Abstract
Background
Meniscal pathology is a commonly seen orthopedic condition that can affect a wide age range of patients. Athletes subject their menisci to an increased amount of stress during their careers and may increase their risk of meniscal pathology.
Objective
The purpose of this systematic review is to evaluate the prevalence of isolated meniscal pathology in asymptomatic athletes.
Methods
A systematic review was undertaken to determine the prevalence of meniscal pathology in asymptomatic athletes. A search of multiple databases was conducted. Recreational and higher-level athletes were included. Fourteen articles including 295 athletes (208 male, 87 female) were identified for inclusion (age range 14–66 years, mean 31.2 years). Meniscal pathology was visualized with magnetic resonance imaging and graded on a 1–4 scale (grades 1 and 2 indicating intrasubstance damage, grades 3 and 4 indicating a tear).
Results
There was an overall prevalence of 27.2 % (105/386) of knees with intrasubstance meniscal damage (grades 1 and 2), and 3.9 % (15/386) of knees with a tear (grades 3 and 4). When athletes were split into those who participate in pivoting sports versus non-pivoting sports, pivoting athletes showed an overall prevalence of 15.3 % (31/202) of knees with intrasubstance meniscal pathology and 2.5 % (5/202) of knees with a tear. Non-pivoting athletes showed a prevalence of 54.5 % (61/112) of knees with intrasubstance meniscal pathology and 5.4 % (6/112) of knees with a tear.
Conclusion
The overall prevalence of isolated meniscal pathology in asymptomatic athletes was 31.1 % (27.2 % with intrasubstance meniscal damage and 3.9 % with a meniscal tear). More studies of age-comparable, non-athletic populations are necessary for direct comparison with these groups.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Meniscal injuries are one of the most common orthopedic injuries, and can affect a wide age range of patients. |
Athletes subject their menisci to an increased amount of stress during their careers and may increase their risk of acute traumatic tear (pivot athletes) or degenerative tear (non-pivot athletes). |
Asymptomatic athletes have a relatively high prevalence of meniscal pathology on magnetic resonance imaging. |
1 Introduction
Meniscal pathology is among the most common orthopedic diagnoses, and can be seen in all age groups [1]. The mean annual incidence of meniscal tears is reported to be 60–70 per 100,000, and a male predominance has been shown in all age groups ranging from 2.5:1 to 4:1 [2–4]. The peak incidence for traumatic meniscal tears in men is between 21 and 30 years, and in women is between 11 and 20 years [3]. Arthroscopic treatment of meniscal injuries has become one of the most common orthopedic surgical procedures in the USA, constituting 10–20 % of all orthopedic surgeries [4, 5].
The meniscus is essential to the long-term health of the knee, providing a surface for transfer of weight and shock absorption, especially during high-impact activities [6–8]. All athletes, whether high-level or recreational, subject their knees to a greater amount of stress than the general population [9]. A potential consequence of this increased demand is an increased risk for osteoarthritis (OA). It has been reported that the incidence of OA in retired athletes is 12 times greater than that in the general population [9–11]. In a study by Creaby et al. [12], peak knee adduction moments were associated with cartilage defects and subchondral bone area in patients with medial knee OA, suggesting that increased mechanical loading may play a role in the pathological changes in articular cartilage and subchondral bone [12]. In addition, injury to the meniscus has a detrimental effect on load transmission, leading to further pressure on the articular cartilage [6]. Furthermore, the cartilage-to-cartilage contact area and peak contact pressure substantially increase following partial, sub-total, and total meniscectomy [12, 13]. These two factors have been shown to be significant parameters for evaluating degenerative OA [13].
