Abstract
Purpose
I. R. Iran has been considered iodine replete since 2000, but iodine nutrition of vulnerable subjects is not clear. The main goal of this study was assessment of iodine nutrition and thyroid function in pregnant Iranian women.
Methods
A total of 1072 pregnant women from ten provinces in the different parts of the country were recruited from November to March 2014. Median urinary iodine concentration (UIC) as the measure of iodine status and serum free T4 (FT4), thyrotropin (TSH), thyroglobulin (Tg), anti-thyroglobulin and anti-thyroid peroxidase antibodies (TgAb and TPOAb) were measured.
Results
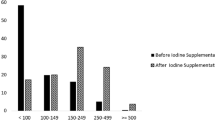
Mean ± SD age of the cohort was 27.0 ± 7.2 years and gestational age was 20.7 ± 10.0 weeks. The median UIC for pregnant women was 87.3 μg/L, being 92.1, 86.0 and 76.8 μg/L, in three trimesters of pregnancy, respectively. Median UIC of <100, 100–149, 150–249, 250–499 and ≥500 μg/L was found in 58.4, 19.8, 16.2, 5.13 and 0.46 % of subjects, respectively. Median (IQR) values in the first, second, and third trimesters were 1.7 (0.9–2.8), 2.1 (1.5–2.9), and 2.1 (1.4–2.8) mIU/L for TSH, and 16.4 (12.21–21.13), 14.34 (12.16–19.69), and 14/07 (12.02–18.64) pmol/L for FT4, respectively. The frequency of elevated serum TSH was 9.0 % (6.6 % subclinical, 2.4 % overt hypothyroidism). The frequency of low serum TSH was 0.6 %. The frequency of positive TPOAb was 7.6 %.
Conclusions
Results of this study have clarified that despite iodine sufficiency of school children in Iran, pregnant women have moderate iodine deficiency and need iodine supplementation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Iodine is the major component of thyroid hormones which are vital for neurocognitive development of the fetus and newborn [1, 2]. Inadequate levels of thyroid hormones during pregnancy may contribute to obstetric complications for the mother and the fetus [3]. Pregnant women are vulnerable to iodine deficiency as the effects of in utero deficiency on the developing fetus may have life-long consequences for the offspring. Severe iodine deficiency during pregnancy is well known to cause cretinism and severe mental retardation [4–7]. Bath et al. have recently found mild-to-moderate iodine deficiency in a large UK cohort of pregnant women and the children of those that had low iodine status in early pregnancy were more likely to have significantly lower IQ and reading scores [8].
The fundamental changes in thyroid function during normal pregnancy and lactation include increased thyroid hormone production, increased renal iodine excretion and iodine secretion into breast milk, and iodine requirement for the fetus [9]. Therefore, a woman needs more iodine during pregnancy and lactation to maintain normal metabolism as well as for the transfer of thyroxine and iodine to the fetus and neonate. Though it is vital that pregnant women meet their iodine requirements, this is not always achieved even in developed countries such as the USA [10]. Ideally, women should have adequate intra-thyroidal iodine stores before conception, which should be ensured by universal salt iodization (UIC) programs, but the American Thyroid Association (ATA) has recommended 150 µg iodine daily as dietary supplement for all pregnant women [11]. The most recent recommendation has been endorsed by World Health Organization (WHO) [12], UNICEF [13] and Iodine Global Network (IGN) formerly ICCIDD [14]. According to recent guidelines of both the ATA [15], and the Endocrine Society [16], daily intake of 250 µg iodine has been advised for all pregnant and breastfeeding women, not only in iodine-deficient areas, but also in iodine-sufficient regions.
