Abstract
Background
The prescription of antidepressant drugs during pregnancy has been steadily increasing for several decades. Meta-analyses (MAs), which increase the statistical power and precision of results, have gained interest for assessing the safety of antidepressant drugs during pregnancy.
Objective
We aimed to provide a meta-review of MAs assessing the benefits and risks of antidepressant drug use during pregnancy.
Methods
Following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, a literature search on PubMed and Web of Science databases was conducted on 25 October, 2021, on MAs assessing the association between antidepressant drug use during pregnancy and health outcomes for the pregnant women, embryo, fetus, newborn, and developing child. Study selection and data extraction were carried out independently and in duplicate by two authors. The methodological quality of included studies was evaluated with the AMSTAR-2 tool. Overlap among MAs was assessed by calculating the corrected covered area. Data were presented in a narrative synthesis, using four levels of evidence.
Results
Fifty-one MAs were included, all but one assessing risks. These provided evidence for a significant increase in the risks for major congenital malformations (selective serotonin reuptake inhibitors, paroxetine, fluoxetine, no evidence for sertraline; eight MAs), congenital heart defects (paroxetine, fluoxetine, sertraline; 11 MAs), preterm birth (eight MAs), neonatal adaptation symptoms (eight MAs), and persistent pulmonary hypertension of the newborn (three MAs). There was limited evidence (only one MA for each outcome) for a significant increase in the risks for postpartum hemorrhage, and with a high risk of bias, for stillbirth, impaired motor development, and intellectual disability. There was inconclusive evidence, i.e., discrepant results, for an increase in the risks for spontaneous abortion, small for gestational age and low birthweight, respiratory distress, convulsions, feeding problems, and for a subsequent risk for autism with an early antidepressant drug exposure. Finally, MAs provided no evidence for an increase in the risks for gestational hypertension, preeclampsia, and for a subsequent risk for attention-deficit/hyperactivity disorder. Only one MA assessed benefits, providing limited evidence for preventing relapse in severe or recurrent depression. Effect sizes were small, except for neonatal symptoms (small to large). Results were based on MAs in which overall methodological quality was low (AMSTAR-2 score = 54.8% ± 12.9%, [19–81%]), with a high risk of bias, notably indication bias. The corrected covered area was 3.27%, which corresponds to a slight overlap.
Conclusions
This meta-review has implications for clinical practice and future research. First, these results suggest that antidepressant drugs should be used as a second-line treatment during pregnancy (after first-line psychotherapy, according to the guidelines). The risk of major congenital malformations could be prevented by observing guidelines that discourage the use of paroxetine and fluoxetine. Second, to decrease heterogeneity and bias, future MAs should adjust for maternal psychiatric disorders and antidepressant drug dosage, and perform analyses by timing of exposure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
This meta-review of 51 meta-analyses on the benefits and risks of antidepressants during pregnancy, all but one assessing risks, found evidence for an increase in the risks for major congenital malformation, congenital heart defects, preterm birth, neonatal adaptation symptoms, and persistent pulmonary hypertension of the newborn. There was limited evidence for an increase in the risks for postpartum hemorrhage, and with a high risk of bias, for stillbirth, impaired motor development, and intellectual disability. There was also limited evidence for preventing relapse in severe or recurrent depression. |
Meta-analyses provided inconclusive evidence for an increase in the risks for spontaneous abortion, small for gestational age and low birthweight, respiratory distress, convulsions, feeding problems, and for a subsequent risk for autism with an early antidepressant exposure. There was no evidence for gestational hypertension, preeclampsia, and for subsequent risks for attention-deficit/hyperactivity disorder. |
These results suggest that anti-depressant drugs should be used as a second-line treatment during pregnancy (after first-line psychotherapy, according to the guidelines). Future meta-analyses should attempt to decrease heterogeneity and bias. |
1 Introduction
The prescription of antidepressant drugs (ADs) during pregnancy has been steadily increasing for several decades. The international prevalence of selective serotonin reuptake inhibitor (SSRI) prescription during pregnancy is actually estimated at around 3% [1], with about 6–6.5% in the USA [2,3,4], 2.3–3.3% in Europe [5, 6], 1.9% in Japan [7], and 1% in China [8]. It is followed by serotonin and norepinephrine reuptake inhibitors (SNRIs) and tricyclic antidepressants (TCAs), at almost 1% and 0.5%, respectively [1]. Antenatal depression is the most common major psychiatric disorder during pregnancy, with an estimated worldwide prevalence exceeding 20% [9] (17% in Western countries [10]), and the leading indication for AD prescriptions [11], which are mostly recommended for severe forms or after first-line psychotherapy [12].
However, because of safety concerns, approximately half of women stop taking ADs during the pre-pregnancy period or the first trimester of pregnancy [1, 13], leading to a risk of relapse of depression. Therefore, assessing the benefits and risks of AD use during pregnancy is a major issue. Numerous studies have been conducted, evidencing outcomes for the pregnant women (i.e., preeclampsia, hemorrhage, and depression relapse after AD discontinuation), the embryo and the fetus in early pregnancy (i.e., spontaneous abortions, birth defects) and in late pregnancy (i.e., preterm birth, low birth weight), the neonate (i.e., withdrawal symptoms, persistent pulmonary hypertension of the newborn), and the developing child (autistic spectrum disorder, attention-deficit without or with hyperactivity disorder, impaired cognitive and motor development) [14,15,16]; these studies mainly focused on adverse drug reactions, showing small absolute risks, with many conflicting results. In this context, the use of meta-analyses (MAs), which increase the statistical power and precision of results, has gained interest for assessing the safety of AD prescriptions with respect to pregnant women, the fetus, and the newborn [17].
As a result, MAs currently represent a significant portion of the literature on AD prescription during pregnancy [14, 18], which can be synthesized through meta-reviewing. The meta-review method indeed allows the synthesizing of evidence from multiple MAs on a given topic into a single available document, and hence tends to be considered the highest level of evidence [19].
We conducted a meta-review of MAs evaluating the benefits and risks associated with AD use during pregnancy. We focused on outcomes for the pregnant woman, the fetus during the first and second half of pregnancy, the newborn, and the offspring. The outcomes assessed were those identified in the included MAs.
2 Methods
We used both the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [20], and the Hennessy, Johnson, and Keenan (2019)’s Guidelines and Essential Methodological Steps to Conduct Rigorous and Systematic Meta-Reviews [21], the latter to address specific issues (i.e., datedness, overlap, quality assessment, and data synthesis). The review was not registered on any database, and a review protocol was not published.
2.1 Literature Source and Search Strategy
A literature search was conducted on PubMed and Web of Science databases on 25 October, 2021. To prevent outdatedness [21], only MAs published since 2010 were included. Keywords used were Medical Subject Heading (MeSH) terms combined with Boolean terms: “antidepressant AND pregnancy AND meta-analysis”.
Inclusion criteria were: (a) MAs of randomized controlled trials, case-control studies, cohort studies, or population-based studies, published in English, and peer reviewed and (b) data on the association between AD use during pregnancy and health outcomes for the pregnant women, or between in utero AD exposure and health outcomes for the embryo, fetus, newborn, or developing child.
Exclusion criteria were: (a) studies other than MAs; (b) no data on the association between AD use by pregnant women or in utero AD exposure and health outcomes; (c) period of AD use outside of pregnancy; and (d) when after requesting a full text, we received no reply within 3 weeks.
2.2 Screening and Data Extraction
Study selection and data extraction were carried out independently and in duplicate by the first two authors (PD and LGE), using an Excel datasheet. Extracted information was consistent with the Participant, Intervention, Comparison, Outcome, and Study (PICOS) design framework [22]: indication of ADs, total number of included studies in MAs, trimester of prescription, classes of ADs or molecules studied, type of comparison group and adjustments, outcomes (i.e., benefit or adverse drug reaction), type of included studies in MAs, main results (i.e., effect size [ES], mean difference [MD], odds ratio [OR], relative risk [RR], or hazard ratio [HR], and their 95% confidence interval [reported in brackets in the text and tables] without and with adjustment), and heterogeneity (I2 values of 25%, 50%, and 75% corresponded to low, medium, and high heterogeneity, respectively [23]). Outcomes were selected a priori, then adapted to those of the included MAs, and subdivided into five categories: (a) outcomes for women of AD use during pregnancy; (b) embryo or fetal outcomes during the first half of pregnancy, of in utero AD exposure; (c) fetal outcomes during the second half of pregnancy, of in utero AD exposure; (d) outcomes for the newborn of in utero AD exposure; and (e) subsequent outcomes for the offspring of in utero AD exposure. Discrepancies were resolved by mutual agreement.
2.3 Quality Assessment of Included MAs
The methodological quality of each MA was assessed by the first author using the AMSTAR-2 tool [24]. AMSTAR-2 is a 16-item questionnaire, seven of which assess critical domains, validated for meta-reviews [21].
2.4 Overlap Among Included MAs
We calculated the corrected covered area (CCA) to assess overlap: \(\mathrm{CCA}=\frac{N - r}{rc - r}\) , where N is the sum of the number of primary studies in each MA, r is the total number of primary studies, and c is the number of MAs. The CCA was interpreted as: slight (0–5%), moderate (6–10%), high (11–15%), or very high (> 15%) overlap [21, 25, 26].
2.5 Data Synthesis
Data were presented in a narrative synthesis [21], using four levels of evidence [27]: (a) evidence: consistent positive significant effects on a specific outcome in at least one MA (based on at least two underlying effect studies); (b) limited evidence: effects on a specific outcome in at least one MA (based on one underlying effect study); (c) inconclusive evidence: inconsistent effects on a specific outcome because at least one MA (including at least two underlying studies) shows positive significant effects, while other MAs included did not find such effects; and (d) no evidence: none of the included MAs reported significant effects on a specific outcome. This method was chosen retrospectively, as the included MAs consisted mostly of non-randomized controlled trials.
3 Results
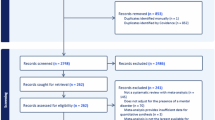
The initial search generated 134 records, of which 51 MAs from different countries were eligible for the meta-review (Fig. 1, and Table S1 of the Electronic Supplementary Material [ESM]). Control groups, apart from specificities, consisted of pregnant women without AD use or embryos/fetuses/offspring without in utero exposure to ADs. Only one MA reported twin or multiple pregnancies as exclusion criteria [28].
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart [20] of study selection in the systematic meta-review. AD antidepressant drug, MA meta-analysis
3.1 MAs Assessing Outcomes for Women of AD Use During Pregnancy
Three MAs respectively assessed the risks for depression relapse associated with AD discontinuation [29], gestational hypertension (hypertension onset after 20 weeks’ gestation) and preeclampsia (hypertension and proteinuria) [30], and postpartum hemorrhage (blood loss ≥ 500 mL after vaginal delivery or ≥ 1000 mL after cesarean delivery) [31], associated with AD use.