Symptoms of meniscal tears include pain, swelling, locking catching, and loss of motion; however, not all meniscal pathology is symptomatic. In Framingham, Massachusetts, USA, meniscal pathology was identified via magnetic resonance imaging (MRI), and the majority (180 of 297; 61 %) of subjects did not have any pain, aching, or stiffness in the previous month (average age 62.3 years, range 50–90 years) [14]. Studies documenting the incidence of meniscal pathology in the general population have largely been performed in the elderly population. The prevalence of meniscal tears has been shown to increase with age and be as high as 56 % in men aged 70–79 years [14]. The Knee Osteoarthritis Initiative (OAI) identified signal change suggestive of mucoid degeneration in over 25 % of patients with an average age of 63.2 years [15]. It is difficult to determine from prior studies which condition (OA or meniscal injuries) precedes the other as they share many of the same risk factors and are associated with the same biological processes such as cartilage defects and alterations in bone size [16]. However, when comparing a group with pre-existing OA versus a control group without radiographic evidence of OA, the incidence of signal change suggestive of mucoid degeneration was reported to be 17.2 % higher in the OA group than in the control group [15].
Full-thickness focal chondral defects in the knee are more common in athletes than the general population, with more than one-half of asymptomatic athletes having a full-thickness defect [16]. Based on the similar loads borne by cartilage meniscal tissue, one would anticipate a relatively high prevalence of meniscus pathology in athletes. The purpose of this systematic review is to evaluate the prevalence of isolated meniscal pathology in asymptomatic athletes.
2 Methods
A systematic review including levels 1 through 4 evidence was performed as determined by the Oxford Centre for Evidence-Based Medicine [17]. A literature search of PubMed, MEDLINE, EMBASE, The Cochrane Collaboration of Systematic Reviews, the Cumulative Index for Nursing and Allied Health Literature (CINAHL), and SPORTDiscus was carried out on 25 February 2014. The authors of this review manually reviewed all articles, and references were reviewed for possible inclusion. Any disagreements among the authors regarding inclusion of an article were resolved by consensus. The search terms were knee, epidemiology, incidence, prevalence, occurrence, athlete(s), athletic, sport(s), meniscus, meniscal, tear(s), torn, injury, injuries, and lesion(s).
2.1 Inclusion Criteria
Inclusion criteria for studies in the analysis were (i) written in English language; (ii) human subject sample; (iii) levels 1–4 evidence; (iv) meniscal tears or signal changes suggestive of mucoid degeneration identified by MRI; and (v) investigated athletes.
2.2 Exclusion Criteria
Criteria for exclusion of articles from the analysis were (i) non-English-language studies; (ii) animal studies; (iii) level 5 evidence; (iv) studies evaluating meniscal pathology only in the setting of anterior cruciate ligament reconstruction; and (v) studies reporting a sample population only of athletes with known meniscal pathology (excluding athletes within the same population without meniscal pathology).
2.3 Imaging Techniques
The description of meniscal pathology in most of the studies identified used a system described by Crues et al. [18] (Table 1). The remaining studies [19, 20] held to the Crues grading system but reported their findings as tear or no tear. The meniscus was evaluated in six locations: the medial anterior horn, medial body, medial posterior horn, lateral anterior horn, lateral body, and lateral posterior horn. Meniscal tears were classified as oblique, horizontal, vertical, or radial.
Although there have been many described MRI techniques for diagnosing meniscal lesions, there is no universally accepted standard. We therefore included all meniscal lesions identified by MRI and documented the specific protocol used in each study. The specific study MRI techniques as well as the level of evidence of the paper are listed in Table 2.
3 Results
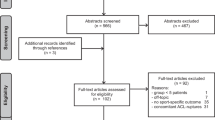
An initial search yielded 1728 citations. Limiting the studies to those including isolated meniscal tears yielded 373 citations. Further limitation to just athletes yielded 179 citations. For the purposes of this review, an “athlete” was defined as a subject who trains and competes in games or exhibitions. Both competitive recreational and high-level (high school, collegiate, and professional) athletes were included. Further limiting of studies to those including asymptomatic populations within this search yielded 30 citations. Limiting duplicate studies found in multiple databases resulted in 19 articles (Fig. 1). Upon further application of exclusion criteria (articles solely reporting on the asymptomatic population, or distinct separation of results from symptomatic/asymptomatic athletes), 14 articles were included in this systematic review (Table 2).
