Abstract
A single bout of resistance training induces residual fatigue, which may impair performance during subsequent endurance training if inadequate recovery is allowed. From a concurrent training standpoint, such carry-over effects of fatigue from a resistance training session may impair the quality of a subsequent endurance training session for several hours to days with inadequate recovery. The proposed mechanisms of this phenomenon include: (1) impaired neural recruitment patterns; (2) reduced movement efficiency due to alteration in kinematics during endurance exercise and increased energy expenditure; (3) increased muscle soreness; and (4) reduced muscle glycogen. If endurance training quality is consistently compromised during the course of a specific concurrent training program, optimal endurance development may be limited. Whilst the link between acute responses of training and subsequent training adaptation has not been fully established, there is some evidence suggesting that cumulative effects of fatigue may contribute to limiting optimal endurance development. Thus, the current review will (1) explore cross-sectional studies that have reported impaired endurance performance following a single, or multiple bouts, of resistance training; (2) identify the potential impact of fatigue on chronic endurance development; (3) describe the implications of fatigue on the quality of endurance training sessions during concurrent training, and (4) explain the mechanisms contributing to resistance training-induced attenuation on endurance performance from neurological, biomechanical and metabolic standpoints. Increasing the awareness of resistance training-induced fatigue may encourage coaches to consider modulating concurrent training variables (e.g., order of training mode, between-mode recovery period, training intensity, etc.) to limit the carry-over effects of fatigue from resistance to endurance training sessions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Several studies have reported that a single bout of resistance training impairs subsequent endurance performance as a result of fatigue. |
Resistance training-induced fatigue may impair the quality of endurance training sessions during concurrent training, thereby limiting optimisation of endurance development. |
Coaches/athletes should be aware of fatigue when undertaking concurrent training to optimise endurance performance development, particularly fatigue induced by resistance training. |
1 Introduction
Incorporating exercises that induce adaptations for resistance and endurance in the one training regime irrespective of whether the training is performed on the same day or on separate days is known as concurrent training [1]. Current literature advises health professionals to prescribe concurrent training in order to improve and sustain health-related fitness [2]. The majority of concurrent training studies to date have shown that resistance training combined with endurance training enhances endurance adaptation to a greater extent than endurance training alone [3]. Common mechanisms that have been proposed to explain this phenomenon include alterations in muscle fibre-type recruitment pattern, greater muscle force generation capacity, increased proportion of type IIA fibres and reduced proportion of type IIB fibres, and a shift toward a fatigue-resistance yet more powerful muscle phenotype, all of which can improve movement economy [4, 5].
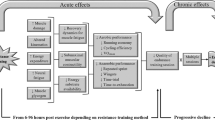
In light of the above, resistance training is highly recommendable for endurance athletes aiming to optimise their performance. However, concurrent training prescription should be undertaken with care given that fatigue induced by resistance training may impair the quality of endurance training sessions, and, possibly, induce sub-optimal endurance development (Fig. 1). In fact, cross-sectional studies have reported attenuated repeated sprint ability [6], running time-to-exhaustion [7], arm cranking time-to-exhaustion [8] and cycling time-trial performance [9] several days following a single bout of resistance training. Whilst this phenomenon would only be observed during resistance training-induced fatigue, if training variables and adequate recovery are not appropriately accounted for, athletes may become subject to non-functional over-reaching or overtraining, which have been reported to impair chronic running [10] and cycling [11] time-trial improvements. Furthermore, whilst overloaded training is used to induce a super-compensation cycle effect [12], studies have shown no greater improvement in running and cycling time-trial performance with several weeks of overloaded training compared to normal-loaded training following tapering [10, 13], suggesting that excessive fatigue in some cases may cause unnecessary physiological stress resulting in no further benefits than normal-loaded training or increased susceptibility to injuries.
To conceptualise the possibilities of concurrent training inducing sub-optimal endurance development, the current review will (1) explore cross-sectional studies that have reported impaired endurance performance following a single, or multiple, bouts, of resistance training, hereafter referred to as resistance training-induced sub-optimisation of endurance performance (RT-SEP); (2) describe the implications of fatigue on the quality of endurance training sessions during concurrent training; (3) identify the potential impact of fatigue on chronic endurance development; and (4) explain the mechanisms contributing to RT-SEP from neurological, biomechanical and metabolic standpoints. Although training variables (i.e. intensity/volume, exercise sequence, recovery period, number of training weeks, etc.) and recovery strategies may affect the type and magnitude of resistance training-induced fatigue, an in-depth exploration of such complex factors in addition to mechanisms in a single review is beyond the scope of this review.
2 Acute Effects of Resistance Exercise on Indices of Endurance Performance and Their Implications
Prior to discussing the mechanisms of RT-SEP, the implications of this phenomenon should be elucidated from a concurrent training standpoint. To date, studies have reported that a single resistance training bout impairs subsequent time-trial and time-to-exhaustion performance of various exercise modes [8, 9, 14,15,16,17,18] for 24–72 h post-training. For example, Doncaster and colleagues [8] examined the acute effects of bench press exercise on arm cranking endurance performance. The results showed that ten sets of six repetitions of bench press exercises at 70% of one repetition maximum (RM) reduced arm cranking time-to-exhaustion for up to 48 h post-exercise. Similarly, Doma and colleagues [14] reported reduced running time-to-exhaustion at 110% of anaerobic threshold for up to 24 h following a bout of lower body resistance training consisting of leg press, leg extension and leg curls at six repetition maximum. Furthermore, several studies have reported impaired running economy (RE) following a single resistance training bout for several days post-training [7, 19, 20]. While RE is not a measure of endurance performance per se, and compromised RE may not likewise compromise the development of endurance performance, there are observations suggesting reduced RE in a laboratory-based condition may be indicative of impaired running time-trial performance in a non-laboratory-based condition. For instance, Hoogkamer et al. [21] showed that shoes with added mass impaired RE and concomitantly slowed 3000-m running time trial performance and demonstrated that laboratory-based RE measures can accurately predict alterations in road-based running performance. Thus, impaired RE may indicate that the quality of actual running sessions could be compromised [22]. For example, attenuation of the physiological cost of aerobic exercise tends to suggest that athletes would experience difficulty covering particular distances or maintaining intensity (e.g. pacing) to meet session goals as a result of residual fatigue from previous training sessions. This implication is also attributable to studies that have shown impaired endurance time-trial performances following a single bout of resistance training [8, 14, 16,17,18]. Studies have also shown that resistance training-induced fatigue increases heart rate and RPE during RE tests [20, 23]. Therefore, if athletes are prescribed to train at an intensity relative to their maximum heart rate or RPE, they may have to reduce their training pace during periods of resistance training-induced fatigue, which may defeat the purpose of the training session.
3 Effects of Fatigue on Chronic Endurance Development
A number of studies have attempted to link acute fatigue responses to training sessions, then to subsequent endurance development. For example, Lamberts et al. [24] retrospectively examined the relationship between changes in heart rate recovery and cycling performance in trained cyclists. Following 4 weeks of high intensity endurance training, the results showed that the level of improvement was greater for those that demonstrated better heart rate recovery, with authors suggesting that training-induced fatigue may have impacted on the level of training adaptation. Furthermore, Halson et al. [13] investigated the effects of cumulative exercise stress and subsequent recovery on performance changes and fatigue indicators in trained cyclists during a 6-week training period. The cyclists completed normal training, intensified training and recovery training for 2 weeks, respectively. The results showed that maximal cycling power output and cycling time-trial performance was worsened following the intensified training period, despite the provision of recovery training. These findings suggest that athletes are unable to perform at optimum levels as a result of cumulative effects of fatigue from intensified training. Whilst speculative, if intensified endurance training alone induces fatigue sufficient to affect endurance development, then combining resistance with endurance training may induce similar to or further levels of cumulative effects of fatigue if proper recovery and/or training variables are not accounted for. This is because the physiological stress caused by a typical resistance training bout (i.e., 40–60 min) can continue for several days post-exercise [15, 19] as opposed to a full recovery observed within 24 h following a typical endurance training bout (i.e. 40–60 min) [14, 25, 26]. However, further studies are warranted to confirm whether endurance development is impacted specifically from over-reaching or overtraining induced by concurrent training.
