Abstract
Hypercholesterolaemia is a highly prevalent condition that has major health and cost implications for society. Pharmacotherapy is an important and effective treatment modality for hypercholesterolaemia, with 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors (‘statins’) the most commonly used class of drugs. Over the past decade, there has been intensive research to identify pharmacogenetic markers to guide treatment of hypercholesterolaemia. This study aimed to review the evidence of incremental cost, effect and cost effectiveness of pharmacogenetic-guided treatment of hypercholesterolaemia. Three cost-effectiveness analyses (CEAs) were identified that studied the value of screening for genotypes of angiotensin I converting enzyme (ACE), cholesteryl ester transfer protein (CETP), and kinesin family member 6 (KIF6) prior to initiating statin therapy. For all three CEAs, a major limitation identified was the reproducibility of the evidence supporting the clinical effect of screening for the pharmacogenetic marker. Associated issues included the uncertain value of pharmacogenetic markers over or in addition to existing approaches for monitoring lipid levels, and the lack of evidence to assess the effectiveness of alternative therapeutic options for individuals identified as poor responders to statin therapy. Finally, the economic context of the market for diagnostic tests (is it competitive or is there market power?) and the practicality of large-scale screening programmes to inform prescribing in a complex and varied market may limit the generalizability of the results of the specific CEAs to policy outcomes. The genotype of solute carrier organic anion transporter family member 1B1 (SLCO1B1) has recently been associated with increased risk of muscle toxicity with statin therapy and the review identified that exploration of cost effectiveness of this pharmacogenetic marker is likely warranted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
• There has been extensive research into pharmacogenetic markers of the therapeutic and adverse effects of statin drugs. Despite this, there are few pharmacogenetic markers that have consistently been associated with important clinical outcomes
• Only three cost-effectiveness analyses of pharmacogenetic markers of statin therapy were identified. Generally, the evidence for the effectiveness of these markers is not strong, and hence the validity of the findings of these studies is questionable
• Further exploration of the cost effectiveness of testing for the solute carrier organic anion transporter family member 1B1 (SLCO1B1) genotype to identify individuals at higher risk of statin toxicity is warranted
• Future cost-effectiveness analyses of pharmacogenetic-guided therapy for hypercholesterolaemia should consider: the quality and reproducibility of evidence for the pharmacogenetic marker, the alternative treatment options available for poor responders, the additional value of information of the marker if lipid monitoring is standard practice, the economic context of the market for diagnostic tests, and the practicality of large-scale screening programmes in an area such as statin prescribing
1 Background
1.1 Hypercholesterolaemia and Pharmacological Treatments
Hypercholesterolaemia (or hyperlipidaemia) generally refers to an increased serum concentration of low-density lipoprotein (LDL) cholesterol. Hypercholesterolaemia is the major cause of atherosclerosis, and is therefore a major risk factor for the development of cardiovascular disease (CVD) including coronary artery disease and ischaemic stroke [1].
There are a number of drugs that may be used to lower LDL cholesterol; however, by far the most commonly used are the ‘statins’, or 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGCR) inhibitors [2, 3]. The first statin drugs were introduced to the market in 1987 (Fig. 1). In the USA, the percentage of adults 45 years of age and over using statin drugs has increased from 2.4 % in 1988–1994 to 25.1 % in 2005–2008, and almost half of males and over 35 % of women aged 65 years or older take a statin [2]. In 2006, one drug alone (atorvastatin) was generating sales of US$8.6 billion in the USA and US$13.6 billion internationally [4]. The Australian Pharmaceutical Benefits Scheme (PBS) subsidy of the top three statins (atorvastatin, rosuvastatin and simvastatin) accounted for 14 % of the entire PBS expenditure for the 2010/2011 financial year [3].
Key developments in the history of statin pharmacogenetics. ACE angiotensin I converting enzyme, CETP cholesteryl ester transfer protein, GWAS genome-wide association study, KIF6 kinesin family member 6, NIH National Institutes of Health, SLCO1B1 solute carrier organic anion transporter family member 1B1
1.1.1 Efficacy of Statin Drugs
Current European and US guidelines state that cholesterol-lowering medications are indicated in individuals with hypercholesterolaemia, usually in addition to other risk factors for CVD, such as presence of coronary heart disease, hypertension, diabetes and age [1, 5]. Statins have been demonstrated to reduce LDL cholesterol, CVD risk and mortality risk [6]. The absolute benefit of statin therapy is dependent upon the baseline risk of CVD [7], and is associated with the extent to which LDL cholesterol is lowered [8]. Thus, guidelines typically suggest initiation of statins on the basis of CVD risk, and monitoring of LDL concentrations is commonly utilized to guide treatment based on targets for LDL concentrations and/or change in LDL concentration [1, 5].
It is important to note, however, that there are a number of other effects of statins (often referred to as pleiotropic effects, e.g. stabilization of atherosclerotic plaques, anti-inflammatory and anti-coagulant effects) that may be important in conferring benefits to cardiovascular health over and above those that are associated with LDL lowering [9–13]. It is unclear if some/all of these are class effects, and to what extent these properties contribute to the reduction in CVD events.
1.2 Statin Pharmacogenetics
1.2.1 Introduction to Pharmacogenetics and Personalized Medicine
Personalized medicine is based on the concept that observable characteristics of a person or a disease may be used to make better treatment decisions. The concept is not new: age, weight, renal function and disease subtype, for example, have been used for many years to guide the selection of drugs and drug doses. Pharmacogenetics is an extension of this concept in which genetic information from the individual is used to guide treatment decisions. The reason why there is considerable excitement in the area of pharmacogenetics is threefold. Firstly, there are hundreds of thousands of genetic differences between individuals, meaning that there is a much greater chance of identifying an observable characteristic of an individual that is strongly predictive of treatment effect. Secondly, genetic differences occur at the molecular level and often result in changes in the amount and/or activity of important proteins such as enzymes, transporters and receptors. Drug effects also occur at the molecular level through interaction with enzymes, transporters and receptors, and hence the existence of a genetic difference that is strongly predictive of an altered effect of a drug is biologically plausible. Lastly, the technologies for identifying genetic differences are rapidly improving, resulting in cheaper screening approaches and the ability to measure hundreds of thousands of potential genetic differences between individuals.
1.2.2 Overview of Statin Pharmacogenetic Markers
There are many proposed pharmacogenetic markers of statin therapy [14]. The most promising are very briefly highlighted (below and in Fig. 2) with respect to the biological pathways for lipid regulation, drug absorption, drug distribution and drug elimination.
Biochemical pathways associated with putative pharmacogenetic markers of statin therapy. ABCB1 ATP-binding cassette sub-family B member 1, ACE angiotensin I converting enzyme, APOE apolipoprotein E, CETP cholesteryl ester transfer protein, CoA coenzyme A, CYP450 cytochrome P450 enzymes, HDL-C high-density lipoprotein cholesterol, HMGCR3-hydroxy-3-methylglutaryl coenzyme A reductase, KIF6 kinesin family member 6, LDL-C low-density lipoprotein cholesterol, LDLR low-density lipoprotein receptor, SLCO1B1 solute carrier organic anion transporter family member 1B1
Markers involved in lipid regulation that have been the most studied include cholesteryl ester transfer protein (CETP), apolipoprotein E (APOE), LDL-Receptor (LDLR) and HMGCR. CETP mediates the exchange of cholesterol and statins are known to reduce CETP activity by up to 30 % [15]. APOE and LDLR are associated with LDL uptake and the APOE genotype is known to influence binding to LDLRs, while HMGCR is the pharmacological target for statin drugs and thus is an obvious candidate for a pharmacogenetic marker of statin response.
