Abstract
Purpose of Review
Longitudinal studies show that the menopausal transition (MT) is associated with poorer self-reported sleep. Increases in sleep disturbances across and beyond the MT are strongly associated with vasomotor symptoms (VMS) but occur even without VMS. We analyzed data from baseline through 13 annual or biennial follow-up assessments from SWAN’s multi-racial/ethnic cohort of midlife women, specifically focusing on patterns of sleep problems in the years preceding and following the final menstrual period (FMP). The FMP demarcated the MT and the postmenopausal period. We addressed the following questions: (1) are there distinct trajectory patterns of sleep problems across the MT, and (2) do pre-FMP sleep trajectories predict sleep problems around the time of FMP (trans-FMP) and post-FMP? Group-based trajectory modeling using repeated-measures log-binomial regression with generalized estimating equation methods was used to describe trajectory patterns of the most prevalent sleep problem, waking several times at least 3 nights weekly during the previous 2 weeks, in 1285 naturally menopausal women.
Recent Findings
We found (1) 4 distinct trajectories for waking several times per night across the MT [low prevalence (n = 487; 37.9%), moderate prevalence (n = 365; 28.4%), increasing prevalence (n = 197; 15.3%), and high prevalence (n = 236; 18.4%)]; (2) the prevalence of sleep problems increased overall, but in one trajectory group (increasing prevalence) more than in the other three; and (3) trouble falling asleep, early morning awakening, and frequent VMS were strongly associated with problems waking several times that persist into postmenopause.
Summary
Using trajectory analysis, we showed that, in general, awakenings were stable or increased slowly from pre-FMP to post-FMP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sleep is a biological imperative—a fundamental behavioral and neurobiological state intrinsic to biological systems [1] and influences women’s health and wellbeing during and after the menopausal transition (MT) [2]. Poor sleep can affect cognition [3, 4], increase risk for depression [5,6,7], increase health-damaging behaviors [8], and may increase risk for cardiometabolic disorders [9,10,11,12]. The MT may be a key inflection point when sleep patterns are altered in a negative way in the short run and perhaps more permanently.
Aging affects sleep, and sleep problems in women are especially prevalent during the late reproductive (perimenopausal) stages and across the MT. The task of teasing apart the relative contributions of chronological and reproductive aging has been challenging. In large surveys, peri- and postmenopausal women report more sleep disturbances than do premenopausal women [13]. However, most studies have been cross-sectional and defined the MT according to self-reported bleeding status rather than by endocrinologic patterns of hormone changes associated with ovarian aging (particularly follicle stimulating hormone (FSH) and estradiol (E2) concentrations) relative to the final menstrual period (FMP) [14]. The FMP, defined as the initial day of the last menstrual period preceding 12 consecutive months of amenorrhea without other obvious pathological or physiological cause [15], is a biological marker of ovarian aging, which is manifested clinically as the MT. Thus, women’s sleep problems may be associated with hormonal changes occurring at the MT.
Although most observational studies of insomnia have found greater prevalence among women than men [16], especially in the postmenopausal years, limited longitudinal studies of sleep complaints across the MT and into the postmenopause have been published [17•, 18, 19, 20•]. A systematic review of eight longitudinal studies published prior to 2013 [20•] involving community dwelling midlife women showed a “small increased risk of self-reported sleep disturbance” through the MT after adjusting for potential confounders. Longitudinal data from the Study of Women’s Health Across the Nation (SWAN) [21] and other studies [22,23,24,25,26] showed that self-reported sleep disturbances increase through the MT and are strongly associated with vasomotor symptoms (VMS) [17•, 21, 27,28,29,30,31,32], but that even among women without VMS, the MT has been associated with poorer self-reported sleep [17•, 33].
A narrative review of both cross-sectional and longitudinal analyses of menopause-related self-reported sleep quality and physiologically monitored (actigraphy or polysomnography) sleep patterns [34•] showed that compared with late reproductive age women, more women in MT stages (i.e., early and late perimenopause) perceived poor sleep. Self-reported awakenings from sleep (including number and duration of bouts of wakefulness during sleep and early morning awakening) as well as increased sleep latency (trouble falling asleep) were the sleep symptoms during the MT that were most often mentioned. Both Shaver and Woods [34•] and Xu et al. [20•] noted differences in conclusions among studies that could be attributed to sample heterogeneity, variability in sleep assessment methods and measures, and incomplete or inconsistent accounting for confounding factors (including comparing broader age ranges or across menopausal status stages that affect sleep).
More recently, Freeman et al. [17•] examined the prevalence and predictors of moderate/severe poor sleep in relation to the FMP in 255 Black and White women followed with approximately annual assessments for 16 years in the Penn Ovarian Aging Study cohort. Except in a small “at-risk” subgroup, they found no significant increase in poor sleep in any year relative to the FMP, and premenopausal sleep status predicted poor sleep around the FMP (i.e., FMP to 3 years after). Concurrent hot flashes contributed to poor sleep independent of baseline sleep status.
SWAN data showed that bleeding-defined menopausal stages and endogenous sex steroid hormones were associated with self-reported problems falling asleep, awakenings from sleep, and early morning awakening after controlling for a broad array of factors that have been associated with these sleep problems [21]. However, drawbacks of these analyses included limited follow-up and modeling strategies, a potentially biased sample of “early transitioners,” and imprecise predictors of FMP.
Herein, we analyzed data from baseline through 13 annual or biennial follow-up assessments from those SWAN participants who had a documented FMP. Analyses focused on patterns of sleep problems in the years preceding the FMP rather than on women’s menopausal transition stage, and on how the trajectories changed at and following the FMP. Because prior SWAN analyses have demonstrated distinct trajectory patterns of VMS [35] as well as of E2 and FSH [36], and because Freeman et al. [17•] found a small at-risk subgroup for poor sleep, we hypothesized the presence of distinct trajectory patterns of sleep problems before and after the FMP.
Our specific hypotheses were that [1] there are distinct trajectory patterns of sleep problems across the MT (Fig. 1, Hypothesis 1) and that [2] pre-FMP sleep trajectories predict sleep problems (a) across the MT (Fig. 1, Hypothesis 2A), (b) around the time of FMP (trans-FMP; Fig. 1, Hypothesis 2B), and (c) post-FMP (Fig. 1, Hypothesis 2C), even after adjusting for time-varying covariates which have been shown to be related to sleep problems.
