Abstract
Background
Sport-related concussion is recognized as a significant injury with variable recovery rates.
Objective
This study defined the acute natural history of sport concussion in male and female collegiate athletes participating in a broad array of sports.
Methods
We conducted a prospective, longitudinal investigation among collegiate student athletes (n = 34,709) from 30 academic institutions. Primary outcomes included the time (days) from injury until initiation of a return to participation (RTP) protocol and time from injury until medical clearance for unrestricted RTP.
Results
Concussed athletes (n = 1751, 19.2 years, 63.2% male) participating in 22 different sports began the RTP protocol in a median 6.4 (IQR 3.7–11.8) days. Time to initiate the RTP protocol was lengthened by less frequent post-injury assessments, greater initial post-injury symptom severity, limited contact sports participation, practice/training injuries, and three or more prior concussions. The median total RTP duration was 12.8 (IQR 8.7–20.1) days. Total RTP duration was shorter with ADHD medication usage, males, and greater assessment frequency; while greater initial post-injury symptom severity, practice-/training-related injuries, and three or more prior concussions had longer recoveries.
Conclusion
Although median recovery times are consistent with previous guidelines, it was not until 1 month post-injury that a preponderance of collegiate athletes were cleared to begin the RTP protocol (92%) or cleared for unrestricted sport participation (85%). Intrinsic and extrinsic factors had a small effect, altering recovery trajectories by up to 2 days, suggesting a largely unified approach to post-injury monitoring and management across all athletes. These data represent a shift from previous classification parameters of normal clinical recovery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Symptom resolution and return to play from sport-related concussion may take up to 1 month in a preponderance of athletes. |
Some intrinsic (e.g., concussion history) and extrinsic (e.g., injury setting) factors may modify recovery, but with minimal impact on the overall time course. |
Normal recovery from sport-related concussion should be viewed as a process taking up to 1 month, double that of previous recommendations. |
1 Introduction
Concussion (i.e., mild traumatic brain injury) occurring during sport participation has been the focus of increasing public concern over the past decade. Despite increased research efforts, the natural history of concussion across diverse cohorts of collegiate athletes remains undefined, as the preponderance of sport concussion research has centered on male athletes participating in collision sports and female soccer athletes.
Sport-related concussion (SRC) recovery is widely viewed as rapid with medical organizations [1, 2] and international consensus [3] demarcating normal clinical recovery within 10–14 days, while those with symptoms persisting beyond 2 weeks categorized as ‘atypical’. Subsequent to symptom resolution is the implementation of a standardized return to participation (RTP) protocol. The most widely employed RTP protocol is a six-step progression that moves the athlete through increasingly more intense exercise before a return to unrestricted sport participation [3]. Unfortunately, generalizability of this recovery classification is limited by a dearth of research on female athletes, those participating in sports traditionally viewed at low risk for injury (e.g., swimming), and other pre- and post-morbid characteristics.
A systematic review of factors influencing concussion recovery among adults identified a history of migraine and mental health diagnoses (e.g., depression), as well as the initial symptom burden, as factors that may prolong the recovery trajectory, while attention-deficit/hyperactivity disorder (ADHD) did not [4]. Other potential moderators of recovery such as concussion history [5], somatization [6], loss of consciousness [5], post-traumatic amnesia [5], injury setting (i.e., practice vs competition), and type of sport participation have been less well studied.
As such, this investigation sought to evaluate the concussion recovery trajectory using three post-injury intervals: (1) time from injury until initiation of an RTP protocol, (2) length of the RTP protocol, and (3) time from injury to unrestricted RTP.
2 Methods
The CARE Consortium is a 30-site investigation designed to delineate the acute natural history and neurobiology of concussion. Standardized methods for each participating site are detailed in a prior publication [7]. All varsity-level athletes at 26 civilian institutions and all varsity athletes and cadets from four military service academies within the United States were eligible to participate. All sites obtained local approval from their respective Institutional Review Board and the United States Army Human Research Protection Office prior to data collection. This study was conducted in accordance with the Declaration of Helsinki.
Following written informed consent, each participant completed a pre-season baseline evaluation consisting of participant demographics and medical history, and evaluations of symptoms, postural control, mental status, and neurocognitive functioning. During the baseline evaluation, participants self-reported medical history (diagnosed conditions when appropriate) and concussion history using a custom-built electronic data capture platform [7] and sports were categorized as Contact (e.g., football, ice hockey), Limited Contact (e.g., baseball/softball), or Non-Contact (e.g., golf, track) [8].
