Abstract
Autoinflammatory diseases are disorders of the innate immune system characterized by uncontrolled inflammation. The most commonly encountered autoinflammatory diseases are the hereditary periodic fever syndromes, which present with fever and other features of the skin, serosal membranes, and musculoskeletal system. The main inherited (monogenic) periodic fever syndromes are familial Mediterranean fever (FMF), cryopyrin-associated periodic syndrome (CAPS), tumor necrosis factor receptor-associated periodic syndrome (TRAPS), and hyperimmunoglobulin D syndrome (HIDS)/mevalonate kinase deficiency (MKD). Recent advances in our understanding of the molecular and pathophysiological basis of autoinflammatory diseases have provided new treatment strategies. Patients with periodic fever syndromes have clearly benefited from anti-interleukin (IL)-1 treatment. Colchicine is still the mainstay of FMF therapy, but IL-1 blockade is also effective if colchicine fails. Early diagnosis and effective treatment can prevent irreversible organ damage. The scope of pathogenic mutations and more targeted therapy for better management of these rare diseases remains to be defined.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Beneficial effects from interleukin (IL)-1-blocking agents have been reported in all monogenic autoinflammatory diseases. |
Treat-to-target strategies are very important to achieve complete response, particularly in cryopyrin-associated periodic syndrome (CAPS). |
Guidelines are required on how and when to use anti-IL-1 agents effectively in these autoinflammatory diseases. |
The prognosis for these patients depends on early diagnosis and treatment. |
1 Introduction
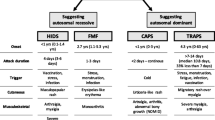
Autoinflammatory diseases are a heterogeneous group of disorders of the innate immune system that present with clinical signs of inflammation such as fevers, skin features, musculoskeletal features, and raised acute-phase inflammatory markers [1]. The most common and well-characterized group of autoinflammatory syndromes are those associated with “periodic fevers”, where unprovoked sterile inflammation manifests mainly as recurrent fever and disease-specific patterns of organ inflammation. The main inherited (monogenic) periodic fever syndromes are familial Mediterranean fever (FMF), cryopyrin-associated periodic syndrome (CAPS), tumor necrosis factor (TNF)-receptor-associated periodic syndrome (TRAPS), and hyperimmunoglobulin D syndrome (HIDS)/mevalonate kinase deficiency (MKD). Periodic fever with aphthous stomatitis, pharyngitis, and adenitis (PFAPA) syndrome is also common. However, as it is not due to a single gene defect it is not discussed in this review, although it should be in the differential diagnosis of all four of the monogenic diseases covered here.
-
Globally, FMF is the most common autoinflammatory disease, caused by mutations of the MEFV gene, which codes for the pyrin protein [2, 3]. Mutated pyrin causes an exaggerated inflammatory response via uncontrolled interleukin (IL)-1 secretion [4]. The typical attack will last <3 days and is characterized by fever and serositis in the form of abdominal or chest pain or arthritis. The most significant complication of FMF is amyloidosis [5]. Fortunately, the majority of cases can be treated with colchicine [6], an oral drug that is generally cheaply available in most countries. However, a minority of patients can be resistant to colchicine. Anti-IL-1 therapy is a promising second-line therapy should colchicine treatment fail [6, 7].
-
CAPS is caused by gain-of-function mutations in the NLRP3 gene encoding cryopyrin, resulting in increased IL-1 secretion [8]. CAPS is clinically characterized by fever, urticaria, musculoskeletal features, and conjunctivitis, as well as hearing loss and neurological symptoms at the more severe end of the disease spectrum. CAPS has traditionally been divided into three diseases: familial cold autoinflammatory syndrome (FCAS), representing the mildest form of the disease; Muckle–Wells syndrome (MWS), which may also cause hearing defects; and neonatal-onset multisystem inflammatory disease (NOMID)/chronic infantile neurological cutaneous articular syndrome (CINCA), which represents the most severe form of the disease [9]. However, a more recent approach is to consider all patients with a CIAS1 mutation under the same heading of CAPS. This is mainly because of the overlap of the underlying mutations and the variation of symptoms with mosaicism and therapy. Diagnosis relies on clinical suspicion followed by genetic testing, but an international group of experts recently suggested classification criteria that would guide clinicians in the diagnosis of these diseases [10].