3.1.1 Depression Relapse Following AD Discontinuation
One MA showed that the risk for depression relapse during pregnancy did not significantly differ between women who discontinued ADs during the anteconceptional period (i.e., 3 months prior to pregnancy) or the first trimester of pregnancy, versus continued ADs [29] (Table S1a of the ESM). Subgroup analyses identified that this non-significant increase in the risk was related to mild-to-moderate non-recurrent depression (n = 3 included studies), whereas the increase in the risk was significant for severe depression, with a medium RR, and only one included study [29].
3.1.2 Gestational Hypertension and Preeclampsia
One MA on SSRIs reported a significantly increased overall risk for gestational hypertension or preeclampsia, with a small RR, and high heterogeneity, persisting after adjustments for main risk factors (i.e., maternal age, anteconceptional body mass index, parity, diabetes mellitus, and smoking) [30] [Table S1b of the ESM]. The increase was no longer significant after adjustment for a psychiatric history or ethnicity. Subgroup analyses showed non-significant increases in the risks for gestational hypertension and for preeclampsia.
3.1.3 Postpartum Hemorrhage
One MA reported a significant increase in the risk for postpartum hemorrhage with AD use during pregnancy, with a small RR and high heterogeneity [31] (Table S1c of the ESM). This increase persisted after adjustment for psychiatric history, and with current AD use, or use during the last month of pregnancy, and was non-significant when ADs were discontinued at least 1 month before birth. This increase was higher for SNRIs than for both SSRIs and non-serotonin reuptake inhibitors, and also higher for a cesarean birth than for a vaginal delivery (no statistical comparisons were performed) [31].
3.2 MAs Assessing Fetal Outcomes During the First Half of Pregnancy, of In Utero AD Exposure
3.2.1 Spontaneous Abortion
The risk for spontaneous abortion, defined as the unwanted expulsion of the fetus before viability (i.e., weighing less than 500 g or expelled before 22 weeks of gestation), and occurring in 10% of known pregnancies [32], was assessed in 3 MAs [33,34,35] (Table S1d of the ESM).
Two MAs reported an increased risk with any classes of ADs, with small-to-medium RRs, and with high heterogeneity, including when data were confined to studies conducted in the first trimester [33], and with SSRIs with a medium OR (n = 7, of which six studies focused on the first-trimester exposure only) [34]. The increase was significant for most ADs prescribed during pregnancy, with the exception of bupropion, i.e., citalopram, escitalopram, fluoxetine, paroxetine, sertraline, and venlafaxine [33]. One MA found non-significant results (n = 3, with one study focusing on the first-trimester exposure only) [35].
3.2.2 Major Congenital Malformations
The malformation risk was assessed in 17 MAs, which distinguished between major congenital malformations (i.e., non-cardiac) [Table S1e of the ESM] and congenital heart defects (Table S1f of the ESM) [34, 36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51]. Major congenital malformations refer to any structural defect present at birth, requiring surgical, medical, or cosmetic intervention, or having a functional impact on social acceptability [51], with a global birth prevalence estimated at 2% [52]. The teratogenic effect of ADs, like any other teratogenic agent, occurs during the first 3 months of development [53].
The two MAs that considered any classes of ADs found no significant increase in the risk for major congenital malformations [36, 51], whatever the type of study design (i.e., prospective and retrospective cohort studies, case-control studies) [36]. Considering SSRIs, four MAs showed a significantly increased risk, with small RRs and ORs [34, 43, 44], including for SSRIs co-prescribed with benzodiazepines [45]. The increase was no longer significant in the only MA that controlled for maternal psychiatric history [44]. This later MA showed in subgroup analyses, without this adjustment, increased risks with small-to-medium RRs, for neural tube defects, abdominal wall defects, and gastroschisis, but no increase for other major malformation other than the heart (i.e., ear, face and neck, urinary, digestive, musculoskeletal, and respiratory systems, hypospadias, limb) [44]. One MA focused specifically on anorectal malformations, and reported no elevation in the relative risk [46].
Four of the five MAs that focused on paroxetine identified a significantly increased risk for major congenital malformation, with small RRs and ORs [43, 44, 47, 48]; one MA found non-significant results [51]. The increase persisted in the only MA that controlled for depression [48].
Similarly, four of the five MAs that focused on fluoxetine identified a significantly increased risk, with small RRs and ORs [43, 44, 49, 51]; one MA found non-significant results [50]. In subgroup analyses, the increase was not significant for each major malformation other than the heart (i.e., eye and nervous, urogenital, digestive, respiratory, and musculoskeletal systems) [49]. All three MAs that focused on sertraline found no evidence of an increased risk for major congenital malformations [43, 44], including organs other than the heart (i.e., eye, ear, face, neck, and nervous, urogenital, digestive, and musculoskeletal systems) [37].
Of the two MAs focusing on citalopram, one reported a significantly increased risk for major congenital malformation with a small RR [44], and the other did not [43]. Finally, one MA concluded that there was no increase in malformation risk with bupropion [38], an atypical AD with wide indications (i.e., adult depression, seasonal affective disorder, depression associated with bipolar disorder, anti-depressant-induced sexual dysfunction, smoking cessation, attention-deficit/hyperactivity disorder [ADHD], and obesity) [54].
3.2.3 Congenital Heart Defects
The global birth prevalence of congenital heart defects is around 1%, consisting mainly of ventricular septal defects [55]. Considering any classes of ADs, two MAs reported a significant increase in the risk for congenital heart defects with small RRs and ORs [39, 51], including septal defects [51], regardless of the study design, i.e., case-control studies, prospective cohort studies, and retrospective cohort studies [39]. Another MA reported non-significant results with both prospective retrospective cohort studies and case-control studies [36]. Among classes of ADs, an increase in the risk has been found with SSRIs and SNRIs, but not with TCAs [39].
Meta-analyses focusing on SSRIs have shown conflicting results. Four MAs reported a significant elevation in the risk for congenital heart defects with small RRs and ORs [39, 40, 44], or only with the most severe cases, with a positive effect–dose relationship [41]. Three MAs showed non-significant results [34, 42, 43]. The increase in the risk persisted after adjustment for relevant confounding factors [40, 41], and became non-significant after adjustment for smoking [41] or a psychiatric history [44]. Subgroup analyses indicated an increase in the risk of septal defects including atrial [44], and right ventricular outflow tract obstruction [40, 44].
Six of the seven MAs that focused on paroxetine reported significant results with small RRs and ORs [39, 43, 44, 47, 51], including without and with adjustment for depression [48]. One MA reported non-significant results [42]. Subgroup analyses showed an increase in the risk for both atrial septal defects and right ventricular outflow defects with medium ORs [48].
With fluoxetine, four of seven MAs reported a significantly increased risk with small RRs and ORs [39, 44, 49, 50], and three did not [42, 43, 51]. Subgroup analyses showed an increase in the risk for septal defects [44, 49], especially atrial [44], and non-septal defects [49], with small-to-medium RRs.
Examining sertraline, three MAs found significant results with small RRs and ORs [37, 39, 44], and two MAs reported non-significant results [42, 43]. In subgroup analyses, this risk was increased for septal defects [37, 44], especially atrial [44], with small-to-medium RRs.
Four MAs evaluated citalopram, among which one reported significant results with a small RR [44], and three others did not [39, 42, 43]. A MA reported a significant result for bupropion with a small OR, but not for venlafaxine [39].
3.3 MAs Assessing Fetal Outcomes During the Second Half of Pregnancy, of in Utero AD Exposure
3.3.1 Preterm Birth
Nine MAs assessed the risk for preterm birth (i.e., birth before 37 weeks of gestation) associated with in utero AD exposure; all of which statistically controlled for several relevant confounding factors [28, 33, 35, 56,57,58,59,60,61] (Table S1g of the ESM). All MAs that considered any class of AD identified a significantly increased risk for preterm birth, with small RRs and ORs and with high heterogeneity, for in utero exposure at any time of pregnancy [28, 33, 35, 56, 57], during the first [33], second, and third trimesters [33], and the third trimester with a medium OR [56]. Anti-depressant drug use would reduce the duration of pregnancy by an average of − 0.45 week ([− 0.64 to − 0.25], n = 15) [35]. The type of study design did not influence the results as all were significant, i.e., overall study designs, retrospective studies, and prospective studies [58]. When ADs were prescribed for depression, the increase in the risk has been shown to be significant [57], or non-significant [28]. Four of these MAs adjusted for depression, of which the increased risk persisted in three MAs [33, 56, 58], and became non-significant in one MA [35].
Examining AD classes, three MAs reported an overall increased risk with SSRIs, with small RRs and ORs, and with medium-to-high heterogeneity [28, 58, 59]. This increase persisted after adjustment for confounders [59], and was higher than with non-SSRIs [58]. This increase was higher for late than for early pregnancy exposure (third trimester vs first trimester: OR = 4.17 [2.75–6.30], n = 3) [59], and persisted after adjustment for depression [59]. Two MAs reported non-significant results [57, 60]. When SSRIs were prescribed for depression, the increase was significant [57] or non-significant [28]. With SSRIs or SNRIs combined, a MA reported an increased risk, without and with adjustment for depression, with a significant reduction in the duration of pregnancy, on average − 0.47 week [− 0.74 to − 0.21], becoming non-significant after adjustment for depression [61]. An increased risk has been shown with TCAs and SNRIs [33].
Focusing on each SSRI, an increase in the risk for preterm birth was reported with citalopram, paroxetine, and sertraline, with small RRs, but not with fluoxetine [33]. For bupropion, a MA showed a mean gestational age at delivery of 39.2 weeks, i.e., birth at term [38].
3.3.2 Small for Gestational Age and Low Birth Weight
Seven MAs addressed the risks for small for gestational age and low birth weight [28, 33, 35, 38, 58, 61, 62] (Table S1h of the ESM), which correspond respectively to an estimated fetal weight below the tenth percentile, and a birth weight < 2500 g, and display a global prevalence of around 25% and 15%, respectively [63]. Considering any class of AD, two MAs found no increase in the risk for small for gestational age [28, 33], of which one identified instead an increased risk for large for gestational age with a small RR [33]. Regarding low birth weight, two MAs reported a significantly increased risk with small RRs and medium-to-large heterogeneity [33, 58], including after adjustment for depression [33, 58]; one MA showed a non-significant increased risk [28]. The type of study design seemed to influence results, as being significant with retrospective studies, and non-significant with prospective studies [58]. Anti-depressant drug use would reduce the mean birth weight by an average of − 74 g [− 117 to − 31], which was no longer significant after controlling for maternal depression [35].
Examining AD classes, one MA concluded, for SSRIs or SNRIs combined, that there were significant increases in the risks for small for gestational age and low birth weight with small ORs, with a significant difference in birth weight, on average − 69.75 g [− 115.51 to − 23.99], becoming non-significant after adjustment for depression [61]. This MA showed a non-significant increase in the risk for large for gestational age [61]. Two MAs that focused on SSRIs reported significantly increased risks for small for gestational age with a small RR and medium heterogeneity [62], and for low birth weight with small RRs and medium heterogeneity [58, 62]. One MA found no increase in these risks, whether SSRIs were prescribed for depression or all indications [28]. The type of study design also seemed to influence results, as being consistent for small for gestational age (i.e., retrospective and prospective cohort studies), and discordant for low birth weight (i.e., significant with retrospective cohort studies, and non-significant for prospective studies) [62]. With non-SSRIs, a MA reported a non-significantly increased risk for small for gestational age [58]. Finally, a MA found a significant increased risk low birth weight with TCAs, and with SNRIs, with small RRs [33].
