0.264), which was independent of the salivary pH level. Sandblasting decreased the R p of Ti–6Al–4V at all pH levels (p < 0.05). The machined surface showed the lowest CPE, followed by the surfaces treated with double acid-etching, and the highest values were observed for the sandblasting (p < 0.05), which were all independent of the pH. It was concluded that a higher corrosion process was observed in the Ti–6Al–4V alloys surface sandblasted with aluminum oxide. Acidic pH tended to reduce total impedance values. Acid-etching may be a suitable treatment for dental implants as it provides good corrosion resistance and increased contact area for bone adhesion.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Titanium (Ti) has essential properties for osseointegration, such as good biocompatibility and good mechanical strength [1–4]. However, situations harmful to biomaterials such as pH changes, the presence of fluoride, thermal, chemical, or mechanical changes, the presence of saliva, and plaque may degrade the oxide layer, releasing ions both externally and internally, as in cases of corrosion of the implant surface [5, 6].
In the oral cavity, various situations promote acidification or alkalinization of the salivary pH [7]. Foods such as milk, nuts, and those with higher concentrations of calcium increase the pH of saliva, whereas sweetened foods, sweets, fruit juices, and soft drinks can decrease salivary pH [7, 8]. These situations increase the corrosion rate of dental implants [5, 6, 9].
Metals such as Ti are highly toxic to the human body and can cause irreversible damage when they are released into the bloodstream [10–12]. In this context, studies on peri-implant mucosa and cadavers [13–18] showed particles of Ti in the peri-implant region and in organs such as the liver, kidney, spleen, and lymph nodes next to the implants or some distance away [12, 19].
In recent decades, dentistry has developed topographic and physical–chemical surface modifications of dental implants [20–23], mainly seeking increased mechanical strength and increased bone-implant contact percentage, particularly in areas of low bone density [20–23]. Several techniques for the surface treatment of Ti implants are commercially available and are described in the literature. The key processes are techniques that promote implant surfaces through addition [coating with titanium plasma spray and hydroxyapatite (HA), HA and other calcium phosphate coatings, deposition of ions [24–26] or subtraction [creating pores and pits on the surface of the implant through electropolishing, mechanical polishing, treatment with acid, associated with the sandblasting of titanium oxide (TiO2) or aluminum oxide (Al2O3) oxidation, and laser irradiation [27, 28].
The electrochemical stability of Ti subjected to different surface treatments is still being debated in the literature. However, it is known that changes in salivary pH influence the corrosion behavior of Ti with machined surfaces [9]. Barao et al. [29] investigated the corrosion properties of machined Ti in the presence of pathogenic components present in the walls of gram-negative bacteria [lipopolysaccharide (LPS) in artificial saliva with three levels of pH: acidic pH 3, neutral pH 6.5, and basic pH 9]. LPS and pH affected the corrosion behavior of Ti. In general, low pH (pH 3) and the highest concentration of LPS accelerated the corrosion process of Ti. Increasing pH significantly decreased the corrosion rate and increased ion exchange between the environment and Ti. Therefore, it is necessary to investigate the effect of different surface modifications on the electrochemical stability of Ti with different levels of salivary pH, since in a previous study, the acidic pH showed higher corrosion potential in machined surfaces [29].
The aim of this study was to evaluate the corrosion kinetics and microstructural changes of a Ti–6Al–4V alloy with machined and treated surfaces (sandblasted with aluminum oxide or double acid-etched) at three salivary pH levels (3, 6.5, and 9). The null hypothesis is that neither surface treatment nor salivary pH would have an effect on the electrochemical behavior of the Ti–6Al–4V alloy.
2 Materials and Methods
Forty-five disks of a Ti–6Al–4V alloy were fabricated. The disks were 15 mm in diameter and 2-mm thick. The specimens were divided into 9 groups (n = 5) according to the surface type (three levels) and salivary pH (three levels), as detailed in Table 1.