-
TRAPS is caused by mutations in the TNFRSF1A gene encoding TNF receptor 1 (TNFR1) and is characterized by attacks of fever, serositis (chest and/or abdominal pain), migratory rash, orbital findings, and musculoskeletal symptoms such as myalgia [11]. Unlike the other three monogenic periodic fever syndromes, the duration of attacks tends to be longer than a week. FMF, TRAPS, and severe CAPS may all lead to secondary amyloidosis as a sequelae of chronic inflammation [12, 13].
-
HIDS or MKD is caused by mutations of the MVK gene. On this basis, it is a metabolic disease; however, the defect leads to increased IL-1 production (discussed elsewhere [14]) and frank inflammation [14]. The main clinical manifestations are attacks of fever, abdominal pain, vomiting, diarrhea, lymphadenopathy, and rashes lasting 3–6 days. Attacks are provoked by immunizations, surgery, trauma, and infections [15].
Better understanding of the molecular basis and pathophysiology of autoinflammatory diseases has led to new treatment targets and strategies. The common feature of the aforementioned autoinflammatory diseases (including colchicine-resistant FMF) is response to IL-1 blockade, which is a promising therapy for the management of these diseases and for the prevention of irreversible organ damage. In this review, we discuss the new treatment options for autoinflammatory diseases.
2 Treatment Approach to Periodic Fevers
Autoinflammatory diseases cause uncontrolled systemic inflammation, leading to irreversible organ damage. A multidisciplinary approach to early diagnosis and control of inflammation is critical in the prevention or stabilization of organ damage (hearing loss, visual loss, chronic meningitis in CAPS, or amyloidosis mostly seen in FMF and TRAPS) and to improve the patient’s quality of life. In 2015, a group of experts from different countries proposed recommendations for the management of CAPS, TRAPS, and HIDS/MKD [16] (Table 1), and in 2016 an international group of experts published the European League Against Rheumatism (EULAR)-endorsed recommendations for the management of FMF [17] (Table 2), to help clinicians, pediatricians, and pediatric rheumatologists diagnose and manage these diseases. In all monogenic autoinflammatory diseases, beneficial effects from IL-1 blockade have been reported [18]. Unfortunately, these drugs are quite expensive. Three forms of anti-IL-1 medications are currently available: anakinra (a recombinant, human IL-1 receptor antagonist), canakinumab (a human immunoglobulin [Ig]-G1 monoclonal antibody directed against IL-1β), and rilonacept (a fully human dimeric fusion protein that binds the extracellular domains of IL-1α and IL-1β). The US FDA has approved anakinra, canakinumab, and rilonacept for the treatment of CAPS, and the European Medicines Agency (EMA) has approved anakinra and canakinumab for the treatment of CAPS. Recently, the FDA also approved canakinumab for the treatment of TRAPS, HIDS/MKD, and (colchicine-resistant) FMF.
2.1 Familial Mediterranean Fever (FMF)
FMF is the most common autoinflammatory disease. An international group of experts recently published recommendations for the management of FMF [17]. According to these recommendations, the main goal of treatment should be complete control of unprovoked attacks and minimizing subclinical inflammation between attacks. Colchicine has been the mainstay of FMF treatment since 1972 [19] and is generally safe and well-tolerated in children [20]. Colchicine can reduce the frequency and severity of attacks and suppress subclinical inflammation between attacks [21,22,23]. Furthermore, it prevents the development of secondary amyloidosis in patients with FMF [24]. It is recommended that colchicine should be started as soon as the patient is clinically diagnosed. Genetic diagnosis may not be enough to start colchicine treatment without clinical manifestations or subclinical inflammation. Physicians should follow-up these patients closely for clinical and subclinical signs of FMF [17]. In countries where amyloidosis is frequent, if the patient has M694V homozygous mutations, physicians tend to start colchicine, since this mutation is associated with the risk of amyloidosis [25, 26].