The local physicians used a common concussion definition [9] for injury diagnosis and post-injury protocol. Following concussion diagnosis, participants were administered research assessments at five additional post-injury time points: at least once within 48 h of injury, at RTP protocol initiation, when cleared for unrestricted RTP, and 6 months post-injury. Post-injury evaluations implemented the same measures as the baseline examination. Between the initial post-injury evaluation and RTP protocol initiation, concussed participants reported symptom status to the study staff in-person daily, for up to 14 days, and then once a week if they had not returned to unrestricted play. Medical teams may have elected to continue administering daily symptom inventories for clinical purposes, but those data were not included in the research database or analysis for this paper. Symptoms were captured using the Sport Concussion Assessment Tool (SCAT) symptom list, a 22-item inventory with severity ranked on a 0–6 scale. Percent completion of the daily symptom reports was used as a metric of clinical care assessment frequency. Consistent with medical [1, 2] and international [3] recommendations, the presiding clinician’s exam served as the gold standard throughout post-injury management, clearing concussed athletes to begin a standardized RTP protocol [3] (RTP protocol initiation) and again for unrestricted RTP (i.e., first available practice or competition following medical clearance). Metrics other than the clinical exam and symptom evaluations may have been used by clinicians to determine the initiation of the RTP protocol and determine the timing of unrestricted RTP, but were not included in this analysis or captured in the study database. Study team members recorded study-related variables on paper case report forms that were later entered into the database by trained research team members.
2.1 Participants
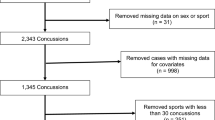
Between Fall 2014 and Spring 2018, 38,888 athletes and cadets were enrolled in the investigation. As the intent of this paper was to characterize the natural history of SRC, service academy cadets who did not participate in varsity sport [10] (n = 10,522) were excluded from this analysis. Following enrollment and intake, 2256 incident concussions were recorded. Concussions occurring outside of varsity sport participation (e.g., motor vehicle accident, n = 340), and repeat sport-related concussions occurring during the study period (n = 207) were excluded. A total of 1709 athletes sustaining a concussion while participating in 22 unique National Collegiate Athletic Association (NCAA) sports were included in the analyses (Fig. 1).
2.2 Data Analysis
Descriptive statistics for continuous variables and percentages for categorical variables were calculated for baseline demographic and examination measures, injury characteristics, and post-injury (within 48 h) measures. The primary outcomes used to characterize SRC recovery trajectory were days from injury until RTP protocol initiation, days of the RTP protocol, and total RTP duration. Athletes who had not reached RTP initiation or full RTP when the season ended and follow-up was not readily available (e.g., practices were no longer scheduled and/or athletes left campus) were right censored for analysis. Thus, an exact time for RTP or RTP protocol initiation could not be determined. Using time-to-event (survival) analysis techniques, censored observations (those where RTP initiation and/or full RTP were not captured) are included in the analysis up until the censoring time. This included 220 athletes at RTP protocol initiation and an additional 28 athletes at the unrestricted RTP point.
Model building for time to RTP protocol initiation, RTP protocol duration, and total RTP duration were completed using a series of univariate Cox proportional hazard models and Kaplan–Meier survival curves fit to screen all of the candidate predictors. The univariate concussion history analysis showed no differences among those with a history of zero (n = 965), one (n = 539) or two (n = 147) concussions; therefore, further analyses used a dichotomous measure (≤ 2 [n = 1651] vs ≥ 3 prior concussions [n = 67]). The candidate variables that had a p-value of ≤ 0.20 in the univariate models were then divided into three groups: baseline, injury, and post-injury. A p-value of ≤ 0.20 was selected to reduce the number of candidate predictors but allow those that may be important in multivariable models to remain. The p-value of < 0.20 for the initial screening of the many candidate independent variables was chosen based on a simulation study [11] indicating that screening variables with significance tests perform well as long as the significance level is set to 0.20 or higher.
Within each of those time domain subsets, the best model was chosen among all possible models based on the Akaike Information Criteria (AIC). Minimizing the AIC returns the best fitting, most parsimonious model. Utilizing minimization of AIC in the analysis to construct multivariable models requires that a dataset with only cases with a complete set of independent variables be used. This is due to the fact that a difference in sample size (because of missing data) changes the number of degrees of freedom, which in turn affects the AIC. Proper comparison cannot occur between the AICs of different models unless the sample size is the same. Because of this, we chose study variables that had lower levels of missing data in order to maximize the complete sample size. The variables identified in the three separate time domain models were then combined and the best overall multivariable model for each time to event interval (RTP protocol initiation, protocol duration, and total RTP duration) was chosen based on the AIC. Separation based on the time point domain was done primarily due to missing values at the post-injury time point. Since AIC requires complete cases of all variables, running them separately and then again together is essentially a sensitivity analysis to ensure that the missing values are not causing the results to be skewed. Time to RTP initiation and total RTP time have a subjective component based on clinical judgement that could induce a correlation among observations at a given institution. Therefore, as a sensitivity analysis, we fit Cox proportional hazards regression models with generalized estimating equations to adjust for any within-school correlation for each of the final models. Although the hazard ratios changed slightly, the statistical significance levels and the interpretation of the results were unchanged. Data were analyzed using R Studio (version 1.1.463) and median values (interquartile range [IQR]), along with hazard ratios and 95% confidence intervals (CI) are reported.