Whilst full restoration of homeostasis is not required between training sessions [27], general consensus suggests that recovery and psychophysiological ‘readiness’ for each training session may impact on the overall magnitude of training adaptation [28]. In fact, the accumulation of specific transcriptional and translational alterations that occur from one session to the next may dictate the course of training adaptation [29]. Thus, the benefits of resistance training on endurance development could be further optimised by limiting the carry-over effects of fatigue from resistance to endurance training sessions by modulating the concurrent training variables (e.g. order of training mode, between-mode recovery period, training intensity, etc.). For example, Chtara et al. [30] reported greater improvement in 4-km running time-trial performance following 12 weeks of concurrent training for the group that undertook endurance training sessions prior to resistance training compared to the group that carried out each mode of training in the reverse order. Izquierdo-Gabarren and colleagues [31] reported improved 20-min rowing time-trial performance following 8 weeks of concurrent training compared to the control group that solely undertook endurance training. However, when concurrent training groups were compared between those that performed resistance training sets to failure to those without failure, a lesser improvement in rowing performance measures was observed for those that performed resistance training sets to failure. Robineau et al. [32] recently showed that 7 weeks of concurrent training induced greater improvement in running peak oxygen consumption (VO2peak) for rugby players who had 24 h of recovery between resistance and endurance training sessions compared to groups that had 0–6 h of rest. Whilst VO2peak does not necessarily equate to endurance performance measures [33], alterations in aerobic capacity tend to suggest that concurrent training methods appear to influence the level of endurance training adaptation. Collectively, these findings [30,31,32] suggest that training methods that minimise residual fatigue between each mode of training (e.g. performing endurance training prior to resistance training or extending periods of between-mode recovery) may further increase the benefits of resistance training on endurance development but by no means indicate that resistance training impairs endurance development (refer to Table 1). This concept is also attributed to cross-sectional studies that have examined various recovery strategies following a single bout of resistance training with the underlying conjecture that insufficient recovery from a strenuous training session may leave individuals with the inability to train at required levels during subsequent training sessions [34,35,36].
Interestingly, Halson and colleagues [37] reported greater improvement in a 4-min cycling time-trial for cyclists that received cold water immersion (CWI) interventions post 11 days of tapering after 21 days of intensified training compared to cyclists without a recovery intervention. Whilst Halson and colleagues [37] did not systematically examine the acute effects of CWI on cycling performance, a recent cross-sectional study reported a greater increase in cycling power output measures for a group that received CWI following a single bout of lower body resistance training compared to a group that received cold air therapy treatment [36]. Collectively, accelerating the recovery process and minimising fatigue between strenuous training sessions may enhance an athlete’s ‘readiness’ for a subsequent training session and optimise training adaptation [28]. Consequently, the potential mechanisms of this phenomenon should be discussed to increase the awareness for and understanding of the implications of RT-SEP.
4 Consequences of Neuromuscular Fatigue
4.1 Neuromuscular Fatigue in Response to Resistance Training
Several studies have reported that a single bout of resistance training impairs muscle force generation capacity (MFGC) for up to 72 h post-exercise in resistance-trained individuals [7, 38,39,40,41]. For example, Michaut et al. [41] examined the effects of five sets of ten maximal eccentric contractions of the biceps brachii. Twitch activation and maximal contraction was significantly reduced from 2 min to 48 h post-exercise. In addition, Stock et al. [38] reported impaired squatting performance from 24 to 72 h following six sets of squatting exercises at 75% of 1RM amongst resistance trained individuals. With repetitive maximal and submaximal contractions, the neural drive declines and the motor neurons become less responsive to synaptic stimulation [42]. This decrement of neural function in response to exercise can originate from peripheral and/or central sites of the nervous system. From a concurrent training standpoint, the attenuation of muscular function due to acute carry-over effects of neuromuscular fatigue between resistance and endurance training sessions may interfere with the training stimuli to optimise endurance development. In fact, the neuromuscular response following long distance running and cycling events has been studied extensively with results demonstrating reductions in maximal voluntary contraction (MVC) due to central and/or peripheral fatigue [43,44,45]. Romer et al. [46] reported that cycling time to exhaustion was less in hypoxia in comparison to normoxia with a concommitant reduction in potentiated twitch force. These findings indicate that cessation of endurance exercise appears to occur as a result of peripheral fatigue. However, it has also been suggested that termination of exercise occurs due to central fatigue as a protective mechanism from potentially harmful metabolic disturbances to preserve muscular function [47]. In light of this hypothesis, Ansley et al. [48] showed that power output and integrated EMG increased comparatively at the end of a 4-km cycling time trial exercise, indicating that fatigue was centrally controlled. Furthermore, Decorte et al. [49] reported that cycling exercise to failure was associated with central rather than peripheral fatigue. Subsequently, how the mechanisms of neuromuscular fatigue contribute to premature cessation of endurance exercises remains unclear. However, given that strength training has been shown to reduce MVC for several days post-exercise [38, 40, 41], neuromuscular fatigue generated by a strength training session may compromise the quality of a subsequent endurance training session.
Whilst resistance training clearly impairs MFGC, it should be noted that the degree of resistance training-induced changes in neuromuscular performance could be influenced by resistance training variables and training background. For example, Paschalis et al. [50] reported significantly greater decrements in muscle performance for up to 72 h post-high intensity eccentric contractions compared to low intensity eccentric contractions when equated for training volume. However, both conditions impaired MFGC and there were similar effects on muscle damage. Similarly, Newton and colleagues [51] showed that eccentric contractions impaired MFGC to a greater extent in resistance-untrained individuals compared to their trained counterparts. These findings demonstrate that resistance training variables and training background impact on the magnitude of resistance training-induced fatigue following a single bout of resistance exercise. Nonetheless, MFGC is still impaired for several days following high intensity resistance training in resistance-trained individuals [50, 51]. Subsequently, irrespective of training variables and training background, prolonged neuromuscular fatigue generated by a bout resistance training may compromise the quality of a subsequent endurance training session.
4.2 Impact of Neuromuscular Fatigue on Running Gait Patterns
The effects of resistance training-induced fatigue have been reported to alter neural recruitment patterns during the performance of various exercises. Plattner et al. [52] recently showed that muscle activity of the biceps brachii decreased during maximal contractions although increased during sub-maximal contractions in response to resistance training-induced fatigue, indicating alterations in neural firing patterns and motor unit activity. Furthermore, eccentric contractions have been demonstrated to impair proprioceptive mechanisms by increasing the perception of force generation in comparison to the actual force recorded [53, 54]. Given that neuromuscular control is imperative for human locomotion [55], neuromuscular dysfunction in response to resistance training could alter kinematics of endurance performance, impair movement efficiency and decrease the economy of movement [56].
There is a growing body of evidence showing alterations in lower extremity running kinematics 24–48 h following resistive-type exercises, such as downhill running [23, 57,58,59] and lower extremity eccentric contractions [60, 61]. The majority of these studies have shown significant reductions in lower extremity joint range of motion during running [23, 57, 59, 61] with suggestions that neuromuscular fatigue, delayed-onset of muscle soreness (DOMS) and exercise-induced muscle damage (EIMD) may have contributed to such kinematic changes. Despite this consensus, however, only two studies have investigated the acute effects of traditional resistance training on running gait patterns. For example, Doma et al. reported reduction in hip range of motion in the saggital plane during sub-maximal running 6 h [7] and 24 h [14] following three sets of six repetitions of leg press, leg extension and leg curls at 6RM (Fig. 1). Interestingly, these findings are similar to those observed by others that have implemented downhill running [23, 57, 59, 61] and eccentric contractions [60, 61], suggesting that resistive-type exercises appear to alter running kinematics during periods of neuromuscular fatigue, muscle damage and soreness.