2.1 Preparation of the Ti Disks
The disks were polished and cleaned using standard metallographic methods [9, 29–31]. The polishing was performed in an automatic polisher (ECOMET 250PRO/AUTOMET 250, Buehler, Lake Bluff, IL, USA) with sandpaper (#320, #400, #600, and #800) (Carbimet 2, Buehler, Lake Bluff, IL, USA). The final polishing was done with diamond paste (MetaDi 9-micron, Buehler, Lake Bluff, IL, USA) in a polishing cloth (Polishing Cloth TextMet, Buehler, Lake Bluff, IL, USA) and a colloidal silica (MASTERMED, Buehler, Lake Bluff, IL, USA) rubber cloth disk (Chemomet I Buheler, Lake Bluff, IL, USA) until the surfaces of the Ti–6Al–4V disks were reflective.
2.2 Surface Treatments
From a total of 45 polished Ti disks, 15 disks were randomly selected for the machined surface group. The others were subjected to surface modification according to market availability. The disks were chemically treated using three acids (nitric, sulfuric, and hydrochloric acids) (n = 15) according to the Military Institute of Engineering – IME (Rio de Janeiro, Brazil) protocol or sandblasted with aluminum oxide (Al2O3) (n = 15) according to Emfils Implants (Itu, Sao Paulo, Brazil) standards.
2.3 Electrochemical Test
The electrochemical test was performed according to previously published protocols [9, 29, 31, 32]. A polysulfone electrochemical cell with a three-electrode cell was used to measure the corrosion parameters, following the guidelines of the American Society for Testing Materials (ASTM) (G61 and G31-72). For each electrochemical test, 10 mL of artificial saliva at three pH levels (3, 6.5, and 9) was used as the electrolyte. The artificial saliva composition was KCl (0.4 g/L), NaCl (0.4 g/L), CaCl2·2H2O (0.906 g/L), NaH2PO4·2H2O (0.690 g/L), (Na2S·9H2O g/L) (0.005 g/L), and urea (1 g/L) according to Vieira et al. [4]. Ti–6Al–4V alloy disks were subjected to a cathodic potential (−0.9 V vs. SCE) to standardize the oxide layer formed on the metal surface. The open-circuit potential was monitored for 3600 s to analyze the potential of the material into the solution and to stabilize the system.
Electrochemical impedance spectroscopy (EIS) was used to evaluate the electrochemical formation of the oxide layer on the surfaces of the Ti–6Al–4V disks and the properties of this oxide layer (corrosion kinetics). EIS measurements were performed at frequencies between 100 kHz and 5 MHz at an AC amplitude of 10 mV applied to the electrode [9, 29, 31]. These values were used to determine the real (Z′) and imaginary (Z″) components of impedance for the Nyquist plot or total impedance (|Z|) and phase angle. Echem Analyst software (Gamry Instruments Warminster, PA, USA) was used to fit the EIS data using a modified Randle’s circuit. In this circuit, the capacitance was represented by a constant phase element (CPE) due to the inhomogeneous passive layer at the material surface. Capacitive behavior can be related with the existence of an electrochemical double layer on the material/electrolyte interface and the dielectric nature of TiO2 [32]. A Chi-square value less than 0.01 was obtained as it indicates excellent agreement between experimental data and the fitted values [33]. Oxide film properties such as CPE and the polarization resistance (R p) were quantified.
2.4 Microstructural Assessment
2.4.1 Microstructural Characterization: SEM and AFM
To characterize the surfaces of the Ti–6Al–4V alloy disks, images of the disks were obtained using scanning electron microscopy (SEM, JEOL JSM-7401F, Oregon, USA). The images before and after corrosion were compared in order to analyze the different surfaces of the disks and salivary pH levels.
The three-dimensional analysis was performed using atomic force microscopy (AFM, Veeco Metrology Inc., Santa Barbara, CA, USA). The images from the microscope were transferred to a microcomputer and analyzed using the Analysis Nanoscope software (2004 Veeco Instruments Inc., Santa Barbara, CA, USA). Finally, three-dimensional images were obtained to allow a comparative analysis between the experimental groups.
2.4.2 Surface Roughness
Superficial changes of the Ti–6Al–4V alloy promoted by the corrosion process were measured by average roughness values (Ra, arithmetic average surface roughness). A profiler (Dektak d-150; Veeco, Plainview, New York, USA) was used, and the measurements were carried out in two stages, before (baseline) and after the electrochemical tests [30, 31]. The Ra values were obtained using a 500 μm cut-off and a time constant of 12 s. Three readings were taken on each surface, and the average was calculated. Random readings were taken from the center of the specimen, as well as two parallel readings, to the right and left of center [30, 31].