3 Results
At the time of enrollment, the 1751 concussed participants (63% male) were 19.2 (± 1.3) years old, participating in Contact (79%, n = 1387), Limited Contact (17%, n = 292), and Non-Contact (4%, n = 72) sports. Overall, concussions occurring during American football (34.6%) were the most prevalent. Concussed female athletes most commonly participated in football (i.e., soccer) (23.4%), volleyball (14.0%), basketball (12.9%), and lacrosse (8.4%). Concussed male athletes most commonly participated in American football (54.7%), football (10.7%), basketball (6.8%), and wrestling (6.4%). Table 1 provides other baseline and post-injury characteristics.
3.1 Time from Injury to Return to Participation Protocol Initiation
Across all athletes participating in all sport and injury settings, the median (IQR) duration from the date of injury to RTP protocol initiation was 6.4 (3.7–11.8) days with 55% (95% CI 52.7–57.3) beginning the RTP protocol by day 7, 81% (95% CI 79.2–82.9) by day 14, 90% (95% CI 88.1–91.0) by day 21, and 92% (95% CI 90.7–93.3) by day 28 post-injury (Fig. 2).
3.1.1 Variable Screening
To identify factors influencing the recovery time course, univariate survival analyses of categorical data identified sex (p = 0.10), concussion history (p = 0.006), ADHD diagnosis (p = 0.10), sport category (p = 0.03), ADHD medication use after injury (p = 0.11), and injury setting (p < 0.004) as having a significant relationship with time to RTP protocol initiation. Table 2 presents the median time to RTP protocol initiation for each variable. Table 3 presents the weekly cumulative percent of athletes initiating the RTP protocol up to 1-month post-injury for each variable. Kaplan–Meier recovery curves for these variables are available in the Electronic Supplemental Material (ESM).
Among the continuous variables, Brief Symptom Inventory (BSI) Somatization score (p = 0.20, hazard ratio [HR] 0.98 [95% CI 0.96–1.01]), symptom severity at baseline (p = 0.09, HR 0.995 [95% CI 0.99–1.00]); post-injury symptom severity (p < 0.001, 0.99 [95% CI 0.99–0.99]), post-injury assessment Balance Error Scoring System (BESS) Firm score (p < 0.001, HR 0.977 [95% CI 0.96–0.99]), Standardized Assessment of Concussion (SAC) total score at post-injury (p < 0.001, HR 1.04 [95% CI 1.02–1.06]), and post-injury assessment frequency (p < 0.001, HR 1.01 [95% CI 1.01–1.02]) were significant predictors of time to RTP protocol initiation and were included in the multivariate models. Self-reported migraine diagnosis (p = 0.44), observed loss of consciousness (p = 0.94), and observed post-traumatic amnesia (p = 0.24) did not reach the level of significance necessary to suggest a relationship with symptom resolution.
3.1.2 Model Building
The best model using baseline measures based on the AIC included three variables that were associated with longer time to RTP protocol initiation: three or more prior concussions, higher baseline symptom severity score, and sport category (limited and non-contact compared with contact sports). For measures recorded at the time of injury, only concussions occurring during practice/training significantly extended symptom resolution. Finally, among post-injury variables, higher post-injury symptom severity scores within 48 h of injury and less frequent daily post-injury symptom evaluation were associated with longer times, while ADHD medication taken after injury was associated with shorter times.
The best overall multivariable proportional hazards model, as determined by minimizing the AIC, included five variables. Athletes participating in limited-contact sports, reporting higher post-injury symptom severity score, injury in practice/training, and self-report of three or more prior concussions were all associated with a longer time to RTP protocol initiation. Those with a higher daily post-injury symptom assessment frequency had shorter times to RTP protocol initiation. Hazard ratios and 95% confidence intervals for the final model are presented in Table 4.
3.2 Return to Participation Protocol Duration
Across all athletes participating in all sport and injury settings, the median duration of the RTP protocol was 6.0 (IQR 4.0–7.3) days, with 73% (95% CI 71–75) completing the protocol within 7 days, 92% (95% CI 91–93) within 14 days, 95% (95% CI 94–96) within 21 days, and 97% (95% CI 96–98) within 28 days.
3.2.1 Variable Screening
To identify factors influencing the recovery time course, univariate survival analyses of identified injury setting (p = 0.004), gender (p = 0.07), concussion history (p = 0.02), ADHD medications after injury (p = 0.20), athletic division (p = 0.03), daily post-injury assessment frequency (p = 0.004), post-injury symptom severity (p < 0.001), BSI somatic raw score at baseline (p = 0.14), and BESS Firm Score (p = 0.19) as having a significant relationship with RTP protocol duration. Table 2 presents the median time of the RTP protocol for each variable.
3.2.2 Model Building
The best model using all possible predictors chosen by the AIC method included eight variables: daily post-injury assessment frequency, post-injury SCAT symptom severity score, injury setting, previous concussions, baseline SCAT symptom severity score, athletic division, ADHD medications after injury, and baseline BSI-18 Somatic score. Hazard ratios and 95% confidence intervals for the final model are presented in Table 4.