Unfortunately, studies of the effects of exercise-induced fatigue on the mechanics of other modes of endurance exercises (e.g. cycling and rowing) are scarce. This may be due to the equipment restricting the movement to a particular plane of motion, hence causing the biomechanics to be predictable. For example, the pedals on a cycling ergometer predominantly execute lower extremity movement in the sagittal plane with limited variation in range of motion for each cadence. On the other hand, there is greater degree of freedom and larger emphasis on balance and proprioception during running, as there is limited equipment controlling gait. Subsequently, it is reasonable to assume that running gait may be more susceptible to movement variation as a result of fatigue in comparison to other modes of endurance exercises.
It has been hypothesised that lower extremity range of motion following strenuous exercises is restricted due to perception of muscle pain [58], altered motor unit activation pattern [62] and reduced reflex sensitivity and/or decreased ability to use the stretch-shortening cycle [59]. This compromise in lower extremity range of motion has been associated with reductions in stride length and an increase in stride frequency during running [62]. Such biomechanical modifications would elevate energy expenditure due to an increase in the number of muscular contractions for a given running velocity [16]. Furthermore, compromise of the stretch-shortening cycle would minimise energy transfer between segments, rendering the locomotion inefficient. Attenuation of movement efficiency will increase energy expenditure during endurance exercise thereby limiting the body’s capability to produce sufficient cardiovascular training stimuli for adaptation [7, 14].
A strong correlation (r = −0.8) has been reported between reduction in stride length and increase in VO2 during a RE test following downhill running [62]. In addition, Bonacci et al. [63] showed that alterations in running kinematics following cycling were related to changes in RE. Subsequently, alterations in running kinematics as a result of exercise-induced fatigue appear to contribute to a decrement in RE. However, Doma and Deakin [7, 14] found no correlation (r = 0.12–0.41) between alterations in RE and running kinematics following a lower body resistance training bout. The authors postulated that the lack of relationship may have been due to differences in the way each participant altered their gait pattern to compensate for muscle fatigue given that running experience varied between each participant. Further, runners tend to choose the optimal stride length and frequency at which they are most economical for a given condition. Subsequently, strong correlations between RE and running gait patterns may not always exist as runners may purposefully change their running gait to be more economical. Nonetheless, given that alterations in both oxygen cost of running and lower extremity kinematics were found, Doma and Deakin [14] suggested that changes in running technique may in part have impaired RE 6 h post a resistance training bout.
4.3 Impact of Neuromuscular Fatigue on Indices of Endurance Performance
A few studies have examined the impact of resistance training on MFGC and subsequent endurance performance. Palmer et al. [64] examined the effects of a whole body resistance training session on MFGC and RE. The results showed that RE was significantly impaired for up to 8 h post-exercise, although torque production was only significantly less immediately following the resistance training session. Doma and colleagues [15] also reported attenuation in RE with a reduction in anaerobic power for up to 48 h following lower body resistance training session in resistance-untrained men. Studies have also reported attenuation of cycling efficiency [65], cycling-specific power output [36], rowing-specific power output [18], running time-to-exhaustion [14, 16], repeated sprints [6] and running time-trial performance [66] with a reduction in MFGC 24–72 h following lower body resistance exercises. These findings suggest that resistance training could compromise the quality of various modes of endurance training sessions at sub-maximal and maximal effort if impaired MFGC is sustained between training sessions during concurrent training.
4.4 Impact of the Repeated Bout Effect of Resistance Training and Training Background on Resistance Training-Induced Fatigue
To this point, the impact of resistance training-induced fatigue following a single bout of resistance training has been discussed with the aim of determining its implications on endurance performance. However, following the initial bout of muscle-damaging exercises, it has been shown that the level of EIMD is reduced following a subsequent identical bout of muscle damaging exercises, known as the repeated bout effect [67]. Subsequently, when considering the repeated bout effect, it could be speculated that repeated resistance training bouts during the course of concurrent training could minimise RT-SEP [9]. Furthermore, by periodising concurrent training, the magnitude of fatigue impacting upon endurance training sessions could be minimised [68]. Indeed, in a study examining the repeated bout effect of lower body resistance training sessions on running performance measures, Doma et al. [19] reported that the magnitude of increase in muscle damage markers (i.e. creatine kinase and DOMS) was attenuated following the second bout of lower body resistance training in resistance-untrained runners. However, no differences were found in RE measures between the first and second lower body resistance training bout, suggesting that the initial bout of resistance training did not provide any protection against the second bout of resistance training for sub-maximal running performance. In a similar cohort, Doma et al. [69] showed that lower extremity resistance training impaired running time-to-exhaustion (i.e. above 110% of anaerobic threshold) for 48 h post-exercise across three lower extremity resistance training bouts, indicating minimal protection from the initial exposure to resistance training. Furthermore, Doma et al. [14] reported impaired running time-to-exhaustion measures 24 h following lower body resistance training in runners despite their having undergone a flush-out period (i.e. performing a number of resistance training sessions to minimise the possibility of a repeated bout effect) prior to the experimental intervention. Similarly, for individuals who completed a flush-out period, Doma and Deakin [15] showed that three lower body resistance training sessions on alternating-days still impaired running time-to-exhaustion for 48 h following each resistance training session. Accordingly, whilst the levels of muscle-damage markers are attenuated following the second bout of muscle-damaging exercises, this protection does not always appear to occur for running performance measures. This phenomenon has been observed by several studies [15, 23, 59], where the trend over time in muscle damage markers (i.e. CK, DOMS and MVC) following muscle-damaging exercises was different to changes in running performance measures. This discrepancy appears to exist as the cause of impairment in running performance may not directly be associated with changes in the indirect markers of muscle damage given the complex neurophysiological factors required for execution of running. Subsequently, resistance training-induced fatigue may still be present irrespective of the number of times runners are exposed to resistance training.
With respect to training background, studies have reported that the magnitude of muscle damage and attenuation in muscle function is greater for resistance-untrained compared to resistance-trained individuals [70]. However, despite the discrepancy in the magnitude of muscle damage markers amongst different resistance training backgrounds, several studies have reported performance decrements in 1RM bench press performance [71], resistance training performance [39, 72] and elbow-flexor torque [73] 24–48 h following resistance training in resistance-trained individuals. Whilst these findings were not based on endurance performance measures, it appears that resistance training-induced fatigue can impair neuromuscular performance measures even in resistance-trained individuals. Furthermore, attenuated neuromuscular properties as a result of resistance training have also been associated with impaired running, cycling and rowing time-trial performance measures [6, 18, 36].
Whilst a number of studies have systematically compared the acute effects of muscle damaging exercises between resistance-trained and resistance-untrained individuals, such investigations between endurance-trained and endurance-untrained individuals are scarce. One study by Snieckus and colleagues [74] did compare the acute effects of eccentric contractions in long distance runners, cyclists and healthy untrained men and did find that the magnitude of muscle damage markers were less in endurance trained than untrained men. However, they reported that the endurance-trained men had ceased their resistance training program for only one month prior to study commencement, indicating that the lesser susceptibility to muscle damage may not have been due to differences in endurance training background, but as a result of their previous resistance training exposure given that protection against muscle damage can be preserved for as long as 6 months Nosaka et al. [75]. Further studies are warranted to systematically compare the susceptibility to muscle damage amongst different endurance training backgrounds with similar previous exposure to resistance training. Nonetheless, previous studies have reported increased muscle damage markers and impaired neuromuscular performance and running performance measures for at least 48 h post-muscle-damaging exercises in both endurance-trained [7, 15, 20] and endurance-untrained individuals [23, 59]. Accordingly, given that impaired physical performance measures are present 24–48 h post-muscle-damaging exercises for both resistance-trained and endurance-trained individuals, coaches should therefore be cautious of resistance training-induced fatigue for individuals of all training backgrounds.
5 Effects of Exercise-induced Muscle Damage and Delayed-Onset of Muscle Soreness on Indices of Endurance Performance
A common acute response following typical resistance training sessions is EIMD, particularly from the repetitive eccentric components of the exercises [67, 76]. One major consequence of EIMD is DOMS that occurs immediately post [77] and peaks over the next 72 h [57, 62]. The production of prostaglandin E2 in response to muscle damage stimulates type III and IV pain afferents whereas neutrophils permeate through the vascular wall to the damaged site due to leukotriene synthesis. These neutrophils cause further muscle damage via production of free radicals [77] and attract an influx of protein-rich fluid into the muscle (i.e. oedema swelling). Consequently, repetitive eccentric contractions cause inflammatory responses [78], increase intramuscular osmotic pressure and generate pain by the activation of group III and IV afferents [79]. The symptoms of EIMD have also been shown to cause marked reduction in MVC [52, 80, 81] and increased muscle stiffness [82] and swelling [83]. Given that indices of endurance performance are highly dependent on the mechanical properties of the musculature [84], symptoms associated with EIMD may need to be considered within the context of the RT-SEP phenomenon.