2.5 Statistical Analysis
The R p and corrosion CPE parameters were compared by two-way ANOVA, factor 1: surface of the disks (machined, double acid-etched, and aluminum oxide sandblasted) and factor 2: pH of artificial saliva (3, 6.5, and 9). The Tukey HSD test was applied as a post hoc technique. One-way ANOVA was used for the analysis of the surface roughness (Ra) of the specimens before and after the corrosion process. The level of significance used for all tests was 0.05.
3 Results
3.1 Electrochemical Data
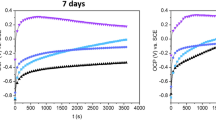
The Bode plot representing the corrosion kinetics of Ti–6Al–4V showed a constant phase angle in all cases. Different phase angle values were noted for the machined surface (approximately 90°), the acid-etched surface (approximately 80°), and the sandblasted surface (approximately 70°). At low frequency, the total impedance values, |Z|, were higher for the machined and etched groups as compared to the sandblasted group, regardless of salivary pH conditions (Fig. 1). Acidic pH tended to reduce total impedance values. The Nyquist plot showed that the sandblasted surface decreased the diameter of the semicircle of the total impedance plot of Ti–6Al–4V (Fig. 2). At pH 3, there was a slight reduction in the diameter of the semicircle as compared to pH 6.5.
Electrochemical impedance spectroscopy (EIS) represented by the Bode plot of the Ti-6Al-4V alloy for acid-etching, sandblasting and machined surface in three pH levels (3, 6.5 and 9). Different phase angle values were noted for the machined surface (approximately 90°), the acid-etched surface (approximately 80°), and the sandblasted surface (approximately 70°)
R p was influenced only by the surface condition of Ti–6Al–V (p = 0.023, ANOVA) (Table 2). The specimens with the machined surface and treated with double acid-etching showed the highest values of R p, regardless of salivary pH (p = 0.264, Tukey HSD test). At pH 3, R p had a tendency to increase for etched Ti–6Al–4V, whereas at pH 6.5 and 9, the machined disks had a higher R p (Fig. 3). The treatment with aluminum oxide decreased the R p value of Ti–6Al–4V at all pH levels (p < 0.05, Tukey HSD).
In relation to the CPE, the surface treatment condition exhibited a statistically significant effect (p < 0.001, ANOVA) (Table 3). In all interactions (sandblasted vs. machined, sandblasted vs. etched, and machined vs. etched), there was a significant change in CPE (p < 0.05, Tukey HSD test) (Table 3; Fig. 4). The machined surfaces had the lowest CPE, which were independent of pH, followed by the disks treated with double acid-etching; the greatest CPE values were observed for the sandblasted surfaces. These data corroborate the results of the Bode plots and Nyquist plots (Figs. 1, 2).
Means and standard deviations of constant phase element (CPE) of the Ti-6Al-4V alloy for acid etching, sandblasting and machined surface in three pH levels (3, 6.5 and 9). Machined surfaces had the lowest CPE, which were independent of pH, followed by the disks treated with double acid-etching; the greatest CPE values were observed for the sandblasted surfaces
3.2 Microstructure Data
The SEM images (Fig. 5) before and after electrochemical assay showed smooth surfaces for the machined samples, good adhesion of the oxide layer on the sandblasted samples, film integrity, and homogeneous morphology. The machined surfaces after electrochemical tests in artificial saliva at pH 3 and 6.5 showed some black spots as a result of artifacts. The sandblasted samples exhibited visible topographic alteration, with the attachment of Al2O3 particles of standard shape and size. The acid-etched samples exhibited uniformly pitted surfaces.
Figure 6 exhibits the AFM images of Ti–6Al–4V before and after corrosion. The machined surfaces were smooth for all conditions. Sandblasting and acid-etching treatments created peaks and valleys on the Ti surface.