3.3 Total Return to Participation Duration
Across all athletes participating in all sport and injury settings, the median total RTP duration was 12.8 (IQR 8.7–20.1) days, with 16% (95% CI 14–18) returning to participation by day 7, 57% (95% CI 55–60) by day 14, 77% (95% CI 75–79) by day 21 and 85% (95% CI 84–87) by day 28 post-injury (Fig. 2).
To identify factors influencing the recovery time course, univariate survival analyses of categorical data identified sex (p = 0.043), concussion history (p = 0.002), ADHD diagnosis (p = 0.10), sport category (p = 0.08), athletic division (p = 0.17), ADHD medication post-injury (p = 0.04), and injury setting (p = 0.0013) as having a significant relationship with total RTP duration. Table 2 presents the median time to return to participation for each variable. Table 3 presents the weekly cumulative percent of athletes for total RTP duration up to 1 month post-injury for each variable. Kaplan–Meier recovery curves for these variables are available in the ESM.
Among the continuous variables, post-injury assessment SAC Total Score (p = 0.0014, HR 1.03 [95% CI 1.01–1.05]), symptom severity at baseline (p = 0.10, HR 0.995 [0.990–1.000]); post-injury symptom severity (p < 0.001, HR 0.988 [0.986–0.991]), post-injury assessment BESS Firm score (p = 0.0042, HR 0.981 [0.968–0.994]), and post-injury assessment frequency (p < 0.001, HR 1.01 [1.008–1.013]) were significant predictors of total RTP duration and included in the multivariate models. History of migraine diagnosis (p = 0.44), loss of consciousness (p = 0.94), and post-traumatic amnesia (p = 0.24) did not reach the level of significance necessary to suggest a relationship with RTP.
3.3.1 Model Building
The best model using baseline measures based on the AIC included three variables that were associated with longer time to RTP: limited and non-contact sports, three or more prior concussions, and athletic Division III. For measures recorded at the time of injury, only concussion during practice/training compared with competition was significant. Finally, among post-injury variables, higher symptom severity score and less frequent daily post-injury symptom assessment were associated with longer RTP times, while ADHD medication usage following injury was associated with shorter times.
The best overall multivariable proportional hazards model, as determined by minimizing the AIC, included six variables. Longer RTP times were associated with higher post-injury symptom severity score, practice/training injuries, and self-report of three or more prior concussions. ADHD medication usage, male sex, and those with higher symptom assessment frequency returned to play sooner. Hazard ratios and 95% confidence intervals for the final model are presented in Table 4.
4 Discussion
Findings from this nationwide prospective study delineate the natural history of SRC recovery and RTP in male and female athletes participating across the full array of collegiate sports. Following a diagnosed concussion, the overall median time to initiate the RTP protocol neared 1 week, followed by a 6-day RTP protocol, resulting in a total median RTP duration approaching 2 weeks. The recovery metrics for time to RTP protocol initiation (6.4 days) and total RTP duration (12.8 days) in our population are skewed towards a rapid improvement, although 19% of the athletes continued to report symptoms at 2 weeks and 8% remained symptomatic 28 days post-injury. Furthermore, nearly half had not returned to play 2 weeks after injury and 15% had not returned to play 1 month after injury. A number of factors altered the recovery trajectory up to 2 days. Collectively, the individual variability in injury recovery documented here (see Fig. 2) and elsewhere [12,13,14,15] suggests that describing ‘normal recovery’ (defined by time for resolution of symptoms and clearance for unrestricted RTP) as occurring within a 2-week timeframe [3] may be too restrictive and does not accurately characterize the entirety of injury recovery. Setting the expectation that symptom resolution and the return to play process may take up to 4 weeks more fully encapsulates ‘normal recovery’ for the preponderance of collegiate athletes and potentially reduces any stigma surrounding those requiring the full duration.
Concussion recovery reported in this study is considerably longer than that found in seminal work conducted in American football athletes, reporting a median time of 2.0 days to RTP protocol initiation and an additional 3.0 days until unrestricted RTP [16]. While this study was not designed to identify the underpinnings of the lengthened timeline, attention around concussive injuries has increased considerably over the previous decade, with enhanced athlete awareness [17] and improved clinical care [14]. These efforts have resulted in an overall upward trend in symptom reporting duration [18], suggesting an increased concussion symptoms awareness and/or willingness to acknowledge them, and the implementation of a standardized RTP protocol [3]. The RTP protocol duration in this study spanned 6.4 days, contributing significantly to the increase in total RTP duration among American football athletes from the 1999–2001 and 2014–2017 academic years (3.0 days vs 12.2 days) [14]. Others have conducted similar evaluations in a pediatric cohort, reporting a slightly smaller percentage of the youth athletes achieving asymptomatic status at 1 month [19, 20]—a finding that is consistent with a meta-analytic review evaluating age-related differences [21].