5.1 Consequences of Exercise-Induced Muscle Damage and Delayed-Onset of Muscle Soreness on Endurance Performance
Attenuation in endurance performance measures has been reported as a result of EIMD [23, 59, 62]. For example, Braun et al. [62] examined the effects of downhill running on RE 48 h post, and demonstrated an increase in VO2 and muscle soreness. The authors speculated that the attenuation of RE may have been attributed to muscle damage as indicated by DOMS. Similarly, Chen et al. [23, 59] examined RE 1–5 days following downhill running. The results showed that VO2 was significantly greater in conjunction with an increase in CK and a reduction in MVC over the course of the 5-day period. Given the increase in myofibre proteins and reduction in MVC, the authors postulated that the increased physiological cost of running following downhill running was associated with EIMD.
In contrast, Paschalis et al. [85] reported significant changes in muscle damage indicators (i.e. CK, DOMS and range-of-motion and MVC) 24–96 h following 120 maximal eccentric contractions although RE parameters were not affected. Other studies have also shown no alterations in RE 6–48 h post-resistance training despite significant changes in muscle damage indicators (i.e. CK, MVC and DOMS) [16, 66]. However, according to studies by Marcora and colleagues [66] and Doma and Deakin [16], running time-trial and time-to-exhaustion performances were impaired as a result of a single bout of resistance training. Furthermore, Byrne and Eston [86] reported a significant reduction in 30-second Wingate performance as a result of EIMD. Collectively, attenuation of endurance performance due to EIMD appears to be more susceptible at higher intensities. In fact, it has been suggested that fast-twitch muscle fibres are more prone to EIMD than slow twitch muscle fibres [6]. Given that fast-twitch muscle fibres are predominantly used above anaerobic threshold (AT), EIMD may have a greater impact on subsequent endurance performance at higher intensities [59].
Whilst the impact that EIMD has on endurance performance is dependent on the intensity of the endurance exercise following a single bout of resistance training, little is known of the effects of EIMD on endurance performance at various intensities following multiple bouts of resistance training. A more recent study by Doma and Deakin [15] examined the acute effects of concurrent training on DOMS and running performance measures across 6 days. Specifically, following a flush out period of three resistance training sessions, one group undertook resistance training on alternating days combined with running training on consecutive days (CCT) versus another group with resistance training on alternating days only (RT). The results showed that DOMS was significantly elevated during the 6-day period for both groups. Furthermore, RPE was significantly increased when running at 90–110% of AT with reduced running time-to-exhaustion during the running sessions although RPE was unaffected at 70% of running AT for the CCT group. When compared between groups, DOMS was significantly greater for the CCT group compared to the RT group. These findings demonstrate that DOMS is consistently elevated across multiple resistance training sessions, and that a combined resistance and endurance training micro-cycle induces greater DOMS. In addition, the concomitant increase in RPE and DOMS with reduced running time-to-exhaustion suggests that DOMS may have caused participants to perceive running to be harder at higher intensities thereby impairing running performance. Studies have also shown impaired running velocity [87], cycling time-trial performance [35] and rowing work rate [88] with increased RPE. Increased perceived exertion may be caused by a number of sensory cues [89] that may be governed by muscular pain, feelings of breathlessness during endurance exercise and increased motor unit recruitment necessary to produce equivalent sub-maximal force production as baseline prior to muscle-damaging exercises [90,91,92].
Of particular interest in the study by Doma and colleagues [15] was elevated DOMS across multiple resistance training sessions despite participants completing a flush-out period. These findings are consistent with those of Hassan et al. [93] who reported significantly elevated DOMS for 4 weeks across four bouts of eccentric knee extensor exercises. Collectively, whilst DOMS is attenuated following the initial bout of resistance training in resistance-untrained individuals, given that this measure is still present in individuals with previous exposure to resistance training, DOMS should be monitored following resistance training irrespective of training background or level of previous resistance training exposure.
The morphological disruption causing EIMD may also contribute to attenuation of endurance performance. A number of studies using magnetic resonance imaging have shown an increase in inorganic-phosphate:phosphocreatine (PCr) ratio as a result of EIMD following resisted eccentric contractions [94,95,96]. This metabolic alteration may be attributed to disruption of the sarcolemma or an increase in metabolic rate [96]. Furthermore, Davies et al. [97] showed an increase in indirect markers of muscle damage (i.e. CK, DOMS and MVC), inorganic phosphate concentration and inorganic-phosphate:PCr ratio with a concomitant reduction in time-to-exhaustion of knee extensors 24 h following knee extensor eccentric contractions. Walsh et al. [98] suggested that metabolic alterations in response to EIMD may result in reduced oxygen diffusion due to restricted blood flow, sub-optimal mitochondrial respiration and adenosine diphosphate desensitization of mitochondrial respiration. Furthermore, EIMD has been shown to inhibit insulin sensitivity which may resist the uptake of glucose into damaged muscles [99, 100]. Studies have also shown a compromise in glycogen synthesis following eccentric contractions that caused EIMD [101,102,103].
6 Effects of Exercise-Induced Muscle Glycogen Depletion on Endurance Performance
The literature suggests that the pathways that impair the neuromuscular properties are strongly dependent on the level of endogenous glycogen [104] and that inadequate energy supply may lead to failure of transformation from the neuromuscular event to mechanical force production [105]. Muscle glycogen has been shown to be the dominant fuel source at workloads above 65–70% of maximal oxygen consumption (VO2max) [106, 107] during endurance exercise. The association between muscle glycogen content and endurance performance has been most apparent during cycling and running exercises performed at intensities from 65–85% of VO2max [108,109,110]. Impaired performance in prolonged and high intensity endurance exercise has been attributed to energy substrate depletion, which includes muscle and liver glycogen [111, 112], blood glucose [113] and PCr [114, 115]. Accordingly, resistance training-induced muscle glycogen depletion may cause RT-SEP with inadequate recovery and nutrition.
6.1 Muscle Glycogen Content Following Resistance Training
There has been limited research examining glycogenolytic effects following resistance training. A study conducted by MacDougall et al. [116] showed that muscle glycogen depleted by 25% in the biceps brachii following three sets of 10 repetitions of elbow flexion/extension exercises. A more recent study by MacDougall and colleagues [117] reported similar findings where muscle glycogen content of the biceps brachii was reduced by approximately 20% following elbow flexion/extension exercises at 80% of 1RM to failure. In addition, Tesch et al. [118] reported that muscle glycogen content of the vastus lateralis decreased by 26% following repeated sets of resistive exercises of the lower extremity.
A study conducted by Robergs et al. [119] examined high and low intensity (i.e. 70 and 35% of 1RM, respectively) resistance training on muscle glycogen content by performing 6 sets of leg extension exercises whilst matching work between the two intensities. The results showed that muscle glycogen content reduced by 38 and 39% following low and high intensity resistance training protocols, respectively. These results show that muscle glycogen content is substantially reduced following resistance training regardless of resistance training intensity provided that work is matched between high and low training intensities. Pascoe et al. [120] reported that muscle glycogen content remained significantly depleted (by approximately 25%) 6 h following leg extension exercises at 70% of 1RM to failure. These findings suggest that muscle glycogen depletion can be sustained for several hours following resistance training, although it should be acknowledged that the degree to which glycogen depletion is sustained or not following RT may depend on post-exercise nutritional intake and activity levels following the session [121, 122]. Overall, the glycogenolytic effect elicited by resistance training could impair endurance performance within the hours following resistance training and which may impact on optimising endurance development during concurrent training.