Methods
Study Design and Participants
SWAN is a multi-ethnic, community-based, cohort study of the menopausal transition. Initiated in 1996, 3302 women were enrolled at seven SWAN sites: Boston, MA; Chicago, IL; Detroit area, MI; Los Angeles and Oakland, CA; Newark, NJ; and Pittsburgh, PA. Study design and recruitment of the SWAN cohort have been described in detail [37]. Briefly, each site recruited Caucasian women and a minority group sample. Eligible women were 42–52 years, premenopausal or early perimenopausal, had an intact uterus and at least one ovary, had at least one menstrual period in the previous 3 months, and they were not pregnant/lactating or using any sex steroid hormones in the 3 months preceding the baseline interview. At study entry, approximately half the women were premenopausal and half were early perimenopausal according to bleeding criteria. Extensive data on psychological, social, and health parameters were collected at baseline and thereafter at follow-up visits scheduled at approximately 12- to 18-month intervals. Institutional review board approval was obtained at each SWAN study site and, after a complete description of the study to the participants, written informed consent was obtained.
Procedure and Measures
As women with 12 months of amenorrhea are considered postmenopausal, we defined the post-FMP period as starting 1 year after the FMP in the naturally menopausal group. Based on our data, pre-FMP was defined as more than 2.5 years before the FMP, and “trans-FMP” was identified as the 3.5-year period from 2.5 years before the FMP to 1 year after the FMP, which includes the late menopausal transition (Stage −1) and the first year of the early postmenopause (Stage +1a) according to the STRAW +10 staging system [38]. Women who had a hysterectomy and/or oophorectomy prior to their FMP were excluded from these analyses.
Participant selection for inclusion in the various data analyses for the hypotheses is shown in Fig. 1. Total time in study for the 1285 participants included in analyses was between 2.9 and 17.1 years, with a median (interquartile range, IQR) of 15.3 years (15.0–15.6). Women had an average of 12.0 ± 2.8 assessments (4.3 ± 2.6 pre-FMP [N = 1285 women; 5467 sleep observations], 3.0 ± 0.8 trans-FMP [N = 1224; 3638 observations], and 5.0 ± 2.6 post-FMP [N = 1256; 6298 observations]).
Dependent Variables
At each assessment, women self-reported trouble falling asleep (sleep initiation problems), waking up several times a night (sleep maintenance problems), and waking up earlier than planned and unable to fall asleep again (early morning awakening). Frequency of sleep problems in the past 2 weeks was recorded as [1] none, [2] less than once a week, [3] one or two times a week, [4] three or four times a week, [5] five or more times a week, and dichotomized as “no” (<3 times a week) or “yes” (≥3 times a week). “Waking several times” represented the best single sleep problem for examining changes across the MT for this naturally menopausal group (Table 1 and Fig. 2) as it was the most frequent symptom, as previously reported in postmenopausal women [39], and is the focus of the analyses for this report.
Prevalence of sleep problems at least three nights per week reported by women with a natural final menstrual period (FMP) (N = 1285). Shaded bar indicates the ‘trans‐FMP,’ which was identified as the 3.5‐year period from 2.5 years before the FMP to 1 year after the FMP. Vertical dashed line centered on the FMP (0)
Independent Variables
The primary independent variables were the pre-FMP sleep trajectory groups for waking several times. Our approach to identifying these trajectory groups is described in Data Analysis.
Covariates
The primary covariates were all time-varying: (a) the other two individual sleep problems (trouble falling asleep, early morning awakening); (b) frequency of vasomotor symptoms in the past 2 weeks (VMS; hot flashes/flushes and/or night sweats), which were analyzed as a three-level categorical summary variable (0, no symptoms; 1, VMS on fewer than 6 days; 2, at least one symptom reported 6–14 days); and (c) serum reproductive hormone concentrations (follicle stimulating hormone, FSH; estradiol, E2), which were measured on days 2–5 of a spontaneous menstrual cycle occurring within 60 days of recruitment at the baseline visit, and annually thereafter; the assay procedure has been described [21]. If a day 2–5 specimen could not be obtained after 2 attempts, or if a participant was post-FMP, a random fasting specimen was taken within 90 days of the annual assessment. Because not all pre-FMP reproductive hormone samples were collected within the intended window, cycle day of blood draw also was included as a covariate (dichotomized as day 2–5 versus not day 2–5 or unknown). E2 and FSH data from the last two assessments, follow-up 12 and 13, were not available due to changes in assay methods.
Other time-varying covariates included marital status (married/living with a partner versus not), body mass index (BMI; calculated as weight divided by the square of height, kg/m2), psychological symptoms (Center for Epidemiologic Studies-Depression scale (CES-D; [40]) score, calculated as the sum of all items minus the sleep item, and anxiety symptoms score, calculated as the sum of 4 items (irritability/grouchiness, feeling tense/nervous, heart pounding/racing, feeling fearful for no reason), scored on a scale from 0 (not at all) to 4 (every day over 14 days) derived from a menopausal symptom questionnaire [41, 42]), psychosocial variables (social support [43], financial resource strain ([44]; very or somewhat hard to pay for basics versus not hard at all)), self-assessed health (very good/excellent versus poor/fair/good [45]), number of medical conditions (diabetes, anemia, hypertension, osteoporosis, hyperlipidemia, migraines, thyroid problems, osteoarthritis, stroke, heart attack, angina, cancer, fibroids; 0 or 1 condition versus 2 or more conditions), the Short Form-36 bodily pain score (percentile, range = 0–100, with higher scores indicating less pain, derived from the sum of the two items assessing the level of pain severity and the extent to which pain has interfered with function; [45]), health-related behaviors (current smoking, alcohol and caffeine use, physical activity), and medications (menopause hormone therapy, medication for nervous conditions [e.g., antidepressants, tranquilizers], sedatives [including medication taken to sleep], and medication taken for pain).
Time-invariant covariates included sociodemographics (self-identified race/ethnicity, study site, education [completed high school or less versus more than a high school diploma], income [6 categories; see Table 2], and age at FMP). Since women were nested within race/ethnicity groups and study site, a combined variable with 14 categories was created, and participants were treated as nested within this variable.