Other than pravastatin and rosuvastatin, statins are metabolized by a variety of cytochrome P450 enzymes (CYP450). Furthermore, some statins are substrates of the transport protein ATP-binding cassette sub-family G member 2 (ABCG2) [16]. As such, variants of these proteins that are associated with higher or lower activity have the potential to predict changes in the systemic exposure to statins, and therefore be predictive of efficacy and toxicity. Solute carrier organic anion transporter family member 1B1 (SLCO1B1) transports all of the statins (other than fluvastatin) into hepatocytes [17] where they act to inhibit hepatic cholesterol biosynthesis.
In addition, there are also a number of putative pharmacogenetic markers of statin therapy that have no known biologically plausible mechanism for influencing statin effect. This includes kinesin family member 6 (KIF6) and angiotensin I converting enzyme (ACE), which have been hypothesized to be associated with one of the unknown pleiotropic effects of statins.
The majority of these markers aim to predict differences in statin efficacy (i.e. LDL cholesterol-lowering effect or morbidity/mortality benefits), but there is also growing interest in the use of pharmacogenetic markers to predict risk of toxicity with statin therapy [18]. Although the majority of studies on statin pharmacogenetics are association (observational) studies, there are also a number of retrospective genetic subgroup analyses based on previously conducted randomized controlled trials (RCTs). These RCT-based analyses provide the highest quality evidence available as there are no RCTs specifically designed to assess the value of statin pharmacogenetic markers.
By identifying markers that identify individuals who are at increased or decreased risk of achieving LDL targets, informed choices may be made about drug selection and/or dosage, which may lead to a faster attainment of these targets and/or use of lower drug doses. On the other hand, surrogate endpoints such as LDL concentration are unable to capture the impact that the pleiotropic effects have on reducing CVD and mortality, and there is no known surrogate marker of efficacy related to these effects. Genetic differences that allow identification of subgroups who will receive greater benefit from the pleiotropic effects of statins may therefore be more useful at tailoring treatment. Furthermore, markers may be able to identify individuals who are at an increased risk of serious adverse events, and therefore where a lower than normal maximum dose should not be exceeded [16] and/or an alternative therapy should be used.
The key issue is whether the additional costs of testing for these markers are justified by the benefits, which could include: (1) health benefits from improved alignment of treatment with a patient’s characteristics; and (2) financial savings from a reduction in prescribing at doses that are higher than necessary or prescribing of drugs for patients for whom pharmacotherapy is neither cost effective nor clinically effective.
This paper aims to review the cost effectiveness of pharmacogenetic markers to guide statin therapy, specifically to:
-
Identify all cost-effectiveness studies that assess screening a specific pharmacogenetic marker prior to initiating treatment of a statin drug
-
Critically review whether the cost effectiveness of screening for a specific pharmacogenetic marker is favourable
-
Identify pharmacogenetic markers for which further study of cost effectiveness would be useful
-
Highlight general methodological issues with assessing the cost effectiveness of pharmacogenetic markers for guiding treatment of hypercholesterolaemia
In Sect. 2, we present a summary and commentary of the cost-effectiveness studies identified with a focus on the key assumptions and potential limitations of the studies. Subsequently, in Sect. 3 we discuss potential future directions of research and general methodological issues identified in assessing the cost effectiveness of statin pharmacogenetic markers.
2 Literature Review of Cost-Effectiveness Studies
2.1 Detailed Literature Search
A detailed database search was performed to find all references relevant to the economic evaluation of pharmacogenetic markers of statin therapy. Major challenges were the lack of use of MEDLINE medical subject heading (MeSH) terms in this field, recognition and inclusion of all gene variants due to nomenclature changes over the past 15 years, and development of an appropriate search strategy that captured genotypes rather than phenotypes. Over 1,500 titles and abstracts were reviewed to guide the search strategy. The following databases were searched: PubMed, Cochrane, EconLit, EMBASE, MEDLINE, International Pharmaceutical Abstracts and SciFinder.
Given that MeSH terms and their equivalent did not produce original articles of relevance, a combination of search terms was used: (exp ‘Hydroxymethylglutaryl CoA Reductases’ [MeSH Terms] OR exp Hydroxymethylglutaryl-CoA Reductase Inhibitors [MeSH Terms] OR statin*) AND (pharmacogen* OR genomic* or genetic*) AND (pharmacoecon* OR cost-effective).
After the removal of duplicates, this algorithm had identified 336 articles requiring further review. To ensure all relevant pharmacoeconomic and pharmacogenomic statin articles had been captured, additional internet searches of Google, Pharmacogenomics Knowledge Base and other internationally recognized pharmacogenomic institution websites were performed. These were further supplemented by hand searching. The majority of articles were excluded; they were about non-pharmacogenomic aspects of familial hypercholesterolaemia (FH), were general reviews or only mentioned the future possibility of performing economic analyses.
The exploratory search identified only three economic evaluations of pharmacogenomic approaches to statin response (Table 1) [19–21]. Two were available as full journal publications, and the remaining evaluation was presented as a conference poster that was in the public domain. All were published in the English language. In addition, a number of economic studies were identified relating to genetic testing in FH. Although this is not classically considered a pharmacogenetic marker, it does influence statin treatment indirectly and, for completeness, a brief summary of these studies is included.
2.2 CETP Genotype (Kemp et al. [19])
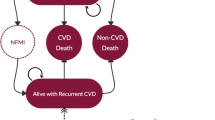
This cost-effectiveness analysis (CEA) was set in the Australian healthcare system and focused on assessing screening for the Taq1B polymorphism in the CETP gene to guide treatment in the secondary prevention of coronary heart disease [19]. Three different CETP Taq1B genotypes (mutually exclusive and complete subgroups) were considered: B2B2, B1B2 and B1B1. The modelling assumed that individuals with the B2B2 genotype received the greatest benefit from the use of statins in terms of reduction in CVD events; individuals with the B1B1 genotype received the smallest benefit from the use of statins; and individuals with the B1B2 genotype had an intermediate statin treatment effect.
2.2.1 Strategies Compared
Three alternative strategies involving the use of the Taq1B CETP genotype were compared with standard practice in this study:
-
1.
No screening for the CETP genotype and the use of a statin for all individuals
-
2.
Screen for the CETP genotype and the use of a statin for individuals with a B2B2 genotype and no lipid therapy for individuals with a B1B2 or B1B1 genotype
-
3.
Screen for the CETP genotype and the use of a statin for individuals with a B2B2 or B1B2 genotype and no lipid therapy for individuals with a B1B1 genotype
-
4.
Screen for the CETP genotype and the use of a statin for individuals with a B2B2 or B1B2 genotype and ezetimibe therapy for individuals with a B1B1 genotype
2.2.2 Study Conclusions
The study concluded that prescribing statins only to individuals carrying at least one B2 allele (i.e. B2B2 or B1B2) is more cost effective than prescribing statins to all patients. This would result in considerable cost savings, but be at the expense of increased CVD events (as statins were assumed to have benefit [albeit reduced] in the B1B1 group). Additionally, the study indicated that using ezetimibe instead of a statin for individuals with the B1B1 genotype would result in a reduced cost and increased effect compared with treating everyone with statins. However, the authors acknowledged that more evidence is required to support such a change to the current guidelines and prescribe ezetimibe as initial therapy in this subgroup of patients.