3.3 Surface Roughness Data
Figure 7 shows the mean values of Ra. Although the surface treatment had a significant influence (p < 0.0001, ANOVA), the corrosion process had no effect on the Ra values (p = 0.284, ANOVA). The machined surfaces exhibited the lowest Ra values before and after corrosion. Similar Ra values were noted for the sandblasted and acid-etched samples.
4 Discussion
The electrochemical behavior of the Ti–6Al–4V alloy was only affected by different surface treatments (p < 0.05). Given these results, the null hypothesis that the electrochemical behavior was similar between the different variables (pH levels and surface treatment) was rejected.
Surprisingly, salivary pH had no effect on the corrosion kinetics of the Ti–6Al–4V alloy. The corrosion behavior was very similar among the analyzed pH levels (3, 6.5, and 9), as seen from the observed values of R p and CPE (Figs. 3, 4). It is clear that the surface of the Ti–6Al–4V alloy promoted oscillations in the material corrosion rate. Previously published studies have shown a higher rate of corrosion in acidic saliva (pH 3) [9, 34, 35]. However, these studies evaluated Ti–6Al–4V disks with mirror-like machined surfaces with no changes in microstructural topography. Even if the results of our study did not show statistically significant changes for the salivary pH, the machined surfaces showed a trend toward higher capacitance values and lower polarization resistance values in acidic saliva. CPE refers to the ions exchange between the material and the environment, whereas R p indicates the protectiveness of the material’s surface against corrosion. The higher the CPE value and the lower the R p value, the greater the corrosion of the material.
With respect to the corrosion kinetics, the surfaces of Ti–6Al–4V sandblasted with Al2O3 had the lowest values of R p and higher CPE values. Additionally, the Nyquist plot showed that this surface decreased the diameter of the semi-circular “loop” capacitance for all situations tested, indicating poor corrosion resistance. Surfaces with a larger Nyquist plot diameter are fitted to circuits with a larger R p. Surfaces with a larger R p have better corrosion resistance and decreased corrosion rates. Surfaces with a higher CPE have an electrical double layer with higher capacitance, promoting higher corrosion rates. Thus, the sandblasted Ti–6Al–4V alloy exhibited poor corrosion behavior as compared to the etched and machined samples. This may have been due an increased surface area and the presence of Al2O3 particles, which can decrease the corrosion resistance through passive layer weakening, as observed by Burnat et al. [36].
According to previous studies [37, 38], the sandblasting procedure prepares the titanium surface more rigorously than polishing (machined surface), creating surfaces with increased activity. This rigorous surface preparation on the titanium substrate generates a surface oxide layer with reduced integrity and thickness [39]. Such a mechanism may be the driving force for the poor corrosion resistance of sandblasted samples in the present study. Energy dispersive spectroscopy analysis showed that the content of titanium (in wt%) decreased from 96 to 33 as compared to the machined versus sandblasted samples (data not shown). Based on that, it could be estimated that approximately 60 % of the surface area of the sandblasted samples had Al2O3 embedded particles.
The modification of the titanium surface is a major factor in osseointegration owing to the higher adhesion property of osteoblast cells to surfaces with a larger contact area [20–23]. However, other factors such as corrosion resistance must be considered in order to achieve successful treatment. Corrosion promotes the release of titanium ions into the bloodstream [10–12], promoting damage in metabolizing organs [12, 19]. Additionally, corrosion causes greater adhesion of microorganisms in dental implants, predisposing the patient to peri-implantitis [31, 43, 44]. Thus, acid-etching may be a suitable treatment for dental implants as it provides increased contact area for bone adhesion and good corrosion resistance in the oral environment.
Given the various techniques for modifying the microstructure of the Ti surface, some may fail during technical processing, resulting smaller microstructural homogeneity. Thus, the mechanical stress caused during the installation of the implants, especially in bones of higher density, can promote the shedding of Ti particles to the peri-implant region, inducing a foreign-body reaction as noted by Tavares et al. [23]. This detachment will expose the surfaces of implants and increase the tendency to corrosion. Another implication proposed is the surface weakness caused by etching-acid technique that can promote electrochemical reactions in oral environment, increasing implant corrosion.