The univariate and multivariate models identified both injury and non-injury (e.g., demographics) factors that influenced the time to RTP protocol initiation and total RTP duration (Tables 2 and 4). While the magnitude of difference between the groups was 1–2 days (Table 2), each analytical approach informs clinical care by alerting clinicians to what factors should be investigated and attended to in understanding and evaluating the recovery trajectory after concussion. For example, those participating in contact/collision sports (e.g., football) had symptom resolution and full RTP status 1–2 days sooner than those in limited contact and non-contact sports. The factors explaining this difference were unexplored, but others have noted that collision sport athletes are the most likely to conceal concussion-related symptoms at the time of injury [22], suggesting they may also mitigate symptoms to accelerate their return to sport.
The context of the concussive injury also appears to play a role in recovery. Concussions occurring during competitions required 1.4 fewer days to initiate the RTP protocol and 1.8 days less for total RTP duration. Shorter recovery times for game injuries may be related to increased medical vigilance resulting in faster identification and removal from play, a factor independently associated with recovery duration [23]. Furthermore, a large proportion of practice-related concussions (33.6%) occurred during the off-season, when time allotments allow the medical staff to be more conservative in progressing an athlete back to participation.
Consistent with prior work, higher initial post-injury symptom burden lengthened the recovery process [12], perhaps serving as a marker of injury severity [24]. Also of interest is the association between symptoms evaluation frequency and reduced symptom resolution time and total RTP duration. It is doubtful that a daily evaluation alters the biological recovery process, but missed symptom checks within the first 14 days may preclude the clinician from identifying the exact day of resolution and initiation of the RTP protocol. In addition, local medical staff may not perform daily checks in athletes with persistent but stable symptoms, but rather ask the athlete to return when the symptom presentation changes.
Concussion history has long been described as a factor influencing recovery, with each successive injury lengthening the recovery time [5]. Our findings noted that only those reporting three or more injuries prior to the incident concussion had a significantly longer recovery process, taking 1.6 and 1.3 days longer to begin the RTP protocol and for total RTP duration, respectively. While cumulative biological injury may have influenced the time to RTP protocol initiation, it did not appear to change the RTP protocol duration, suggesting clinicians are managing those with and without a significant concussion history similarly.
The biological foundation for shorter recoveries among those reporting ADHD medication use is beyond the scope of this investigation. ADHD medication use decreased from baseline (32%) to post-injury (28%) among those reporting an ADHD diagnosis. Regardless, ADHD medications are known to improve concentration [25], which may have assisted athletes in a post-concussion state to perform normally on concussion assessments. These findings, however, are consistent with a systematic review addressing concussion recovery in those with ADHD [4], but differ from work done in pediatric participants [26].
A shortening of the recovery timeline was noted between sexes, whereby male athletes reported RTP protocol initiation 1 day earlier than females (6 vs 7 days) and a total RTP duration that was abbreviated by 1.0 day (12.2 vs 13.2 days). Women are known to have higher concussion rates when participating in comparable sporting events (e.g., soccer) [27] and larger post-concussion deficits [28], but the literature directly addressing concussion recovery duration between the sexes is sparse and the findings are mixed [15, 29]. Females aged < 18 years are at greater risk for protracted symptoms [30], but the reasons are not clear, although some have speculated that recovery differences may have biological underpinnings [31] or females may have a greater willingness to disclose symptoms [22].
This study is not without limitations. As with all investigations, selection bias (e.g., by institution, sport, division) is a potential concern. The large and diverse sample of collegiate sports and athletes minimizes this concern. In addition, the data are regularly screened and compared against national collegiate participation rates, which mirrors the expected distribution of athletes with regard to attributes such as year (freshman, sophomore etc.), sport, position, etc. Further, concussion remains a clinical diagnosis with no objective gold standard, making it likely for there to be variation in how injuries were diagnosed and managed both within and between the 30 participating sites. A standard definition [9] was provided to all study personnel and slight variation in its interpretation strengthens the clinical applicability of our findings. Some athletes may also have been motivated to mitigate their symptom experience, unnaturally shortening the time to RTP protocol initiation. While this is certainly possible, recent findings demonstrate RTP protocol initiation duration has increased over the previous two decades [14], likely showing a more accurate representation of the recovery timeline. Notably, this investigation addresses symptom resolution and tolerance to resumption of full activity, which may not equate with neurobiological recovery. Additional research is needed to evaluate the recovery trajectory of concussed athletes on standard and emerging post-concussion metrics as there is some emerging evidence that return to pre-injury baseline measures of brain structure and function may lag behind both resolution of symptoms and tolerance of full participation [32,33,34]. The factors investigated herein do not preclude others from influencing the recovery trajectory. For example, pre-existing factors and post-injury rehabilitation may influence injury recovery, but were not adequately captured to allow for inclusion in the recovery models. Lastly, we categorized sports based on injury risk [8], leaving room for additional research investigating recovery profiles for athletes participating in specific sports and events outside of collegiate athletics. Conversely, this investigation has several strengths, notably the size, breadth, and prospective study design that is nationally representative of young adults participating in sports across the entire spectrum of collegiate athletics.