2.2.3 Key Assumptions and Evidence
The first major implicit assumption of the model is that the poor statin response associated with individuals having the CETP B1B1 genotype would not be reflected by a smaller reduction in LDL cholesterol. If this assumption does not hold, the incremental value of the genotyping CETP would be reduced considerably as poor response may be detected during regular monitoring of LDL cholesterol and the additional information provided by the Taq1B polymorphism would be attenuated. Furthermore, dose increases to achieve a set LDL cholesterol reduction may then mitigate any reduction in statin response that was modelled.
The second major assumption was that individuals with the B1B1 genotype taking ezetimibe would have a greater reduction in CVD events than if they had used a statin. This comprises two assumptions: (1) that the benefit of ezetimibe is not associated with the CETP genotype, and (2) that the use of ezetimibe reduces the risk of CVD. The ability of ezetimibe to reduce CVD events is not well accepted [22], and the assumption that ezetimibe is not affected by the Taq1B CETP genotype has not been substantiated. Thus, an important alternative to consider is whether increasing the dose of statin drugs for individuals identified to be poor responders (B1B1 genotype) will partially or fully overcome the reduced therapeutic effect. If so, an important alternative that needs to be modelled is starting individuals with a B1B1 genotype at a higher statin dose. However, in order to model the advantage of additional information on genotype, it would be necessary to compare this with a strategy where the dose was increased if there was insufficient reduction of LDLs (as previously discussed).
The estimates of the therapeutic effect of statins for CETP genotype subgroups were derived solely from an observational study of 2,531 patients with significant coronary artery disease who underwent coronary arteriography between 1994 and 1998 [23]. The effect of statins compared with no statin therapy for different genotypes suggested the differential effect size; however, the subgroups’ statins or no statins were based on prescribing at discharge and did not take into account the dose and type of statin or changes in these over time, including people who were not prescribed a statin at discharge but prescribed one at a later stage.
2.2.4 Updated Estimates
There are other studies of the association between the Taq1B CETP genotype and statin effect and these generally show little effect or inconsistent direction of the association [24–31]. A 2005 meta-analysis of three studies found that there was no statistically or clinically significant interaction between the Taq1B CETP genotype and outcomes of pravastatin therapy [32]. A 2008 review of the literature identified five studies and concluded that it is unlikely that the Taq1B CETP genotype modifies the effect of statins on CVD event reduction [33].
The final consideration is the cost of statin therapy. The study estimated the annual cost of 40 mg simvastatin therapy in 2003 to be 866 Australian dollars (A$) [19], whereas the current cost is substantially less (A$335), reflecting the loss of exclusivity and emergence of competition between generic versions of simvastatin. Thus, even if the clinical effect of the Taq1B CETP genotype is as large as assumed in the study, the estimates of cost savings would need to be significantly reduced to reflect changes in drug pricing since the time the study was undertaken.
2.3 ACE Genotype (Maitland-van der Zee et al. [20])
This CEA assessed the value of screening men for their ACE genotype prior to initiating statin therapy from the healthcare payer perspective in The Netherlands [20]. Men with the DD genotype were assumed to receive no benefit from using statin therapy in terms of reduction in CVD events (relative risk [RR] 1.00), men with the ID genotype were assumed to have a modest statin treatment effect (RR 0.87), and men with the II genotype were assumed to have a profound statin treatment effect (RR 0.23). It was further assumed that the ACE genotype did not influence the treatment effect of alternative lipid-lowering agents (fibrates, nicotinic acid and bile acid sequestrants) [20].
2.3.1 Strategies Compared
The study compared two strategies for statin therapy:
-
1.
No screening for the ACE genotype and the use of a statin in all men
-
2.
Screening of the ACE genotype, the use of a statin for men with a II or ID ACE genotype, and the use of either no therapy or an alternative lipid-lowering agent for men with the DD genotype
2.3.2 Study Conclusions
The study results generally indicated that the screening strategy was dominant—resulting in reduced cost with no reduction in effect (life-years) [20]. The sensitivity analysis explored the issue of the reduction in future costs of statins due to patent expiry and found that, even if the price of stain therapy were reduced by 50 %, the screening strategy would remain cost saving. The authors acknowledged that confirmatory evidence of the effect of the ACE genotype on statin effectiveness was required.
2.3.3 Key Assumptions and Evidence
The authors made implicit assumptions analogous to those identified in Kemp et al. [19]. The model design did not consider the possibility that in regular clinical practice men who were identified by genotype would have otherwise been identified by the poor LDL response and have their dose or type of lipid therapy changed as a consequence.
The estimates of statin treatment effect for men with different ACE genotypes were sourced from the Rotterdam Study, a population-based prospective cohort study [34]. Differences in statin treatment effect on CVD outcomes were identified for men, but not for women. Although these results were used to model the effect of the ACE genotype, two prior studies suggested that the effect of the genotype was uncertain [35, 36]. A study published in 2000 suggested that the treatment effect of statins (in terms of LDL reduction and progression of coronary artery disease) was greatest for the DD genotype rather than the II genotype [35]. In addition, a case-control study published in 2001 based on a subset of 486 participants in the CARE (Cholesterol And Recurrent Events) trial indicated that the treatment effect of pravastatin was unrelated to the ACE genotype alone [36].
2.3.4 Updated Estimates
In 2007, the same research group that undertook the CEA subsequently published a retrospective subgroup analysis of the ALLHAT (Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack Trial) RCT with respect to the ACE genotype [37]. The ALLHAT genetic sub-study indicated that the ACE genotype did not appear to significantly influence the statin effect on CVD events [37]. In 2009, a meta-analysis focusing on the ACE genotype concluded that there was little evidence to support a statistically and clinically important difference in statin treatment effect between individuals with different ACE genotypes [38]. The results and conclusions of this CEA [20] must therefore be carefully interpreted in light of the current lack of evidence to support the influence of the ACE genotype on statin efficacy.
2.4 KIF6 Genotype (Parthan et al. [21])
Parthan and colleagues [21] undertook a CEA to assess the value of screening individuals for the KIF6 genotype prior to the selection of moderate or intensive statin therapy from a third-party payer perspective in the USA. Currently only a conference abstract and poster report are available for the study, and hence details are lacking on some of the methods and assumptions used [21]. The modelling was primarily based on a genetic sub-study of the PROVE IT-TIMI 22 (Pravastatin or Atorvastatin Evaluation and Infection Therapy—Thrombolysis in Myocardial Infarction 22) RCT, which compared atorvastatin 80 mg/day (intensive statin therapy) with pravastatin 40 mg/day (moderate statin therapy) for individuals with acute coronary syndrome [39, 40]. In the overall study population, atorvastatin 80 mg/day was found to have a statistically significant reduction in the risk of CVD events compared with pravastatin 40 mg/day [39]. The genetic sub-study of the PROVE IT-TIMI 22 trial comprised 1,778 individuals (approximately 40 % of the main trial population) and identified that individuals carrying the KIF6 Trp719Arg variant received a much greater benefit from using intensive statin therapy instead of using moderate statin therapy [40]. In contrast, non-carriers of the Trp719Arg variant received little or no benefit from using intensive rather than moderate statin therapy.