Data Analysis
Analyses for this paper included all SWAN participants with an FMP and sleep data for at least one time point pre-FMP and at least one time point post-FMP. Analyses were anchored/centered on the FMP (time 0). VMS was included as a covariate in all analyses because VMS are associated with subjective sleep disturbances, particularly nighttime awakenings, across the MT [30, 32, 46, 47]. E2 and FSH values were set to missing for visits 12 and 13 because these data were not available. Although the New Jersey (NJ) site did not complete full in-person clinic visits for SWAN follow-up visits 8–11, in-person assessments resumed at visit 12 so NJ data were included in the analyses. All analyses were completed in SAS 9.3 (SAS, Cary, NC). Two-tailed P values <0.05 were considered statistically significant.
Hypothesis 1: For the 43 women with a hysterectomy or oophorectomy after their FMP, observations were censored at the time of surgery. To describe trajectory patterns of problems waking several times over the menopausal transition, group-based trajectory modeling was used (PROC TRAJ; [48,49,50]). Time-varying binary indicators of sleep problems as a function of the FMP were analyzed. The date of FMP was set as time 0. Time pre- and post-FMP was calculated by substracting annual visit date and the date of FMP. We examined whether 3 or 4 groups fit the data best, allowing linear, quadratic, and cubic terms for time in each group. Higher-order terms were eliminated one by one, starting with the least significant term in any group. Bayes Information Criterion (BIC) was used to select the number of distinct trajectory groups [48]. Age at FMP as well as the site/race groups were adjusted in these analyses.
Hypothesis 2: To answer the question of whether pre-FMP sleep predicts sleep problems at the time of FMP (i.e., trans-FMP) and beyond (i.e., post-FMP), we first grouped each woman’s sleep reports into three time periods relative to FMP, as described above. Trajectory analyses as described in Hypothesis 1 were repeated for the pre-FMP period, adjusted for age at FMP and the site/race indicators. As in the analysis (H1) using the entire time of the MT transition, 4 groups were identified: Group 1, low prevalence of sleep problems; Group 2, moderate prevalence of sleep problems; Group 3, increasing prevalence of sleep problems; and Group 4, high prevalence of sleep problems throughout. In the best-fitting trajectory model for this restricted time to pre-FMP, only one non-constant term was retained, and it was for Group 3, as anticipated. Trajectories of sleep problems, shown in Fig. 3, were analyzed using repeated-measures log-binomial regression analysis with the generalized estimating equations (GEE) method (PROC GENMOD; [51]). The log-binomial model, rather than logistic regression, was chosen because of the high prevalence of the outcome (>10%). The repeated measurements were nested within participants, i.e., the same individuals contributed more than one observation to the dataset, and the dependence of the within-participant observations was taken into account in estimating the standard errors. The main effects of group, time, and the interaction of group and time were included in Model 1. Model 2 was adjusted for time-varying early morning awakening and trouble falling asleep. Model 3 included time-varying VMS. Model 4 additionally adjusted for time-varying hormones (E2 and FSH). In modeling the hormone effect, we adjusted for whether the blood draw occurred in the 2- to 5-day window. Models without the group by time interaction fit the data as well as those with the interaction. Therefore, we present only main effects models. For ease of interpretation, we chose to use models with an intercept for each group separately instead of choosing one group as a reference. Single covariates (listed in Table 2) were added to Model 4, and those which were significant at level 0.10 were added into the final model. Financial resource strain rather than income was added to the model due to their collinearity and less missing data for the former. In addition to the combined trans- and post-FMP (Hypothesis 2A), we present separate models for the trans-FMP (Hypothesis 2B) and post-FMP (Hypothesis 2C) time periods as well.
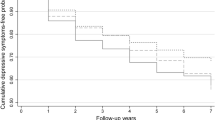
Trajectories for waking up several times at least three nights per week reported by women with a natural final menstrual period (FMP) (N = 1285). Groups were determined by trajectory analysis (see text for details) using the entire range of observed data. Group 1 (N=487; 37.9%): low prevalence of problems waking several times; Group 2 (N=365; 28.4%): moderate prevalence of problems waking several times; Group 3 (N=197; 15.3%): increasing prevalence of problems waking several times; Group 4 (N=236; 18.4%): high prevalence of problems waking several times. Shaded bar indicates the ‘trans-FMP,’ which was identified as the 3.5-year period from 2.5 years before the FMP to 1 year after the FMP. Vertical dashed line centered on the FMP (0)
Results
Sample Description
As seen in the CONSORT diagram (Fig. 1) and in Tables S1 and S2 (Appendix), 1699 of the 3302 women who participated in SWAN were excluded; 1398 had no data on either FMP or surgery, and 283 had a hysterectomy and/or oophorectomy that preceded their FMP, and 18 did not have data on sleep problems at baseline. Compared with the 1603 women with a natural FMP, a statistically significantly larger proportion of the 1699 women excluded from the analysis were from the New Jersey site, of Hispanic or White race/ethnicity, and reported more frequent VMS. Excluded women reported a small (5%) but statistically higher percentage of any sleep problems, and ranged from 3.1% for waking several times to 4.4% for early morning awakening, even after eliminating missing values from the comparison.
Baseline Characteristics
Table 2 displays the baseline characteristics of the 1285 naturally menopausal women. Their mean age was 45.9 years, and FMP occurred on average 6.7 years later. Among these women, 65% were married, more than three quarters had completed at least some college, modal household income was $50,000–$75,000, and most reported no significant financial resource strain. Although the majority rated their overall health as excellent/good, mean BMI was in the overweight category, just over one-third reported having two or more medical conditions, and one-third had taken medication for pain. Alcohol use was moderate or less for most women in the sample and only 15% were current smokers. In this predominantly premenopausal sample, more than two-thirds reported no VMS and only 7.4% reported frequent VMS. At baseline, 27% of the sample reported a sleep problem occurring at least three times a week, mainly waking several times, which was reported by 87% of the 348 women who indicated a sleep problem.
Prevalence of Sleep Problems
Prevalence is graphically presented in Fig. 2 for the 1285 women. Table 1 shows these data separately for the pre-, trans-, and post-FMP periods; numbers of women available vary due to missed visits. Waking several times was the most prevalent sleep problem pre-FMP and increased over time, parallel to the trend for any sleep problem. Its overall prevalence in each time period was almost double the prevalence for either trouble falling asleep or early morning awakening, and both its prevalence and its pattern across time paralleled that of the composite “any sleep problem” variable. The trend is less steep for trouble falling asleep and early morning awakening. Moreover, many women who reported waking several times also reported trouble falling asleep and/or early morning awakening.