6.2 Impact of Muscle Glycogen Depletion on Endurance Performance
A number of studies have examined the impact of muscle glycogen depletion on endurance performance [123,124,125]. For example, Suriano et al. [125] examined the effects of cycling-induced muscle glycogen depletion on RE. Following 60 min of cycling, the results showed a significant reduction in muscle glycogen content with a concomitant increase in oxygen consumption (VO2) during the RE test. In addition, a significant relationship (r = 0.57) was found between changes in VO2 and the magnitude of total glycogen depletion. Furthermore, the reduction in muscle glycogen content that claimed to have impaired RE was approximately 20%, which is less than previous reports that have examined muscle glycogen content immediately post to 6 h following resistance training (i.e. 25–39% loss of muscle glycogen content) [119, 126]. This relationship between alterations in muscle glycogen content and VO2 kinetics reported by Suriano et al. [125] demonstrated that muscle glycogen depletion as a result of strenuous exercises (e.g. resistance training) may contribute to attenuation of subsequent endurance performance. However, there is still limited evidence about the relationship between alterations in muscle glycogen content and endurance performance following resistance training which warrants investigation.
Despite the possibility of RT-SEP from reduction in glycogen content, it should also be noted that energy restriction and glycogen depletion enhances AMPK activity both under basal conditions and following acute exercise [127, 128], suggesting that exercising in glycogen-depleted conditions may produce a positive metabolic environment for endurance adaptation. However, as Knuiman and colleagues [122] recently mentioned in their review, training sessions repetitively commencing in a low glycogen state may increase the risk of an overtraining syndrome [129], and as a result, impair training capacity [130]. Accordingly, the role of glycogen status in chronic training adaptation remains a matter of debate warranting further research, particularly from a concurrent training perspective.
7 Conclusion
In summary, resistance training impairs muscular contractility via numerous physiological mechanisms which may have profound detrimental effects on the quality of subsequent endurance training sessions. Overall, RT-SEP may be a complex phenomenon that may occur from multiple neuropsychophysiological factors, including neuromuscular fatigue, alterations in running gait patterns, DOMS, and muscle glycogen depletion, all of which can be induced following a single bout of resistance training and acutely impair endurance performance. However, it should be acknowledged that the acute RT-induced attenuation on subsequent endurance performance is a short-term effect following a single RT session. In addition, the quality of endurance training may only be compromised if any RT-induced residual fatigue is not attenuated by aspects of program design (e.g., exercise order, length of between-mode recovery, etc.), particularly when endurance training is performed following resistance training. It is also critical to understand that the purpose of this review was to discuss the possibility that resistance training-induced fatigue may limit optimisation of endurance development. This is by no means to suggest that concurrent training impairs endurance development, but rather, even greater benefits of resistance training may be experienced by endurance athletes if potential fatigue between each mode of training session is accounted for, particularly with respect to resistance training-induced fatigue on subsequent endurance training sessions during a concurrent training program. Subsequently, resistance training should be encouraged for endurance athletes given the plethora of evidence indicating the benefits of resistance training for endurance development. Furthermore, whilst the level of interference that fatigue may have on the quality of endurance training session may be dictated by the nature of the training program, discussing training variables was beyond the scope of this review. However, we hope that the review encourages practitioners to become more aware of resistance training-induced fatigue for endurance athletes undertaking concurrent training.
Change history
27 July 2017
An erratum to this article has been published.
References
Hickson RC. The interference effects of training for strength and endurance simultaneously. Eur J Appl Physiol Occup Physiol. 1980;45(2–3):255–63.
Garber CE, Blissmer B, Deschenes MR, et al. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc. 2011;43(7):1334–59. doi:10.1249/MSS.0b013e318213fefb.
Ronnestad BR, Mujika I. Optimizing strength training for running and cycling endurance performance: a review. Scand J Med Sci Sports. 2014;24(4):603–12. doi:10.1111/sms.12104.
Ronnestad BR, Hansen EA, Raastad T. Strength training improves 5-min all-out performance following 185 min of cycling. Scand J Med Sci Sports. 2011;21(2):250–9. doi:10.1111/j.1600-0838.2009.01035.x.
Aagaard P, Andersen JL, Bennekou M, et al. Effects of resistance training on endurance capacity and muscle fiber composition in young top-level cyclists. Scand J Med Sci Sports. 2011;21(6):e298–307. doi:10.1111/j.1600-0838.2010.01283.x.
Twist C, Eston R. The effects of exercise-induced muscle damage on maximal intensity intermittent exercise performance. Eur J Appl Physiol Occup Physiol. 2005;94(5–6):652–8. doi:10.1007/s00421-005-1357-9.
Doma K, Deakin GB. The effects of combined strength and endurance training on running performance the following day. Int J Sport Health Sci. 2013;11:1–9.
Doncaster GG, Twist C. Exercise-induced muscle damage from bench press exercise impairs arm cranking endurance performance. Eur J Appl Physiol. 2012;112(12):4135–42. doi:10.1007/s00421-012-2404-y.
Burt DG, Twist C. The effects of exercise-induced muscle damage on cycling time-trial performance. J Strength Cond Res. 2011;25(8):2185–92. doi:10.1519/JSC.0b013e3181e86148.
Coutts AJ, Wallace LK, Slattery KM. Monitoring changes in performance, physiology, biochemistry, and psychology during over-reaching and recovery in triathletes. Int J Sports Med. 2007;28(2):125–34. doi:10.1055/s-2006-924146.
Aubry A, Hausswirth C, Louis J, et al. Functional over-reaching: the key to peak performance during the taper? Med Sci Sports Exerc. 2014;46(9):1769–77. doi:10.1249/MSS.0000000000000301.
Bompa TO. Theory and methodology of training. The key to athletic performance. 3rd ed. Dubuque: Kendall/Hunt Publishing Company; 2005.
Halson SL, Bridge MW, Meeusen R, et al. Time course of performance changes and fatigue markers during intensified training in trained cyclists. J Appl Physiol (1985). 2002;93(3):947–56. doi:10.1152/japplphysiol.01164.2001.
Doma K, Deakin GB. The effects of strength training and endurance training order on running economy and performance. Appl Physiol Nutr Metab. 2013;38(6):651–6. doi:10.1139/apnm-2012-0362.
Doma K, Deakin G. The acute effect of concurrent training on running performance over 6 days. Res Q Exerc Sport. 2015;86(4):387–96. doi:10.1080/02701367.2015.1053104.
Doma K, Deakin GB. The acute effects intensity and volume of strength training on running performance. Eur J Sport Sci. 2014;14(2):107–15. doi:10.1080/17461391.2012.726653.
Burt D, Lamb K, Nicholas C, et al. Lower-volume muscle-damaging exercise protects against high-volume muscle-damaging exercise and the detrimental effects on endurance performance. Eur J Appl Physiol. 2015;115(7):1523–32. doi:10.1007/s00421-015-3131-y.
Gee TI, French DN, Howatson G, et al. Does a bout of strength training affect 2,000 m rowing ergometer performance and rowing-specific maximal power 24 h later? Eur J Appl Physiol. 2011;111(11):2653–62. doi:10.1007/s00421-011-1878-3.
Doma K, Schumann M, Sinclair WH, et al. The repeated bout effect of typical lower body strength training sessions on sub-maximal running performance and hormonal response. Eur J Appl Physiol. 2015;115(8):1789–99. doi:10.1007/s00421-015-3159-z.
Burt D, Lamb K, Nicholas C, et al. Effects of repeated bouts of squatting exercise on submaximal endurance running performance. Eur J Appl Physiol. 2013;113(2):285–93. doi:10.1007/s00421-012-2437-2.
Hoogkamer W, Kipp S, Spiering BA, et al. Altered running economy directly translates to altered distance-running performance. Med Sci Sports Exerc. 2016;48(11):2175–80. doi:10.1249/MSS.0000000000001012.
Doma K, Deakin BD, Leicht AS, et al. The reliability of running economy among trained distance runners and field-based players. J Exerc Sci Fit. 2012;10(2):90–6.
Chen TC, Nosaka K, Lin MJ, et al. Changes in running economy at different intensities following downhill running. J Sports Sci. 2009;27(11):1137–44. doi:10.1080/02640410903062027.