2.4.1 Strategies Compared
The CEA therefore compared the following treatment strategies:
-
1.
The use of pravastatin 40 mg/day for all patients
-
2.
The use of atorvastatin 80 mg/day for all patients
-
3.
Screening for the KIF6 genotype with the use of atorvastatin 80 mg/day for Trp719Arg carriers and pravastatin 40 mg/day for Trp719Arg non-carriers
2.4.2 Study Conclusions
The CEA found that the use of atorvastatin for all patients was the dominant strategy in the base case [21]. However, this assumes that atorvastatin 80 mg and pravastatin 40 mg are of equivalent cost on the basis of the impending loss of exclusivity of atorvastatin. Sensitivity analyses demonstrated that the cost of atorvastatin is a very important factor influencing whether screening for the KIF6 genotype is cost effective. The CEA modelled a sensitivity analysis in which screening for KIF6 would increase adherence from 50 % to 60 %. In this sensitivity analysis, screening for KIF6 was found to be dominant—resulting in the lowest cost and greatest number of QALYs [21].
2.4.3 Key Assumptions and Evidence
As in the previous two CEAs [19, 20], the possibility that this group of patients could otherwise have been identified as requiring more intensive therapy was not addressed. Specifically, this would have required the model design to reflect the usual practice for patients who start with less intensive therapy to be placed on more intensive therapy if their LDL cholesterol levels do not respond sufficiently. The model could then have explored the benefits of using an alternative or additional piece of information on genotype.
Adherence is a major issue limiting the effectiveness of pharmacotherapy [41] and it has been proposed that pharmacogenetic testing may improve adherence by ensuring that medications are prescribed to individuals with the greatest likelihood of benefit and the least risk of toxicity. Although no evidence was cited in the report to justify the assumption that KIF6 screening would increase adherence from 50 % to 60 %, the review authors are aware of a recent prospective, non-randomized intervention trial, which assessed the effect of providing patients with information about KIF6 carrier status on statin adherence [42]. The study found that providing individuals with knowledge of their KIF6 genotype significantly improved statin adherence at 6 months after the initiation of the statin (63 % vs. 45 %) [42]. The full report on this study is not yet available and it is not clear whether improved adherence would continue beyond 6 months. This issue is important as the available information suggests that the modelling may be based on the assumption that the improved adherence associated with KIF6 genotype screening would continue for the duration of the model.
Recently, the claim that the KIF6 genotype is association with CVD risk and statin treatment effect size has been questioned [14, 43]. Although retrospective analyses of the CARE, WOSCOPS (West Of Scotland COronary Prevention Study) and PROSPER (PROspective Study of Pravastatin in the Elderly at Risk) trials have found that only carriers of the Trp719Arg allele receive benefit from pravastatin therapy [44–46], retrospective analyses of the HPS (Heart Protection Study), 4D (Deutsche Diabetes Dialyse Studie) and JUPITER (Justification for the Use of statins in Prevention: an Intervention Trial Evaluating Rosuvastatin) trials did not find a significant interaction between the KIF6 genotype and statin effectiveness [47–49]. Similarly, although the genetic sub-study of the PROVE IT-TIMI 22 study found that only carriers of the Trp719Arg allele gain additional benefit from using intensive statin therapy (atorvastatin 80 mg/day) over moderate statin therapy (pravastatin 40 mg/day) [40], this was not confirmed in the TNT (Treating to New Targets) [atorvastatin 80 mg/day vs. 10 mg/day] and IDEAL (Incremental Decrease in End points through Aggressive Lipid lowering) [high-dose atorvastatin vs. moderate simvastatin dose] studies [50].
2.5 Familial Hypercholesterolaemia
A number of studies [51–56] considered the cost effectiveness of genetic screening programmes in FH. A large proportion of individuals with FH have mutations, most commonly in the gene encoding the LDLR [57]. Both genetic testing and LDL cholesterol testing may be used for the diagnosis and screening of FH and individuals with FH are generally started on statin therapy due to their high CVD risk. Thus, screening for FH-causing mutations may impact on the decision to treat with statin therapy [58]. However, screening for FH-causing mutations is not classically thought to be pharmacogenetic because its primary purpose is to aid diagnosis of a disease (i.e. a diagnostic marker) and its influence on the selection of therapy is secondary [59]. Additionally, in contrast to the pharmacogenetic markers previously discussed, FH-causing mutations primarily act to modify prognosis (increase CVD risk) [59], but there is no evidence that they significantly modify the RR of clinical events with statin therapy.
In general, the cost-effectiveness studies have compared strategies to screen for affected relatives following identification of an index case of FH, and generally screening for the FH-causing mutations was estimated to be cost effective [52–54, 56]. The additional utility in this strategy is primarily derived from the identification of individuals who are at high CVD risk and will benefit from statin therapy and/or higher-intensity statin therapy.
3 Findings and Implications
In this section, we present our findings relating to the extent and nature of the evidence of association and cost effectiveness for statin pharmacogenetics; a statin pharmacogenetic marker that is a candidate for further research; and implications for future CEAs of screening for pharmacogenetic markers for statins.
3.1 Reproducibility of Evidence Supporting Pharmacogenetic Markers
A common issue identified in the CEAs reviewed related to the evidence supporting the assertion that the pharmacogenetic marker significantly influenced statin efficacy. Specifically, the pharmacogenetic marker effect size utilized was often found to be controversial when systematic reviews of the evidence were subsequently undertaken. This issue has more generally been identified by others [60, 61]. An analysis of biomarker studies found that highly cited biomarker studies frequently report larger effect sizes than those subsequently estimated in systematic reviews [60]. Optimistic early estimates of the biomarker effect size and methodological shortcomings result in hype that rarely translates into clinical practice [62, 63]. Similarly, it has been shown that significant between-study heterogeneity is common, and that the effect size found by the initial association studies is often greater than the effect size subsequently reported by replication studies [61]. Similar issues regarding the replication of initial findings have also recently been highlighted in the context of the pharmacogenetics of the antiplatelet effect of clopidogrel [64].
3.2 Current Literature on the Pharmacogenetics of Statins
To further understand and ensure we captured all pharmacogenetic CEAs in the statin field, we performed a search for known individual pharmacogenetic markers involved with statin efficacy or tolerability up until 2012 (Fig. 3). One hundred and twenty-eight published studies were identified (excluding reviews, in vitro and animal studies, FH trials without statin response reported, and Alzheimer’s disease-related studies). Although pharmacogenetic studies are classically considered to be those in which variation in DNA characteristics alter statin response [65], a wide range of studies have been carried out. The majority of trials investigated genetic influences on one or more plasma lipid fractions, for example, Bailey et al. [66] and Poduri et al. [67], but the interaction of genetic variation and statins has also been investigated for the following: coronary disease [68], coronary event [45] or coronary mortality risk [69]; atherosclerosis progression or regression [49]; plaque characteristics [70]; side effects [71]; plasma protein biomarker levels or activities [72]; and gene expression (messenger RNA [mRNA]) levels [73].