In this context, another important factor to be noted in the results of this work is the clinical implication of the exchange of Ti ions with the electrolyte environment. In addition to the microstructural changes, the release of Ti ions, Al, and V to the peri-implant region may cause greater damage at the systemic level. These metals are highly toxic to the body, with the potential to cause damage in metabolizing organs and lead to neurological disorders [12, 19]. Reviews of the peri-implant mucosa have shown particles of Ti with foreign-body reaction, as evidenced by macrophages, including some particles and other scattered in the tissue [15–17, 40]. The question remains whether the foreign-body reaction is capable of engulfing the Ti particles in the peri-implant environment, or whether there will be metabolic changes in vital organs such as the liver and kidney and neurological disorders, depending on the degree of metal poisoning.
The large R p for surfaces conditioned with the double acid-etching, even in acidic pH, was an unexpected result. It was expected that a higher R p would be observed in the etched surface than in the machined surface. However, these data were not consistent with the CPE values, wherein the etched disks showed a higher CPE than the machined disks. It was observed that at least initially, most of the metals conditioned with double acid-etching were resistant to attack from an electrolytic environment, although the reason for this is unclear. Over time, the oxide layer that forms on the surface of the Ti–6Al–4V alloy, which is porous and has irregularities, may be degraded, thus, increasing the ability to exchange ions with the electrolyte environment (greater CPE values) [33, 34, 41, 42].
The Ra data were not affected by the corrosion process, and no changes were observed in the AFM and SEM images. It is important to note that the release of Ti ions in the corrosion process may not cause a significant increase in Ra. Another factor to consider is the immersion time of the Ti–6Al–4V surface in acid pH and its clinical implications. It is thought that the immersion time (approximately 2.5 h) was too short to cause changes on the Ti microstructure. Additionally, monitoring the open-circuit potential and then performing EIS analysis are not generally expected to change the surface very much for passive alloys. Future studies should investigate the corrosion behavior of titanium material subjected to long-term immersion, thus simulating acidic diets or systemic changes that would modify the salivary pH for a long period of time.
From the results obtained in this study, it is evident that there is a need for future studies investigating different techniques to treat the surface of Ti dental implants in order to improve not only their corrosion stability but also their osseointegration potential. It will reduce the possibility of particle detachment for the peri-implant region and the Ti toxicity, thus reducing failures of dental implants.
5 Conclusions
Within the limitations of this in vitro study, it can be concluded that
-
(1)
Higher corrosion kinetics were observed in the Ti–6Al–4V alloy treated with Al2O3 sandblasting.
-
(2)
Acid-etching may be a suitable treatment for dental implants as it provides good corrosion resistance and increased contact area for bone adhesion.
-
(3)
The pH of saliva had no effect on the electrochemical stability of the Ti–6Al–4V alloy for all surface conditions (machined, sandblasted, and etched).
References
Cortada M, Giner L, Costa S, Gil FJ, Rodriguez D, Planell JA (2000) Galvanic corrosion behavior of titanium implants coupled to dental alloys. J Mater Sci Mater Med 11:287–293
Schiff N, Grosgogeat B, Lissac M, Dalard F (2002) Influence of fluoride content and pH on the corrosion resistance of titanium and its alloys. Biomaterials 23(9):1995–2002