Focusing on each SSRI, an increase in the risk for low birth weight was reported with citalopram, paroxetine, and sertraline, with small RRs, but not with fluoxetine [33]. For bupropion, an MA showed a mean birth weight in the normal range [38].
3.3.3 Stillbirth
Two MAs assessed the risk of stillbirth [33, 41] (Table S1i of the ESM), defined as intrauterine death after 22 weeks of gestation, the prevalence of which is estimated at 1.17% in developing countries and 0.5% in more developed countries [64]. The first MA assessed the composite adverse outcome of a congenital anomaly or stillbirth, which was significantly increased for SSRIs with a small OR, including after adjustments for smoking, and for socioeconomic status, with a positive effect–dose relationship [41]. The second MA identified a significantly increased risk for stillbirth with any class of AD, with a small RR, being no longer significant in subgroup analyses for citalopram and fluoxetine [33].
3.4 MAs Assessing the Outcomes for the Newborn of in Utero AD Exposure
3.4.1 Neonatal Adaptation Symptoms
The main neonatal complication of in utero AD exposure is the poor neonatal adaptation syndrome, which consists of mild clinical signs of serotonin withdrawal (mainly hyper-hypotonus, respiratory distress, hyperreflexia, restlessness, irritability, and tremor). As it has no consensus definition, this symptom and its syndromes have been found from 25 to 75% of exposed newborns [65,66,67]. Neonatal adaptation syndrome and symptoms were assessed in eight MAs [28, 33, 35, 60, 61, 68,69,70] (Table S1j of the ESM).
The high prevalence of the poor neonatal adaptation syndrome was confirmed by one MA identifying a significant and large increase for this risk, with all AD classes [68]. The increase persisted after adjustment for co-prescription and substance use, and late exposure [68].
Four MAs reported an increase in the risk for neonatal respiratory symptoms, with medium-to-large ORs. These showed an increased risk for respiratory distress with all AD classes (including after adjustment for confounders, and late exposure) [68], SSRIs (including after adjustment for depression) [60], TCAs [60], SSRIs, and venlafaxine [69]. These also showed an increased risk for rapid breathing, with SSRIs and venlafaxine [69], and for respiratory problems, with SSRIs or SNRIs, including after adjustment for depression [61]. The increase in the risk for respiratory distress was non-significant in two MAs, with any classes of ADs [33], and with SSRIs [69].
Four MAs identified an increase in the risk for neuromuscular symptoms, with large ORs. These included tremor, with all AD classes [68], and with SSRIs and venlafaxine, tremor, hypertonia, and hypotonia [69].
For neurologic signs, three MAs reported an increased risk for neonatal convulsions with medium-to-large RRs and ORs and high heterogeneity [33, 61, 70], being higher for late exposure than for early exposure (no statistical comparisons were performed) [70]. One MA found no increase in this risk [69].
Two MAs reported increased risks for autonomic nervous system dysfunctions with SSRIs or SNRIs, with medium-to-large RRs and ORs. These included feeding problems [61], hypoglycemia [61, 69], jaundice [61], temperature dysregulation [61], and tachycardia [69]. The increase in the risk for feeding problems was non-significant in one MA [33].
Results were discordant regarding Apgar scores. For the 1′ Apgar score, two MAs reported a significantly decreased score, with any classes (MD = − 0.19 [− 0.30 to − 0.08], n = 10) [35], and with SSRIs or SNRIs (MD = − 0.38 [− 0.68 to − 0.08], n = 10) [61]. Two MAs showed a significantly increased risk for score ≤ 7, with SSRIs [69], and with SSRIs or SNRIs [61], becoming non-significant after adjustment for depression [61]. One MA showed a non-significant risk [33]. For the 5′ Apgar score, two MAs reported a significantly decreased score, with any classes (MD = − 0.33 [− 0.47 to − 0.20], n = 14) [35], and with SSRIs or SNRIs after adjustment for depression (MD = −0.32 [− 0.54 to − 0.11], n = 3) [61]. Three MAs identified a significantly increased risk for a score ≤ 7 with small-to-medium RRs and ORs, with any classes [28, 33], SSRIs [28], SSRIs, or SNRIs [61]. The increase in the risk for a score ≤ 7 was non-significant in one MA [69], and when ADs were indicated for depression [28], and after adjustment for depression [61]. Finally, two MAs identified an increased risk for neonatal intensive care unit admissions with small-to-medium RRs and ORs, both without and with adjustment for depression [33, 61].
3.4.2 Persistent Pulmonary Hypertension in the Newborn
This relatively rare (about 0.2% of newborns) but serious condition (a leading cause of neonatal morbidity and mortality, responsible for reduced pulmonary blood flow) [71], was assessed in three MAs [72,73,74] (Table S1k of the ESM). These MAs, focusing on SSRIs [72, 74], SSRIs, and SNRIs [73], have shown an increased risk for in utero exposure at any time in pregnancy, [72, 73], including after adjustment for main confounders [73], and during most or all of pregnancy [74], and late pregnancy [73, 74], all with medium-to-large ORs. This increase was non-significant for an exposure limited to early pregnancy and at any time in pregnancy in one MA [74]. The absolute risk difference has been estimated at 0.619 per 1000 livebirths and a number needed to harm of 1615 women [72]. Among the network’s specific comparison, these risks were significantly lower for sertraline than for fluoxetine (OR = 0.34 [0.11–0.96], n = 5) [73].
3.5 MAs Assessing the Subsequent Outcomes for the Offspring of In Utero ADs
3.5.1 Autism Spectrum Disorder (ASD)
The risk for ASD, which has an estimated worldwide prevalence of around 1% [75, 76], was evaluated in 12 MAs [77,78,79,80,81,82,83,84,85,86,87,88] (Table S1l of the ESM). The four MAs that investigated exposure to any AD at any time of pregnancy identified a significantly increased risk for subsequent ASD, with small-to-medium ORs, RR, and HR [77, 78, 81, 82]. Three of these MAs investigated exposure during the first trimester of pregnancy, and all showed a significant risk for ASD, with small-to-medium ORs and RR [78, 81, 82]. Two of the MAs investigated exposure during the second trimester and during the third trimester. Both found a significant risk regarding the second trimester, with small-to-medium ORs and RR [78, 81]. Contradictory findings were observed regarding the third trimester, i.e., significant with a medium OR [78], and non-significant [81].
Six MAs focused on SSRIs, and all reported an increased risk for subsequent ASD for an exposure at any time of pregnancy, with small-to-medium ORs and RR [82,83,84,85,86,87]. A network MA among SSRIs reported no significant difference in ASD risk among any of the comparison pairs [88].
Three MAs have looked at study designs, by distinguishing between case-control and cohort studies [78,79,80]. Two of them [78, 79] considered all ADs, and reported significant results with case-control studies for an exposure at any time of pregnancy, with small-to-medium ORs [78, 79]. In contrast, all results in cohort studies were non-significant, for an AD exposure at any time of pregnancy [78, 79]. The two MAs that focused on SSRIs reported significant results with case-control studies for an exposure at any time of pregnancy, with small-to-medium ORs [79, 80]. Regarding cohort studies of SSRIs, these two MAs showed contradictory conclusions for an exposure at any time of pregnancy, i.e., significant with a small HR [80], and non-significant [79].
The fact that the link between AD use during pregnancy and subsequent ASD in the offspring is stronger in case-control studies than cohort studies suggests the existence of confounding biases in available studies, among which the indication bias seems particularly probable: as pregnant women taking an AD usually have a mental disorder, primarily depression, this factor may need to be statistically controlled for [89]. Five of the 12 MAs considered provided results without and with statistical adjustment for a maternal psychiatric history: three for all mental disorders [79, 82, 85], one for affective disorders [81], and one specifically for depression [78]. For any AD at any time of pregnancy, initially significant results disappeared after adjustment in two MAs [81, 82]; the OR decreased but stayed significant in the remaining MA with a small OR [78], which focused only on case-control studies, and a concomitant MA focusing on cohort studies and adjusted for maternal depression showed non-significant results [78]. The same MA looked at the different trimesters of pregnancy within case-control studies: it found a decreased but still significant risk for ASD regarding the first trimester and the second trimester, with small-to-medium ORs, and the risk became non-significant for the third trimester [78]. Another of the three MAs on any AD [82] investigated the risk for ASD with an exposure during the first trimester, which remained significant, with a small OR. Three MAs investigated the same topic specifically in SSRIs [79, 82, 85], all of which reported non-significant results for an exposure at any time of pregnancy [79, 82, 85]. Finally, three MAs performed sibling-matched comparisons, which control for time-stable shared factors including maternal genetics and neuropsychiatric traits [90]: for an AD exposure at any time of pregnancy, all OR/RRs were non-significant [79, 81, 82]; for a first-trimester exposure, results were non-significant for SSRIs, and significant for any AD (OR = 0.62 [0.40–0.96], n = 1), with OR < 1 indicating a protective effect, and only one included study [82].
3.5.2 ADHD
The risk for ADHD, which prevalence averages 5% worldwide [91], associated with in utero AD exposure was assessed in five MAs [80, 81, 88, 92, 93] (Table S1m of the ESM). The three MAs on any ADs reported an increase in the risk for subsequent ADHD for an exposure at any time of pregnancy, with small RRs [81, 92, 93]. These three MAs investigated exposure during the first trimester of pregnancy, and all showed a significant risk for ADHD, with small RRs [81, 92, 93]. Of the two MAs that specifically investigated second- and third-trimester AD exposures [81, 93], one reported significant results with a second-trimester exposure with a small RR [93]. The remaining MA investigated the second and third trimesters of pregnancy merged, and found a significant risk for ADHD, with a small RR [92].
Focusing on SSRIs, a MA reported a significant result for an exposure at any time of pregnancy, with a small RR [92]. With case-control studies, a MA reported significant results with first-trimester exposure with a small-to-medium OR, and non-significant with exposures at any of pregnancy, second trimester, and third trimester [80]. With cohort studies, the same MA reported significant results with exposures at any time of pregnancy or during the first trimester, with small HRs, and non-significant results during second/third trimesters [80]. For SSRIs and SNRIs merged, a MA found an increased risk for an exposure at any time of pregnancy, with a small OR [88].
One MA adjusted for maternal psychiatric history [92], within which the initially significant results regarding any ADs at any time of pregnancy became non-significant [92]. Finally, three MAs performed sibling-matched comparisons regarding any ADs at any time of pregnancy, and all reported non-significant results [81, 92, 93].
3.5.3 Impaired Motor Development
One MA assessed the association between in utero AD exposure and subsequent impaired motor development (included studies reported a measure of motor performance, in children from birth to 9 years of age), identifying a significant and positive association, with a small ES [94] (Table S1n of the ESM).