Iodine deficiency has been aggressively addressed in I. R. Iran by the production and consumption of iodized salt since 1970 and three national surveys have shown iodized salt consumption by >95 % of the population and an adequate UIC of 232, 190, and 140 µg/L among schoolchildren in 1996, 2001, and 2006, respectively [17–19]. In our latest national survey in 2014, the median UIC of 18,000 schoolchildren was 161 µg/L, which indicates sustainability of iodine sufficiency in I. R. Iran (unpublished data). However, pregnant as well as lactating women and their newborns (up to early infancy) are among the populations the most vulnerable to the detrimental effects of iodine deficiency. Therefore, the aim of this study was to assess iodine intake and the prevalence of thyroid dysfunction in a national representative sample of pregnant women in Iran.
Materials and methods
Study population
This is an observational, cross-sectional study. A total of 1200 pregnant women attending antenatal care clinics in the mother and child health care centers in 10 provinces, randomly selected from 31 provinces of the country, included women aged 20–40 years who had lived in the city for more than 5 years with pre-gestational regular menses and were pregnant with a singleton pregnancy and in the first, second and third trimester of their pregnancy (400 pregnant women in each trimester) were consecutively recruited from November to March 2014. Excluded were smokers and those women with a history of thyroid disease or any other chronic diseases, and subjects on any medical regimen before pregnancy that may affect thyroid function, such as glucocorticoids, dopamine, or antiepileptic drugs. All women were asked to complete the study questionnaire at their first visit and written informed consent was obtained. The study was approved by the human research committee of the Research Institute for Endocrine Sciences. Of these 1200 women, 128 subjects were excluded, because of inadequate urine samples for measurement of urinary iodine concentration, and finally 338, 389 and 295 pregnant women recruited at the first, second and third trimester, respectively.
Procedures
Trained midwives explained the rationale of the study to the pregnant women and collected demographic data (including age, parity and gestational age) and medical history (including thyroid medications and history of previous thyroid disorders). Gestational age was calculated from the first day of the last normal menstrual period and gestational ages <12, 12–23.9, and >24 weeks comprised the first, second, and third trimesters of pregnancy.
Laboratory measurements
Urinary iodine concentration (UIC) was measured in random urine samples using a manual method based on the Sandell–Kolthoff technique [20] and the results were expressed as microgram of iodine per liter of urine (µg/L). The urine samples were not dipsticked for other assays. The analytical sensitivity for iodine was 1.39 µg/L and the intra-assay and interassay coefficients of variation were 4.4 and 3.9 %, respectively.
Venous blood samples were collected from pregnant women, using anticoagulant-free tubes and after centrifugation the serum samples were stored at −80 °C until analyses. Serum FT4, TSH, and thyroglobulin (Tg) were measured by the electrochemiluminescence immunoassay (ECLIA) method, using Roche Diagnostics kits and the Roche/Hitachi Cobas e-411 analyzer (GmbH, Mannheim, Germany). Anti-thyroid peroxidase (TPOAb) and anti-thyroglobulin (TgAb) antibodies were determined by immunoenzymometric assay (IEMA) using commercial kit (Monobind, Costa Mesa, CA, USA). All tests performing by enzyme-linked immunosorbent assays were done by a Sunrise ELISA reader (Tecan Co. Salzburg, Austria). Lyophilized quality control material (Lyphochek Immunoassay Plus Control, Bio-Rad Laboratories, Irvine, CA, USA) in three different concentrations was used to check accuracy of assays; also a pooled serum was prepared, aliquoted and stored at −80° and assayed in all runs for monitoring precision of measurements. The intra assay coefficients of variation (CV) of serum FT4, TSH, TPOAb, TgAb, and Tg were 1.49–4.18 %, 2.1–3.3 %, 2.40–6.1 %, 1.3–4.7 % and 1.9–5.0 %, respectively. The interassay CV values for above mentioned parameters were 4.65–7.99 %, 1.30–7.85 %, 2.2–6.7 %, and 3.8–5.8 %, respectively.