5 Conclusions
This investigation highlights the enhanced understanding of concussion, concussion recovery, and influence of modern approaches to concussion management across a diverse cohort of male and female athletes participating in multiple collegiate sports. Consistent with prior work, our findings demonstrate variability in injury recovery duration, but the normal recovery window from concussion for a majority of athletes may extend up to 1 month. Both intrinsic and extrinsic factors slightly altered that timeline, but the magnitude of influence on the recovery trajectory between cohorts was ultimately small, suggesting a largely unified approach to recovery monitoring and concussion management across all athletes in all sports. As such, clinicians should continue implementing a conservative management strategy based on contemporary guidelines, but remain aware of the reality of individual athlete recovery trajectories and associated risk factors.
References
Broglio SP, Cantu RC, Gioia GA, Guskiewicz KM, Kutcher J, Palm M, et al. National Athletic Trainers’ Association position statement: management of sport concussion. J Athl Train. 2014;49(2):245–65.
Harmon KG, Clugston JR, Dec K, Hainline B, Herring S, Kane SF, et al. American Medical Society for Sports Medicine position statement on concussion in sport. Br J Sports Med. 2019;53(4):213–25.
McCrory P, Meeuwisse W, Dvorak J, Aubry M, Bailes J, Broglio S, et al. Consensus statement on concussion in sport-the 5(th) international conference on concussion in sport held in Berlin, October 2016. Br J Sports Med. 2017;51(11):838–47.
Iverson GL, Gardner AJ, Terry DP, Ponsford JL, Sills AK, Broshek DK, et al. Predictors of clinical recovery from concussion: a systematic review. Br J Sports Med. 2017;51(12):941–8.
Guskiewicz KM, McCrea M, Marshall SW, Cantu RC, Randolph C, Barr W, et al. Cumulative effects associated with recurrent concussion in collegiate football players: the NCAA Concussion Study. JAMA. 2003;290(19):2549–55.
Nelson LD, Tarima S, LaRoche AA, Hammeke TA, Barr WB, Guskiewicz K, et al. Preinjury somatization symptoms contribute to clinical recovery after sport-related concussion. Neurology. 2016;86(20):1856–63.
Broglio SP, McCrea M, McAllister T, Harezlak J, Katz B, Hack D, et al. A National Study on the effects of concussion in collegiate athletes and US Military Service Academy Members: The NCAA-DoD Concussion Assessment, Research and Education (CARE) Consortium structure and methods. Sports Med. 2017;47(7):1437–51.
Rice SG. American Academy of Pediatrics Council on Sports M, Fitness. Medical conditions affecting sports participation. Pediatrics. 2008;121(4):841–8.
Carney N, Ghajar J, Jagoda A, Bedrick S, Davis-O’Reilly C, du Coudray H, et al. Concussion guidelines step 1: systematic review of prevalent indicators. Neurosurgery. 2014;75(Suppl 1):S3-15.
Van Pelt KL, Allred D, Cameron KL, Campbell DE, D’Lauro CJ, He X, et al. A cohort study to identify and evaluate concussion risk factors across multiple injury settings: findings from the CARE Consortium. Inj Epidemiol. 2019;6(1):1.
Mickey RM, Greenland S. The impact of confounder selection criteria on effect estimation. Am J Epidemiol. 1989;129(1):125–37.
McCrea M, Guskiewicz K, Randolph C, Barr WB, Hammeke TA, Marshall SW, et al. Incidence, clinical course, and predictors of prolonged recovery time following sport-related concussion in high school and college athletes. J Int Neuropsychol Soc. 2013;19(1):22–33.
Master CL, Katz BP, Arbogast KB, McCrea MA, McAllister TW, Pasquina PF, Lapradd M, Zhou W, Broglio SP, CARE Consortium Investigators. Differences in sport-related concussion for female and male athletes in comparable collegiate sports: a study from the NCAA-DoD Concussion Assessment, Research and Education (CARE) Consortium. Br J Sports Med. 2020;. https://doi.org/10.1136/bjsports-2020-103316.
McCrea M, Broglio S, McAllister T, Zhou W, Zhao S, Katz B, et al. Return to play and risk of repeat concussion in collegiate football players: comparative analysis from the NCAA Concussion Study (1999–2001) and CARE Consortium (2014–2017). Br J Sports Med. 2020;54(2):102–9.
Henry LC, Elbin RJ, Collins MW, Marchetti G, Kontos AP. Examining recovery trajectories after sport-related concussion with a multimodal clinical assessment approach. Neurosurgery. 2016;78(2):232–41.
McCrea M, Guskiewicz KM, Marshall SW, Barr W, Randolph C, Cantu RC, et al. Acute effects and recovery time following concussion in collegiate football players: the NCAA Concussion Study. JAMA. 2003;290(19):2556–63.