3.5.4 Intellectual Disability
One MA reported a significantly increased risk for intellectual disability in offspring after in utero exposure to SSRIs, with a small HR [80] (Table S1o of the ESM). However, authors reported that a confounding by indication bias was identified in the included studies.
3.6 Methodological Quality
The mean AMSTAR-2 percentage score was 54.8% (standard deviation 12.9%), ranking from 19 to 81% (Figs. 2 and 3). Based on the AMSTAR-2 critical domains (Figs. 2 and 3, and Table S2 of the ESM), the methodological quality of reviewed MAs was considered low (i.e., one critical flaw with or without non-critical weaknesses), or very low (i.e., more than one critical flaw with or without non-critical weaknesses).
Methodological quality of the 51 meta-analyses (MAs) according to the 16 items of A MeaSurement Tool to Assess systematic Reviews 2nd version (AMSTAR-2) [for reasons of quality, the original questions have been reduced to some extent]. AMSTAR-2 critical domains are marked with an asterisk (*). PICO Participant, Intervention, Comparison, Outcome
3.7 Overlap
The corrected covered area was 3.27%, which corresponds to a slight overlap (0–5%) [21, 25, 26].
3.8 Data Synthesis
A summary of evidence for outcomes assessed in this meta-review is provided in Table 1.
4 Discussion
Of the 51 MAs reviewed, all but one assessed risks associated with AD use during pregnancy for pregnant and parturient women, fetuses, newborns, and offspring. The remaining MA assessed the risk of depression relapse associated with AD discontinuation.
Of the MAs that addressed risks for pregnant and parturient women, one reported with SSRIs, non-significant results for gestational hypertension and preeclampsia [30], pointing to no evidence for these risks. As gestational hypertension has been reported with untreated depression [95,96,97], and with prolonged SNRI or TCA use [98, 99], further MAs could assess these AD classes, controlling for use duration and for depression. For parturient women, a single MA identified an increased risk for postpartum hemorrhage with SSRIs, SNRIs, non-serotonin reuptake inhibitors, with small ESs, being higher with SNRIs than with SSRIs [31]. This single MA therefore provided limited evidence for this risk. Pathophysiology may involve an AD-induced decrease in intraplatelet serotonin, and a hypertensive effect regarding SNRIs [100].
Meta-anlayses assessing the fetal outcomes occurring in the first half of pregnancy focused on spontaneous abortion, major congenital malformations, and congenital heart defects. Two of the three MAs reported an increased risk for spontaneous abortion with any AD class[33, 34], with high heterogeneity [34], and one MA did not [35], therefore pointing to inconclusive evidence. None controlled for depression, which is a risk factor of spontaneous abortion [101], through elevated pro-inflammatory factors and cortisol, and decreased immunity [101, 102], resulting in a confounding bias by indication, which further MAs should control.
For major congenital malformations, an increased risk with small ESs and low heterogeneity was reported in all MAs with SSRIs [34, 43,44,45], most of the MAs with paroxetine [43, 44, 47, 48], and fluoxetine [43, 44, 49, 51], one of the two MAs on citalopram [44]. This indicated evidence for SSRIs, paroxetine, and fluoxetine, inconclusive evidence for citalopram, and no evidence for sertraline [37, 43, 44]. Teratogenicity could involve an imbalance in the concentration of serotonin, as it acts as an organ growth factor in the fetus [103], and safety with sertraline may involve its low placental transfer to the fetus [104]. Effect size became non-significant in the only MA with SSRIs that adjusted for depression [44], which may imply overlapping risk factors between congenital malformations and depression (e.g., as substance use [105, 106], or overweight and obesity [107, 108]), which should be assessed in subsequent MAs.
For congenital heart defects, an increased risk with small ESs and low-to-medium heterogeneity was identified in most of MAs with paroxetine [39, 43, 44, 47, 48, 51], fluoxetine [39, 44, 49, 50], and sertraline [37, 39, 44], in two of the three MAs with any AD class [39, 51], four of the seven MAs with SSRIs [39,40,41, 44], one of the four MAs with citalopram [44], in the only MA with SNRIs [39] and bupropion [39], and not in the only MA with TCAs [39]. This indicated evidence for paroxetine, fluoxetine, and sertraline, inconclusive evidence for any AD class, SSRIs, and citalopram, and limited evidence for SNRIs and bupropion, and no evidence for TCAs. Atrial septal defects were the most commonly reported heart defects [44, 48], which usually evolve to spontaneous closure or regression; exceptionally, safe surgery can be considered [109]. The lack of significance in the only MA adjusted for depression [44] also suggests overlapping risk factors. Pathogenicity may entail altered cardiac morphogenesis, disruption of laterality in heart cells, or abnormal intracardiac blood flow [110]. Heterogeneity of these results suggests risks with some individual ADs, paroxetine, fluoxetine, and sertraline, which subsequent MAs should further assess.
Meta-analyses assessing the fetal outcomes during the second half of pregnancy, of in utero AD exposure, focused on preterm birth, small for gestational age and low birth weight, and stillbirth. Most or all of MAs reported an increased risk for preterm birth, with any AD class [28, 33, 35, 56,57,58], SSRIs [57,58,59], SSRIs and SNRIs [61], and non-SSRIs [33, 59], with small ESs and medium-to-high heterogeneity. This pointed to consistent evidence. The increase persisted after adjustment for depression with small-to-medium ESs [33, 56, 58], and was higher for late than for early pregnancy exposure with SSRIs [59]. However, a possible indication bias cannot be completely ruled out. An MA evidenced, in subgroup analyses limited to pregnant women with depression, a significant increase in the risk for preterm birth in those not receiving ADs, and a non-significant increase in those receiving ADs [28]. Another MA showed a significantly increased risk for preterm birth with SSRIs when indicated for depression, while non-significant with all indications combined [57]. Indeed, uncontrolled antenatal depression is a risk factor for prematurity and low birth weight [111, 112], through an increased synthesis of corticotrophin-releasing hormone from the placenta initiating premature labor, and a high risk of inflammation or infection [112]. The mean reduction in the pregnancy duration was estimated at half a week, unadjusted [28, 35]—a result close to that reported in a review with untreated antenatal depression [113]—suggesting a low frequency of prematurity associated with AD exposure.
For fetal and birth weight outcomes, discrepant results were reported with medium-to-high heterogeneity, i.e., increased risks for small for gestational age [61, 62] and large for gestational age [33], diminished mean birth weight [35, 61] and low birth weight [33, 58, 61, 62], persisting [33, 58] or not [35, 61] after adjustment for depression. This indicated inconclusive evidence. Unmeasured indication biases can be discussed. An MA also reported increased risks for small for gestational age and low birth weight in depressed pregnant women without AD use [28]. A review concluded a similar risk level for birth weight between treated and untreated antenatal depression [113]. Antenatal depression may result in fetal growth restriction through elevated cortisol inducing high fetal caloric expenditure, and reduced oxygen and nutrient delivery to the fetus by vasoconstriction of uterine arteries [114].
One MA concluded an increased risk for stillbirth [33], including seven small cohort studies [115,116,117,118,119,120,121], and two population-based studies [122, 123]. The first study, involving more than 1 million births, identified a similar rate of stillbirth (i.e., 0.4%) in pregnant women with AD use, and in untreated and undepressed pregnant women [122]. The second study, involving more than 1.6 million births, identified a higher rate of stillbirths in pregnant women with versus without SSRI use (0.462% vs 0.369%), becoming non-significant after adjustment for a history of psychiatric hospitalization [123]. Prior psychiatric hospitalization, possibly a marker of depression severity or recurrence, may be the risk factor of stillbirth rather than AD use, and given the lack of adjustment, this MA [33] provided limited evidence, with a high risk of bias.
Considering neonatal adaptation symptoms, MAs provided: evidence for tremor with a high ES [68, 69], hypoglycemia [61, 69], decreased 1’ and 5′ Apgar scores [35, 61], and neonatal intensive care unit admissions [33, 61]; limited evidence for rapid breathing [69], respiratory problems [61], hypertonia [69], hypotonia [69], jaundice [61], temperature dysregulation [61], and tachycardia [69]. These also provided inconclusive evidence for respiratory distress [33, 60, 68, 69], neonatal convulsions [33, 61, 69, 70], feeding problems [33, 61], and low 1’ and 5′ Apgar scores [28, 33, 61, 69]. A single MA on the neonatal adaptation syndrome provided limited evidence, but with a large ES and low heterogeneity [68]. This mild and transient condition, involving serotonin withdrawal, evolves favorably with supportive care [124], although long-term neurodevelopmental outcomes have been suspected [125]. All three MAs on persistent pulmonary hypertension of the newborn reported an increase in the risk, with late pregnancy exposure to SSRIs or SNRIs, with small ESs and high heterogeneity [72,73,74], providing consistent evidence. Prognosis of this syndrome depends on its cause, and lacks evaluation for in utero AD exposure. Pathophysiology may include serotonin-induced pulmonary vasoconstriction and smooth cell proliferation [126].
Meta-analyses assessing the subsequent outcomes for the offspring focused on ASD, ADHD, impaired motor development, and intellectual disability. Though an increased risk for ASD with small ESs was initially found in all MAs [77,78,79,80,81,82,83,84,85,86,87,88], this risk became non-significant for an exposure to any AD or SSRI at any time of pregnancy in all MAs that controlled for a maternal psychiatric history [78, 79, 81, 82, 85]. However, in two of them investigating any AD [78, 82], an increased risk with a small ES remained regarding the first [82] or the two first [78] trimesters of pregnancy, and a decreased risk was shown in a sibling-matched comparison regarding the first trimester [82], but with only one study included. Therefore, these results provide no evidence for an increase in the risk for subsequent ASD with in utero AD exposure at any time of pregnancy, and inconclusive evidence with AD exposure during early pregnancy. On the one hand, an association with early exposure remains plausible on the physiopathological level, as serotonin acts on brain maturation from the beginning of prenatal life [127, 128]. On the other hand, insufficient control of confusion bias is also possible, as pregnant women’s stress during early pregnancy is a known risk factor for ASD [129]. Further studies are needed using sibling-matched comparisons, i.e., taking into account a potential indication bias and other time-stable confounding factors.
Although an increased risk for ADHD with small ESs was initially found in all MAs [80, 81, 88, 92, 93], this risk became non-significant in the MA that controlled for a maternal psychiatric history [92], and in the three MAs that performed sibling-matched comparisons [81, 92, 93]. It can therefore be said that there is no reliable evidence for an increased risk for subsequent ADHD in the offspring after in utero AD exposure. None of these MAs screened or adjusted for ADHD in mothers or fathers, although this disorder is highly heritable [130] and associated with depression in adults [131], which may have resulted in a confounding by indication bias. In addition, it must be stressed that no MA provided sound results regarding the different trimesters of pregnancy. Thus, further cohort studies and MAs adjusting for parental diagnosis of ADHD, and individualizing the risk for ADHD by trimester of pregnancy, in particular the first trimester, would be useful.