We applied our local trimester-specific reference ranges of TSH and FDefinitions and diagnostic criterion for thyroid disorders
T4 made by our laboratory based on Guideline 22 of the National Academy of Clinical Biochemistry [21]. A total of 466 Iranian pregnant women, who had the same ethnicity of Persians, in the first trimester of pregnancy attending antenatal care clinics in the mother and child health care centers of two general hospitals of Tehran were consecutively recruited from November 2004 to November 2006. Only women with singleton pregnancies were enrolled. Inclusion criteria required documentation that thyroid-related measurements were available in all of the three trimesters. Of 466 women who were referred in the first trimester, 147 subjects were excluded because of preexisting thyroid disorders or nodules; those taking medications affecting thyroid function and those not available in all trimesters or lost to follow-up (referring elsewhere for delivery, nonviable pregnancy) were excluded, and 219 healthy pregnant women were selected. A further 20 subjects were excluded due to laboratory results of positive serum thyroid peroxidase antibodies (TPOAb > 40 IU/mL), low urinary iodine level (<150 μg/dL in two out of three sample measurements in the first trimester) (34 subjects), and enlarged thyroid gland (thyroid volume greater than 30 mL) by ultrasonography (nine subjects). None had overt hypo- (TSH > 4.5 mIU/L and T4 < 5.5) or hyperthyroidism (TSH < 0.1 mIU/L and T4 > 14.5) or subclinical hypothyroidism (TSH > 10 mIU/L). Those with subclinical hyperthyroidism (serum TSH levels under 0.1 mIU/L and normal T4) were not excluded due to normal TSH suppression at pregnancy. Finally, 152 healthy iodine-sufficient women with viable, singleton pregnancies comprised the cohort study. Reference intervals in the first, second, and third trimesters were as follows: TSH 0.2–3.9, 0.5–4.1, and 0.6–4.1 mIU/L, respectively [22]. Subclinical hypothyroidism was defined as TSH between 3.9 and 10 mIU/L in the first and between 4.1 and 10 mIU/L in both the second and third trimester, with normal FT4 in all trimesters of pregnancy. Overt hypothyroidism was defined as TSH > 10 mIU/L or TSH > 3.9 mIU/L and FT4 < 12.5 pmol/L in the first, TSH > 4.1 mIU/L and FT4 < 12.8 pmol/L in the second, and TSH > 4.1mIU/L and FT4 < 12.0 pmol/L in the third trimesters of pregnancy. The reference of TPOAb 0–35 IU/ml, and TgAb 0–115 IU/ml was provided by the manufacturer. According to UIC values, pregnant women were divided into five groups: UIC <100 µg/L, UIC 100–149 µg/L, UIC 150–249 µg/L, UIC 250–499 µg/L, and UIC ≥500 µg/L, but for defining nutritional status, they stratified in tow groups with UIC < 150 and UIC ≥ 150 µg/L.
Statistical analysis
The Kolmogorv–Smirnov method was used to test data distribution normality. Normally distributed values are reported as mean ± SD, or if not so, were reported as median and interquartile range (IQR). Repeated measure test was used to compare FT4 between three trimesters and the Friedman test was used for TSH comparison. McNemar, s test was applied for comparison of prevalence of hypothyroidism based on two different cut points of TSH; P values <0.05 denoted statistical significance. One-way ANOVA with post hoc Scheffe correction was used to test differences of serum Tg between groups of participants. Linear regression was used between UIC and thyroid function parameters. Mann–Whitney test was used to compare differences in continuous variables between trimesters. The statistical software SPSS version 20 was used for data analysis.
Results
A total of 1072 participants were recruited in this study. Table 1 shows demographic and biochemical parameters in participating women.