Fourteen studies were identified for inclusion, primarily case series (level 4 evidence). Within all studies, there were 295 athletes (386 knees) identified. MRI was used to identify meniscal pathology in all 14 studies. Images were reviewed by at least one musculoskeletal or experienced staff radiologist in all studies. The meniscal changes were classified according to the system proposed by Crues et al. [18] (Table 1). Of the subjects, 208 were male and 87 female. The age range of the subjects was 14–66 years, and the average age was approximately 31.2 years; however, there was one study that did not include an average age/range (Table 3).
The overall prevalence of meniscal signal change suggestive of mucoid degeneration that did not have a complete tear (grade 1 or 2 lesions) was 27.2 % (105/386) of knees (range 0–61 %), and the overall prevalence of meniscus tears (grade 3 or 4 lesions) was 3.9 % (15/386) of knees (range 0–37.5 %). Seven of the 14 studies focused on endurance athletes (marathon runners/Ironman athletes). Other types of athletes ranged from recreational to professional (i.e., National Basketball Association players) and various collegiate athletes (Table 3). In the seven studies that included only runners/long distance athletes, 54.5 % (61/112) of knees showed meniscal signal change suggestive of mucoid degeneration and 5.4 % (6/112) of knees showed a tear. Six of the studies reported primarily on athletes who participated in mostly pivoting/cutting types of sports. In this population, 15.3 % (31/202) of knees showed meniscal signal change suggestive of mucoid degeneration and 2.5 % (5/202) of knees showed a tear (Table 4).
4 Discussion
The most important finding of this study is that asymptomatic athletes have a high incidence of meniscus pathology. There is a paucity of epidemiologic data on the prevalence of meniscal pathology in the general population in this age group. Further research is needed in this area to place these numbers in more complete context (Table 5).
The prevalence of meniscal tears has been well-documented in the elderly. The menisci degenerate with increasing age and are at increased risk for tears. The prevalence of meniscal tears for women from aged 50–59 years was reported to be 19 %, and for men aged 70–79 years as high as 56 %, showing a progression with age [14]. A study conducted through the Knee OAI found the incidence of meniscal myxoid degeneration (grade 1 and 2) in patients with an average age of 63.2 years to be 26.6 % [15]. In the same study, those with OA had a 17.2 % increased incidence of meniscal signal change suggestive of mucoid degeneration compared with those without OA [15]. As expected, the average age of patients in the current review (31.2 years) was younger than the average age of patients in the aforementioned studies of meniscal tears and signal change suggestive of mucoid degeneration. Interestingly, this review demonstrates a higher prevalence of meniscal signal change suggestive of mucoid degeneration (grade 1 or 2) in this younger athletic population [14, 21]. In a subset of patients without radiographic evidence of OA from the ages of 45 to 60 years there was a 27.6 % prevalence of meniscal pathology, lower than that of our younger, athletic population [22].
There are concerning implications of the relatively high prevalence of meniscal pathology in the asymptomatic athlete population. Numerous clinical and biomechanical studies have shown the long-term consequences of the meniscus-deficient knee, which include increased loading of the cartilage, and meniscal injuries have long been associated with osteoarthritic changes and increased morbidity [8, 14, 23]. Incidental (asymptomatic) meniscal findings on MRI were found in 95 % of persons with radiographic evidence of severe OA (Kellgren and Lawrence grade 3 or higher) [14]. The prevalence of incidental meniscal signal change suggestive of mucoid degeneration decreased with less severe OA (Kellgren and Lawrence grade 2) and normal knees (82 vs. 25 %, respectively; P < 0.001) [14, 24]. This suggests that asymptomatic meniscal signal changes suggestive of mucoid degeneration are at least one factor in the development of OA. Symptomatic meniscal injuries have also been shown to be associated with higher rates of OA, and a high prevalence of meniscal damage (67–91 %) among patients with symptomatic OA of the knee has previously been reported [25–28]. Knees with meniscal damage (grade 2 intrasubstance change) have been shown to have higher rates of symptomatic knee OA [29]. Previous studies have also documented the influence of meniscectomy on the likelihood of progression to radiographic OA [30–32]. Another study investigated patients with symptomatic OA and found a strong association between meniscal tears, signal change suggestive of mucoid degeneration, and cartilage loss [29]. This points to meniscus integrity (symptomatic or asymptomatic) as a key factor in the development of OA.