The starting dose of colchicine has been suggested as ≤0.5 mg/day for children aged <5 years, 0.5–1 mg/day in children aged 5–10 years, and 1–1.5 mg/day in children aged >10 years and adults [17]. The optimal dosage of colchicine varies between clinical practices. Treatment is generally started at the sub-therapeutic dose of 0.5 mg/day and monitored according to disease activity and the patient’s tolerance. Higher doses up to 2 mg/day in children and up to 3 mg/day in adults can be used to control ongoing disease activity and amyloidosis [17]. The side effects of colchicine are gastrointestinal, with vomiting, diarrhea, and transient elevation of transaminases occurring in up to 5% of patients with FMF. A minority of patients with FMF do not respond to colchicine or are intolerant to the drug because of its side effects [27]. These patients will experience frequent attacks and, more importantly, ongoing inflammation. The chronic inflammation may lead to many sequelae, the most frequent and worrisome being the development of secondary amyloidosis. Since the mutation in the pyrin protein has been clearly associated with increased IL-1 production, anti-IL-1 treatment has emerged as a promising option in patients with resistant disease [7, 28]. Several studies have reported successful results in colchicine-resistant patients with IL-1-blocking agents, including anakinra, canakinumab, and rilonacept [29, 30]. A literature review of anti-IL-1 treatment in FMF reported complete response in 76.5% of patients receiving anakinra treatment and 67.5% of those receiving canakinumab treatment [30]. In a small randomized controlled study, rilonacept treatment resulted in complete response in two and partial response in eight of 14 patients [31]. IL-1 blockade can also reverse proteinuria in patients with secondary amyloidosis [30, 32]. However, there is no evidence for using anti-IL-1 treatment without colchicine to prevent amyloidosis. Thus, a maximal tolerated dose of colchicine is recommended with anti-IL-1 treatment [17]. A cluster study evaluating the efficacy and safety of canakinumab in patients with hereditary periodic fevers, including colchicine-resistant FMF, is underway (NCT02059291) [33]. Etanercept has also been used to treat FMF before administration of anti-IL-1 agents [34]. Anti-TNF treatment can be successful, especially in patients with FMF and chronic arthritis and sacroiliitis [35].
2.2 Cryopyrin-Associated Periodic Syndrome (CAPS)
In CAPS, corticosteroids and non-steroidal anti-inflammatory drugs (NSAIDs) can be used for symptom relief [36]. However, no evidence is available relating to the efficacy and safety of disease-modifying anti-rheumatic drugs or biological therapy, except for IL-1-blocking agents [36]. Early diagnosis and initiation of treatment are very important because hearing loss, joint deformities, and neurological damage are the leading cause of morbidity and can be stabilized or improved with anti-IL-1 treatment [37,38,39,40]. Elegant research in CAPS has clearly identified the excess production of IL-1 in this disease; thus, it is no surprise that anti-IL-1 drugs have emerged as the appropriate treatment [8]. In the published series for CAPS, patients have often been defined as NOMID/CINCA or MWS or FCAS [8], and these conditions are discussed accordingly.
Anakinra was the first anti-IL-1 drug to be tried in these patients. Sibley et al. [40] showed that, in 26 patients with NOMID/CINCA (aged 8 months to 41 years), anakinra provided up to 5 years of sustained efficacy. The authors observed that anakinra not only prevented systemic inflammation but also reduced steroid use, improved patient growth, and improved or stabilized hearing and vision. However, there was no clear effect on bony lesions such as clubbing or patellar overgrowth [40]. Increasing doses of anakinra reduced the cerebrospinal fluid (CSF) leukocyte count and opening pressure, which are indicators of central nervous system (CNS) inflammation. The authors also demonstrated that cochlear enhancement on magnetic resonance imaging correlated with continued hearing loss and low optic nerve size correlated with poor visual field [40]. In another study, anakinra treatment improved not only clinical and laboratory features but also cochlear and leptomeningeal lesions [41]. In 2015, Ross et al. [42] showed the clinical and laboratory efficacy of anakinra in eight adult members of the same family with FCAS. All signs and symptoms of FCAS resolved in all patients within 24 h after the first injection of anakinra [42]. In 2015, Sibley et al. [43] demonstrated that canakinumab, a long-acting anti-IL-1 treatment, improved both symptoms and serum inflammatory markers in six patients with NOMID with appropriate dose adjustments; however, CSF pleocytosis persisted in five of six patients. Rodriguez-Smith et al. [44] recently showed that levels of IL-6, interferon γ-induced protein (IP)-10 (also known as C-X-C motif chemokine 10 [CXCL10]), and IL-18 were elevated in the CSF of patients with NOMID and could serve as markers for CNS inflammation. CSF studies showed that cytokine levels, as well as monocyte and granulocyte counts, in CSF decreased with anakinra treatment. CSF levels of IL-6, IP-10/CXCL10, and IL-18 and CSF white blood cell counts were significantly higher when patients received canakinumab than when they received anakinra, despite similar serum cytokine levels. The authors thus suggested that anakinra may be more likely to pass through the blood–brain barrier than canakinumab [44].