Urinary iodine concentration (UIC)
In total cohorts, the median (IQR) of UIC was 87.3 (46.5–139.1) µg/L, it was 92.1 (48.8–145.5), 86.0 (46.2–137.5), and 76.8 (42.8–132.8) µg/L in the first, second, and third trimesters, respectively. 838 (78.2 %) of women had UIC < 150 µg/L and 234 (21.8) had UIC < 150 µg/L. According to WHO criteria, 19.8 % of all pregnant women had mild (UIC = 100–150 µg/L) and 58.4 % had moderate (UIC = 50–100 µg/L) iodine deficiency. Table 2 shows the percent of frequency distribution of median UIC by trimester of pregnancy. The mean, median, 10th and 90th percentiles of UIC in the study population are depicted in Table 3.
Thyroid tests results
The median (IQR) of serum TSH concentration was 1.70 (0.90–2.80), 2.10 (1.50–2.90) and 2.10 (1.40–2.80) mIU/L in the first, second, and third trimester, respectively, (p < 0.001). The median (IQR) of FT4 was 16.40 (12.21–21.13), 14.34 (12.16–19.69), and 14.07 (12.02–18.64) pmol/L in the first, second and third trimester, respectively (NS). TPOAb was positive in 82 subjects (7.6 %) of whom 44 (4.0 %), 24 (2.3 %), and 14 (1.3 %) subjects who were in their first, second, and third trimesters, respectively. Table 4 depicts serum concentrations of TSH, FT4, Tg and positive TPOAb in different UIC groups.
Prevalence of thyroid dysfunction
Considering our local trimester-specific reference range for TSH (0.2–3.9 mIU/L in the first and 0.3–4.1 mIU/L in the second and third trimesters) 0.6 % (seven subjects) had low TSH and the frequency of elevated serum TSH was 9.0 % (96 subjects). 97.9 % of subjects with elevated TSH had subclinical hypothyroidism, but when the ATA recommended trimester-specific reference ranges were used, 33.9 % (359 subjects) had TSH >2.5 mIU/L in the first, while 10.6 % (114 subjects) had TSH >3.0 mIU/L in the second and third trimesters (Fig. 1). In total cohorts, there was significant difference in the prevalence of subclinical hypothyroidism based on two different cut points (p = 0.001). Overt hypothyroidism was detected in 6, 5, and 7 women in the first, second and third trimesters of pregnancy. No body had isolated hypothyroxinemia. In the total population, the frequency of positive TPOAb was 7.6 %. Median serum Tg was not significantly different in women with UIC ≥ 150 µg/L and those with UIC < 150 µg/L. Table 5 depicts prevalence of thyroid disorders in different UIC groups.
Discussion
The median UIC of subjects in this study indicates moderate iodine deficiency among pregnant women in Iran, a country considered as iodine sufficient based on school-aged children UIC for one decade. The overall prevalence of thyroid dysfunction in pregnant women in this study was 9.6 %. Mean FT4 was not significantly different in three trimesters of pregnancy (P = 0.20). TPOAb was positive in 7.6 % of women and nobody had increased in Tg levels. There are currently no validated thresholds for serum Tg values in pregnancy with regard to identifying population iodine status.