Trajectories for Waking Several Times across the Menopausal Transition
Hypothesis 1: As depicted in Fig. 3, we found distinct trajectory patterns for waking several times across the whole data range, including the MT and into the postmenopause (i.e., pre-, trans-, and post-FMP). The best-fitting trajectory model for waking several times included four groups. Groups 1 and 2 had a linear term, Group 3 a cubic term, and Group 4 a quadratic term. Figure 3 shows the trajectory patterns for the 1285 women with a natural FMP: a group with a low pre-FMP prevalence of waking several times (Group 1), a group with a moderate prevalence (Group 2), a group with an increasing prevalence from pre- through post-FMP (Group 3), and a group with a high prevalence of this sleep problem throughout the observation period (Group 4).
Pre-FMP trajectory groups: Table 3 displays the baseline comparisons between the four groups for the 1057 women included in the analyses for Hypothesis 2. Overall, these groups differed significantly on a number of symptoms, health indices and behaviors, and medication use characteristics, but notably not on FSH or E2 concentrations. The groups also differed on site and race/ethnic distributions as well as on prevalence of each of the three individual sleep problems but not on other sociodemographic characteristics.
Pre-FMP Trajectories as Predictors of Trans- and Post-FMP Trajectories
Hypothesis 2 is addressed in Tables 4 and 5. In the unadjusted model that included only the pre-FMP trajectory groups and time, each of the trajectory groups for waking several times pre-FMP shown in Fig. 4 was significantly associated with similar problems after the FMP in the combined trans- + post-FMP as well as in each of these two time periods separately. The effect of time was highly significant in the combined periods, reflecting that sleep problems increase across the MT, as is also illustrated in Fig. 2, in unadjusted models as well as in models adjusted for all pre-selected covariates (trouble falling asleep, early morning awakening, VMS, and reproductive hormones). Trouble falling asleep and early morning awakening contributed significantly to problems with waking several times during all three time periods except in the trans-FMP in the model also adjusted for VMS and hormones. Frequent VMS (6–14 days/2 weeks) were significantly associated with waking several times during all three time periods. Lower FSH concentrations were significantly associated with waking several times only during the post-FMP, and E2 concentrations had no significant association with either trans- or post-FMP problems waking several times.
Table 5 shows that the addition of other covariates had little impact on the sleep trajectory parameter estimates for waking several times. After accounting for the effects of trouble falling asleep, early morning awakening, VMS, E2 and FSH, and the covariates that entered into each model, each of the four trajectory groups remained significantly associated with problems waking several times after the FMP in each of the three time periods. In these final adjusted models, time was significant only in the model for predicting post-FMP sleep, and both trouble falling asleep and early morning awakening were significant in all three time periods, as were VMS (except less frequent VMS during the trans-FMP), but neither E2 nor FSH concentrations showed a significant association. None of the covariates was significantly associated with waking several times in the combined trans- + post-FMP period. Medication for a nervous condition was the only additional covariate that was significantly associated with waking several times during the trans-FMP period. Of the additional covariates added to the post-FMP model, only anxiety symptoms were significantly (but negatively) associated with waking several times.
Discussion
Nagin [48] defined “trajectory” as “the evolution of an outcome over age or time.” In this report, we examined trajectories of self-reported sleep problems across the menopausal transition in an aging cohort of midlife women. Specifically, we examined sleep maintenance problems in SWAN women who had a natural transition to menopause. Note that the term “sleep maintenance” refers to these nighttime awakenings, i.e., waking from sleep several times, as noted under the “Dependent variables” section.
Overall, the reported prevalence for all sleep problems increased across the MT, particularly for waking several times. This is consistent with Shaver and Woods [34•] review, that for women during the MT, the most consistently observed sleep problems are those related to wakefulness during sleep episodes. As hypothesized (Hypothesis 1), across the MT, there were distinct trajectory patterns for waking several times (Fig. 3). Hypothesis 2 examined whether the pre-FMP trajectories (Fig. 4) predicted the post-FMP trajectories (Tables 4 and 5). Within each trajectory group, pre-FMP trajectories for waking several times were related to persistent sleep maintenance problems during the trans- and post-FMP periods, even after covariate adjustment.
The good news for women during the MT is that a low prevalence of waking several times at least three nights per week before the FMP, which accounted for the largest proportion of women (53%; Fig. 4), predicted a pattern of continued low prevalence. Also, the negative parameter estimates indicate that fewer than half of the women in each trajectory group had a sleep maintenance problem at the time of the FMP. For women in the moderate and high trajectory groups, unfortunately, the problem did not seem to remit with time during postmenopause. Notably, in one trajectory group, which included the smallest proportion of the women (17%; 176 of 1057 women), problems waking several times nightly increased from the pre-FMP into the post-FMP (Fig. 4). Except for the serum concentrations of reproductive hormones E2 and FSH, the covariates most consistently and significantly associated with awakenings around the FMP and during the postmenopause were those selected a priori to be included in all models: trouble falling asleep, early morning awakening, and frequent VMS.
Joffe et al. [52] noted that women are most likely to report sleep problems as they transition from late reproductive stage to perimenopause. This is consistent with cross-sectional observations by Woods and Mitchell [26], based on diary data from the Seattle Midlife Women’s Health Study, that peak reporting of severe poor sleep symptoms (especially night-time awakening) occurred during late MT stages and early postmenopause. Freeman et al.’s [17•] analysis of poor sleep in relation to the natural menopause showed relative consistency in prevalence across study years (28–35%); overall, they found no significant differences in any year relative to the FMP. The annual prevalence for any sleep problems and waking several times in our cohort was 20–45% from pre-FMP through post-FMP (Table 1). Both studies reported sleep symptoms and neither our study nor that of Freeman et al. [17•] included an impairment criterion to define clinical insomnia, the absence of which could contribute to these high prevalence levels [53]. After Freeman et al. [17•] stratified by sleep problem severity level at the premenopausal baseline, premenopausal sleep severity status strongly predicted poor sleep around the FMP. They also indicated that a small at-risk subgroup had a significant increase in poor sleep in relation to the FMP, perhaps similar to the group we identified as having an increasing trajectory. Among women with mild sleep problems in premenopause, Freeman et al. [17•] noted that only those who reported moderate/severe sleep problems during the later postmenopause years (i.e., 3–10 years postmenopause) had a significant increase in sleep problems around the FMP.