Studies reporting the use of canakinumab in CAPS are increasing. In two randomized controlled trials, canakinumab induced rapid and sustained remission [45, 46]. Subcutaneous canakinumab 2 mg/kg (children) and 150 mg (adults) once per 8 weeks provided sustained remission in patients with CAPS [47, 48]. Supporting the effectiveness of canakinumab in CAPS, Kuemmerle-Deschner et al. [48] found that seven patients with CAPS treated with canakinumab achieved complete response. Furthermore, in a multicenter phase III study [47], they demonstrated that higher canakinumab doses were required to obtain complete response in younger patients and in more severe disease. Dose escalation was not associated with an increase in adverse events. The same group drew attention to the real-life effectiveness of canakinumab in CAPS. They showed that centers using a standard dose of canakinumab have fewer complete responders than centers using ‘treat-to-target’ strategies that adjust the canakinumab dose according to patient age and disease severity to achieve complete response [49]. Similarly, according to the Eurofever Registry, the number of patients with CAPS responding to anti-IL-1 drug therapy was lower than the literature described [35]. Yokota et al. [50] recently described 19 Japanese patients with CAPS (MWS 7; NOMID 12), who were treated with subcutaneous canakinumab 2–8 mg/kg every 8 weeks. In total, 15 (79%) patients achieved a complete response by day 15, 18 (94.7%) by week 24, and all by week 48. Thirteen (68%) patients (MWS 4; NOMID 9) required a dose increase during the trial [50].
The efficacy of rilonacept was demonstrated in two double-blind, placebo-controlled trials of 6 and 18 weeks duration in 41 patients with FCAS and three with MWS: rilonacept decreased the number of disease flare days and levels of C-reactive protein (CRP) and serum amyloid A (SAA), improved the physician’s and patient’s global assessments of disease activity, and reduced limitations in patients’ daily activities. The treated patients also experienced a significant reduction in mean symptom score (84%) compared with placebo (13%) [51]. During the 96-week follow-up, the effect of rilonacept was sustained [52].
2.3 Tumor Necrosis Factor Receptor-Associated Periodic Syndrome (TRAPS)
The treatment of TRAPS is determined by the severity of the disease. NSAIDs are usually used as on-demand therapy to relieve symptoms, but they are not effective in suppressing ongoing inflammation [36]. For patients with mild disease, corticosteroids can be an option to end attacks, but the corticosteroid doses may need to be escalated, and steroid withdrawal may be difficult [53]. Anti-TNF therapy with etanercept is effective in controlling attacks and reducing steroid requirements for some patients [53]. In the Eurofever Registry, etanercept was effective in 87% of patients with TRAPS, but a complete response was seen only in 30% [36]. Etanercept is a fusion protein containing two copies of the TNF receptor bound to the Fc region of human IgG1. Subsequent studies have shown that the responsiveness to etanercept is not usually complete and may decrease over time [54]. It has been shown that treatment with infliximab and adalimumab might trigger inflammatory attacks, and they are therefore not recommended for the treatment of TRAPS [16]. These latter two drugs are monoclonal anti-TNF antibodies. The increased proinflammatory response after infliximab administration is thought to be a result of the failure to shed infliximab-bound TNF/TNFR1 from the cell surface [55].