Inadequate levels of thyroid hormone during pregnancy may contribute to obstetric complications for the mother and neurodevelopmental deficits in the fetus [4, 5]. This topic received increased attention during last few decades and in 2012, the Endocrine Society guideline recommended: “As long as possible before pregnancy and during pregnancy and breastfeeding, women should increase their daily iodine intake to 250 µg on average by daily oral iodine supplement of 150–200 µg/days irrespective of historical iodine nutrition status” [16]. As a component of thyroid hormones, iodine is essential for regulation of metabolic processes in most cells, besides playing a vital role in the development of most organs, in particular the brain for adequate thyroid hormone synthesis throughout life, therefore assuring adequate iodine nutrition, especially during pregnancy is essential. Many physicians who encounter pregnant women do not have sufficient information about management of thyroid disorders and providing iodine support during pregnancy and lactation [23]. There are a few well conducted studies on iodine nutrition in pregnancy, based on median UIC [24–28]. Different studies have shown that median UIC in children cannot be used to define normal iodine nutrition in pregnancy and it is suggested that even in an iodine-sufficient area, an additional iodine supply might be necessary during pregnancy and lactation. The Yozen Fuse et al. study [29] in Japan, which is regarded as an iodine-sufficient or even excessive country, a substantial percentage of pregnant and lactating women had UICs below 100 µg/L and could be at risk for iodine deficiency if there is a restriction of iodine-rich foods. In the present study conducted in an area with iodine sufficiency, it was shown that despite a legislated national program of iodization of all household salts and the consumption of iodized salt by more than 95 % of the population, with sustained adequate median UIC among schoolchildren in all provinces of Iran during the years after national salt iodization, the median UIC of pregnant women was 87.3 µg/L, indicating moderate iodine deficiency. Only 21.8 % of subjects had UIC > 150 µg/L. In our previous study, we found variations of urinary iodine concentration (UIC) during a 4-week period among pregnant and non-pregnant women in Tehran, capital city of Iran. There was a wide intrapersonal variation in UIC values among pregnant and non-pregnant women residing in an iodine-replete area, supporting the notion that causal UIC would not reflect the iodine status of the individual [30]. Similar findings have been demonstrated in Ireland [31], Switzerland [32], Tasmania (Australia) [33], and Japan [34],whereas studies from Spain [35], France [36], and Hong Kong [37] have shown that with the progression of pregnancy, the median UIC was increased; however, a statistically significant change was observed only in Hong Kong study.
Based on trimester-specific reference ranges of thyroid parameters derived from our local study, the overall prevalence of thyroid dysfunction in Iranian pregnant women in present study was 9.5 %. According to recently published guidelines [15, 16] which recommend normal TSH value less than 2.5 mIU/L in the first trimester and 3.0 mIU/L in the second and third trimesters, in our study 395 subjects (66.8 %) had elevated TSH which was significantly higher than those using our local reference range (p < 0.001) (Fig. 1). In the present study, the frequency of positive TPOAb was 7.6 %. Pregnant women diagnosed with thyroid autoimmunity early in pregnancy should be monitored for further changes in serum TSH during the subsequent trimesters, since these women are considered to be at higher risks of early miscarriage and also of developing hypothyroidism later. There is no universal consensus regarding to prescribe levothyroxine in euthyroid women with autoimmune thyroid disease, but a few studies have addressed this issue [38].
In this study, we have found mild-to-moderate iodine deficiency among a pregnant population by measuring UIC, yet this deficiency was not reflected by other indicators (FT4 and TSH), supporting the notion that thyroid function is not considered to be a valid marker for mild-to-moderate iodine deficiency in adult. Study by Moleti and his co-worker has shown that the neuro-intellectual outcomes in children appear to be more dependent on their mothers’ nutritional iodine status than on maternal thyroid function. These results support the growing body of evidence that prenatal, mild-to-moderate iodine deficiency adversely affects cognitive development later in life, with a seemingly greater impact on verbal abilities [39].
The causal UIC reflects iodine intake over a short time period and the variation is huge, which affects the reliability of studies of iodine nutrition and UIC should not be used for the purpose of individual diagnosis and treatment. Also, some studies have confirmed that if the spot urine samples are randomly collected from a sufficient number of individuals, the median UIC obtained would be a reliable indicator of iodine status of that population and there would be no need for cumbersome and less feasible methods of measurement of iodine and/or creatinine in 24 h urine samples. The number of spot urine samples required to estimate iodine status in a population with a 95 % confidence interval within precision range of 10 and 5 % should be approximately 125 and 500, respectively [40]. A study by Antonin Korenek et al. of 168 pregnant women of the Czech Republic shows that although all participants had sufficient UIC measured as the amount of iodine in urine over 24 h during the first trimester of pregnancy, 14 subjects had elevated TSH levels signaling subclinical or incipiently clinical hypothyroidism [41]. A similar study by Raverot et al. from Lyon has shown that healthy pregnant French women are iodine deficient, and overall 11 % of them had abnormal TSH or positive TPOAb [42]. These data raise the question of what is the most appropriate indicator for assessing iodine status during pregnancy. Maternal free T4 during pregnancy is an important risk factor for impaired psychomotor development in infants and in an individual pregnant women. The best surrogate for measuring iodine sufficiency is maternal thyroid function. It seems that TSH and freeT4 may be better indicators for iodine deficiency in the first trimester, but this would require the development of method-specific and trimester-specific reference intervals for free T4 estimates in pregnancy. On the other hand, most experts would disagree with this concept.