Finally, a MA reported an association between in utero AD exposure with subsequent impaired motor development in children [94]. However, there was no adjustment for depression during pregnancy, which is a known risk factor for poor psychomotor developmental outcomes in offspring [132], resulting in a limited evidence with a high risk of bias. One MA also reported an association with subsequent intellectual disability, with a small ES, but only two studies were included, and each entailed an indication bias [80], which points to limited evidence, with a high risk of bias, for this risk.
Assessing benefits, a MA showed a risk for depression relapse during pregnancy that was no higher for women with mild-to-moderate non-recurrent depression who discontinued ADs during the anteconception period or pregnancy, than for those who continued their use [29]. As half of women discontinue AD during the preconception period or the first trimester, especially those with the fewest psychiatric comorbidities [1, 13], this result is to be considered as reassuring. This MA also evidenced a significant risk of relapse after AD discontinuation with severe or recurrent depression, but with only one study included. Therefore, these results show, with limited evidence, a benefit of AD use during pregnancy for treating severe or recurrent depression. Clinicians should advise their pregnant or intended-pregnant patients to continue ADs in these forms of depression. Moreover, AD discontinuation during pregnancy is also a risk factor for postpartum depression [133], with potential long-term negative outcomes on both maternal health and child development [134, 135]. This single result [29] hence provides limited insight on the benefits of AD prescriptions in pregnant women, and points to the need for further evaluations of AD effectiveness during pregnancy.
This meta-review has several limitations. First, there was no information on co-prescriptions, especially benzodiazepines that are frequent used during pregnancy [13] and could induce similar neonatal risks [136], nor information on dosage, which prevents any analysis of the dose–effect relationship, nor on adherence, although it is greatly reduced with ADs during pregnancy [137]. Second, as it is unethical to conduct randomized controlled trials with ADs during pregnancy, the revised MAs mostly involved non-randomized studies. Third, moderate-to-high heterogeneity was present in many of the reviewed MAs, which suggests uncontrolled biases, including selection, classification, publication, and confusion biases. Selection bias could entail the definition of AD exposure (the exact duration of in utero AD exposure is not controlled), the definition of the event evaluated (some MAs reported inconsistent definitions between included studies), and the methodologies used (the type of study included, e.g., cohort or case-control studies, being poorly controlled). Different classes of ADs or different molecules were pooled in MAs, which makes it impossible to differentiate their specific risks, and also induces a selection bias. In utero AD exposure in most studies is based on prescriptions, and may be overestimated because of the indistinction between adherent and non-adherent pregnant women, which creates a classification bias. Some MAs included only a small number of articles, suggesting publication bias, which should be better assessed. The most important bias could be confounding by indication, resulting from poorly measured (co-prescriptions), unmeasured (intensity and level of control of depression), or unknown (genetic and environmental factors) variables. Few MAs have adjusted for depression and none has adjusted for the level of control of depression, while insufficiently controlled antenatal depression may expose the pregnant woman and the fetus to the consequences of both residual depressive symptoms and the AD. Similarly, confounding by indication (i.e., the possibility that the indication of the AD is the cause of the risk, rather than the AD itself) was inadequately controlled for in the revised MAs, even though antenatal depression is a known risk factor for obstetric and neonatal complications [138]. The quality of revised MAs was low to moderate, using the AMSTAR-2 tool [24], which is a similar pattern to that identified by another meta-review assessing ADs [139]. Fourth, although this meta-review examined a large amount of information covering a large number of outcomes, it aimed to provide a comprehensive synthesis on the efficacy and safety of antidepressants prescribed during pregnancy, in order to inform clinicians, and to identify areas needing further research.
4.1 Implications for Clinicians
Consistent evidence highlighted in this meta-review relates to the risks for fetuses and neonates, i.e., major congenital malformations (SSRIs, paroxetine, and fluoxetine; no evidence for sertraline), congenital heart defects (paroxetine, fluoxetine, and sertraline), preterm birth with a high risk of bias, tremor, hypoglycemia, decreased 1’ and 5′ Apgar scores, neonatal intensive care unit admissions, and pulmonary hypertension of the newborn. Atrial septal defects—the most commonly reported heart defects [44, 48]—and tremor are mild conditions [65, 66, 109, 140], but others are potentially serious. These results are however limited by the scarcity of data specifying the indication for ADs, and using an appropriate comparison group, which leads to a potential confounding bias by indication. There are also limited by unmeasured variables such as maternal psychopathology and the environment, contributing to residual confounding. By contrast, there was only limited evidence for the benefits of AD use, i.e., to prevent a relapse of severe or resistant depression during pregnancy [29]. There was however no data on the risk of depression relapse postpartum, following AD discontinuation during pregnancy.
Together, these results strengthen the guidelines indicating first-line psychotherapy for mild-to-moderate depression, and ADs for severe depression [12]. The risk of major congenital malformations could be prevented by observing guidelines that discourage the use of paroxetine and fluoxetine, and prefer sertraline [12]; in the case of paroxetine use in early pregnancy, fetal echocardiography should be performed [141]. Obstetricians and pediatricians should be informed of AD use. Pregnant women using ADs should give birth in hospitals with a neonatal intensive care unit available, if possible.
4.2 Implications for Research
This meta-review also has implications for research, particularly in attempting to decrease heterogeneity and bias in MAs. To better control for indication bias, future MAs should adjust for maternal psychiatric disorders and distinguish between depression and other indications, and, if possible, adjust on the level of control of the disorders. To better control for selection bias, MAs should perform analyses by trimester, to better match the period of AD use with the period of occurrence of risks, and to better assess neurodevelopmental risks in the offspring. Limited evidence for the risks for postpartum hemorrhage, stillbirth, subsequent impaired motor development and intellectual disability in offspring, and for benefits, highlights the lack of studies and MAs, which calls for more evaluations of these outcomes. Finally, inconclusive evidence for the risks for spontaneous abortion, fetal and birth weight outcomes, and ASD with early AD exposure suggests an uncontrolled bias that future studies and MAs should better assess.
5 Conclusions
This meta-review primarily highlights the risks of AD use during pregnancy, with consistent evidence for fetuses and neonates, and limited evidence for parturient women. It also highlights limited (with a high risk of bias), or inconclusive, or no evidence, for neurodevelopmental disorders in the offspring. There was only limited evidence for a benefit of AD use for pregnant women. These results strengthen guidelines that recommend ADs for severe depression during pregnancy, and also highlight the need for further research, particularly to reduce heterogeneity and bias in risk assessments, and to evaluate the efficacy of ADs in pregnant women.
References
Molenaar NM, Bais B, Lambregtse-van den Berg MP, et al. The international prevalence of antidepressant use before, during, and after pregnancy: a systematic review and meta-analysis of timing, type of prescriptions and geographical variability. J Affect Disord. 2020;264:82–9. https://doi.org/10.1016/j.jad.2019.12.014.
Andrade SE, Reichman ME, Mott K, et al. Use of selective serotonin reuptake inhibitors (SSRIs) in women delivering liveborn infants and other women of child-bearing age within the U.S. Food and Drug Administration’s Mini-Sentinel program. Arch Womens Ment Health. 2016;19(6):969–77. https://doi.org/10.1007/s00737-016-0637-1.
Cooper WO, Willy ME, Pont SJ, Ray WA. Increasing use of antidepressants in pregnancy. Am J Obstet Gynecol. 2007;196(6):544.e1-5. https://doi.org/10.1016/j.ajog.2007.01.033.
Andrade SE, Raebel MA, Brown J, et al. Use of antidepressant medications during pregnancy: a multisite study. Am J Obstet Gynecol. 2008;198(2):194.e1-5. https://doi.org/10.1016/j.ajog.2007.07.036.
Charlton R, Jordan S, Pierini A, et al. Selective serotonin reuptake inhibitor prescribing before, during and after pregnancy: a population-based study in six European regions. BJOG An Int J Obstet Gynaecol. 2015;122(7):1010–20.
Zoega H, Kieler H, Nørgaard M, et al. Use of SSRI and SNRI antidepressants during pregnancy: a population-based study from Denmark, Iceland, Norway and Sweden. PLoS ONE. 2015;10(12):e0144474. https://doi.org/10.1371/journal.pone.0144474.
Ishikawa T, Obara T, Kikuchi S, et al. Antidepressant prescriptions for prenatal and postpartum women in Japan: a health administrative database study. J Affect Disord. 2020;264:295–303. https://doi.org/10.1016/j.jad.2020.01.016.
Hung C, Chan JKN, Wong CSM, Fung VSC, Lee KCK, Chang WC. Antidepressant utilization patterns and predictors of treatment continuation in pregnant women: a 16-year population-based cohort. Aust N Z J Psychiatry. 2022. https://doi.org/10.1177/00048674221109443.
Yin X, Sun N, Jiang N, et al. Prevalence and associated factors of antenatal depression: systematic reviews and meta-analyses. Clin Psychol Rev. 2021;83:101932. https://doi.org/10.1016/j.cpr.2020.101932.
Underwood L, Waldie K, Souza SD, Peterson ER, Morton S. A review of longitudinal studies on antenatal and postnatal depression. Arch Womens Ment Health. 2016;19(5):711–20. https://doi.org/10.1007/s00737-016-0629-1.
Schwalm M, Miotti H, Hellard C, Bounit L, Trehony J, Jouaville S. Treatment indications for antidepressants prescribed in primary care in France, 2006–2015. Value Health. 2016;19(7):A529. https://doi.org/10.1016/j.jval.2016.09.1062.
Molenaar NM, Kamperman AM, Boyce P, Bergink V. Guidelines on treatment of perinatal depression with antidepressants: an international review. Aust N Z J Psychiatry. 2018;52(4):320–7. https://doi.org/10.1177/0004867418762057.
Cabaillot A, Bourset A, Mulliez A, et al. Trajectories of antidepressant drugs during pregnancy: a cohort study from a community-based sample. Br J Clin Pharmacol. 2021;87(3):965–87. https://doi.org/10.1111/bcp.14449.
Fischer Fumeaux CJ, Morisod Harari M, Weisskopf E, et al. Risk-benefit balance assessment of SSRI antidepressant use during pregnancy and lactation based on best available evidence: an update. Expert Opin Drug Saf. 2019;18(10):949–63. https://doi.org/10.1080/14740338.2019.1658740.
Bellantuono C, Vargas M, Mandarelli G, Nardi B, Martini MG. The safety of serotonin-noradrenaline reuptake inhibitors (SNRIs) in pregnancy and breastfeeding: a comprehensive review. Hum Psychopharmacol Clin Exp. 2015;30(3):143–51. https://doi.org/10.1002/hup.2473.
Gentile S. Tricyclic antidepressants in pregnancy and puerperium. Expert Opin Drug Saf. 2014;13(2):207–25. https://doi.org/10.1517/14740338.2014.869582.
Biffi A, Cantarutti A, Rea F, Locatelli A, Zanini R, Corrao G. Use of antidepressants during pregnancy and neonatal outcomes: an umbrella review of meta-analyses of observational studies. J Psychiatr Res. 2020;124:99–108. https://doi.org/10.1016/j.jpsychires.2020.02.023.
Ornoy A, Koren G. SSRIs and SNRIs (SRI) in pregnancy: effects on the course of pregnancy and the offspring: how far are we from having all the answers? Int J Mol Sci. 2019;20(10):2370. https://doi.org/10.3390/ijms20102370.