These same studies generated 265 ‘pharmacogenetic marker evaluations’ across a total of 62 genes (see Fig. 3). One or more pharmacogenetic markers could be evaluated in one study, for example, Chien et al. [74], Hamrefors et al. [75] and Trompet et al. [76], and one pharmacogenetic marker could be evaluated in multiple studies in one year, for example, Cerda et al. [73], Baptista et al. [77] and Davies et al. [78]. The number of pharmacogenetic markers evaluated under-represents the number of gene variants examined as some genes had significant amounts of variation. The markers evaluated most frequently were: APOE (38), CETP (20), LDLR (15), HMGCR (15), SLCO1B1 (12), KIF6 (12) and ACE (6). No two of these frequently evaluated makers were evaluated in the same study. This result is not surprising for the genes with pivotal roles in statin efficacy (APOE, CETP, LDLR and HMGCR), but the importance of the KIF6 and ACE genes is less straightforward to explain given their lack of clear involvement in statin or lipid metabolism pathways. Figure 4 presents the number of pharmacogenetic studies published over time for the markers that have been studied for cost effectiveness and for SLCO1B1 (see Sect. 3.3).
3.3 Potential of the SLCO1B1 Transporter
All of the cost-effectiveness studies to date have focused on genetic markers that may provide insight into the effectiveness of statin therapy. However, it is arguable that the statin pharmacogenetic marker with the strongest evidence is actually predictive of statin toxicity (specifically muscle toxicities such as muscle pain and degradation) rather than efficacy. These toxicities are most common for individuals using higher doses of statins, in particular high-dose simvastatin [79–81]. A polymorphism of the SLCO1B1 gene is thought to increase the risk of muscle toxicity, particularly for individuals taking high-dose simvastatin [18, 79, 82–84]. The impact of the SLCO1B1 genotype on adherence to statin therapy is also being explored, but the relationship is not yet clear [81, 82].
Although this is currently the most promising pharmacogenetic marker of statin therapy, the overall impact of genotyping SLCO1B1 prior to initiating statins may be relatively modest. The major risk of muscle toxicity that is identified by the SLCO1B1 genotype relates to the use of simvastatin 80 mg. However, it is likely that use of simvastatin 80 mg will become increasingly infrequent with time due to a recent recommendation by the US FDA to avoid the use of simvastatin 80 mg [80] and the recent availability of generic atorvastatin. It is possible that the SLCO1B1 genotype may be useful to inform the risk of myopathy with the use of simvastatin 40 mg [79]; however, both the clinical utility and the cost effectiveness of such an approach require further exploration. Given that approximately four individuals would need to be screened to identify one individual at higher risk of myopathy [79], it may well be less expensive and more convenient to simply start individuals on statin drugs/doses that are less likely to have muscle toxicities without undertaking genotyping.
3.4 Implications for Future CEAs of Screening for Pharmacogenetic Markers for Statins
Four sets of implications for future CEAs of statin pharmacogenetics were identified.
3.4.1 Model Structure
As in conventional economic evaluations of screening, it is important to include the best alternative treatment for patients identified by screening as being suboptimal responders to usual care. Furthermore, the effect of this alternative therapy, conditional on genotype, needs to be considered. Ezetimibe was identified as an alternative to statin therapy for patients who tested positive for the B1B1 genotype in the CETP genotype CEA [19]; however, it was implicitly assumed that the effect of ezetimibe derived from a study that did not provide the results of subgroup analyses by genotype would be appropriate as an estimate of effect for this subgroup.
Additionally, the information already used to refine treatment decisions (for example, lipid levels) [5] needs to be included in the model in order to assess the incremental impact of an additional piece of information (genotype). None of the three studies identified the incremental effect of an additional piece of (pharmacogenetic) information and instead implicitly assumed that, in the absence of screening for pharmacogenetic markers, there would be no information used to refine therapy [19–21]. In an extreme case, it is possible that the use of genotype as a guide to treatment decisions could be consistent with the treatment decisions made solely from information provided by changes in LDLs. In this case, there would be no incremental effect on treatment decisions of the additional pharmacogenetic marker information.
The possibility that the treatment strategies compared in the CEAs do not represent the majority of treatment options currently faced by clinicians needs to be considered. Consider the example of the screening for KIF6 [21]. The implications of prescribers having multiple treatment options in addition to the two specified in the model for the cost effectiveness of widespread use of KIF6 screening should be considered. For example, should the screening test be used if patients would otherwise have been started on a statin other than atorvastatin or pravastatin?
The analytical validity of genetic testing must be considered in the model structure [85]. If the commercially available test does not replicate the results of the test used in the trial, then the results of that trial are not generalizable more widely. If there are multiple commercial tests or multiple sites at which the test occurs, if commercial tests are not yet available or if there are rapid changes in the testing technologies, then there is reason to anticipate limited generalizability of study results.
3.4.2 The Economic Context of the Statin and Diagnostic Test Markets
An important issue identified with respect to the CEAs of statin pharmacogenetics was the cost of statin therapy. Most statin drugs are now off patent and statin costs are, or are soon expected to be, relatively low per person treated. At the time some of the CEAs were undertaken, the price of statins and the difference in price between statins were greater and thus there was a greater incentive to reduce the use of statins, or to maximize the use of lower-cost statins [19, 20].
In most cases, the price of a commercial test for the pharmacogenetic markers was not available in the public domain. In addition to a simple sensitivity analysis, considerations about the degree of competition in the market for that diagnostic test, and the implication for price, in particular, above marginal cost pricing must be considered. The additional costs of the infrastructure for wide uptake of pharmacogenetic testing for such a large group of patients should also be considered.
3.4.3 Current Utilization and Maturity of the Market
If the result of a CEA is that screening for a pharmacogenetic marker in a specific decision context is cost effective, these results should be qualified by the context of the current utilization and market. There are a number of issues that would qualify implementing pharmacogenetic screening as a policy or guideline recommendation, despite a favourable result from a CEA, in addition to those discussed previously.
A policy of screening for a pharmacogenetic marker would need to be accommodated in the context of current usage. Would a screening rule apply prospectively (for incident cases), or should patients who are currently being treated be screened also? How should prescribers review the quality of their patients’ care if there is a mixture of screened and unscreened patients? If there are many potential markers, and the current marker has an uncertain result, what are the implications for policy, best practice guidelines and prescriber education of changing the preferred pharmacogenetic marker or accommodating multiple pharmacogenetic markers?
Treatment algorithms are complex in large markets with multiple drugs in a class, patient variation in response and long-term use of therapies. The role of evidence of the cost effectiveness of pharmacogenetic screening in the context of a choice between two treatment options in informing a policy of screening when there are many treatment options currently available is unclear. Given that there do not appear to be any pharmacogenetic markers that conclusively predict response to statins, there does not appear to be justification to alter current guidelines or policy.
Adherence is an important issue in drugs for chronic conditions and the statin market is no exception. If genetic tests could improve adherence for patients, this would be a useful result; however, the implications of reduced adherence for patients who do not have a specific genotype also needs to be considered, as does the additional service use that could occur if that genotype has implications for other therapeutic decisions.