Mabboux F, Ponsonnet L, Morrier JJ, Jaffrezic N, Barsotti O (2004) Surface free energy and bacterial retention to saliva-coated dental implant materials–an in vitro study. Colloids Surf B 39:199–205
Vieira AC, Ribeiro AR, Rocha LA, Celis JP (2006) Influence of pH and corrosion inhibitors on the tribocorrosion of titanium in artificial saliva. Wear 261(9):994–1001
Nikolopoulou F (2006) Saliva and dental implants. Implant Dent 15:372–376
Correa CB, Pires JR, Fernandes-Filho RB, Sartori R, Vaz LG (2009) Fatigue and fluoride corrosion on Streptococcus mutans adherence to titanium-based implant/component surfaces. J Prosthodont 18:382–387
Murrell S, Marshall TA, Moynihan PJ, Qian F, Wefel JS (2010) Comparison of in vitro erosion potentials between beverages available in the United Kingdom and the United States. J Dent 38(4):284–289
Dong YM, Pearce EI, Yue L, Larsen MJ, Gao XJ, Wang JD (1999) Plaque pH and associated parameters in relation to caries. Caries Res 33(6):428–436
Barao VA, Mathew MT, Assuncao WG, Yuan JC, Wimmer MA, Sukotjo C (2012) Stability of cp-Ti and Ti-6Al-4V alloy for dental implants as a function of saliva pH—an electrochemical study. Clin Oral Implants Res 23(9):1055–1062
Mjoberg B, Hellquist E, Mallmin H, Lindh U (1997) Aluminum, Alzheimer’s disease and bone fragility. Acta Orthop Scand 68(6):511–514
Karrholm J, Frech W, Nivbrant B, Malchau H, Snorrason F, Herberts P (1998) Fixation and metal release from the Tifit femoral stem prosthesis. 5-year follow-up of 64 cases. Acta Orthop Scand 69(4):369–378
Jacobs JJ, Skipor AK, Patterson LM, Hallab NJ, Paprosky WG, Black J et al (1998) Metal release in patients who have had a primary total hip arthroplasty. A prospective, controlled, longitudinal study. J Bone Joint Surg Am 80(10):1447–1458
Lalor PA, Revell PA, Gray AB, Wright S, Railton GT, Freeman MA (1991) Sensitivity to titanium. A cause of implant failure? J Bone Joint Surg Br 73(1):25–28
Matthew IR, Frame JW, Browne RM, Millar BG (1996) In vivo surface analysis of titanium and stainless steel miniplates and screws. Int J Oral Maxillofac Surg 25(6):463–468
Olmedo D, Guglielmotti MB, Cabrini RL (2002) An experimental study of the dissemination of titanium and zirconium in the body. J Mater Sci Mater Med 13:793–796
Olmedo DG, Paparella ML, Brandizzi D, Cabrini RL (2010) Reactive lesions of peri-implant mucosa associated with titanium dental implants: a report of 2 cases. Int J Oral Maxillofac Surg 39:503–507
Olmedo DG, Paparella ML, Spielberg M, Brandizzi D, Guglielmotti MB, Cabrini RL (2012) Oral mucosa tissue response to titanium cover screws. J Periodontol 83(8):973–980
Flatebo RS, Hol PJ, Leknes KN, Kosler J, Lie SA, Gjerdet NR (2011) Mapping of titanium particles in peri-implant oral mucosa by laser ablation inductively coupled plasma mass spectrometry and high-resolution optical darkfield microscopy. J Oral Pathol Med 40(5):412–420
Urban RM, Jacobs JJ, Tomlinson MJ, Gavrilovic J, Black J, Peoc’h M (2000) Dissemination of wear particles to the liver, spleen, and abdominal lymph nodes of patients with hip or knee replacement. J Bone Joint Surg Am 82(4):457–476
Wennerberg A, Albrektsson T (2008) Effects of titanium surface topography on bone integration: a systematic review. Clin Oral Implants Res 20(4):172–184
Xavier SP, Carvalho PS, Beloti MM, Rosa AL (2003) Response of rat bone marrow cells to commercially pure titanium submitted to different surface treatments. J Dent 31:173–180
Buser D, Broggini N, Wieland M, Schenk RK, Denzer AJ, Cochran DL et al (2004) Enhanced bone apposition to a chemically modified SLA titanium surface. J Dent Res 83(7):529–533
Tavares MG, de Oliveira PT, Nanci A, Hawthorne AC, Rosa AL, Xavier SP (2007) Treatment of a commercial, machined surface titanium implant with H2SO4/H2O2 enhances contact osteogenesis. Clin Oral Implants Res 18:452–458
Gotfredsen K, Berglundh T, Lindhe J (2001) Bone reactions adjacent to titanium implants with different surface characteristics subjected to static load. A study in the dog (II). Clin Oral Implants Res 12(3):196–201