Biondi-Zoccai G. Umbrella reviews. In: Evidence synthesis with overviews of reviews and meta-epidemiologic studies. Cham: Springer International; 2016. https://doi.org/10.1007/978-3-319-25655-9.
Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. https://doi.org/10.1136/bmj.n71.
Hennessy EA, Johnson BT, Keenan C. Best practice guidelines and essential methodological steps to conduct rigorous and systematic meta-reviews. Appl Psychol Health Well Being. 2019;11(3):353–81. https://doi.org/10.1111/aphw.12169.
Linde K. Systematic reviews and metaanalyses. In: Clinical research in complementary therapies. Churchill Livingstone; 2002, p. 187–97. https://doi.org/10.1016/B978-0-443-06367-1.50015-6.
Higgins JPT, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539–58. https://doi.org/10.1002/sim.1186.
Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. https://doi.org/10.1136/bmj.j4008.
Hennessy EA, Johnson BT. Examining overlap of included studies in meta-reviews: guidance for using the corrected covered area index. Res Synth Methods. 2020;11(1):134–45. https://doi.org/10.1002/jrsm.1390.
Pieper D, Antoine S-L, Mathes T, Neugebauer EAM, Eikermann M. Systematic review finds overlapping reviews were not mentioned in every other overview. J Clin Epidemiol. 2014;67(4):368–75. https://doi.org/10.1016/j.jclinepi.2013.11.007.
Huis in het Veld JG, Verkaik R, Mistiaen P, van Meijel B, Francke AL. The effectiveness of interventions in supporting self-management of informal caregivers of people with dementia; a systematic meta review. BMC Geriatr. 2015;15(1):147. https://doi.org/10.1186/s12877-015-0145-6.
Vlenterie R, van Gelder MMHJ, Anderson HR, et al. Associations between maternal depression, antidepressant use during pregnancy, and adverse pregnancy outcomes. Obstet Gynecol. 2021;138(4):633–46. https://doi.org/10.1097/AOG.0000000000004538.
Bayrampour H, Kapoor A, Bunka M, Ryan D. The risk of relapse of depression during pregnancy after discontinuation of antidepressants. J Clin Psychiatry. 2020. https://doi.org/10.4088/JCP.19r13134.
Guan H-B, Wei Y, Wang L-L, Qiao C, Liu C-X. Prenatal selective serotonin reuptake inhibitor use and associated risk for gestational hypertension and preeclampsia: a meta-analysis of cohort studies. J Women’s Heal. 2018;27(6):791–800. https://doi.org/10.1089/jwh.2017.6642.
Jiang H, Xu L, Li Y, Deng M, Peng C, Ruan B. Antidepressant use during pregnancy and risk of postpartum hemorrhage: a systematic review and meta-analysis. J Psychiatr Res. 2016;83:160–7. https://doi.org/10.1016/j.jpsychires.2016.09.001.
ACOG. Early pregnancy loss. 2015. https://www.acog.org/womens-health/faqs/early-pregnancy-loss. Accessed 17 Feb 2023.
Xing D, Wu R, Chen L, Wang T. Maternal use of antidepressants during pregnancy and risks for adverse perinatal outcomes: a meta-analysis. J Psychosom Res. 2020;137:110231. https://doi.org/10.1016/j.jpsychores.2020.110231.
Nikfar S, Rahimi R, Hendoiee N, Abdollahi M. Increasing the risk of spontaneous abortion and major malformations in newborns following use of serotonin reuptake inhibitors during pregnancy: a systematic review and updated meta-analysis. DARU J Pharm Sci. 2012;20(1):75. https://doi.org/10.1186/2008-2231-20-75.
Ross LE, Grigoriadis S, Mamisashvili L, et al. Selected pregnancy and delivery outcomes after exposure to antidepressant medication. JAMA Psychiat. 2013;70(4):436. https://doi.org/10.1001/jamapsychiatry.2013.684.
Einarson TR, Kennedy D, Einarson A. Do findings differ across research design? The case of antidepressant use in pregnancy and malformations. J Popul Ther Clin Pharmacol. 2012;19(2):e334–48.
Shen Z, Gao S, Li SX, et al. Sertraline use in the first trimester and risk of congenital anomalies: a systemic review and meta-analysis of cohort studies. Br J Clin Pharmacol. 2017;83(4):909–22. https://doi.org/10.1111/bcp.13161.
Turner E, Jones M, Vaz LR, Coleman T. Systematic review and meta-analysis to assess the safety of bupropion and varenicline in pregnancy. Nicotine Tob Res. 2019;21(8):1001–10. https://doi.org/10.1093/ntr/nty055.
De Vries C, Gadzhanova S, Sykes MJ, Ward M, Roughead E. A systematic review and meta-analysis considering the risk for congenital heart defects of antidepressant classes and individual antidepressants. Drug Saf. 2021;44(3):291–312. https://doi.org/10.1007/s40264-020-01027-x.
Selmer R, Haglund B, Furu K, et al. Individual-based versus aggregate meta-analysis in multi-database studies of pregnancy outcomes: the Nordic example of selective serotonin reuptake inhibitors and venlafaxine in pregnancy. Pharmacoepidemiol Drug Saf. 2016;25(10):1160–9. https://doi.org/10.1002/pds.4033.
Jordan S, Morris JK, Davies GI, et al. Selective serotonin reuptake inhibitor (SSRI) antidepressants in pregnancy and congenital anomalies: analysis of linked databases in Wales, Norway and Funen, Denmark. PLoS ONE. 2016;11(12):e0165122. https://doi.org/10.1371/journal.pone.0165122.
Wang S, Yang L, Wang L, Gao L, Xu B, Xiong Y. Selective serotonin reuptake inhibitors (SSRIs) and the risk of congenital heart defects: a meta-analysis of prospective cohort studies. J Am Heart Assoc. 2015;4(5):1–7. https://doi.org/10.1161/JAHA.114.001681.
Myles N, Newall H, Ward H, Large M. Systematic meta-analysis of individual selective serotonin reuptake inhibitor medications and congenital malformations. Aust N Z J Psychiatry. 2013;47(11):1002–12. https://doi.org/10.1177/0004867413492219.
Gao S-Y, Wu Q-J, Sun C, et al. Selective serotonin reuptake inhibitor use during early pregnancy and congenital malformations: a systematic review and meta-analysis of cohort studies of more than 9 million births. BMC Med. 2018;16(1):205. https://doi.org/10.1186/s12916-018-1193-5.
Grigoriadis S, Graves L, Peer M, et al. Benzodiazepine use during pregnancy alone or in combination with an antidepressant and congenital malformations. J Clin Psychiatry. 2019. https://doi.org/10.4088/JCP.18r12412.
Zwink N, Jenetzky E. Maternal drug use and the risk of anorectal malformations: systematic review and meta-analysis. Orphanet J Rare Dis. 2018;13(1):75. https://doi.org/10.1186/s13023-018-0789-3.
Wurst KE, Poole C, Ephross SA, Olshan AF. First trimester paroxetine use and the prevalence of congenital, specifically cardiac, defects: a meta-analysis of epidemiological studies. Birth Defects Res Part A Clin Mol Teratol. 2010;88(3):159–70. https://doi.org/10.1002/bdra.20627.
Bérard A, Iessa N, Chaabane S, Muanda FT, Boukhris T, Zhao J-P. The risk of major cardiac malformations associated with paroxetine use during the first trimester of pregnancy: a systematic review and meta-analysis. Br J Clin Pharmacol. 2016;81(4):589–604. https://doi.org/10.1111/bcp.12849.
Gao S-Y, Wu Q-J, Zhang T-N, et al. Fluoxetine and congenital malformations: a systematic review and meta-analysis of cohort studies. Br J Clin Pharmacol. 2017;83(10):2134–47. https://doi.org/10.1111/bcp.13321.
Riggin L, Frankel Z, Moretti M, Pupco A, Koren G. The fetal safety of fluoxetine: a systematic review and meta-analysis. J Obstet Gynaecol Can. 2013;35(4):362–9. https://doi.org/10.1016/S1701-2163(15)30965-8.
Grigoriadis S, VonderPorten EH, Mamisashvili L, et al. Antidepressant exposure during pregnancy and congenital malformations: is there an association? J Clin Psychiatry. 2013;74(04):e293-308. https://doi.org/10.4088/JCP.12r07966.
Moorthie S, Blencowe H, Darlison MW, et al. Estimating the birth prevalence and pregnancy outcomes of congenital malformations worldwide. J Community Genet. 2018;9(4):387–96. https://doi.org/10.1007/s12687-018-0384-2.
van Gelder MMHJ, van Rooij IALM, Miller RK, Zielhuis GA, de Jong-van den Berg LTW, Roeleveld N. Teratogenic mechanisms of medical drugs. Hum Reprod Update. 2010;16(4):378–94. https://doi.org/10.1093/humupd/dmp052.
Huecker MR, Smiley A, Saadabadi A. Bupropion. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 (PMID: 29262173). https://www.ncbi.nlm.nih.gov/books/NBK470212
Liu Y, Chen S, Zühlke L, et al. Global birth prevalence of congenital heart defects 1970–2017: updated systematic review and meta-analysis of 260 studies. Int J Epidemiol. 2019;48(2):455–63. https://doi.org/10.1093/ije/dyz009.
Huybrechts KF, Sanghani RS, Avorn J, Urato AC. Preterm birth and antidepressant medication use during pregnancy: a systematic review and meta-analysis. PLoS ONE. 2014;9(3):e92778. https://doi.org/10.1371/journal.pone.0092778.
Chang Q, Ma XY, Xu XR, Su H, Wu QJ, Zhao YH. Antidepressant use in depressed women during pregnancy and the risk of preterm birth: a systematic review and meta-analysis of 23 cohort studies. Front Pharmacol. 2020;11(May):1–12. https://doi.org/10.3389/fphar.2020.00659.
Huang H, Coleman S, Bridge JA, Yonkers K, Katon W. A meta-analysis of the relationship between antidepressant use in pregnancy and the risk of preterm birth and low birth weight. Gen Hosp Psychiatry. 2014;36(1):13–8. https://doi.org/10.1016/j.genhosppsych.2013.08.002.
Eke AC, Saccone G, Berghella V. Selective serotonin reuptake inhibitor (SSRI) use during pregnancy and risk of preterm birth: a systematic review and meta-analysis. BJOG An Int J Obstet Gynaecol. 2016;123(12):1900–7. https://doi.org/10.1111/1471-0528.14144.
McDonagh MS, Matthews A, Phillipi C, et al. Depression drug treatment outcomes in pregnancy and the postpartum period. Obstet Anesth Dig. 2015;35(3):126–7. https://doi.org/10.1097/01.aoa.0000469461.45993.de.
Kautzky A, Slamanig R, Unger A, Höflich A. Neonatal outcome and adaption after in utero exposure to antidepressants: a systematic review and meta-analysis. Acta Psychiatr Scand. 2022;145:6–28. https://doi.org/10.1111/acps.13367.