In our analysis across the entire MT (Fig. 3), we found that the prevalence of sleep problems increased in all groups, as is also reflected in Table 1 and in Fig. 2. This increase was slow in 3 of the 4 groups, which accounted for almost 85% of the women, as shown in Fig. 3. Using the pre-FMP group membership as a predictor for problems waking several times during the combined periods of trans-FMP and post-FMP, we found an overall significant time trend of worsening sleep problems with no difference between groups (lack of interaction, data not shown). However, in the final adjusted model (Table 5), time was significant only in the post-FMP model.
We anticipated that our preselected covariates, i.e., trouble falling asleep, early morning awakening, VMS, and reproductive hormones, would show significant associations with our sleep outcomes, based on our previous studies as well as on Freeman et al. [17•] and two reviews [20•, 34•]. After including these variables in the models for problems waking several times, few other covariates were significantly associated with the sleep outcome variable, and even those covariates did not significantly affect the robustness of the main associations. Notably, physical activity, sleep medications, alcohol, and caffeine did not demonstrate significant associations with trajectories for waking several times.
Studies included in the reviews generally concurred that VMS are significantly associated with sleep problems in women transitioning to menopause and beyond, particularly when sleep problems are assessed by self-report. In an experimental menopause model, Joffe et al. [47] demonstrated that nighttime VMS correlated with increased sleep fragmentation on polysomnography. However, the VMS—sleep association has been observed less consistently when sleep is assessed objectively. Moreover, the associations we found between the pre-FMP trajectories and post-FMP sleep maintenance problems persisted even after adjusting for VMS. Shaver and Woods [34•] commented that nighttime wakefulness influences how nighttime VMS are reported upon awakening, suggesting that this association may be dependent more on the subjective perception of sleep than on the physiologically assessed sleep state.
Although VMS can persist well beyond the FMP, perhaps at least as long as a decade [35, 54] and continue to be associated with sleep problems, variability in reproductive hormone concentrations (i.e., E2, FSH) do not share a similarly strong association with sleep complaints in midlife women. Freeman et al. [17•] suggested that this lack of association could also be attributed to their analytic models, which included time relative to the FMP, which reflects changing hormone concentrations during the MT. Alternatively, other hormones that we did not measure may be more relevant, such as inhibin B [28], which may be a more specific and reliable marker of menopausal status and ovarian aging. Data reviewed by Shaver and Woods [34•] demonstrating that exogenous hormone replacement therapy improves sleep would seem to suggest that ovarian hormones may have a direct effect on sleep regulation and/or an indirect effect via alleviation of other sleep-interfering symptoms, but this remains an open but important clinical question.
Different from most prior studies, which analyzed subjective sleep problems/disturbance during the MT as a single-item global construct of sleep, we included all three types of sleep problems in the same analysis to examine trajectories of the most common problem, sleep maintenance. Noting that about 75–80% or more of the women who experienced problems falling asleep or early morning awakening also reported problems waking sleep several times (Table 1), it was not surprising to see that both of these time-varying covariates demonstrated independent associations with sleep maintenance problems. Nevertheless, the main effect for pre-FMP trajectories for waking several times predicting post-FMP trajectories remains highly significant across the post-FMP time periods.
More surprising was that depressive symptoms did not contribute significantly to the sleep problem after we controlled for the main independent variable and preselected covariates, and anxiety symptoms were significantly associated only in the post-FMP, but in the direction opposite to what would be anticipated, i.e., lower anxiety symptom score was associated with reporting more sleep maintenance problems during the post-FMP period. Freeman et al. [17•] found that higher anxiety symptom and perceived stress scores were significantly associated with poor sleep, but depression was not a significant risk factor in their adjusted analysis either. We did not include perceived stress in our analyses due to its collinearity with depressive symptoms. A possible explanation for the lack of association with the CES-D score may be due to collinearity with VMS, because the latter has been linked to depressed mood [55, 56]. Moreover, anxiety, which Freeman et al. [17•] noted has been identified as the strongest predictor of poor sleep quality in previous studies [57, 58], commonly coexists with depressed mood and with VMS in midlife women [59]. We cannot explain why anxiety symptoms were negatively associated with waking several times, but what does seem clear is that associations between depressed mood, anxiety, VMS, and poor sleep across the MT and into postmenopause involve complex and likely bidirectional interrelationships.
Strengths of the SWAN and our longitudinal analysis include a long follow-up in a large community sample of women unselected for sleep problems, which enhances the generalizability of our sample and decreases the risk of selection bias. Although use of self-report measures of sleep problems carries a risk of information bias, it is a clinical gold standard for sleep fragmentation problems as insomnia treatment is initiated based on patients’ self-report. The same three questionnaire items, which are based on the Women’s Health Initiative Insomnia Rating Scale [60], a valid and reliable self-report measure of sleep disturbance in menopausal women [61], have been administered repeatedly since the SWAN baseline assessment in 1996–1997. Rather than using menopausal stages based on bleeding criteria as in our previous analysis [21], we defined pre-FMP, trans-FMP, and FMP according to time centered on the FMP date, providing time periods that were consistent with the STRAW +10 staging system. Using trajectory models centered on the FMP, we examined how sleep maintenance patterns varied across the MT and distinguish subgroups of SWAN women with distinct transition experiences in sleep maintenance. It is important to note that our findings may not generalize to surgically menopausal women.
SWAN is one of the largest and longest followed-up racial/ethnically diverse studies of the MT. Use of group-based trajectory modeling accounted for heterogeneity within the sample, allowing us to identify groupings that are both statistically sound and scientifically valid [35]. Using the available premenopausal and postmenopausal observations and within-woman duration (i.e., time), we were able to evaluate sleep maintenance trajectories pre- and post-FMP. The large sample and number of observations provided sufficient statistical power to control for the effects of a number of covariates. Nevertheless, to achieve convergence of the models, we needed to limit the covariates included in order to obtain a parsimonious model that both converged and fit the data best. Also, although SWAN is a multiethnic/multiracial cohort, because race and site are confounded due to study design and because the race/ethnicity distribution in the various trajectory groups led to some sparse cells, we statistically adjusted for but could not examine the contribution of race/ethnicity in this analysis.