Gibson TB, Herring SA, Kutcher JS, Broglio SP. Analyzing the effect of state legislation on health care utilization for children with concussion. JAMA Pediatr. 2015;169(2):163–8.
Wasserman EB, Kerr ZY, Zuckerman SL, Covassin T. Epidemiology of sports-related concussions in National Collegiate Athletic Association Athletes from 2009–2010 to 2013–2014: symptom prevalence, symptom resolution time, and return-to-play time. Am J Sports Med. 2016;44(1):226–33.
Zemek R, Barrowman N, Freedman SB, Gravel J, Gagnon I, McGahern C, et al. Clinical risk score for persistent postconcussion symptoms among children with acute concussion in the ED. JAMA. 2016;315(10):1014–25.
Ledoux AA, Tang K, Yeates KO, Pusic MV, Boutis K, Craig WR, et al. Natural progression of symptom change and recovery from concussion in a pediatric population. JAMA Pediatr. 2019;173(1): e183820.
Williams RM, Puetz TW, Giza CC, Broglio SP. Concussion recovery time among high school and collegiate athletes: a systematic review and meta-analysis. Sports Med. 2015;45(6):893–903.
Kerr ZY, Register-Mihalik JK, Kroshus E, Baugh CM, Marshall SW. Motivations associated with nondisclosure of self-reported concussions in former collegiate athletes. Am J Sports Med. 2016;44(1):220–5.
Asken BM, Bauer RM, Guskiewicz KM, McCrea MA, Schmidt JD, Giza CC, et al. Immediate removal from activity after sport-related concussion is associated with shorter clinical recovery and less severe symptoms in collegiate student-athletes. Am J Sports Med. 2018;46(6):1465–74.
Mustafi SM, Harezlak J, Koch KM, Nencka AS, Meier TB, West JD, et al. Acute white-matter abnormalities in sports-related concussion: a diffusion tensor imaging study from the NCAA-DoD CARE consortium. J Neurotrauma. 2018;35(22):2653–64.
Taylor E, Dopfner M, Sergeant J, Asherson P, Banaschewski T, Buitelaar J, et al. European clinical guidelines for hyperkinetic disorder—first upgrade. Eur Child Adolesc Psychiatry. 2004;13(Suppl 1):I7-30.
Beauchamp MH, Aglipay M, Yeates KO, Desire N, Keightley M, Anderson P, et al. Predictors of neuropsychological outcome after pediatric concussion. Neuropsychology. 2018;32(4):495–508.
Zuckerman SL, Kerr ZY, Yengo-Kahn A, Wasserman E, Covassin T, Solomon GS. Epidemiology of sports-related concussion in NCAA athletes from 2009–2010 to 2013–2014: incidence, recurrence, and mechanisms. Am J Sports Med. 2015;43(11):2654–62.
Covassin T, Elbin RJ, Harris W, Parker T, Kontos A. The role of age and sex in symptoms, neurocognitive performance, and postural stability in athletes after concussion. Am J Sports Med. 2012;40(6):1303–12.
Black AM, Sergio LE, Macpherson AK. The epidemiology of concussions: number and nature of concussions and time to recovery among female and male Canadian varsity athletes 2008 to 2011. Clin J Sport Med. 2017;27(1):52–6.
Yeates KO, Tang K, Barrowman N, Freedman SB, Gravel J, Gagnon I, et al. Derivation and initial validation of clinical phenotypes of children presenting with concussion acutely in the emergency department: latent class analysis of a multi-center, prospective cohort. Observational Study. J Neurotrauma. 2019;36(11):1758–67.
Bazarian JJ, Blyth B, Mookerjee S, He H, McDermott MP. Sex differences in outcome after mild traumatic brain injury. J Neurotrauma. 2010;27(3):527–39.
Stephens JA, Liu P, Lu H, Suskauer SJ. Cerebral blood flow after mild traumatic brain injury: associations between symptoms and post-injury perfusion. J Neurotrauma. 2018;35(2):241–8.
Kamins J, Bigler E, Covassin T, Henry L, Kemp S, Leddy JJ, et al. What is the physiological time to recovery after concussion? A systematic review. Br J Sports Med. 2017;51(12):935–40.
McCrea M, Meier T, Huber D, Ptito A, Bigler E, Debert CT, et al. Role of advanced neuroimaging, fluid biomarkers and genetic testing in the assessment of sport-related concussion: a systematic review. Br J Sports Med. 2017;51(12):919–29.
Acknowledgements
The authors would like to thank Ryan Tierney (Temple University), Patrick O'Donnell (United States Coast Guard Academy), Josh Goldman (University of California at Los Angeles), Kevin Guskiewicz (University of North Carolina at Chapel Hill), Mickey Collins (University of Pittsburgh), Jeff Bazarian (University of Rochester), Laura Lintner (Wake Forest University), Brian Dyhuizen (Wilmington College), Paul Pasquina (Uniformed Services University), Jaroslaw Harezlak, Jody Harland, Janetta Matesan, and Larry Riggen (Indiana University), Nicole L’Heureux and Ashley Rettmann (University of Michigan), Melissa Koschnitzke (Medical College of Wisconsin), Michael Jarrett, Vibeke Brinck, and Bianca Byrne (Quesgen), Thomas Dompier, Christy Collins, Melissa Baker, and Sara Quetant (Datalys Center for Sports Injury Research and Prevention), and the research and medical staff at each of the participating sites.