Zhao X, Liu Q, Cao S, et al. A meta-analysis of selective serotonin reuptake inhibitors (SSRIs) use during prenatal depression and risk of low birth weight and small for gestational age. J Affect Disord. 2018;241:563–70. https://doi.org/10.1016/j.jad.2018.08.061.
Black RE. Global prevalence of small for gestational age births. Nestle Nutr Inst Workshop Ser. 2015;81:1–7. https://doi.org/10.1159/000365790.
Say L, Donner A, Gülmezoglu AM, Taljaard M, Piaggio G. The prevalence of stillbirths: a systematic review. Reprod Health. 2006;3(1):1. https://doi.org/10.1186/1742-4755-3-1.
Corti S, Pileri P, Mazzocco MI, et al. Neonatal outcomes in maternal depression in relation to intrauterine drug exposure. Front Pediatr. 2019;7:1–8. https://doi.org/10.3389/fped.2019.00309.
Galbally M, Spigset O, Johnson AR, Kohan R, Lappas M, Lewis AJ. Neonatal adaptation following intrauterine antidepressant exposure: assessment, drug assay levels, and infant development outcomes. Pediatr Res. 2017;82(5):806–13. https://doi.org/10.1038/pr.2017.156.
Forsberg L, Navér L, Gustafsson LL, Wide K. Neonatal adaptation in infants prenatally exposed to antidepressants: clinical monitoring using neonatal abstinence score. PLoS ONE. 2014;9(11):e111327. https://doi.org/10.1371/journal.pone.0111327.
Grigoriadis S, VonderPorten EH, Mamisashvili L, et al. The effect of prenatal antidepressant exposure on neonatal adaptation. J Clin Psychiatry. 2013;74(04):e309–20. https://doi.org/10.4088/JCP.12r07967.
Wang J, Cosci F. Neonatal withdrawal syndrome following late in utero exposure to selective serotonin reuptake inhibitors: a systematic review and meta-analysis of observational studies. Psychother Psychosom. 2021;90(5):299–307. https://doi.org/10.1159/000516031.
Leung MTY, Wong KH, Ho PWH, et al. Gestational exposure to antidepressants and risk of seizure in offspring: a systematic review and meta-analysis. Neurosci Biobehav Rev. 2021;131:345–59. https://doi.org/10.1016/j.neubiorev.2021.09.040.
Martinho S, Adão R, Leite-Moreira AF, Brás-Silva C. Persistent pulmonary hypertension of the newborn: pathophysiological mechanisms and novel therapeutic approaches. Front Pediatr. 2020;8:342. https://doi.org/10.3389/fped.2020.00342.
Ng QX, Venkatanarayanan N, Ho CYX, Sim WS, Lim DY, Yeo W-S. Selective serotonin reuptake inhibitors and persistent pulmonary hypertension of the newborn: an updated meta-analysis. J Womens Health. 2019;28(3):331–8. https://doi.org/10.1089/jwh.2018.7319.
Masarwa R, Bar-Oz B, Gorelik E, Reif S, Perlman A, Matok I. Prenatal exposure to selective serotonin reuptake inhibitors and serotonin norepinephrine reuptake inhibitors and risk for persistent pulmonary hypertension of the newborn: a systematic review, meta-analysis, and network meta-analysis. Am J Obstet Gynecol. 2019;220(1):57.e1-57.e13. https://doi.org/10.1016/j.ajog.2018.08.030.
Grigoriadis S, VonderPorten EH, Mamisashvili L, et al. Prenatal exposure to antidepressants and persistent pulmonary hypertension of the newborn: systematic review and meta-analysis. BMJ. 2014;348:f6932. https://doi.org/10.1136/bmj.f6932.
Lai MC, Lombardo MV, Baron-Cohen S. Autism Lancet. 2014;383(9920):896–910. https://doi.org/10.1016/S0140-6736(13)61539-1.
Xu G, Strathearn L, Liu B, et al. Prevalence and treatment patterns of autism spectrum disorder in the United States, 2016. JAMA Pediatr. 2019;173(2):153–9. https://doi.org/10.1001/jamapediatrics.2018.4208.
Rais TB, Rais A. Association between antidepressants use during pregnancy and autistic spectrum disorders: a meta-analysis. Innov Clin Neurosci. 2014;11(5–6):18–22.
Mezzacappa A, Lasica P-A, Gianfagna F, et al. Risk for autism spectrum disorders according to period of prenatal antidepressant exposure. JAMA Pediatr. 2017;171(6):555. https://doi.org/10.1001/jamapediatrics.2017.0124.
Zhou X-H, Li Y-J, Ou J-J, Li Y-M. Association between maternal antidepressant use during pregnancy and autism spectrum disorder: an updated meta-analysis. Mol Autism. 2018;9(1):21. https://doi.org/10.1186/s13229-018-0207-7.
Halvorsen A, Hesel B, Østergaard SD, Danielsen AA. In utero exposure to selective serotonin reuptake inhibitors and development of mental disorders: a systematic review and meta-analysis. Acta Psychiatr Scand. 2019;139(6):493–507. https://doi.org/10.1111/acps.13030.
Morales DR, Slattery J, Evans S, Kurz X. Antidepressant use during pregnancy and risk of autism spectrum disorder and attention deficit hyperactivity disorder: systematic review of observational studies and methodological considerations. BMC Med. 2018;16(1):6. https://doi.org/10.1186/s12916-017-0993-3.
Vega ML, Newport GC, Bozhdaraj D, Saltz SB, Nemeroff CB, Newport DJ. Implementation of advanced methods for reproductive pharmacovigilance in autism: a meta-analysis of the effects of prenatal antidepressant exposure. Am J Psychiatry. 2020;177(6):506–17. https://doi.org/10.1176/appi.ajp.2020.18070766.
Man KKC, Tong HHY, Wong LYL, Chan EW, Simonoff E, Wong ICK. Exposure to selective serotonin reuptake inhibitors during pregnancy and risk of autism spectrum disorder in children: a systematic review and meta-analysis of observational studies. Neurosci Biobehav Rev. 2015;49(6):82–9. https://doi.org/10.1016/j.neubiorev.2014.11.020.
Kaplan YC, Keskin-Arslan E, Acar S, Sozmen K. Prenatal selective serotonin reuptake inhibitor use and the risk of autism spectrum disorder in children: a systematic review and meta-analysis. Reprod Toxicol. 2016;66:31–43. https://doi.org/10.1016/j.reprotox.2016.09.013.
Kobayashi T, Matsuyama T, Takeuchi M, Ito S. Autism spectrum disorder and prenatal exposure to selective serotonin reuptake inhibitors: a systematic review and meta-analysis. Reprod Toxicol. 2016;65:170–8. https://doi.org/10.1016/j.reprotox.2016.07.016.
Andalib S, Emamhadi MR, Yousefzadeh-Chabok S, et al. Maternal SSRI exposure increases the risk of autistic offspring: a meta-analysis and systematic review. Eur Psychiatry. 2017;45:161–6. https://doi.org/10.1016/j.eurpsy.2017.06.001.
Kaplan YC, Keskin-Arslan E, Acar S, Sozmen K. Maternal SSRI discontinuation, use, psychiatric disorder and the risk of autism in children: a meta-analysis of cohort studies. Br J Clin Pharmacol. 2017;83(12):2798–806. https://doi.org/10.1111/bcp.13382.
Leshem R, Bar-Oz B, Diav-Citrin O, et al. Selective serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) during pregnancy and the risk for autism spectrum disorder (ASD) and attention deficit hyperactivity disorder (ADHD) in the offspring: a true effect or a bias? A systematic review & meta-analysis. Curr Neuropharmacol. 2021;19(6):896–906. https://doi.org/10.2174/1570159X19666210303121059.
Csizmadi I, Collet J-P, Boivin J-F. Bias and confounding in pharmacoepidemiology. In: Pharmacoepidemiology. Chichester, UK: John Wiley & Sons, Ltd; p. 791–809. https://doi.org/10.1002/9780470059876.ch47.
Frisell T. Invited commentary: sibling-comparison designs, are they worth the effort? Am J Epidemiol. 2021;190(5):738–41. https://doi.org/10.1093/aje/kwaa183.
Sayal K, Prasad V, Daley D, Ford T, Coghill D. ADHD in children and young people: prevalence, care pathways, and service provision. Lancet Psychiatry. 2018;5(2):175–86. https://doi.org/10.1016/S2215-0366(17)30167-0.
Jiang H-Y, Peng C-T, Zhang X, Ruan B. Antidepressant use during pregnancy and the risk of attention-deficit/hyperactivity disorder in the children: a meta-analysis of cohort studies. BJOG An Int J Obstet Gynaecol. 2018;125(9):1077–84. https://doi.org/10.1111/1471-0528.15059.
Man KKC, Chan EW, Ip P, et al. Prenatal antidepressant exposure and the risk of attention-deficit hyperactivity disorder in children: a systematic review and meta-analysis. Neurosci Biobehav Rev. 2018;86:1–11. https://doi.org/10.1016/j.neubiorev.2017.12.007.
Grove K, Lewis AJ, Galbally M. Prenatal antidepressant exposure and child motor development: a meta-analysis. Pediatrics. 2018;142(1):e20180356. https://doi.org/10.1542/peds.2018-0356.
Nisell H, Larsson G, Wager J. The relation between life stress and hypertensive complications during pregnancy. Acta Obstet Gynecol Scand. 1989;68(5):423–7. https://doi.org/10.3109/00016348909021014.
Kurki T. Depression and anxiety in early pregnancy and risk for preeclampsia. Obstet Gynecol. 2000;95(4):487–90. https://doi.org/10.1016/S0029-7844(99)00602-X.
Price SM, Caughey AB. The impact of prenatal care on pregnancy outcomes in women with depression. J Matern Neonatal Med. 2022;35(20):3948–54. https://doi.org/10.1080/14767058.2020.1844655.
De Ocampo MPG, Araneta MRG, Macera CA, Alcaraz JE, Moore TR, Chambers CD. Risk of gestational hypertension and preeclampsia in women who discontinued or continued antidepressant medication use during pregnancy. Arch Womens Ment Health. 2016;19(6):1051–61. https://doi.org/10.1007/s00737-016-0655-z.
Bernard N, Forest J-C, Tarabulsy GM, Bujold E, Bouvier D, Giguère Y. Use of antidepressants and anxiolytics in early pregnancy and the risk of preeclampsia and gestational hypertension: a prospective study. BMC Pregnancy Childbirth. 2019;19(1):146. https://doi.org/10.1186/s12884-019-2285-8.
Andrade C, Sandarsh S, Chethan KB, Nagesh KS. Serotonin reuptake inhibitor antidepressants and abnormal bleeding. J Clin Psychiatry. 2010;71(12):1565–75. https://doi.org/10.4088/JCP.09r05786blu.
Arck PC, Rücke M, Rose M, et al. Early risk factors for miscarriage: a prospective cohort study in pregnant women. Reprod Biomed Online. 2008;17(1):101–13. https://doi.org/10.1016/S1472-6483(10)60300-8.