Conclusion
Our analytic approach using pre-FMP sleep trajectories to predict sleep trajectories in the time periods after the FMP allowed us to examine how sleep changes across the MT and into the postmenopause by separating the effects of menopause from pre-existing sleep problems. Our findings add to current knowledge regarding patterns and persistence of sleep complaints among midlife women transitioning to menopause and during the early postmenopausal years. Clinicians might find it reassuring that for the majority of women, their symptom levels do not get worse across the MT, yet they should be vigilant for those midlife women whose symptoms do worsen as well as those who continue to have a high level of persistent sleep maintenance problems.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Buysse DJ. Introduction. In: Buysse DJ, editor. Sleep disorders and psychiatry (review of psychiatry series, volume 24, number 2; Oldham JM and Riba MB, series editors). Washington, DC: American Psychiatric Publishing; 2005. p. 1–28.
Greenblum CA, Rowe MA, Neff DF, Greenblum JS. Midlife women: symptoms associated with menopausal transition and early postmenopause and quality of life. Menopause. 2013;20(1):22–7.
Ferrie JE, Shipley MJ, Akbaraly TN, Marmot MG, Kivimaki M, Singh-Manoux A. Change in sleep duration and cognitive function: findings from the Whitehall II study. Sleep. 2011;34(5):565–73.
Scullin MK, Bliwise DL. Sleep, cognition, and normal aging: integrating a half century of multidisciplinary research. Perspect Psychol Sci. 2015;10(1):97–137.
Alvaro PK, Roberts RM, Harris JK. A systematic review assessing bidirectionality between sleep disturbances, anxiety, and depression. Sleep. 2013;36(7):1059–68.
Baglioni C, Battagliese G, Feige B, Spiegelhalder K, Nissen C, Voderholzer U, et al. Insomnia as a predictor of depression: a meta-analytic evaluation of longitudinal epidemiological studies. J Affect Disord. 2011;135:10–9.
Spoormaker VI, Van Den Bout J. Depression and anxiety complaints; relations with sleep disturbances. Eur Psychiatry. 2005;20:243–5.
Clark AJ, Salo P, Lange T, Jennum P, Virtanen M, Pentti J, et al. Onset of impaired sleep as a predictor of change in health-related behaviours; analysing observational data as a series of non-randomized pseudo-trials. Int J Epidemiol. 2015;44(3):1027–37. doi:10.1093/ije/dyv063.
Elwood P, Hack M, Pickering J, Hughs J, Gallacher J. Sleep disturbance, stroke, and heart disease events: evidence from the Caerphilly cohort. J Epidemiol Community Health. 2006;60(1):69–73.
Chien K-L, Chen PC, Hsu HC, Su TC, Sung FC, Chen MF, et al. Habitual sleep duration and insomnia and the risk of cardiovascular events and all-cause death: report from a community-based cohort. Sleep. 2010;33(2):177–84.
Cappuccio F, Taggart F, Kandala N-B, Currie A, Peile E, Stranges S, et al. Meta-analysis of short sleep duration and obesity in children and adults. Sleep. 2008;31:619–26.
Gangwisch J, Malaspina D, Boden-Albala B, Heymsfield S. Inadequate sleep as a risk factor for obesity: analysis of NHANES I. Sleep. 2005;28:1289–96.
Parry BL, Fernando Martinez L, Maurer EL, Lopez AM, Sorenson D, Meliska CJ. Sleep, rhythms and women’s mood. Part II. Menopause. Sleep Med Rev. 2006;10(3):197–208. doi:10.1016/j.smrv.2005.09.004.
Randolph JF Jr, Zheng H, Sowers MR, Crandall C, Crawford S, Gold EB, et al. Change in follicle stimulating hormone and estradiol across the menopausal transition: effect of age at the final menstrual period. J Clin Endocrinol Metab. 2011;96:746–54.
World Health Organization Scientific Group. Research on the Menopause in the 1990s (1994): Geneva, Switzerland. Report of a WHO Scientific Group. WHO Technical Report Series; 866. Geneva: World Health Organization; 1996. p. 1–107.
National Institutes of Health. National Institutes of Health state of the science conference statement: manifestations and management of chronic insomnia in adults June 13–15, 2005. Sleep. 2005;28(9):1049–57.
• Freeman EW, Sammel MD, Gross SA, Pien GW. Poor sleep in relation to natural menopause: a population-based 14-year follow-up of midlife women. Menopause. 2015;22(7):719–26. The importance of this paper is that it is the first published prospective longitudinal study designed to estimate prevalence and predictors of moderate/severe poor sleep in midlife women in relation to their final menstrual period (FMP), which could be identified with minimal recall bias, to determine whether the pattern of poor sleep was associated with this marker of menopause. Annual assessments were conducted, beginning with a randomly identified population-based inception cohort of 255 premenopausal women who reached natural menopause during follow-up into the postmenopausal years.
NIH State-of-the-Science Panel. National Institutes of Health state-of-the-science conference statement: management of menopause-related symptoms. Ann Intern Med. 2005;142(12, part 1):1003–13.
Woods NF, Mitchell ES. Symptoms during the perimenopause: prevalence, severity, trajectory, and significance in women’s lives. Am J Med. 2005;118(12B):14S–24S.
• Xu Q, Lang CP, Rooney N. A systematic review of the longitudinal relationships between subjective sleep disturbance and menopausal stage. Maturitas. 2014;79:401–12. This paper systematically reviewed the longitudinal relationship between sleep disturbance and the menopausal transition among community dwelling women to critique the methodological quality of the 8 eligible studies and to interpret the overall findings. Due to a high level of heterogeneity across the included studies, the authors used a narrative synthesis approach to demonstrate that there is reasonable evidence for an increased risk of sleep disturbance among midlife women, albeit small in magnitude, that can be attributed to the menopausal transition, beyond the effect of age, health behaviors, physical health, psychological health, vasomotor symptoms, previous sleep disturbance, and estradiol and follicle stimulating hormone.
Kravitz HM, Zhao X, Bromberger JT, Gold EB, Hall MH, Matthews KA, et al. Sleep disturbance during the menopausal transition in a multi-ethnic community sample of women. Sleep. 2008;31(7):979–90.