CARE Consortium Investigators include (alphabetical by institution): April Hoy MS, ATC, CSCS (Azusa Pacific University), Joseph B. Hazzard, EdD, ATC (Bloomsburg University), Louise A. Kelly, PhD (California Lutheran University), John DiFiori, MD (Hospital for Special Surgery), Justus D. Ortega, PhD (Humbolt State University), Nicholas Port, PhD (Indiana University), Margot Putukian (Princeton University), Dianne Langford, PhD and Jane McDevitt PhD, ATC, CSCS (Temple University), Darren Campbell, MD, Jonathan C. Jackson, MD, and Gerald McGinty, PT, DPT (United States Air Force Academy), Carlos Estevez, DPT (United States Coast Guard Academy), Kenneth L. Cameron, PhD, MPH, ATC, Megan N. Houston, PhD, ATC, and Steven J. Svoboda, MD (United States Military Academy), Adam James Susmarski, DO (United States Naval Academy), Chris Giza, MD (University of California at Los Angeles), Holly J. Benjamin, MD (University of Chicago), Thomas W. Kaminski, PhD, ATC and Thomas Buckley, EdD, ATC (University of Delaware), James R. Clugston, MD, MS, CAQSM (University of Florida), Julianne Schmidt, PhD, ATC (University of Georgia), Luis A. Feigenbaum, PT, DPT, ATC (University of Miami), J. T. Eckner, MD (University of Michigan), Jason Mihalik, PhD, CAT(C), ATC (University of North Carolina at Chapel Hill), Jessica Dysart Miles, PhD, ATC (University of North Georgia), Scott Anderson, ATC (University of Oklahoma), Kristy Arbogast, PhD and Christina L. Master, MD, CAQSM (University of Pennsylvania), Anthony P. Kontos, PhD (University of Pittsburgh), Sara P. D. Chrisman, MD, MPH (University of Washington), M. Alison Brooks, MD MPH (University of Wisconsin-Madison), Steve Rowson, PhD and Stefan M. Duma, PhD (Virginia Tech), Chris Miles, MD (Wake Forest University).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Funding
Data collection and sharing for this project was conducted with support from the National Collegiate Athletic Association (NCAA) and the Department of Defense (DOD). The U.S. Army Medical Research Acquisition Activity, 820 Chandler Street, Fort Detrick MD 21702-5014, is the awarding and administering acquisition office. This work was supported by the Office of the Assistant Secretary of Defense for Health Affairs, through the Combat Casualty Care Research Program, endorsed by the Department of Defense, through the Joint Program Committee 6/Combat Casualty Care Research Program—Psychological Health and Traumatic Brain Injury Program under Award No. W81XWH1420151. Opinions, interpretations, conclusions and recommendations are those of the authors and are not necessarily endorsed by the Department of Defense.
Author Contributions
SB, TM, MM contributed to the design and implementation of the investigation, drafting and revision of the manuscript; BK contributed to the design and implementation of the investigation, statistical analyses and interpretation, and drafting and revision of the manuscript; MLP and WZ contributed to the statistical analyses and interpretation, and drafting and revision of the manuscript; CARE Consortium Investigators contributed to the implementation of the investigation and revision of the manuscript.
Conflicts of interest/Competing interests
Steven Broglio, Thomas McAllister, Barry Katz, Michelle LaPradd, Wenxian Zhou, and Michael McCrea received funding from the NCAA and the Department of Defense to complete this investigation and to cover travel costs related to the study.
Ethics approval
This study was completed following approval from respective local Institutional Review Boards and the United States Army Human Research Protection Office. This study was conducted in accordance with the Declaration of Helsinki.
Availability of data
Data used in this investigation are available in the Federal Interagency Traumatic Brain Injury Research (FITBIR) Informatics System (https://fitbir.nih.gov/).
Code availability
The specific analytic coding that was used relies on the data set prepared from the full data set of over 38,000 participants, but the analytic techniques used are standard (e.g. Cox proportional hazards models) and available in all statistical software packages. For those interested, the authors can be contacted for the code and with questions.
Additional information
This article is part of a Collection on The NCAA-DoD Concussion Assessment, Research and Education (CARE) Consortium.
The members of “CARE Consortium Investigators” is present in Acknowledgements section.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Broglio, S.P., McAllister, T., Katz, B.P. et al. The Natural History of Sport-Related Concussion in Collegiate Athletes: Findings from the NCAA-DoD CARE Consortium. Sports Med 52, 403–415 (2022). https://doi.org/10.1007/s40279-021-01541-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-021-01541-7