Marinescu IP, Foarfă MC, Pîrlog MC, Turculeanu A. Prenatal depression and stress: risk factors for placental pathology and spontaneous abortion. Rom J Morphol Embryol. 2014;55:1155–60.
Gentile S. Early pregnancy exposure to selective serotonin reuptake inhibitors, risks of major structural malformations, and hypothesized teratogenic mechanisms. Expert Opin Drug Metab Toxicol. 2015;11(10):1585–97. https://doi.org/10.1517/17425255.2015.1063614.
Heinonen E, Blennow M, Blomdahl-Wetterholm M, et al. Sertraline concentrations in pregnant women are steady and the drug transfer to their infants is low. Eur J Clin Pharmacol. 2021;77(9):1323–31. https://doi.org/10.1007/s00228-021-03122-z.
Chaabane S, Berard A. Epidemiology of major congenital malformations with specific focus on teratogens. Curr Drug Saf. 2013;8(2):128–40. https://doi.org/10.2174/15748863112079990011.
Swendsen J. The comorbidity of depression and substance use disorders. Clin Psychol Rev. 2000;20(2):173–89. https://doi.org/10.1016/S0272-7358(99)00026-4.
Persson M, Cnattingius S, Villamor E, et al. Risk of major congenital malformations in relation to maternal overweight and obesity severity: cohort study of 1.2 million singletons. BMJ. 2017;357:j2563. https://doi.org/10.1136/bmj.j2563.
Sharafi SE, Garmaroudi G, Ghafouri M, et al. Prevalence of anxiety and depression in patients with overweight and obesity. Obes Med. 2020;17:100169. https://doi.org/10.1016/j.obmed.2019.100169.
Le Gloan L, Legendre A, Iserin L, Ladouceur M. Pathophysiology and natural history of atrial septal defect. J Thorac Dis. 2018;10(S24):S2854–63. https://doi.org/10.21037/jtd.2018.02.80.
Daud A, Bergman J, Kerstjens-Frederikse W, Groen H, Wilffert B. The risk of congenital heart anomalies following prenatal exposure to serotonin reuptake inhibitors: is pharmacogenetics the key? Int J Mol Sci. 2016;17(8):1333. https://doi.org/10.3390/ijms17081333.
Benatar S, Cross-Barnet C, Johnston E, Hill I. Prenatal depression: assessment and outcomes among Medicaid participants. J Behav Health Serv Res. 2020;47(3):409–23. https://doi.org/10.1007/s11414-020-09689-2.
Fekadu Dadi A, Miller ER, Woodman RJ, Azale T, Mwanri L. Effect of antenatal depression on adverse birth outcomes in Gondar town, Ethiopia: a community-based cohort study. PLoS ONE. 2020;15(6):e0234728. https://doi.org/10.1371/journal.pone.0234728.
Mitchell J, Goodman J. Comparative effects of antidepressant medications and untreated major depression on pregnancy outcomes: a systematic review. Arch Womens Ment Health. 2018;21(5):505–16. https://doi.org/10.1007/s00737-018-0844-z.
Diego MA, Field T, Hernandez-Reif M, Schanberg S, Kuhn C, Gonzalez-Quintero VH. Prenatal depression restricts fetal growth. Early Hum Dev. 2009;85(1):65–70. https://doi.org/10.1016/j.earlhumdev.2008.07.002.
Goldstein DJ, Corbin LA, Sundell KL. Effects of first-trimester fluoxetine exposure on the newborn. Obstet Gynecol. 1997;89(5):713–8. https://doi.org/10.1016/S0029-7844(97)00070-7.
Kulin NA, Pastuszak A, Sage SR, et al. Pregnancy outcome following maternal use of the new selective serotonin reuptake inhibitors; a prospective controlled multicenter study. JAMA. 1998;279(8):609–10. https://doi.org/10.1001/jama.279.8.609.
Einarson A, Bonari L, Voyer-Lavigne S, et al. A multicentre prospective controlled study to determine the safety of trazodone and nefazodone use during pregnancy. Can J Psychiatry. 2003;48(2):106–10. https://doi.org/10.1177/070674370304800207.
Sivojelezova A, Shuhaiber S, Sarkissian L, Einarson A, Koren G. Citalopram use in pregnancy: prospective comparative evaluation of pregnancy and fetal outcome. Am J Obstet Gynecol. 2005;193(6):2004–9. https://doi.org/10.1016/j.ajog.2005.05.012.
Chun-Fai-Chan B, Koren G, Fayez I, et al. Pregnancy outcome of women exposed to bupropion during pregnancy: a prospective comparative study. Am J Obstet Gynecol. 2005;192(3):932–6. https://doi.org/10.1016/j.ajog.2004.09.027.
Colvin L, Slack-Smith L, Stanley FJ, Bower C. Dispensing patterns and pregnancy outcomes for women dispensed selective serotonin reuptake inhibitors in pregnancy. Birth Defects Res A Clin Mol Teratol. 2011;91(3):142–52.
Klieger-Grossmann C, Weitzner B, Panchaud A, et al. Pregnancy outcomes following use of escitalopram: a prospective comparative cohort study. J Clin Pharmacol. 2012;52(5):766–70. https://doi.org/10.1177/0091270011405524.
Kjaersgaard MIS, Parner ET, Vestergaard M, et al. Prenatal antidepressant exposure and risk of spontaneous abortion: a population-based study. PLoS ONE. 2013;8(8):e72095. https://doi.org/10.1371/journal.pone.0072095.
Stephansson O, Kieler H, Haglund B, et al. During pregnancy and risk of stillbirth description of sample. JAMA. 2013;309(1):48–54.
Wachman EM, Schiff DM, Silverstein M. Neonatal abstinence syndrome. JAMA. 2018;319(13):1362. https://doi.org/10.1001/jama.2018.2640.
Klinger G, Frankenthal D, Merlob P, et al. Long-term outcome following selective serotonin reuptake inhibitor induced neonatal abstinence syndrome. J Perinatol. 2011;31(9):615–20. https://doi.org/10.1038/jp.2010.211.
Alwan S, Bandoli G, Chambers C. Maternal use of selective serotonin-reuptake inhibitors and risk of persistent pulmonary hypertension of the newborn. Clin Pharmacol Ther. 2016;100(1):34–41. https://doi.org/10.1002/cpt.376.
Daly E, D. Tricklebank M, Wichers R. Neurodevelopmental roles and the serotonin hypothesis of autism spectrum disorder. In: The serotonin system. Academic Press, 2019; p. 23–44. https://doi.org/10.1016/B978-0-12-813323-1.00002-5.
Kinast K, Peeters D, Kolk SM, Schubert D, Homberg JR. Genetic and pharmacological manipulations of the serotonergic system in early life: neurodevelopmental underpinnings of autism-related behavior. Front Cell Neurosci. 2013;7:72. https://doi.org/10.3389/fncel.2013.00072.
Caparros-Gonzalez RA, de la Torre-Luque A, Romero-Gonzalez B, Quesada-Soto JM, Alderdice F, Peralta-Ramírez MI. Stress during pregnancy and the development of diseases in the offspring: a systematic-review and meta-analysis. Midwifery. 2021;97(Jan):102939. https://doi.org/10.1016/j.midw.2021.102939.
Faraone SV, Larsson H. Genetics of attention deficit hyperactivity disorder. Mol Psychiatry. 2019;24(4):562–75. https://doi.org/10.1038/s41380-018-0070-0.
Mayer JS, Bernhard A, Fann N, et al. Cognitive mechanisms underlying depressive disorders in ADHD: a systematic review. Neurosci Biobehav Rev. 2021;121:307–45. https://doi.org/10.1016/j.neubiorev.2020.12.018.
Gentile S. Untreated depression during pregnancy: short- and long-term effects in offspring. A systematic review. Neuroscience. 2017;342:154–66. https://doi.org/10.1016/j.neuroscience.2015.09.001.
Molenaar NM, Brouwer ME, Kamperman AM, et al. Recurrence of depression in the perinatal period: clinical features and associated vulnerability markers in an observational cohort. PLoS ONE. 2019;14(2):e0212964. https://doi.org/10.1371/journal.pone.0212964.
Netsi E, Pearson RM, Murray L, Cooper P, Craske MG, Stein A. Association of persistent and severe postnatal depression with child outcomes. JAMA Psychiat. 2018;75(3):247. https://doi.org/10.1001/jamapsychiatry.2017.4363.
Slomian J, Honvo G, Emonts P, Reginster J-Y, Bruyère O. Consequences of maternal postpartum depression: a systematic review of maternal and infant outcomes. Womens Health (Lond). 2019;15:174550651984404. https://doi.org/10.1177/1745506519844044.
Grigoriadis S, Graves L, Peer M, et al. Pregnancy and delivery outcomes following benzodiazepine exposure: a systematic review and meta-analysis. Can J Psychiatry. 2020;65(12):821–34. https://doi.org/10.1177/0706743720904860.
Källén B, Nilsson E, Olausson PO. Antidepressant use during pregnancy: comparison of data obtained from a prescription register and from antenatal care records. Eur J Clin Pharmacol. 2011;67(8):839–45. https://doi.org/10.1007/s00228-011-1021-8.
Habecker E, Freeman MP. Awareness and management of obstetrical complications of depression. Curr Psychiatr. 2015;14(12):39–44.
Matthias K, Rissling O, Pieper D, et al. The methodological quality of systematic reviews on the treatment of adult major depression needs improvement according to AMSTAR 2: A cross-sectional study. Heliyon. 2020;6(9):e04776. https://doi.org/10.1016/j.heliyon.2020.e04776.
Kieviet N, van Keulen V, van de Ven PM, Dolman KM, Deckers M, Honig A. Serotonin and poor neonatal adaptation after antidepressant exposure in utero. Acta Neuropsychiatr. 2017;29(1):43–53. https://doi.org/10.1017/neu.2016.30.
ACOG. ACOG practice bulletin no 92: use of psychiatric medications during pregnancy and lactation. Obstet Gynecol. 2008;111(4):1001–20. https://doi.org/10.1097/AOG.0b013e31816fd910.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The authors received no financial support for the research, authorship, and publication of this article.
Conflicts of Interest/Competing Interests
Pierre Desaunay, Léa-Gabrielle Eude, Michel Dreyfus, Cénéric Alexandre, Sophie Fedrizzi, Joachim Alexandre, Faruk Uguz, and Fabian Guénolé have no conflicts of interest that are directly relevant to the content of this article.
Ethics Approval
Not applicable.
Consent to Participate.
Not applicable.
Consent for Publication
Not applicable.
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
Authors’ Contributions
PD and LGE carried out the study selection and data extraction. All authors contributed equally to interpretation of the results, and revising the manuscript. All authors read and approved the final manuscript, and agreed to be accountable for the work. All authors meet the four ICMJE criteria for authorship.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Desaunay, P., Eude, LG., Dreyfus, M. et al. Benefits and Risks of Antidepressant Drugs During Pregnancy: A Systematic Review of Meta-analyses. Pediatr Drugs 25, 247–265 (2023). https://doi.org/10.1007/s40272-023-00561-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40272-023-00561-2