Dennerstein L, Lehert P, Guthrie JR, Burger HG. Modeling women’s health during the menopausal transition: a longitudinal analysis. Menopause. 2007;14(1):53–62. doi:10.1097/01.gme.0000229574.67376.ba.
Freeman EW, Sammel MD, Lin H, Gracia CR, Pien GW, Nelson DB, et al. Symptoms associated with menopausal transition and reproductive hormones in midlife women. Obstet Gynecol. 2007;110(2, Part 1):230–40. doi:10.1097/01.AOG.0000270153.59102.40.
Pien GW, Sammel MD, Freeman EW, Lin H, DeBlasis TL. Predictors of sleep quality in women in the menopausal transition. Sleep. 2008;31(7):991–9.
Tom SE, Kuh D, Guralnik JM, Mishra GD. Self-reported sleep difficulty during the menopausal transition: results from a prospective cohort study. Menopause. 2010;17(6):1128–35. doi:10.1097/gme.0b013e3181dd55b0.
Woods NF, Mitchell ES. Sleep symptoms during the menopausal transition and early postmenopause: observations from the Seattle midlife women’s health study. Sleep. 2010;33(4):539–49.
Dennerstein L, Dudley EC, Hopper JL, Guthrie JR, Burger HG. A prospective population-based study of menopausal symptoms. Obstet Gynecol. 2000;96(3):351–8.
Hollander LE, Freeman EW, Sammel MD, Berlin JA, Grisso JA, Battistini M. Sleep quality, estradiol levels, and behavioral factors in late reproductive age women. Obstet Gynecol. 2001;98(3):391–7.
Kravitz HM, Janssen I, Santoro N, Bromberger JT, Schocken M, Everson-Rose SA, et al. Relationship of day-to-day reproductive hormone levels to sleep in midlife women. Arch Intern Med. 2005;165(20):2370–6. doi:10.1001/archinte.165.20.2370.
Ohayon MM. Severe hot flashes are associated with chronic insomnia. Arch Intern Med. 2006;166(12):1262–8. doi:10.1001/archinte.166.12.1262.
Thurston RC, Blumenthal JA, Babyak MA, Sherwood A. Association between hot flashes, sleep complaints, and psychological functioning among healthy menopausal women. Int J Behav Med. 2006;13(2):163–72. doi:10.1207/s15327558ijbm1302_8.
Young T, Rabago D, Zgierska A, Austin D, Laurel F. Objective and subjective sleep quality in premenopausal, perimenopausal, and postmenopausal women in the Wisconsin sleep cohort study. Sleep. 2003;26(6):667–72.
Kravitz HM, Ganz PA, Bromberger J, Powell LH, Sutton-Tyrrell K, Meyer PM. Sleep difficulty in women at midlife: a community survey of sleep and the menopausal transition. Menopause. 2003;10(1):19–28.
• Shaver JL, Woods NF. Sleep and menopause: a narrative review. Menopause. 2015;22(8):899–915. This narrative review critically examined and interpreted studies, both longitudinal and cross-sectional, published in the past decade for evidence of the impact of menopause-related self-reported sleep quality and recorded sleep patterns on reproductive aging, in the context of self-reported vasomotor symptoms (VMS), recorded vasomotor activity (VMA) events, depressed mood, and ovarian hormones. The authors concluded that beyond anticipated age effects we still have an incomplete picture of what contributes to poor sleep quality in women transitioning to menopause, but multiple factors are at the core, and mixed outcomes probably are related to inconsistent accounting of known influential sleep factors, especially VMS, mood state, primary sleep-related disorders, and inherent sleep disorders.
Tepper PG, Brooks MM, Randolph JF Jr, Crawford SL, El Khoudary SR, Gold EB, et al. Characterizing the trajectories of vasomotor symptoms across the menopausal transition. Menopause. 2016;23(10):1067–74.
Tepper PG, Randolph JF Jr, McConnell DS, Crawford SL, El Khoudary SR, Joffe H, et al. Trajectory clustering of estradiol and follicle-stimulating hormone during the menopausal transition among women in the study of women’s health across the nation (SWAN). J Clin Endocrinol Metab. 2012;97:2872–80.
Sowers MF, Crawford SL, Sternfeld B, Morganstein D, Gold EB, Greendale GA, et al. SWAN: a multicenter, multiethnic, community-based cohort study of women and the menopausal transition. In: Lobo R, Marcus R, Kelsey J, editors. Menopause: biology and pathobiology. San Diego: Academic Press; 2000. p. 175–88.
Harlow SD, Gass M, Hall JE, Lobo R, Maki P, Rebar RW, et al. Executive summary of the stages of reproductive aging workshop + 10: addressing the unfinished agenda of staging reproductive aging. J Clin Endocrinol Metab. 2012;97:1159–68.
Kripke DF, Brunner R, Freeman R, Hendrix SL, Jackson RD, Masaki K, et al. Sleep complaints of postmenopausal women. Clin J Women’s Health. 2001;1(5):244–52.
Radloff LS. The CES-D scale: a self-report depression scale for research in the general population. Appl Psychol Meas. 1977;1:385–401.
Neugarten BL, Kraines RJ. “Menopausal symptoms” in women of various ages. Psychosom Med. 1965;27:266–73.
Bromberger JT, Kravitz HM, Chang Y, Randolph JF Jr, Avis NE, Gold EB, et al. Does risk for anxiety increase during the menopausal transition? Study of women’s health across the nation (SWAN). Menopause. 2013;20(5):488–95.
Sherbourne CD, Stewart AL. The MOS social support survey. Soc Sci Med. 1991;32:705–14.
Hall MH, Matthews KA, Kravitz HM, Gold EB, Buysse DJ, Bromberger JT, et al. Race and financial strain are independent correlates of sleep in midlife women: the SWAN sleep study. Sleep. 2009;32(1):73–82.
Ware JE, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30:473–83.
Bianchi MT, Kim S, Galvan T, White DP, Joffe H. Nocturnal hot flashes: relationship to objective awakenings and sleep stage transitions. J Clin Sleep Med. 2016;12(7):1003–9.