Age and body mass index (BMI) likely play a critical role in the degeneration of the meniscus. Limitations of the available data precluded stratification of data by patient age. A study by Hwang et al. [33] showed a 4.9-fold increase in medial meniscus posterior root tears in patients with a BMI over 30 kg/m2. Although most of the studies included in this review did not document BMI, those that did had no subjects with a BMI of 30 kg/m2 or greater. With the recent increase in the popularity of distance cross training and running, further studies are needed to elucidate the effects of age and BMI on the athlete’s meniscus.
This review documented a higher prevalence of asymptomatic meniscal pathology in the non-pivot athletes (60 %) (distance runners, Ironman athletes, etc.) than in athletes whose sport requires consistent pivoting (18 %) (football, basketball, etc.) This finding likely highlights the effect of recurrent impact activities (distance running, endurance sports) on the meniscus. Interestingly, meniscal tears were also higher in the non-pivot athletes, suggesting that early meniscal degeneration may predispose the meniscus to unrecognized (asymptomatic) tears and further propagate the risk of early-onset OA.
Limitations of this review include those derived from the included studies, which were primarily small series of sport-/training-specific populations. This review found that classification of MRI meniscal findings in asymptomatic athletes was often not as specific as in the general population. There are also methodological differences such as various MRI protocols used in the studies that may influence their sensitivity for detection of meniscal mucoid degeneration and meniscal tears. The studies in this review clearly defined grade 1 and 2 lesions as a meniscal signal change suggestive of mucoid degeneration, and grades 3 or 4 as tears. Some studies in the general population either did not report grade 1 and 2 changes or reported them and grade 3/4 tears as one group.
5 Conclusions
The overall prevalence of meniscal pathology in asymptomatic athletes was 31.1 % (27.2 % with meniscal change suggestive of mucoid degeneration and 3.9 % with a meniscal tear). More studies of the general population in this age group are necessary for direct comparison of these groups.
References
Alatakis S, Naidoo P. MR imaging of meniscal and cartilage injuries of the knee. Magn Reson Imaging Clin N Am. 2009;17:741–56, vii.
Nielsen AB, Yde J. Epidemiology of acute knee injuries: a prospective hospital investigation. J Trauma. 1991;31:1644–8.
Greis PE, Bardana DD, Holmstrom MC, et al. Meniscal injury: I. Basic science and evaluation. J Am Acad Orthop Surg. 2002;10:168–76.
Gu YL, Wang YB. Treatment of meniscal injury: a current concept review. Chin J Traumatol. 2010;13:370–6.
Renstrom P, Johnson RJ. Anatomy and biomechanics of the menisci. Clin Sports Med. 1990;9:523–38.
Kurosawa H, Fukubayashi T, Nakajima H. Load-bearing mode of the knee joint: physical behavior of the knee joint with or without menisci. Clin Orthop Relat Res. 1980;149:283–90.
Shrive NG, O’Connor JJ, Goodfellow JW. Load-bearing in the knee joint. Clin Orthop Relat Res. 1978;131:279–87.
Walker PS, Erkman MJ. The role of the menisci in force transmission across the knee. Clin Orthop Relat Res. 1975;109:184–92.