3.4.4 International Generalizability
Two aspects of international generalizability of these findings are relevant to this paper: the generalizability of the results of the three CEAs [19–21] and the findings of the review. In relation to the first aspect, the usual limitations of cross-country generalizability of the results of a CEA apply [86]. In the case of the CEAs of interest [19–21], drivers of cross-country differences include: the prevalence of the genotypes; the existing patterns of prescribing and the information used to guide dose and therapeutic choice; the relative prices of inputs; and the timing of drugs going off patent. Three further limitations apply: the differences in the capacity for countries to (1) implement a screening strategy, particularly if there is a risk that new markers could be identified, and (2) change screening and treatment decisions in a mature complex and heterogeneous market; and (3) the competition in the market for the test, which will in turn influence price and the capacity for the manufacturer of the test to appropriate the savings from reduced prescribing of statins [87]. In relation to the second point, the findings about the value of CEAs of pharmacogenetic-guided statin therapy are generalizable internationally: the fundamental issue is that these can only provide information about the cost effectiveness under assumptions about the therapy’s clinical value that cannot necessarily be supported by the data. The finding that it is unlikely for an effective or cost-effective pharmacogenetic marker to emerge in this area of prescribing at this stage of the market’s maturity is generalizable across countries, with the exception of a country that has a very limited range of statins used, very little variation in the way they are prescribed and an existing infrastructure for routine screening on a large scale.
4 Conclusion
Only a relatively small number of CEAs have studied the value of pharmacogenetic-guided treatment of hypercholesterolaemia. Of these CEAs, significant limitations were identified with respect to the reproducibility of the evidence for differential statin treatment effect in pharmacogenetic subgroups, uncertainty regarding the nature and value of alternative treatments available, and uncertainty regarding the incremental benefit over-and-above monitoring LDL cholesterol levels. Exploration of the cost effectiveness of SLCO1B1 for statin toxicity was identified as a future research direction.
References
Grundy SM, Cleeman JI, Merz CN, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines. J Am Coll Cardiol. 2004;44(3):720–32.
National Center for Health Statistics. Health, United States, 2010: with special feature on death and dying [DHHS Publication No. 2011-1232]. Hyattsville: US Department of Health and Human Services; 2011.
Department of Health and Ageing. Pharmaceutical Benefits Scheme (PBS): expenditure and prescriptions twelve months to 30 June 2011. Canberra: Australian Government. 2012. http://www.health.gov.au/internet/main/publishing.nsf/content/pbs-stats-pbexp-jun11. Accessed 19 Aug 2012.
Aitken M, Berndt ER, Cutler DM. Prescription drug spending trends in the United States: looking beyond the turning point. Health Aff. 2009;28(1):w151–60.
Reiner Z, Catapano AL, De Backer G, et al. ESC/EAS guidelines for the management of dyslipidaemias: the task force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS). Eur Heart J. 2011;32(14):1769–818.
Baigent C, Keech A, Kearney PM, et al. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet. 2005;366(9493):1267–78.
Mihaylova B, Emberson J, Blackwell L, et al. The effects of lowering LDL cholesterol with statin therapy in people at low risk of vascular disease: meta-analysis of individual data from 27 randomised trials. Lancet. 2012;380(9841):581–90.
Baigent C, Blackwell L, Emberson J, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376(9753):1670–81.
Laufs U, La Fata V, Plutzky J, et al. Upregulation of endothelial nitric oxide synthase by HMG CoA reductase inhibitors. Circulation. 1998;97(12):1129–35.
Mitsios JV, Papathanasiou AI, Goudevenos JA, et al. The antiplatelet and antithrombotic actions of statins. Curr Pharm Des. 2010;16(34):3808–14.
Jain MK, Ridker PM. Anti-inflammatory effects of statins: clinical evidence and basic mechanisms. Nat Rev Drug Discov. 2005;4(12):977–87.
Abela GS, Vedre A, Janoudi A, et al. Effect of statins on cholesterol crystallization and atherosclerotic plaque stabilization. Am J Cardiol. 2011;107(12):1710–7.
Shishehbor MH, Brennan ML, Aviles RJ, et al. Statins promote potent systemic antioxidant effects through specific inflammatory pathways. Circulation. 2003;108(4):426–31.
Superko HR, Momary KM, Li Y. Statins personalized. Med Clin N Am. 2012;96(1):123–39.
Guerin M, Egger P, Soudant C, et al. Dose-dependent action of atorvastatin in type IIB hyperlipidemia: preferential and progressive reduction of atherogenic apoB-containing lipoprotein subclasses (VLDL-2, IDL, small dense LDL) and stimulation of cellular cholesterol efflux. Atherosclerosis. 2002;163(2):287–96.
Niemi M. Transporter pharmacogenetics and statin toxicity. Clin Pharmacol Ther. 2010;87(1):130–3.
Romaine SPR, Bailey KM, Hall AS, et al. The influence of SLCO1B1 (OATP1B1) gene polymorphisms on response to statin therapy. Pharmacogenomics J. 2010;10(1):1–11.
Link E, Parish S, Armitage J, et al. SLCO1B1 variants and statin-induced myopathy: A Genomewide Study. N Engl J Med. 2008;359(8):789–99.
Kemp LK, Doran CM, Vos T, et al. Cost-effectiveness analysis of genetic screening for the Taq1B polymorphism in the secondary prevention of coronary heart disease. Expert Rev Pharmacoecon Outcomes Res. 2007;7(2):119–28.
Maitland-van der Zee AH, Klungel OH, Stricker BHC, et al. Pharmacoeconomic evaluation of testing for angiotensin-converting enzyme genotype before starting [beta]-hydroxy-[beta]-methylglutaryl coenzyme A reductase inhibitor therapy in men. Pharmacogenetics. 2004;14(1):53–60.
Parthan A, Iakoubova O, Leahy K, et al. Cost effectiveness of targeted statin therapy following genotype testing among acute coronary syndrome patients [abstract no. 1538 plus poster]. 16th World Congress on Heart Disease; 23–26 July 2011; Vancouver.
Doggrell SA. The ezetimibe controversy: can this be resolved by comparing the clinical trials with simvastatin and ezetimibe alone and together? Expert Opin Pharmacother. 2012;13(10):1469–80.
Carlquist JF, Muhlestein JB, Horne BD, et al. The cholesteryl ester transfer protein Taq1B gene polymorphism predicts clinical benefit of statin therapy in patients with significant coronary artery disease. Am Heart J. 2003;146(6):1007–14.
Kakko S, Tamminen M, Päivänsalo M, et al. Variation at the cholesteryl ester transfer protein gene in relation to plasma high density lipoproteins cholesterol levels and carotid intima-media thickness. Eur J Clin Invest. 2001;31(7):593–602.
Arca M, Montali A, Ombres D, et al. Lack of association of the common TaqIB polymorphism in the cholesteryl ester transfer protein gene with angiographically assessed coronary atherosclerosis. Clin Genet. 2001;60(5):374–80.
Eiriksdottir G, Bolla MK, Thorsson B, et al. The -629C>A polymorphism in the CETP gene does not explain the association of TaqIB polymorphism with risk and age of myocardial infarction in Icelandic men. Atherosclerosis. 2001;159(1):187–92.
Marschang P, Sandhofer A, Ritsch A, et al. Plasma cholesteryl ester transfer protein concentrations predict cardiovascular events in patients with coronary artery disease treated with pravastatin. J Intern Med. 2006;260(2):151–9.
Mohrschladt MF, Van der Sman-de Beer F, Hofman MK, et al. TaqIB polymorphism in CETP gene: the influence on incidence of cardiovascular disease in statin-treated patients with familial hypercholesterolemia. Eur J Hum Genet. 2005;13(7):877–82.