Joffe H, Crawford S, Economou N, Kim S, Regan S, Hall JE, et al. A gonadotropin-releasing hormone agonist model demonstrates that nocturnal hot flashes interrupt objective sleep. Sleep. 2013;36(12):1977–85. doi:10.5665/sleep.3244.
Nagin DS. Group-based modeling of development. Cambridge: Harvard University Press; 2005.
Jones BL, Nagin DS. Advances in group-based trajectory modeling and an SAS procedure for estimating them. Sociol Methods Res. 2007;35:542–71.
Nagin DS, Odgers CL. Group-based trajectory modeling in clinical research. Annu Rev Clin Psychol. 2010;6:109–38.
Spiegelman D, Hertzmark E. Easy SAS calculations for risk or prevalence ratios and differences. Am J Epidemiol. 2005;162(3):199–200.
Joffe H, Massler A, Sharkey KM. Evaluation and management of sleep disturbance during the menopause transition. Semin Reprod Med. 2010;28:404–21.
Ohayon MM. Epidemiology of insomnia: what we know and what we still need to learn. Sleep Med Rev. 2002;6(2):97–111.
Avis NE, Crawford SL, Greendale G, Bromberger JT, Everson-Rose SA, Gold EB, et al. For the study of women’s health across the nation (SWAN). Duration of menopausal vasomotor symptoms over the menopause transition. JAMA Intern Med. 2015;175(4):531–9.
Cohen LS, Soares CN, Vitonis AF, Otto MW, Harlow BL. Risk for new onset of depression during the menopausal transition: the Harvard study of moods and cycles. Arch Gen Psychiatry. 2006;63:385–90.
Gibson CJ, Thurston RC, Bromberger JT, Kamarck T, Matthews KA. Negative affect and vasomotor symptoms in the study of women’s health across the nation daily hormone study. Menopause. 2011;18:1270–7.
Freedman RR, Roehrs TA. Sleep disturbance in menopause. Menopause. 2007;14:1–4.
Cheng MH, Hsu CY, Wang SJ, Lee SJ, Wang PH, Fuh JL. The relationship of self-reported sleep disturbance, mood and menopause in a community study. Menopause. 2008;15:958–62.
Kravitz HM, Schott LL, Joffe H, Cyranowski JM, Bromberger JT. Do anxiety symptoms predict major depressive disorder in midlife women? The study of women’s health across the nation (SWAN) mental health study (MHS). Psychol Med. 2014;44:2593–602.
Levine DW, Kripke DF, Kaplan RM, Lewis MA, Naughton MJ, Bowen DJ, et al. Reliability and validity of the women’s health initiative insomnia rating scale. Psychol Assess. 2003;15:137–48.
Levine DW, Dailey ME, Rockhill B, Tipping D, Naughton MJ, Shumaker SA. Validation of the women’s health initiative insomnia rating scale in a multicenter controlled clinical trial. Psychosom Med. 2005;67(1):98–104.
Acknowledgements
The Study of Women’s Health Across the Nation (SWAN) has grant support from the National Institutes of Health (NIH), DHHS, through the National Institute on Aging (NIA), the National Institute of Nursing Research (NINR), and the NIH Office of Research on Women’s Health (ORWH) (Grants U01NR004061, U01AG012505, U01AG012535, U01AG012531, U01AG012539, U01AG012546, U01AG012553, U01AG012554, U01AG012495). Dr. Kravitz also was supported by The Stanley G. Harris Family Chair of Psychiatry. The content of this article is solely the responsibility of the authors and does not necessarily represent the official views of the NIA, NINR, ORWH, or the NIH.
Clinical Centers: University of Michigan, Ann Arbor—Siobán Harlow, PI 2011–present; MaryFran Sowers, PI 1994–2011; Massachusetts General Hospital, Boston, MA—Joel Finkelstein, PI 1999–present; Robert Neer, PI 1994–1999; Rush University, Rush University Medical Center, Chicago, IL—Howard Kravitz, PI 2009–present; Lynda Powell, PI 1994–2009; University of California, Davis/Kaiser—Ellen Gold, PI; University of California, Los Angeles—Gail Greendale, PI; Albert Einstein College of Medicine, Bronx, NY—Carol Derby, PI 2011–present, Rachel Wildman, PI 2010–2011; Nanette Santoro, PI 2004–2010; University of Medicine and Dentistry—New Jersey Medical School, Newark—Gerson Weiss, PI 1994–2004; and the University of Pittsburgh, Pittsburgh, PA—Karen Matthews, PI.
NIH Program Office: National Institute on Aging, Bethesda, MD—Chhanda Dutta 2016–present; Winifred Rossi 2012–2016; Sherry Sherman 1994–2012; Marcia Ory 1994–2001; National Institute of Nursing Research, Bethesda, MD—Program Officers.
Central Laboratory: University of Michigan, Ann Arbor—Daniel McConnell (Central Ligand Assay Satellite Services).
Coordinating Center: University of Pittsburgh, Pittsburgh, PA—Maria Mori Brooks, PI 2012–present; Kim Sutton-Tyrrell, PI 2001–2012; New England Research Institutes, Watertown, MA - Sonja McKinlay, PI 1995–2001.
Steering Committee: Susan Johnson, Current Chair.
Chris Gallagher, Former Chair.
We thank the study staff at each site and all the women who participated in SWAN.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Martica H. Hall, Karen A. Matthews, Kristine Ruppert, and Joyce T. Bromberger declare no conflicts of interest.
Hadine Joffe reports grants from NIH during the conduct of the study, grants and personal fees from Merck, grants and personal fees from SAGE, personal fees from Mitsubishi Tanabe, and personal fees from NeRRe.
Howard M. Kravitz reports grants from National Institutes of Health (NIH), during the conduct of the study.
Imke Janssen reports grants from NIH during the conduct of the study.
Human and Animal Rights and Informed Consent
Institutional review board approval was obtained at each SWAN study site and, after a complete description of the study to the participants, written informed consent was obtained.
Additional information
This article is part of the Topical Collection on Women and Sleep
Rights and permissions
About this article
Cite this article
Kravitz, H.M., Janssen, I., Bromberger, J.T. et al. Sleep Trajectories Before and After the Final Menstrual Period in the Study of Women’s Health Across the Nation (SWAN). Curr Sleep Medicine Rep 3, 235–250 (2017). https://doi.org/10.1007/s40675-017-0084-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40675-017-0084-1