Arendt E, Dick R. Knee injury patterns among men and women in collegiate basketball and soccer. NCAA data and review of literature. Am J Sports Med. 1995;23:694–701.
Drawer S, Fuller CW. Propensity for osteoarthritis and lower limb joint pain in retired professional soccer players. Br J Sports Med. 2001;35:402–8.
Roos H. Are there long-term sequelae from soccer? Clin Sports Med. 1998;17:819–31, viii.
Creaby MW, Wang Y, Bennell KL, et al. Dynamic knee loading is related to cartilage defects and tibial plateau bone area in medial knee osteoarthritis. Osteoarthr Cartil. 2010;18:1380–5.
Segal NA, Anderson DD, Iyer KS, et al. Baseline articular contact stress levels predict incident symptomatic knee osteoarthritis development in the MOST cohort. J Orthop Res. 2009;27:1562–8.
Englund M, Guermazi A, Gale D, et al. Incidental meniscal findings on knee MRI in middle-aged and elderly persons. N Engl J Med. 2008;359:1108–15.
Badlani JT, Borrero C, Golla S, et al. The effects of meniscus injury on the development of knee osteoarthritis: data from the osteoarthritis initiative. Am J Sports Med. 2013;41:1238–44.
Flanigan DC, Harris JD, Trinh TQ, et al. Prevalence of chondral defects in athletes’ knees: a systematic review. Med Sci Sports Exerc. 2010;42:1795–801.
Obremskey WT, Pappas N, Attallah-Wasif E, et al. Level of evidence in orthopaedic journals. J Bone Jt Surg Am. 2005;87:2632–8.
Crues JV 3rd, Mink J, Levy TL, et al. Meniscal tears of the knee: accuracy of MR imaging. Radiology. 1987;164:445–8.
Soder RB, Simoes JD, Soder JB, et al. MRI of the knee joint in asymptomatic adolescent soccer players: a controlled study. AJR Am J Roentgenol. 2011;196:W61–5.
Major NM, Helms CA. MR imaging of the knee: findings in asymptomatic collegiate basketball players. AJR Am J Roentgenol. 2002;179:641–4.
Boden SD, Davis DO, Dina TS, et al. A prospective and blinded investigation of magnetic resonance imaging of the knee. Abnormal findings in asymptomatic subjects. Clin Orthop Relat Res. 1992;282:177–85.
Sharma L, Chmiel JS, Almagor O, et al. Significance of preradiographic magnetic resonance imaging lesions in persons at increased risk of knee osteoarthritis. Arthritis Rheumatol. 2014;66:1811–9.
Khetia EA, McKeon BP. Meniscal allografts: biomechanics and techniques. Sports Med Arthrosc. 2007;15:114–20.
Park HJ, Kim SS, Lee SY, et al. A practical MRI grading system for osteoarthritis of the knee: association with Kellgren-Lawrence radiographic scores. Eur J Radiol. 2013;82:112–7.
Englund M, Niu J, Guermazi A, et al. Effect of meniscal damage on the development of frequent knee pain, aching, or stiffness. Arthritis Rheum. 2007;56:4048–54.
Kornaat PR, Bloem JL, Ceulemans RY, et al. Osteoarthritis of the knee: association between clinical features and MR imaging findings. Radiology. 2006;239:811–7.
Link TM, Steinbach LS, Ghosh S, et al. Osteoarthritis: MR imaging findings in different stages of disease and correlation with clinical findings. Radiology. 2003;226:373–81.
Bhattacharyya T, Gale D, Dewire P, et al. The clinical importance of meniscal tears demonstrated by magnetic resonance imaging in osteoarthritis of the knee. J Bone Jt Surg Am. 2003;85-A:4–9.
Hunter DJ, Zhang YQ, Niu JB, et al. The association of meniscal pathologic changes with cartilage loss in symptomatic knee osteoarthritis. Arthritis Rheum. 2006;54:795–801.