Lamberts RP, Swart J, Capostagno B, et al. Heart rate recovery as a guide to monitor fatigue and predict changes in performance parameters. Scand J Med Sci Sports. 2010;20(3):449–57. doi:10.1111/j.1600-0838.2009.00977.x.
Bentley DJ, Zhou S, Davie AJ. The effect of endurance exercise on muscle force generating capacity of the lower limbs. J Sci Med Sport. 1998;1(3):179–88.
Bentley DJ, Smith PA, Davie AJ, et al. Muscle activation of the knee extensors following high intensity endurance exercise in cyclists. Eur J Appl Physiol. 2000;81(4):297–302. doi:10.1007/s004210050046.
Stanley J, Peake JM, Buchheit M. Cardiac parasympathetic reactivation following exercise: implications for training prescription. Sports Med. 2013;43(12):1259–77. doi:10.1007/s40279-013-0083-4.
Mann TN, Lamberts RP, Lambert MI. High responders and low responders: factors associated with individual variation in response to standardized training. Sports Med. 2014;44(8):1113–24. doi:10.1007/s40279-014-0197-3.
Fluck M. Functional, structural and molecular plasticity of mammalian skeletal muscle in response to exercise stimuli. J Exp Biol. 2006;209(Pt 12):2239–48. doi:10.1242/jeb.02149.
Chtara M, Chamari K, Chaouachi M, et al. Effects of intra-session concurrent endurance and strength training sequence on aerobic performance and capacity. Br J Sports Med. 2005;39(8):555–60. doi:10.1136/bjsm.2004.015248.
Izquierdo-Gabarren M, De Txabarri Gonzalez, Exposito R, Garcia-Pallares J, et al. Concurrent endurance and strength training not to failure optimizes performance gains. Med Sci Sports Exerc. 2010;42(6):1191–9. doi:10.1249/MSS.0b013e3181c67eec.
Robineau J, Babault N, Piscione J, et al. Specific training effects of concurrent aerobic and strength exercises depend on recovery duration. J Strength Cond Res. 2016;30(3):672–83. doi:10.1519/JSC.0000000000000798.
Joyner MJ, Coyle EF. Endurance exercise performance: the physiology of champions. J Physiol. 2008;586(1):35–44. doi:10.1113/jphysiol.2007.143834.
Robson-Ansley PJ, Gleeson M, Ansley L. Fatigue management in the preparation of Olympic athletes. J Sports Sci. 2009;27(13):1409–20. doi:10.1080/02640410802702186.
Rowsell GJ, Reaburn P, Toone R, et al. Effect of run training and cold-water immersion on subsequent cycle training quality in high-performance triathletes. J Strength Cond Res. 2014;28(6):1664–72. doi:10.1519/JSC.0000000000000455.
Hayter KJ, Doma K, Schumann M, et al. The comparison of cold-water immersion and cold air therapy on maximal cycling performance and recovery markers following strength exercises. PeerJ. 2016;4:e1841. doi:10.7717/peerj.1841.
Halson SL, Bartram J, West N, et al. Does hydrotherapy help or hinder adaptation to training in competitive cyclists? Med Sci Sports Exerc. 2014;46(8):1631–9.
Stock MS, Young JC, Golding LA, et al. The effects of adding leucine to pre and postexercise carbohydrate beverages on acute muscle recovery from resistance training. J Strength Cond Res. 2010;24(8):2211–9. doi:10.1519/JSC.0b013e3181dc3a10.
Bird SP, Mabon T, Pryde M, et al. Triphasic multinutrient supplementation during acute resistance exercise improves session volume load and reduces muscle damage in strengthtrained athletes. Nutr Res. 2013;33(5):376–87. doi:10.1016/j.nutres.2013.03.002.
Hakkinen K, Pakarinen A, Alen M, et al. Daily hormonal and neuromuscular responses to intensive strength training in 1 week. Int J Sports Med. 1988;9(6):422–8. doi:10.1055/s-2007-1025044.
Michaut A, Pousson M, Ballay Y, et al. Effects of an eccentric exercise session short-term recovery of muscle contractility. J Soc Biol. 2000;194(3–4):171–6.
Taylor JL, Todd G, Gandevia SC. Evidence for a supraspinal contribution to human muscle fatigue. Clin Exp Pharmacol Physiol. 2006;33(4):400–5.
Amann M. Central and peripheral fatigue: interaction during cycling exercise in humans. Med Sci Sports Exerc. 2011;43(11):2039–45. doi:10.1249/MSS.0b013e31821f59ab.
Millet GY, Lepers R. Alterations of neuromuscular function after prolonged running, cycling and skiing exercises. Sports Med. 2004;34(2):105–16 (pii:3424).
Petersen K, Hansen CB, Aagaard P, et al. Muscle mechanical characteristics in fatigue and recovery from a marathon race in highly trained runners. Eur J Appl Physiol. 2007;101(3):385–96. doi:10.1007/s00421-007-0504-x.
Romer LM, Haverkamp HC, Amann M, et al. Effect of acute severe hypoxia on peripheral fatigue and endurance capacity in healthy humans. Am J Physiol Regul Integr Comp Physiol. 2007;292(1):R598–606. doi:10.1152/ajpregu.00269.2006.
Girard O, Millet GP. Neuromuscular fatigue in racquet sports. Neurol Clin. 2008;26(1):181–94. doi:10.1016/j.ncl.2007.11.011.
Ansley L, Schabort E, St Clair Gibson A, et al. Regulation of pacing strategies during successive 4-km time trials. Med Sci Sports Exerc. 2004;36(10):1819–25.
Decorte N, Lafaix PA, Millet GY, et al. Central and peripheral fatigue kinetics during exhaustive constant-load cycling. Scand J Med Sci Sports. 2012;22(3):381–91. doi:10.1111/j.1600-0838.2010.01167.
Paschalis V, Koutedakis Y, Jamurtas AZ, et al. Equal volumes of high and low intensity of eccentric exercise in relation to muscle damage and performance. J Strength Cond Res. 2005;19(1):184–8. doi:10.1519/R-14763.1.
Newton MJ, Morgan GT, Sacco P, et al. Comparison of responses to strenuous eccentric exercise of the elbow flexors between resistance-trained and untrained men. J Strength Cond Res. 2008;22(2):597–607. doi:10.1519/JSC.0b013e3181660003.
Plattner K, Baumeister J, Lamberts RP, et al. Dissociation in changes in EMG activation during maximal isometric and submaximal low force dynamic contractions after exerciseinduced muscle damage. J Electromyogr Kinesiol. 2011;21(3):542–50. doi:10.1016/j.jelekin.2011.01.008.
Brockett C, Warren N, Gregory JE, et al. A comparison of the effects of concentric versus eccentric exercise on force and position sense at the human elbow joint. Brain Res. 1997;771(2):251–8.
Saxton JM, Clarkson PM, James R, et al. Neuromuscular dysfunction following eccentric exercise. Med Sci Sports Exerc. 1995;27(8):1185–93.
Doma K, Deakin GB, Ness KF. Kinematic and electromyographic comparisons between chin-ups and lat-pull down exercises. Sports Biomech. 2013;12(3):302–13. doi:10.1080/14763141.2012.760204.
Williams KR, Cavanagh PR, Ziff JL. Biomechanical studies of elite female distance runners. Int J Sports Med. 1987;8(Suppl 2):107–18.
Dutto DJ, Braun WA. DOMS-associated changes in ankle and knee joint dynamics during running. Med Sci Sports Exerc. 2004;36(4):560–6 (pii:00005768-200404000-00002).
Hamill J, Freedson PS, Clarkson PM. Muscle soreness during running: biomechanical and physiological considerations. Int J Sport Biomech. 1991;7(2):125–37.
Chen TC, Nosaka K, Tu JH. Changes in running economy following downhill running. J Sports Sci. 2007;25(1):55–63. doi:10.1080/02640410600718228.
Kellis E, Liassou C. The effect of selective muscle fatigue on sagittal lower limb kinematics and muscle activity during level running. J Orthop Sports Phys Ther. 2009;39(3):210–20. doi:10.2519/jospt.2009.2859.