Lin A, Wang CJ, Kelly J, Gubbi P, Nishimura I (2009) The role of titanium implant surface modification with hydroxyapatite nanoparticles in progressive early bone-implant fixation in vivo. Int J Oral Maxillofac Implants 24(5):808–816
Wennerberg A, Albrektsson T (2009) Effects of titanium surface topography on bone integration: a systematic review. Clin Oral Implants Res 20(4):172–184
Trisi P, Lazzara R, Rebaudi A, Rao W, Testori T, Porter SS (2003) Bone-implant contact on machined and dual acid-etched surfaces after 2 months of healing in the human maxilla. J Periodontol 74(7):945–956
Pai PS, Mathew MT, Stack MM, Rocha LA (2008) Some thoughts on neural network modelling of microabrasion-corrosion processes. Tribol Int 41(7):672–681
Barao VA, Mathew MT, Assuncao WG, Yuan JC, Wimmer MA, Sukotjo C (2011) The role of lipopolysaccharide on the electrochemical behavior of titanium. J Dent Res 90(5):613–618
Faverani LP, Barao VA, Ramalho-Ferreira G, Ferreira MB, Garcia-Junior IR, Assuncao WG (2013) Effect of bleaching agents and soft drink on titanium surface topography. J Biomed Mater Res B 102:22–30
Faverani LP, Assuncao WG, de Carvalho PS, Yuan JC, Sukotjo C, Mathew MT et al (2014) Effects of dextrose and lipopolysaccharide on the corrosion behavior of a Ti-6Al-4V alloy with a smooth surface or treated with double-acid-etching. PLoS ONE 9(3):e93377
Mohedano M, Matykina E, Arrabal R, Pardo A, Merino MC (2014) Metal release from ceramic coatings for dental implants. Dent Mater 30:e28–e40
Zhang SM, Qiu J, Tian F, Guo XK, Zhang FQ, Huang QF (2013) Corrosion behavior of pure titanium in the presence of Actinomyces naeslundii. J Mater Sci Mater Med 24:1229–1237
Nakagawa M, Matsuya S, Udoh K (2002) Effects of fluoride and dissolved oxygen concentrations on the corrosion behavior of pure titanium and titanium alloys. Dent Mater J 21(2):83–92
Souza ME, Lima L, Lima CR, Zavaglia CA, Freire CM (2009) Effects of pH on the electrochemical behaviour of titanium alloys for implant applications. J Mater Sci Mater Med 20(2):549–552
Burnat B, Walkowiak-Przybyło M, Błaszczyk T, Klimek L (2013) Corrosion behaviour of polished and sandblasted titanium alloys in PBS solution. Acta Bioeng Biomech 15(1):87–95
Cai Z, Nakajima H, Woldu M, Berglund A, Bergman M, Okabe T (1999) In vitro corrosion resistance of titanium made using different fabrication methods. Biomaterials 20(2):183–190
Cai Z, Shafer T, Watanabe I, Nunn ME, Okabe T (2003) Electrochemical characterization of cast titanium alloys. Biomaterials 24(2):213–218
Johansson BI, Bergman B (1995) Corrosion of titanium and amalgam couples: e.ect of ßuoride, area size, surface preparation and fabrication procedures. Dent Mater 11:41–46
Olmedo DG, Tasat DR, Duffo G, Guglielmotti MB, Cabrini RL (2009) The issue of corrosion in dental implants: a review. Acta Odontol Latinoam 22(1):3–9
Abey S, Mathew MT, Lee DJ, Knoernschild KL, Wimmer MA, Sukotjo C (2014) Electrochemical behavior of titanium in artificial saliva: influence of pH. J Oral Implantol 40(1):3–10
Assis SLWS, Costa I (2006) Corrosion characterization of titanium alloys by electrochemical techniques. Electrochem Acta. 51:1815–1819
Messer RL, Tackas G, Mickalonis J, Brown Y, Lewis JB, Wataha JC (2009) Corrosion of machined titanium dental implants under inflammatory conditions. J Biomed Mater Res B 88(2):474–481
Messer RL, Seta F, Mickalonis J, Brown Y, Lewis JB, Wataha JC (2010) Corrosion of phosphate-enriched titanium oxide surface dental implants (TiUnite) under in vitro inflammatory and hyperglycemic conditions. J Biomed Mater Res B 92(2):525–534
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Faverani, L.P., Fogaça, J.F., Machado, T. et al. Does Surface Topography Improve Electrochemical Behavior of Ti–6Al–4V Alloy in Different Saliva pH Levels?. J Bio Tribo Corros 1, 20 (2015). https://doi.org/10.1007/s40735-015-0020-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40735-015-0020-1