IL-1 blocking may be more effective than anti-TNF agents in TRAPS [36, 54, 56]. Anakinra has been a promising therapy for patients with severe disease who require long-term steroids and have an insufficient response to etanercept [56, 57]. Consistently, in the Eurofever Registry, 79% of patients experienced complete responses with anakinra [36]. However, a number of studies have shown that anakinra failed in TRAPS [36, 58]. Recent studies have shown that the efficacy of canakinumab appeared rapidly and provided long-term clinical benefits [57, 59]. The randomized phase III cluster study (NCT02059291) of canakinumab in patients with TRAPS, HIDS, or colchicine-resistant FMF also confirmed the efficacy of this drug in TRAPS [33].
Finally, three observational studies suggested tocilizumab, a humanized monoclonal antibody agent against the IL-6 receptor, was also effective for the treatment of patients with TRAPS. However, two of the three patients did not have pathogenic TRAPS mutations. Further controlled trials are necessary to assist with understanding the effect of tocilizumab on TRAPS [60,61,62].
2.4 Hyperimmunoglobulin D Syndrome (HIDS)/Mevalonate Kinase Deficiency (MKD)
NSAIDs can be effective in HIDS/MKD, and corticosteroids have been tried for HIDS attacks. In the Eurofever Registry, NSAIDs were mostly used as on-demand therapy for attacks, with five (13%) of the 39 patients experiencing complete response and 25 (64%) experiencing a partial response [36]. NSAIDs can relieve symptoms in most patients but may not end the attacks and inflammation [16]. High-dose corticosteroids administered at the onset of an attack can reduce the severity and duration of febrile episodes. In the Eurofever Registry, treatment with corticosteroids resulted in a complete response in eight (24%) and a partial response in 22 (67%) of the 33 patients [36]. Colchicine is not an effective therapy in patients with HIDS/MKD. In the Eurofever Registry, colchicine was used in 17 patients, but 65% experienced no response [36]. As the mutant enzyme in HIDS/MKD is a member of the cholesterol and isoprene pathway, statins have been used for treatment, but results were poor [53]. Moreover, there might be a detrimental effect on attack severity [63]. Because of the increased IL-1β secretion in patients with HIDS/MKD, IL-1 blockade has emerged as a therapeutic option [64]. In the Eurofever Registry, 89% of 27 patients receiving anakinra and 65% of 17 patients receiving etanercept experienced a response. Complete remission was observed in 22% of patients receiving anakinra and in 6% of those receiving etanercept [36]. A small prospective study found a ≥50% reduction in the severity and duration of febrile episodes in patients with HIDS/MKD using on-demand therapy with anakinra when administered in the first 24 h of an attack [65].
Continuous therapy with IL-1-blocking agents or anti-TNF drugs should be preferred in patients with HIDS/MKD with frequent attacks and those with ongoing subclinical inflammation between attacks and long-term complications [16, 65]. Ter Haar et al. [16] found that neither anakinra nor etanercept were superior to each other as first-line biological therapies in HIDS/MKD. For anti-IL-1 treatment, dose escalation should be tried first before switching to other biological therapies. In a recent literature review [66] of the treatment of pediatric patients with HIDS/MKD, 21 patients were treated with anakinra, 16 with etanercept, and five with canakinumab; response rates were 90%, 50%, and 100%, respectively. Galeotti et al. [67] treated six patients with HIDS with canakinumab; three patients achieved a complete response and the remaining three achieved a partial response. If anti-IL-1 and anti TNF therapy do not provide significant improvement, tocilizumab may be a treatment option. Shendi et al. [68] reported on a patient with HIDS/MKD resistant to colchicine, corticosteroids, etanercept, and anakinra who was treated successfully with tocilizumab. In very severe cases, hematopoietic stem cell transplantation may be indicated, and suppression of systemic inflammation and improvement of neurologic symptoms has been observed in such patients [69,70,71]. Again, canakinumab was also shown to be effective in the treatment of patients with HIDS in the cluster study (NCT02059291) [33].
3 Conclusions
Autoinflammatory diseases are rare, chronic disorders characterized by high inflammatory states. The scope of pathogenic mutations and more targeted therapy for better management of these rare diseases remains to be defined. The prognosis for these patients depends on early diagnosis and early treatment. Colchicine is still the main therapy in FMF, but if colchicine fails, IL-1 blockade is a promising second-line therapy. In CAPS, the effect of anti-IL-1 agents is clear cut. In TRAPS and HIDS/MKD, IL-1 blockade seems more effective than other biologics. We can now offer patients with CAPS, TRAPS, HIDS/MKD, or colchicine-resistant FMF a much better quality of life while protecting them from irreversible organ damage.