Kuivenhoven J, Jukema J, Zwinderman A, et al. The role of a common variant of the cholesteryl ester transfer protein gene in the progression of coronary atherosclerosis. The Regression Growth Evaluation Statin Study Group. N Engl J Med. 1998;338(2):86–93.
de Grooth GJ, Zerba KE, Huang SP, et al. The cholesteryl ester transfer protein (CETP) TaqIB polymorphism in the cholesterol and recurrent events study: no interaction with the response to pravastatin therapy and no effects on cardiovascular outcome—a prospective analysis of the CETP TaqIB polymorphism on cardiovascular outcome and interaction with cholesterol-lowering therapy. J Am Coll Cardiol. 2004;43(5):854–7.
Freeman DJ, Samani NJ, Wilson V, et al. A polymorphism of the cholesteryl ester transfer protein gene predicts cardiovascular events in non-smokers in the West of Scotland Coronary Prevention Study. Eur Heart J. 2003;24(20):1833–42.
Boekholdt SM, Sacks FM, Jukema JW, et al. Cholesteryl ester transfer protein TaqIB variant, high-density lipoprotein cholesterol levels, cardiovascular risk, and efficacy of pravastatin treatment. Circulation. 2005;111(3):278–87.
Dullaart RP, Sluiter WJ. Common variation in the CETP gene and the implications for cardiovascular disease and its treatment: an updated analysis. Pharmacogenomics. 2008;9(6):747–63.
Maitland-van der Zee AH, Stricker BHC, Klungela OH, et al. Effectiveness of HMG-CoA reductase inhibitors is modified by the ACE insertion deletion polymorphism. Atherosclerosis. 2004;175(2):377–9.
Marian AJ, Safavi F, Ferlic L, et al. Interactions between angiotensin-I converting enzyme insertion/deletion polymorphism and response of plasma lipids and coronary atherosclerosis to treatment with fluvastatin: the Lipoprotein and Coronary Atherosclerosis Study. J Am Coll Cardiol. 2000;35(1):89–95.
Bray PF, Cannon CP, Goldschmidt-Clermont P, et al. The platelet Pl (A2) and angiotensin-converting enzyme (ACE) D allele polymorphisms and the risk of recurrent events after acute myocardial infarction. Am J Cardiol. 2001;88:347–52.
Maitland-van der Zee A, Boerwinkle E, Arnett D, et al. Absence of an interaction between the angiotensin-converting enzyme insertion-deletion polymorphism and pravastatin on cardiovascular disease in high-risk hypertensive patients: the Genetics of Hypertension-Associated Treatment (GenHAT) study. Am Heart J. 2007;153(1):54–8.
Kitsios G, Zintzaras E. ACE (I/D) polymorphism and response to treatment in coronary artery disease: a comprehensive database and meta-analysis involving study quality evaluation. BMC Med Genet. 2009;10:50.
Cannon CP, Braunwald E, McCabe CH, et al. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med. 2004;350(15):1495–504.
Iakoubova O, Sabatine M, Rowland CM, et al. Polymorphism in KIF6 gene and benefit from statins after acute coronary syndromes: results from the PROVE IT-TIMI 22 Study. J Am Coll Cardiol. 2008;51(4):449–55.
Osterberg L, Blaschke T. Adherence to medication. N Engl J Med. 2005;353(5):487–97.
Charland SL, Agatep BC, Schrader BJ, et al. Statin adherence in males and females, and the impact of knowledge of a genetic test: results from the AKROBATS Trial [abstract no. 290 plus poster]. Circ Cardiovasc Qual Outcomes. 2012;5:A290.
Assimes TL, Hólm H, Kathiresan S, et al. Lack of association between the Trp719Arg polymorphism in kinesin-like protein-6 and coronary artery disease in 19 case-control studies. J Am Coll Cardiol. 2010;56(19):1552–63.
Iakoubova OA, Tong CH, Rowland CM, et al. Association of the Trp719Arg polymorphism in kinesin-like protein 6 with myocardial infarction and coronary heart disease in 2 prospective trials: the CARE and WOSCOPS trials. J Am Coll Cardiol. 2008;51(4):435–43.
Iakoubova OA, Robertson M, Tong CH, et al. KIF6 Trp719Arg polymorphism and the effect of statin therapy in elderly patients: results from the PROSPER study. Eur J Cardiovasc Prev Rehabil. 2010;17(4):455–61.
Shiffman D, Sabatine MS, Louie JZ, et al. Effect of pravastatin therapy on coronary events in carriers of the KIF6 719Arg allele from the cholesterol and recurrent events trial. Am J Cardiol. 2010;105(9):1300–5.
Hopewell JC, Parish S, Clarke R, et al. No impact of KIF6 genotype on vascular risk and statin response among 18,348 randomized patients in the Heart Protection Study. J Am Coll Cardiol. 2011;57(20):2000–7.
Ridker PM, MacFadyen JG, Glynn RJ, et al. Kinesin-like protein 6 (KIF6) polymorphism and the efficacy of rosuvastatin in primary prevention. Circ Cardiovasc Genet. 2011;4(3):312–7.
Hoffmann MM, Marz W, Genser B, et al. Lack of association between the Trp719Arg polymorphism in kinesin-like protein-6 and cardiovascular risk and efficacy of atorvastatin among subjects with diabetes on dialysis: the 4D study. Atherosclerosis. 2011;219(2):659–62.
Arsenault BJ, Boekholdt SM, Hovingh GK, et al. The 719Arg variant of KIF6 and cardiovascular outcomes in statin-treated, stable coronary patients of the treating to new targets and incremental decrease in end points through aggressive lipid-lowering prospective studies. Circ Cardiovasc Genet. 2012;5(1):51–7.
Marks D, Thorogood M, Neil HAW, et al. Comparing costs and benefits over a 10 year period of strategies for familial hypercholesterolaemia screening. J Public Health. 2003;25(1):47–52.
Marks D, Wonderling D, Thorogood M, et al. Cost effectiveness analysis of different approaches of screening for familial hypercholesterolaemia. BMJ. 2002;324(7349):1303.
Wonderling D, Umans-Eckenhausen MA, Marks D, et al. Cost-effectiveness analysis of the genetic screening program for familial hypercholesterolemia in The Netherlands. Semin Vasc Med. 2004;4(1):97–104.
Marang-van de Mheen P, ten Asbroek A, Bonneux L, et al. Cost-effectiveness of a family and DNA based screening programme on familial hypercholesterolaemia in The Netherlands. Eur Heart J. 2002;23(24):1922–30.
Oliva J, Lopez-Bastida J, Moreno SG, et al. Cost-effectiveness analysis of a genetic screening program in the close relatives of Spanish patients with familial hypercholesterolemia [in Spanish]. Rev Esp Cardiol. 2009;62(1):57–65.
Nherera L, Marks D, Minhas R, et al. Probabilistic cost-effectiveness analysis of cascade screening for familial hypercholesterolaemia using alternative diagnostic and identification strategies. Heart. 2011;97(14):1175–81.
Minhas R, Humphries SE, Qureshi N, et al. Controversies in familial hypercholesterolaemia: recommendations of the NICE Guideline Development Group for the identification and management of familial hypercholesterolaemia. Heart. 2009;95(7):584–7.
Vergopoulos A, Knoblauch H, Schuster H. DNA testing for familial hypercholesterolemia: improving disease recognition and patient care. Am J Pharmacogenomics. 2002;2(4):253–62.
Humphries SE, Neil HAW. Developing and applying clinically useful approaches to identify individuals with familial hypercholesterolemia in the UK. Clin Lipidol. 2010;5(4):497–507.