Paschalis V, Giakas G, Baltzopoulos V, et al. The effects of muscle damage following eccentric exercise on gait biomechanics. Gait Posture. 2007;25(2):236–42. doi:10.1016/j.gaitpost.2006.04.002.
Braun WA, Dutto DJ. The effects of a single bout of downhill running and ensuing delayed onset of muscle soreness on running economy performed 48 h later. Eur J Appl Physiol. 2003;90(1–2):29–34. doi:10.1007/s00421-003-0857-8.
Bonacci J, Green D, Saunders PU, et al. Change in running kinematics after cycling are related to alterations in running economy in triathletes. J Sci Med Sport. 2010;13(4):460–4. doi:10.1016/j.jsams.2010.02.002.
Palmer CD, Sleivert GG. Running economy is impaired following a single bout of resistance exercise. J Sci Med Sport. 2001;4(4):447–59.
Deakin BD. Concurrent training in endurance athletes: the acute effects on muscle recovery capacity, physiological, hormonal and gene expression responses post-exercise. Lismore: Southern Cross University; 2004.
Marcora SM, Bosio A. Effect of exercise-induced muscle damage on endurance running performance in humans. Scand J Med Sci Sports. 2007;17(6):662–71. doi:10.1111/j.1600-0838.2006.00627.x.
Brentano MA, Martins Kruel LF. A review on strength exercise-induced muscle damage: applications, adaptation mechanisms and limitations. J Sports Med Phys Fit. 2011;51(1):1–10 (pii:R40113061).
Austin KG, Deuster P. Monitoring training for human performance optimization. J Spec Oper Med. 2015;15(2):102–8.
Doma K, Schumann M, Leicht AS, et al. The repeated bout effect of lower body resistance exercises on running performance across three bouts. Appl Physiol Nutr Metab. 2017. doi:10.1139/apnm-2017-0214 (in press).
Skurvydas A, Brazaitis M, Venckunas T, et al. The effect of sports specialization on musculus quadriceps function after exercise-induced muscle damage. Appl Physiol Nutr Metab. 2011;36(6):873–80. doi:10.1139/h11-112.
Meneghel AJ, Verlengia R, Crisp AH, et al. Muscle damage of resistance-trained men after two bouts of eccentric bench press exercise. J Strength Cond Res. 2014;28(10):2961–6. doi:10.1519/JSC.0000000000000494.
Chaves CP, Simao R, Miranda H, et al. Influence of exercise order on muscle damage during moderate-intensity resistance exercise and recovery. Res Sports Med. 2013;21(2):176–86. doi:10.1080/15438627.2012.738439.
Soares S, Ferreira-Junior JB, et al. Dissociated time course of muscle damage recovery between single- and multi-joint exercises in highly resistance-trained men. J Strength Cond Res. 2015;29(9):2594–9. doi:10.1519/JSC.0000000000000899.
Snieckus A, Kamandulis S, Venckūnas T, et al. Concentrically trained cyclists are not more susceptible to eccentric exercise-induced muscle damage than are stretch-shortening exercise-trained runners. Eur J Appl Physiol. 2013;113(3):621–8. doi:10.1007/s00421-012-2470-1.
Nosaka K, Sakamoto K, Newton M, et al. How long does the protective effect on eccentric exercise-induced muscle damage last? Med Sci Sport Exerc. 2001;33(9):1490–5.
Howatson G, van Someren KA. The prevention and treatment of exercise-induced muscle damage. Sports Med. 2008;38(6):483–503 (pii:3864).
Nguyen D, Brown LE, Coburn JW, et al. Effect of delayed-onset muscle soreness on elbow flexion strength and rate of velocity development. J Strength Cond Res. 2009;23(4):1282–6. doi:10.1519/JSC.0b013e3181970017.
Evans WJ, Meredith CN, Cannon JG, et al. Metabolic changes following eccentric exercise in trained and untrained men. J Appl Physiol (1985). 1986;61(5):1864–8.
Friden J, Sfakianos PN, Hargens AR. Muscle soreness and intramuscular fluid pressure: comparison between eccentric and concentric load. J Appl Physiol (1985). 1986;61(6):2175–9.
Molina R, Denadai BS. Dissociated time course recovery between rate of force development and peak torque after eccentric exercise. Clin Physiol Funct Imaging. 2012;32(3):179–84. doi:10.1111/j.1475-097X.2011.01074.x.
Skurvydas A, Brazaitis M, Kamandulis S, et al. Peripheral and central fatigue after muscle-damaging exercise is muscle length dependent and inversely related. J Electromyogr Kinesiol. 2010;20(4):655–60. doi:10.1016/j.jelekin.2010.02.009.
Thompson HS, Maynard EB, Morales ER, et al. Exercise-induced HSP27, HSP70 and MAPK responses in human skeletal muscle. Acta Physiol Scand. 2003;178(1):61–72. doi:10.1046/j.1365-201X.2003.01112.x.
Nosaka K, Clarkson PM. Changes in indicators of inflammation after eccentric exercise of the elbow flexors. Med Sci Sports Exerc. 1996;28(8):953–61.
Bentley DJ, Millet GP, Vleck VE, et al. Specific aspects of contemporary triathlon: implications for physiological analysis and performance. Sports Med. 2002;32(6):345–59 (pii:320601).
Paschalis V, Koutedakis Y, Baltzopoulos V, et al. The effects of muscle damage on running economy in healthy males. Int J Sports Med. 2005;26(10):827–31. doi:10.1055/s-2005-837461.
Byrne C, Eston R. Maximal-intensity isometric and dynamic exercise performance after eccentric muscle actions. J Sports Sci. 2002;20(12):951–9. doi:10.1080/026404102321011706.
Smith MR, Marcora SM, Coutts AJ. Mental fatigue impairs intermittent running performance. Med Sci Sports Exerc. 2015;47(8):1682–90. doi:10.1249/MSS.0000000000000592.
DellaValle DM, Haas JD. Quantification of training load and intensity in female collegiate rowers: validation of a daily assessment tool. J Strength Cond Res. 2013;27(2):540–8. doi:10.1519/JSC.0b013e3182577053.
Hampson DB, St Clair Gibson A, Lambert MI, et al. The influence of sensory cues on the perception of exertion during exercise and central regulation of exercise performance. Sports Med. 2001;31(13):935–52.
Elmer SJ, McDaniel J, Martin JC. Alterations in neuromuscular function and perceptual responses following acute eccentric cycling exercise. Eur J Appl Physiol. 2010;110(6):1225–33. doi:10.1007/s00421-010-1619-z.
Jameson C, Ring C. Contributions of local and central sensations to the perception of exertion during cycling: effects of work rate and cadence. J Sports Sci. 2000;18(4):291–8. doi:10.1080/026404100365027.
Scott KE, Rozenek R, Russo AC, et al. Effects of delayed onset muscle soreness on selected physiological responses to submaximal running. J Strength Cond Res. 2003;17(4):652–8 (pii:R-12582).
Hassan ES. Muscle damage and adaptation after the second bout of eccentric exercise of the knee extensors. J Sports Med Phys Fit. 2014;54(5):644–50.
Lund H, Vestergaard-Poulsen P, Kanstrup IL, et al. Isokinetic eccentric exercise as a model to induce and reproduce pathophysiological alterations related to delayed onset muscle soreness. Scand J Med Sci Sports. 1998;8(4):208–15.
Lund H, Vestergaard-Poulsen P, Kanstrup IL, et al. The effect of passive stretching on delayed onset muscle soreness, and other detrimental effects following eccentric exercise. Scand J Med Sci Sports. 1998;8(4):216–21.
McCully K, Posner J. Measuring exercise-induced adaptations and injury with magnetic resonance spectroscopy. Int J Sports Med. 1992;13(Suppl 1):S147–9. doi:10.1055/s-2007-1024621.
Davies RC, Eston RG, Fulford J, et al. Muscle damage alters the metabolic response to dynamic exercise in humans: a 31P-MRS study. J Appl Physiol. 2011;111(3):782–90. doi:10.1152/japplphysiol.01021.2010.