References
Masters SL, Simon A, Aksentijevich I, Kastner DL. Horror autoinflammaticus: the molecular pathophysiology of autoinflammatory disease (*). Annu Rev Immunol. 2009;27:621–68.
French FMFC. A candidate gene for familial Mediterranean fever. Nat Genet. 1997;17(1):25–31.
Ancient missense mutations in a new member of the RoRet gene family are likely to cause familial Mediterranean fever. The International FMF Consortium. Cell. 1997;90(4):797–807.
Berkun Y, Padeh S, Reichman B, Zaks N, Rabinovich E, Lidar M, et al. A single testing of serum amyloid a levels as a tool for diagnosis and treatment dilemmas in familial Mediterranean fever. Semin Arthritis Rheum. 2007;37(3):182–8.
Heller H, Sohar E, Gafni J, Heller J. Amyloidosis in familial Mediterranean fever. An independent genetically determined character. Arch Intern Med. 1961;107:539–50.
Meinzer U, Quartier P, Alexandra JF, Hentgen V, Retornaz F, Kone-Paut I. Interleukin-1 targeting drugs in familial Mediterranean fever: a case series and a review of the literature. Semin Arthritis Rheum. 2011;41(2):265–71.
Ozen S, Bilginer Y, Aktay Ayaz N, Calguneri M. Anti-interleukin 1 treatment for patients with familial Mediterranean fever resistant to colchicine. J Rheumatol. 2011;38(3):516–8.
Hoffman HM, Mueller JL, Broide DH, Wanderer AA, Kolodner RD. Mutation of a new gene encoding a putative pyrin-like protein causes familial cold autoinflammatory syndrome and Muckle–Wells syndrome. Nat Genet. 2001;29(3):301–5.
Aksentijevich I, Putnam CD, Remmers EF, Mueller JL, Le J, Kolodner RD, et al. The clinical continuum of cryopyrinopathies: novel CIAS1 mutations in North American patients and a new cryopyrin model. Arthritis Rheum. 2007;56(4):1273–85.
Kuemmerle-Deschner JB, Ozen S, Kone-Paut I, Goldbach-Mansky R, Lachmann H, Blank N, et al. Diagnostic criteria for cryopyrin-associated periodic syndrome (CAPS). Ann Rheum Dis. 2016. doi:10.1136/annrheumdis-2016-209686.
McDermott MF, Aksentijevich I, Galon J, McDermott EM, Ogunkolade BW, Centola M, et al. Germline mutations in the extracellular domains of the 55 kDa TNF receptor, TNFR1, define a family of dominantly inherited autoinflammatory syndromes. Cell. 1999;97(1):133–44.
Aksentijevich I, Galon J, Soares M, Mansfield E, Hull K, Oh HH, et al. The tumor-necrosis-factor receptor-associated periodic syndrome: new mutations in TNFRSF1A, ancestral origins, genotype-phenotype studies, and evidence for further genetic heterogeneity of periodic fevers. Am J Hum Genet. 2001;69(2):301–14.
Feldmann J, Prieur AM, Quartier P, Berquin P, Certain S, Cortis E, et al. Chronic infantile neurological cutaneous and articular syndrome is caused by mutations in CIAS1, a gene highly expressed in polymorphonuclear cells and chondrocytes. Am J Hum Genet. 2002;71(1):198–203.
Park YH, Wood G, Kastner DL, Chae JJ. Pyrin inflammasome activation and RhoA signaling in the autoinflammatory diseases FMF and HIDS. Nat Immunol 2016;17(8):914-21.
van der Hilst JC, Bodar EJ, Barron KS, Frenkel J, Drenth JP, van der Meer JW, et al. Long-term follow-up, clinical features, and quality of life in a series of 103 patients with hyperimmunoglobulinemia D syndrome. Medicine (Baltimore). 2008;87(6):301–10.
ter Haar NM, Oswald M, Jeyaratnam J, Anton J, Barron KS, Brogan PA, et al. Recommendations for the management of autoinflammatory diseases. Ann Rheum Dis. 2015;74(9):1636–44.