Our study highlights the need for ongoing surveillance of the iodine status in various communities and supports the call to raise awareness of the need for pregnant women to increase their iodine intake. Many controlled iodine supplementation trials in pregnancy have been conducted in different countries which have shown no adverse effects of 50–300 µg daily iodine supplementation in moderately iodine-deficient women. These studies suggest that in areas of mild-to-moderate iodine deficiency, the maternal thyroid is able to adapt to meet the increased thyroid hormone requirements of pregnancy. Whether or not mild-to-moderate maternal iodine deficiency produces more subtle changes in cognitive and/or neurologic function in the offspring is uncertain. Mild maternal iodine deficiency may be associated with low child IQ even in the absence of TSH elevations or low FT4 [8]. Other studies using different measures of impaired maternal thyroid function also have reported developmental impairment in the offspring of affected mothers, even if maternal hypothyroidism is mild and asymptomatic [8, 43]. In a meta-analysis of 37 studies (n = 12,292 children), it was shown that the children of mothers supplemented by oral iodine either before or during pregnancy had higher scores of Intelligence Quotient (IQ) compared to children whose mothers were living in severely iodine-deficient areas [44]. The most recent NHANES survey (2005–2008) conducted in the USA indicated that 35.3 % of pregnant women had UIC less than 100 µg/L [45]. Regarding concerns about mild iodine deficiency in at least some pregnant women, the ATA recommends that all women need 150–200 µg iodine daily as dietary supplements during pregnancy and lactation [11]. WHO has also advocated daily iodine supplementation or annual iodized oil supplements for pregnant women in region where only less than 30 % of households use iodized salt. In Iran, the main source of iodine supply is iodized salt for household use and it is mandated to be fortified in the country. As the universal salt iodization may not be adequate during pregnancy, we strongly recommend supplementation of 150 µg of iodine during pregnancy, and if possible for months before conception.
References
Delange F (2000) The role of iodine in brain development. Proc Nutr Soc 59:75–79
Zimmermann MB (2009) Iodine deficiency. Endocr Rev 30:376–408
Krassas GE, Popoe K, Glinor D (2010) Thyroid function and human reproductive health. Endocr Rev 31:702–755
de Escobar GM, Ares S, Berbel P et al (2008) The changing role of maternal thyroid hormone in fetal brain development. Semin Perinatol 32:380–386
Bath SC, Walter A, Taylor A et al (2014) Iodine deficiency in pregnant women living in the South-East of the UK: the influence of diet and nutritional supplements on iodine status. Br J Nutr 111(9):1622–1631
Haddow JE, Palomaki GE, Allan WC et al (1999) Maternal thyroid deficiency during pregnancy and subsequent neuropsychological development of the child. N Engl J Med 341:549–555
Teng W, Shan Z, Patil-Sisodia K, Cooper DS (2013) Hypothyroidism in pregnancy. Lancet Diabetes Endocrinol. 1:228–237
Bath SC, Steer CD, Golding J et al (2013) Effect of inadequate iodine status in UK pregnant women on cognitive outcomes in their children: results from the Avon Longitudinal Study of Parents and Children (ALSPAC). Lancet 382:331–337
Smyth PPA (1999) Variation in iodine handling during normal pregnancy. Thyroid 9:637–642
Caldwell K, Pan Y, Mortensen ME et al (2013) Iodine status in pregnant women in the United States: National Children’s Study and National Health and Nutrition Examination Survey. Thyroid. doi:10.1089/thy.2013.0012