Macnicol MF, Thomas NP. The knee after meniscectomy. J Bone Jt Surg Br. 2000;82:157–9.
Johnson RJ, Kettelkamp DB, Clark W, et al. Factors effecting late results after meniscectomy. J Bone Jt Surg Am. 1974;56:719–29.
Englund M, Roos EM, Lohmander LS. Impact of type of meniscal tear on radiographic and symptomatic knee osteoarthritis: a sixteen-year followup of meniscectomy with matched controls. Arthritis Rheum. 2003;48:2178–87.
Hwang BY, Kim SJ, Lee SW, et al. Risk factors for medial meniscus posterior root tear. Am J Sports Med. 2012;40:1606–10.
Kaplan LD, Schurhoff MR, Selesnick H, et al. Magnetic resonance imaging of the knee in asymptomatic professional basketball players. Arthroscopy. 2005;21:557–61.
Krampla W, Mayrhofer R, Malcher J, et al. MR imaging of the knee in marathon runners before and after competition. Skeletal Radiol. 2001;30:72–6.
LaPrade RF, Burnett QM 2nd, Veenstra MA, et al. The prevalence of abnormal magnetic resonance imaging findings in asymptomatic knees. With correlation of magnetic resonance imaging to arthroscopic findings in symptomatic knees. Am J Sports Med. 1994;22:739–45.
Ludman CN, Hough DO, Cooper TG, et al. Silent meniscal abnormalities in athletes: magnetic resonance imaging of asymptomatic competitive gymnasts. Br J Sports Med. 1999;33:414–6.
Schueller-Weidekamm C, Schueller G, Uffmann M, et al. Incidence of chronic knee lesions in long-distance runners based on training level: findings at MRI. Eur J Radiol. 2006;58:286–93.
Schueller-Weidekamm C, Schueller G, Uffmann M, et al. Does marathon running cause acute lesions of the knee? Evaluation with magnetic resonance imaging. Eur Radiol. 2006;16:2179–85.
Shellock FG, Deutsch AL, Mink JH, et al. Do asymptomatic marathon runners have an increased prevalence of meniscal abnormalities? An MR study of the knee in 23 volunteers. AJR Am J Roentgenol. 1991;157:1239–41.
Stahl R, Luke A, Ma CB, et al. Prevalence of pathologic findings in asymptomatic knees of marathon runners before and after a competition in comparison with physically active subjects-a 3.0 T magnetic resonance imaging study. Skeletal Radiol. 2008;37:627–38.
Walczak BE, McCulloch PC, Kang RW, et al. Abnormal findings on knee magnetic resonance imaging in asymptomatic NBA players. J Knee Surg. 2008;21:27–33.
Reinig JW, McDevitt ER, Ove PN. Progression of meniscal degenerative changes in college football players: evaluation with MR imaging. Radiology. 1991;181:255–7.
Hagemann GJ, Rijke AM, Corr PD. Do knees survive the Comrades Marathon? S Afr Med J. 2008;98:873–6.
Shellock FG, Hiller WD, Ainge GR, et al. Knees of Ironman triathletes: magnetic resonance imaging assessment of older (>35 years old) competitors. J Magn Reson Imaging. 2003;17:122–30.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No sources of funding were used to assist in the preparation of this article.
Conflict of interest
Corey Beals, Robert Magnussen, William Graham, and David Flanigan declare that they have no conflicts of interest relevant to the content of this review. David Flanigan is a consultant for Smith & Nephew, Vericel, Conmed, Depuy-Mitek, and Zimmer.
Rights and permissions
About this article
Cite this article
Beals, C.T., Magnussen, R.A., Graham, W.C. et al. The Prevalence of Meniscal Pathology in Asymptomatic Athletes. Sports Med 46, 1517–1524 (2016). https://doi.org/10.1007/s40279-016-0540-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-016-0540-y