Ozen S, Demirkaya E, Erer B, Livneh A, Ben-Chetrit E, Giancane G, et al. EULAR recommendations for the management of familial Mediterranean fever. Ann Rheum Dis. 2016;75(4):644–51.
Jesus AA, Goldbach-Mansky R. IL-1 blockade in autoinflammatory syndromes. Annu Rev Med. 2014;65:223–44.
Goldfinger SE. Colchicine for familial Mediterranean fever. N Engl J Med. 1972;287(25):1302.
Padeh S, Gerstein M, Berkun Y. Colchicine is a safe drug in children with familial Mediterranean fever. J Pediatr. 2012;161(6):1142–6.
Livneh A, Langevitz P. Diagnostic and treatment concerns in familial Mediterranean fever. Baillieres Best Pract Res Clin Rheumatol. 2000;14(3):477–98.
Majeed HA, Rawashdeh M, El-Shanti H, Qubain H, Khuri-Bulos N, Shahin HM. Familial Mediterranean fever in children: the expanded clinical profile. QJM. 1999;92(6):309–18.
Zemer D, Livneh A, Danon YL, Pras M, Sohar E. Long-term colchicine treatment in children with familial Mediterranean fever. Arthritis Rheum. 1991;34(8):973–7.
Zemer D, Pras M, Sohar E, Modan M, Cabili S, Gafni J. Colchicine in the prevention and treatment of the amyloidosis of familial Mediterranean fever. N Engl J Med. 1986;314(16):1001–5.
Shinar Y, Obici L, Aksentijevich I, Bennetts B, Austrup F, Ceccherini I, et al. Guidelines for the genetic diagnosis of hereditary recurrent fevers. Ann Rheum Dis. 2012;71(10):1599–605.
Ben-Chetrit E, Backenroth R. Amyloidosis induced, end stage renal disease in patients with familial Mediterranean fever is highly associated with point mutations in the MEFV gene. Ann Rheum Dis. 2001;60(2):146–9.
Cerquaglia C, Diaco M, Nucera G, La Regina M, Montalto M, Manna R. Pharmacological and clinical basis of treatment of Familial Mediterranean Fever (FMF) with colchicine or analogues: an update. Curr Drug Targets Inflamm Allergy. 2005;4(1):117–24.
Park H, Bourla AB, Kastner DL, Colbert RA, Siegel RM. Lighting the fires within: the cell biology of autoinflammatory diseases. Nat Rev Immunol. 2012;12(8):570–80.
Grattagliano I, Bonfrate L, Ruggiero V, Scaccianoce G, Palasciano G, Portincasa P. Novel therapeutics for the treatment of familial Mediterranean fever: from colchicine to biologics. Clin Pharmacol Ther. 2014;95(1):89–97.
van der Hilst J, Moutschen M, Messiaen PE, Lauwerys BR, Vanderschueren S. Efficacy of anti-IL-1 treatment in familial Mediterranean fever: a systematic review of the literature. Biologics. 2016;10:75–80.
Hashkes PJ, Spalding SJ, Giannini EH, Huang B, Johnson A, Park G, et al. Rilonacept for colchicine-resistant or -intolerant familial Mediterranean fever: a randomized trial. Ann Intern Med. 2012;157(8):533–41.
Topaloglu R, Batu ED, Orhan D, Ozen S, Besbas N. Anti-interleukin 1 treatment in secondary amyloidosis associated with autoinflammatory diseases. Pediatr Nephrol. 2016;31(4):633–40.
Benedetti FD, Anton J, Gattorno M, Lachmann H, Kone-Paut I, Ozen S. A phase III pivotal umbrella trial of canakinumab in patients with autoinflammatory periodic fever syndromes (colchicine resistant FMF, HIDS/MKD and TRAPS). Ann Rheum Dis. 2016;75(suppl 2):615–6.
Seyahi E, Ozdogan H, Celik S, Ugurlu S, Yazici H. Treatment options in colchicine resistant familial Mediterranean fever patients: thalidomide and etanercept as adjunctive agents. Clin Exp Rheumatol. 2006;24(5 Suppl 42):S99–103.