Becker DV, Braverman LE, Delange F et al (2006) Iodine supplementation for pregnancy and lactation—United States and Canada: recommendations of the American Thyroid Association. Thyroid 16:949–951
WHO Secretariat, Andersson M, de Benoist B, Delange F, Zupan J (2007) Prevention and control of iodine deficiency in pregnant and lactating women and in children less than 2-years-old: conclusions and recommendations of the Technical Consultation. Public Health Nutr 10:1606–1611
WHO/UNICEF (2007) Reaching optimal iodine nutrition in pregnant and lactating women and young children. Joint Statement by the WHO and the UNICEF. World Health Organization, Geneva
International Council for Control of Iodine Deficiency Disorders (2007) Iodine requirements in pregnancy and infancy. IDD Newslett 23(1):1–2
Stagnaro-Green A, Abalovich M, Alexander E et al (2012) Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid 21:1081–1125
De Groot L, Abalovich M, Alexander EK et al (2012) Management of thyroid dysfunction during pregnancy and postpartum: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 97:2543–2565
Azizi F, Sheikholeslam R, Hedayati M et al (2002) Sustainable control of iodine deficiency in Iran: beneficial results of the implementation of the mandatory law on salt iodization. J Endocrinol Invest 25(5):409–413
Azizi F, Mehran L, Sheikholeslam R et al (2008) Sustainability of a well-monitord salt iodization program in Iran: marked reduction in goiter prevalence and eventual normalization of urinary iodine concentrations without alteration in iodine content of salt. J Endocrinol Invest 32(5):422–431
Delshad H, Amouzegar A, Mirmiran P et al (2012) Eighteen years of continuously sustained elimination of iodine deficiency in the Islamic Republic of Iran: the vitality of periodic monitoring. Thyroid 22:415–421
Dunn JT, Crutchfield HE, Gutekunst R, Dunn AD (1993) Methods for measuring iodine in urine. ICCIDD/UNICEF/WHO Publications, The Netherlands
Baloch Z, Carayon P, Conte-Devolx B et al (2003) Laboratory medicine practice guidelines. Laboratory support for the diagnosis and monitoring of thyroid disease. Thyroid 13:3–126
Mehran L, Amouzegar A, Delshad H et al (2013) Trimester-specific reference ranges for thyroid hormones in Iranian pregnant women. J Thyroid Res 65:15–17
Kut A, Kalli H, Anil C, Mousa U, Gursoy A (2015) Knowledge, attitudes and behaviors of physicians towards thyroid disorders and iodine requirements in pregnancy. J Endocrinol Invest 38(10):1057–1064
Lise Brantsæter A, Hope Abel M, Haugen M, Meltzer HM (2013) Risk of suboptimal iodine intake in pregnant Norwegian women. Nutrients 5(2):424–440
Michael Hambidge K, Krebs NF, Westcott JE et al (2014) Preconception maternal nutrition: a multi-site randomized controlled trial. BMC Pregnancy Childbirth 14:111
Bath SC, Furmidge-Owen VL, Redman CWG, Rayman MP (2015) Gestational changes in iodine status in a cohort study of pregnant women from the United Kingdom: season as an effect modifier. Am J Clin Nutr 101(6):1180–1187
Bath SC, Walter A, Taylor A et al (2014) Iodine deficiency in pregnant women living in the South-East of the UK: the influence of diet and nutritional supplements on iodine status. Br J Nutr 111(9):1622–1631
Kibirige M, Hutchison S, Owen C, Delves H (2004) Prevalence of maternal dietary iodine insufficiency in the north east of England: implications for the fetus. Arch Dis Child Fetal Neonatal Ed 89(5):436–439
Yozen F, Yoshimasa S, Minoru I (2013) Gestational changes of thyroid function and urinary iodine in thyroid antibody-negative Japanese women. Endocr J 60(9):1095–1106