Ioannidis J, Panagiotou O. Comparison of effect sizes associated with biomarkers reported in highly cited individual articles and in subsequent meta-analyses. JAMA. 2011;305(21):2200–10.
Ioannidis JPA, Ntzani EE, Trikalinos TA, et al. Replication validity of genetic association studies. Nat Genet. 2001;29(3):306–9.
Bossuyt PM. The thin line between hope and hype in biomarker research. JAMA. 2011;305(21):2229–30.
Veenstra DL, Higashi MK, Phillips KA. Assessing the cost-effectiveness of pharmacogenomics. AAPS PharmSci. 2000;2(3):80–90.
Talameh JA, McLeod HL. PON1 Q192R and clopidogrel: a case of the winner’s curse or inadequate replication? Clin Pharmacol Ther. 2011;90(6):771–4.
US Food and Drug Administration. Guidance for industry: E15 definitions for genomic biomarkers, pharmacogenomics, pharmacogenetics, genomic data and sample coding categories. Rockville: FDA. 2008. http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM073162.pdf. Accessed 14 Aug 2012.
Bailey KM, Romaine SP, Jackson BM, et al. Hepatic metabolism and transporter gene variants enhance response to rosuvastatin in patients with acute myocardial infarction: the GEOSTAT-1 Study. Circ Cardiovasc Genet. 2010;3(3):276–85.
Poduri A, Khullar M, Bahl A, et al. Common variants of HMGCR, CETP, APOAI, ABCB1, CYP3A4, and CYP7A1 genes as predictors of lipid-lowering response to atorvastatin therapy. DNA Cell Biol. 2010;29(10):629–37.
Maitland-van der Zee A-H, Lynch A, Boerwinkle E, et al. Interactions between the single nucleotide polymorphisms in the homocysteine pathway (MTHFR 677C>T, MTHFR 1298 A>C, and CBSins) and the efficacy of HMG-CoA reductase inhibitors in preventing cardiovascular disease in high-risk patients of hypertension: the GenHAT study. Pharmacogenet Genomics. 2008;18(8):651–6.
Chiodini BD, Franzosi MG, Barlera S, et al. Apolipoprotein E polymorphisms influence effect of pravastatin on survival after myocardial infarction in a Mediterranean population: the GISSI-Prevenzione study. Eur Heart J. 2007;28(16):1977–83.
Kral A, Kovarnik T, Kralik L, et al. Genetic variants in haem oxygenase-1 and endothelial nitric oxide synthase influence the extent and evolution of coronary artery atherosclerosis. Folia Biol (Praha). 2011;57(5):182–90.
Marciante KD, Durda JP, Heckbert SR, et al. Cerivastatin, genetic variants, and the risk of rhabdomyolysis. Pharmacogenet Genomics. 2011;21(5):280–8.
Dubuc G, Tremblay M, Pare G, et al. A new method for measurement of total plasma PCSK9: clinical applications. J Lipid Res. 2010;51(1):140–9.
Cerda A, Genvigir FDV, Willrich MAV, et al. Apolipoprotein E mRNA expression in mononuclear cells from normolipidemic and hypercholesterolemic individuals treated with atorvastatin. Lipids Health Dis. 2011;10:206.
Chien K-L, Wang K-C, Chen Y-C, et al. Common sequence variants in pharmacodynamic and pharmacokinetic pathway-related genes conferring LDL cholesterol response to statins. Pharmacogenomics. 2010;11(3):309–17.
Hamrefors V, Orho-Melander M, Krauss RM, et al. A gene score of nine LDL and HDL regulating genes is associated with fluvastatin-induced cholesterol changes in women. J Lipid Res. 2010;51(3):625–34.
Trompet S, de Craen AJM, Postmus I, et al. Replication of LDL GWAs hits in PROSPER/PHASE as validation for future (pharmaco) genetic analyses. BMC Med Genet. 2011;12:131.
Baptista R, Rebelo M, Decq-Mota J, et al. Apolipoprotein E epsilon-4 polymorphism is associated with younger age at referral to a lipidology clinic and a poorer response to lipid-lowering therapy. Lipids Health Dis. 2011;10:48.
Davies NM, Windmeijer F, Martin RM, et al. Use of genotype frequencies in medicated groups to investigate prescribing practice: APOE and statins as a proof of principle. Clin Chem. 2011;57(3):502–10.
Wilke RA, Ramsey LB, Johnson SG, et al. The clinical pharmacogenomics implementation consortium: CPIC Guideline for SLCO1B1 and simvastatin-induced myopathy. Clin Pharmacol Ther. 2012;92:112–7.
US Food and Drug Administration. FDA drug safety communication: new restrictions, contraindications, and dose limitations for Zocor (simvastatin) to reduce the risk of muscle injury. Rockville: FDA. 2011. http://www.fda.gov/Drugs/DrugSafety/ucm256581.htm. Accessed 19 Aug 2012.
Donnelly L, Doney A, Tavendale R, et al. Common nonsynonymous substitutions in SLCO1B1 predispose to statin intolerance in routinely treated individuals with type 2 diabetes: a Go-DARTS study. Clin Pharmacol Ther. 2011;89(2):210–6.
Voora D, Shah SH, Spasojevic I, et al. The SLCO1B1*5 genetic variant is associated with statin-induced side effects. J Am Coll Cardiol. 2009;54(17):1609–16.
Puccetti L, Ciani F, Auteri A. Genetic involvement in statins induced myopathy. Preliminary data from an observational case-control study. Atherosclerosis. 2010;211(1):28–9.
Brunham LR, Lansberg PJ, Zhang L, et al. Differential effect of the rs4149056 variant in SLCO1B1 on myopathy associated with simvastatin and atorvastatin. Pharmacogenomics J. 2012;12(3):233–7.
Teutsch SM, Bradley LA, Palomaki GE, et al. The evaluation of genomic applications in practice and prevention (EGAPP) initiative: methods of the EGAPP working group. Genet Med. 2009;11(1):3–14.
Drummond M, Manca A, Sculpher M. Increasing the generalizability of economic evaluations: Recommendations for the design, analysis, and reporting of studies. Int J Technol Assess Health Care. 2005;21(02):165–71.
Davis JC, Furstenthal L, Desai AA, et al. The microeconomics of personalized medicine: today’s challenge and tomorrow’s promise. Nat Rev Drug Discov. 2009;8(4):279–86.
Acknowledgments
This work was supported by the National Heart Foundation of Australia [G11A5902]. The authors gratefully acknowledge the assistance of Sunjay Pekarsky-Norman in the preparation of Fig. 3. Michael Sorich and Brita Pekarsky are members of the Economics Sub-Committee of the Australian Pharmaceutical Benefits Advisory Committee, but the views expressed in this article are solely those of the authors. Rebekah O’Shea was employed by Bayer Australia Ltd until November 2009. All authors contributed to the conception and planning of the manuscript, drafting and critical revision of the manuscript, and approval of the final submitted version of the manuscript. Michael Sorich acts as guarantor for the overall content of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sorich, M.J., Wiese, M.D., O’Shea, R.L. et al. Review of the Cost Effectiveness of Pharmacogenetic-Guided Treatment of Hypercholesterolaemia. PharmacoEconomics 31, 377–391 (2013). https://doi.org/10.1007/s40273-013-0045-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40273-013-0045-6