Walsh B, Tonkonogi M, Malm C, et al. Effect of eccentric exercise on muscle oxidative metabolism in humans. Med Sci Sports Exerc. 2001;33(3):436–41.
Del Aguila LF, Krishnan RK, Ulbrecht JS, et al. Muscle damage impairs insulin stimulation of IRS-1, PI 3-kinase, and Akt-kinase in human skeletal muscle. Am J Physiol Endocrinol Metab. 2000;279(1):E206–12.
Tuominen JA, Ebeling P, Bourey R, et al. Postmarathon paradox: insulin resistance in the face of glycogen depletion. Am J Physiol. 1996;270(2 Pt 1):E336–43.
Asp S, Kristiansen S, Richter EA. Eccentric muscle damage transiently decreases rat skeletal muscle GLUT-4 protein. J Appl Physiol. 1995;79(4):1338–45.
Kristiansen S, Asp S, Richter EA. Decreased muscle GLUT-4 and contraction-induced glucose transport after eccentric contractions. Am J Physiol. 1996;271(2 Pt 2):R477–82.
Kristiansen S, Jones J, Handberg A, et al. Eccentric contractions decrease glucose transporter transcription rate, mRNA, and protein in skeletal muscle. Am J Physiol. 1997;272(5 Pt 1):C1734–8.
Green HJ. How important is endogenous muscle glycogen to fatigue in prolonged exercise? Can J Physiol Pharmacol. 1990;69(2):290–7.
Luckin KA, Biedermann MC, Jubrias SA, et al. Muscle fatigue: conduction or mechanical failure? Biomech Med Metabol Biol. 1991;46(3):299–316.
Romijn JA, Coyle EF, Sidossis LS, et al. Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am J Physiol. 1993;265(3 Pt 1):E380–91.
van Loon LJ, Greenhaff PL, Constantin-Teodosiu D, et al. The effects of increasing exercise intensity on muscle fuel utilisation in humans. J Physiol. 2001;536(Pt 1):295–304 (pii:PHY_12382).
Laurent D, Schneider KE, Prusaczyk WK, et al. Effects of caffeine on muscle glycogen utilization and the neuroendocrine axis during exercise. J Clin Endocrinol Metab. 2000;85(6):2170–5.
Williams MB, Raven PB, Fogt DL, et al. Effects of recovery beverages on glycogen restoration and endurance exercise performance. J Strength Cond Res. 2003;17(1):12–9.
Wilson RJ, Gusba JE, Robinson DL, et al. Glycogenin protein and mRNA expression in response to changing glycogen concentration in exercise and recovery. Am J Physiol Endocrinol Metab. 2007;292(6):E1815–22. doi:10.1152/ajpendo.00598.2006.
Bosch AN, Weltan SM, Dennis SC, et al. Fuel substrate kinetics of carbohydrate loading differs from that of carbohydrate ingestion during prolonged exercise. Metabolism. 1996;45(4):415–23.
Lima-Silva AE, De-Oliveira FR, Nakamura FY, et al. Effect of carbohydrate availability on time to exhaustion in exercise performed at two different intensities. Braz J Med Biol Res. 2009;42(5):404–12.
McConell G, Snow RJ, Proietto J, et al. Muscle metabolism during prolonged exercise in humans: influence of carbohydrate availability. J Appl Physiol. 1999;87(3):1083–6.
Coyle EF, Montain SJ. Carbohydrate and fluid ingestion during exercise: are there tradeoffs? Med Sci Sports Exerc. 1992;24(6):671–8.
Dupont G, Blondel N, Berthoin S. Performance for short intermittent runs: active recovery vs. passive recovery. Eur J Appl Physiol. 2003;89(6):548–54. doi:10.1007/s00421-003-0834-2.
MacDougall JD, Ray S, McCarteny N. Substrate utilization during weight lifting [abstract]. Med Sci Sports Exerc. 1988;20(2):S66.
MacDougall JD, Ray S, Sale DG, et al. Muscle substrate utilization and lactate production during weightlifting. Can J Appl Physiol. 1999;24(3):209–15.
Tesch PA, Colliander EB, Kaiser P. Muscle metabolism during intense, heavy-resistance exercise. Eur J Appl Physiol Occup Physiol. 1986;55(4):362–6.
Robergs RA, Pearson DR, Costill DL, et al. Muscle glycogenolysis during differing intensities of weight-resistance exercise. J Appl Physiol. 1991;70(4):1700–6.
Pascoe DD, Costill DL, Fink WJ, et al. Glycogen resynthesis in skeletal muscle following resistive exercise. Med Sci Sports Exerc. 1993;25(3):349–54.
Campos-Ferraz PL, Bozza T, Nicastro H, et al. Distinct effects of leucine or a mixture of the branched-chain amino acids (leucine, isoleucine, and valine) supplementation on resistance to fatigue, and muscle and liver-glycogen degradation, in trained rats. Nutrition. 2013;29(11–12):1388–94. doi:10.1016/j.nut.2013.05.003.
Knuiman P, Hopman MT, Mensink M. Glycogen availability and skeletal muscle adaptations with endurance and resistance exercise. Nutr Metab (Lond). 2015;12:59. doi:10.1186/s12986-015-0055-9.
Carter H, Pringle JS, Boobis L, et al. Muscle glycogen depletion alters oxygen uptake kinetics during heavy exercise. Med Sci Sports Exerc. 2004;36(6):965–72.
Gualano AB, Bozza T, Lopes De Campos P, et al. Branched-chain amino acids supplementation enhances exercise capacity and lipid oxidation during endurance exercise after muscle glycogen depletion. J Sports Med Phys Fit. 2011;51(1):82–8.
Suriano R, Edge J, Bishop D. Effects of cycle strategy and fibre composition on muscle glycogen depletion pattern and subsequent running economy. Br J Sports Med. 2010;44(6):443–8.
Barry BK, Riley ZA, Pascoe MA, et al. A spinal pathway between synergists can modulate activity in human elbow flexor muscles. Exp Brain Res. 2008;190(3):347–59. doi:10.1007/s00221-008-1479-5.
Philp A, MacKenzie MG, Belew MY, et al. Glycogen content regulates peroxisome proliferator activated receptor- partial differential (PPAR- partial differential) activity in rat skeletal muscle. PLoS One. 2013;8(10):e77200. doi:10.1371/journal.pone.0077200.
Wojtaszewski JF, MacDonald C, Nielsen JN, et al. Regulation of 5′AMP-activated protein kinase activity and substrate utilization in exercising human skeletal muscle. Am J Physiol Endocrinol Metab. 2003;284(4):E813–22. doi:10.1152/ajpendo.00436.2002.
Petibois C, Cazorla G, Poortmans JR, et al. Biochemical aspects of overtraining in endurance sports: the metabolism alteration process syndrome. Sports Med. 2003;33(2):83–94.
Achten J, Halson SL, Moseley L, et al. Higher dietary carbohydrate content during intensified running training results in better maintenance of performance and mood state. J Appl Physiol (1985). 2004;96(4):1331–40. doi:10.1152/japplphysiol.00973.2003.
Gravelle BL, Blessing DL. Physiological adaptation in women concurrently training for strength and endurance. J Strength Cond Res. 2000. Abstract proceeding.
Schumann M, Mykkänen OP, Doma K, et al. Effects of endurance training only versus same-session combined endurance and strength training on physical performance and serum hormone concentrations in recreational endurance runners. Appl Physiol Nutr Metab. 2015;40(1):28–36. doi:10.1139/apnm-2014-0262.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No sources of funding were used to assist in the preparation of this article
Conflict of interest
Kenji Doma, Glen Deakin and David Bentley declare that they have no conflicts of interest relevant to the content of this review.
Additional information
The original version of this article was revised: The title of the article in the Online version is incorrect. This Error has been corrected.
An erratum to this article is available at https://doi.org/10.1007/s40279-017-0766-3.
Rights and permissions
About this article
Cite this article
Doma, K., Deakin, G.B. & Bentley, D.J. Implications of Impaired Endurance Performance following Single Bouts of Resistance Training: An Alternate Concurrent Training Perspective. Sports Med 47, 2187–2200 (2017). https://doi.org/10.1007/s40279-017-0758-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-017-0758-3