Amouzegar A, Azizi F (2013) Variations of urinary iodine during the first trimester of pregnancy in an iodine-replete area. Comparison with non-pregnant women. Hormones 12(1):111-11
Smyth PPA, Hetherton AMT, Smith DF et al (1997) Maternal iodine status and thyroid volume during pregnancy: correlation with neonatal iodine intake. J Clin Endocrinol Metab 82:2840–2843
Brander L, Als C, Buess H et al (2003) Urinary iodine concentration during pregnancy in an area of unstable dietary iodine intake in Switzerland. J Endocrinol Invest 26:389–396
Stilwell G, Reynolds PJ, Parameswaran V et al (2008) The influence of gestational stage on urinary iodine excretion in pregnancy. J Clin Endocrinol Metab 93:1737–1742
Fuse Y, Ohashi T, Yamaguchi S et al (2011) Iodine status of pregnant and postpartum Japanese women: effect of iodine intake on maternal and neonatal thyroid function in an iodine-sufficient area. J Clin Endocrinol Metab 96:3846–3854
Alvarez-Pedrerol M, Guxens M, Mendez M et al (2009) Iodine levels and thyroid hormones in healthy pregnant women and birth weight of their offspring. Eur J Endocrinol 160:423–429
Luton D, Alberti C, Vuillard E et al (2011) Iodine deficiency in Northern Paris area: impact on fetal thyroid mensuration. PLoS One 6(2):e14707
Kung AWC, Lao TT, Chau MT et al (2000) Goitrogenesis during pregnancy and neonatal hypothyroxinaemia in a borderline iodine sufficient area. Clin Endocrinol (Oxf) 53:725–731
Negro R, Formoso G, Mangieri T et al (2006) Levothyroxine treatment in euthyroid pregnant women with autoimmune thyroid disease: effects on obstetrical complications. J Clin Endocrinol Metab 91:2587–2591
Moleti M, Trimarchi F, Tortorella G, Candia Longo A, Giorgianni G, Sturniolo G, Alibrandi A, Vermiglio F (2016) Effects of maternal iodine nutrition and thyroid status on cognitive development in offspring: a pilot study. Thyroid 26(2):296–305
Andersen S, Karmisholt J, Pedersen KM, Laurberg P (2008) Reliability of studies of iodine intake and recommendations for number of samples in groups and individuals. Br J Nutr 99:813–818
Korenek A, Prochazka M (2008) Iodine in early pregnancy—is there enough? Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 152(1):97–99
Raverot V, Bournaud C, Sassolas G et al (2012) Pregnant French women living in the Lyon area are iodine deficient and have elevated serum thyroglobulin concentrations. Thyroid 22:522–528
Zimmermann MB (2009) Iodine deficiency in pregnancy and the effects of maternal iodine supplementation on the offspring: a review. Am J Clin Nutr 89:668S–672S
Qian M, Wang D, Watkins WE et al (2005) The effects of iodine on intelligence in children: a meta-analysis of studies conducted in China. Asia Pac J Clin Nutr 14:32–42
Caldwell KL, Makhmudov A, Ely E, Jones RL, Wang RY (2011) Iodine Status of the U.S Population, National Health and Nutrition Examination Survey, 2005–2006 and 2007–2008. Thyroid 21:419–427
Acknowledgments
The authors wish to thank the participants for their cooperation in this study. The authors also wish to acknowledge Ms. Niloofar Shiva for critical editing of English grammar and syntax of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any commercial associations which might create a conflict of interest in connection with this manuscript.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
No informed consent.
Rights and permissions
About this article
Cite this article
Delshad, H., Touhidi, M., Abdollahi, Z. et al. Inadequate iodine nutrition of pregnant women in an area of iodine sufficiency. J Endocrinol Invest 39, 755–762 (2016). https://doi.org/10.1007/s40618-016-0438-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-016-0438-4