Bilgen SA, Kilic L, Akdogan A, Kiraz S, Kalyoncu U, Karadag O, et al. Effects of anti-tumor necrosis factor agents for familial mediterranean fever patients with chronic arthritis and/or sacroiliitis who were resistant to colchicine treatment. J Clin Rheumatol. 2011;17(7):358–62.
Ter Haar N, Lachmann H, Ozen S, Woo P, Uziel Y, Modesto C, et al. Treatment of autoinflammatory diseases: results from the Eurofever Registry and a literature review. Ann Rheum Dis. 2013;72(5):678–85.
Koitschev A, Gramlich K, Hansmann S, Benseler S, Plontke SK, Koitschev C, et al. Progressive familial hearing loss in Muckle–Wells syndrome. Acta Otolaryngol. 2012;132(7):756–62.
Kuemmerle-Deschner JB, Koitschev A, Ummenhofer K, Hansmann S, Plontke SK, Koitschev C, et al. Hearing loss in Muckle–Wells syndrome. Arthritis Rheum. 2013;65(3):824–31.
Neven B, Marvillet I, Terrada C, Ferster A, Boddaert N, Couloignier V, et al. Long-term efficacy of the interleukin-1 receptor antagonist anakinra in ten patients with neonatal-onset multisystem inflammatory disease/chronic infantile neurologic, cutaneous, articular syndrome. Arthritis Rheum. 2010;62(1):258–67.
Sibley CH, Plass N, Snow J, Wiggs EA, Brewer CC, King KA, et al. Sustained response and prevention of damage progression in patients with neonatal-onset multisystem inflammatory disease treated with anakinra: a cohort study to determine three- and five-year outcomes. Arthritis Rheum. 2012;64(7):2375–86.
Goldbach-Mansky R, Dailey NJ, Canna SW, Gelabert A, Jones J, Rubin BI, et al. Neonatal-onset multisystem inflammatory disease responsive to interleukin-1beta inhibition. N Engl J Med. 2006;355(6):581–92.
Ross JB, Finlayson LA, Klotz PJ, Langley RG, Gaudet R, Thompson K, et al. Use of anakinra (Kineret) in the treatment of familial cold autoinflammatory syndrome with a 16-month follow-up. J Cutan Med Surg. 2008;12(1):8–16.
Sibley CH, Chioato A, Felix S, Colin L, Chakraborty A, Plass N, et al. A 24-month open-label study of canakinumab in neonatal-onset multisystem inflammatory disease. Ann Rheum Dis. 2015;74(9):1714–9.
Rodriguez-Smith J, Lin YC, Li Tsai W, Kim H, Montealegre-Sanchez G, Chapelle D, et al. CSF cytokines correlate with aseptic meningitis and blood brain barrier function in neonatal-onset multisystem inflammatory disease (NOMID). Arthritis Rheumatol. 2017. doi:10.1002/art.40055.
Kone-Paut I, Lachmann HJ, Kuemmerle-Deschner JB, Hachulla E, Leslie KS, Mouy R, et al. Sustained remission of symptoms and improved health-related quality of life in patients with cryopyrin-associated periodic syndrome treated with canakinumab: results of a double-blind placebo-controlled randomized withdrawal study. Arthritis Res Ther. 2011;13(6):R202.
Lachmann HJ, Kone-Paut I, Kuemmerle-Deschner JB, Leslie KS, Hachulla E, Quartier P, et al. Use of canakinumab in the cryopyrin-associated periodic syndrome. N Engl J Med. 2009;360(23):2416–25.
Kuemmerle-Deschner JB, Hachulla E, Cartwright R, Hawkins PN, Tran TA, Bader-Meunier B, et al. Two-year results from an open-label, multicentre, phase III study evaluating the safety and efficacy of canakinumab in patients with cryopyrin-associated periodic syndrome across different severity phenotypes. Ann Rheum Dis. 2011;70(12):2095–102.
Kuemmerle-Deschner JB, Ramos E, Blank N, Roesler J, Felix SD, Jung T, et al. Canakinumab (ACZ885, a fully human IgG1 anti-IL-1beta mAb) induces sustained remission in pediatric patients with cryopyrin-associated periodic syndrome (CAPS). Arthritis Res Ther. 2011;13(1):R34.
Kuemmerle-Deschner JB, Hofer F, Endres T, Kortus-Goetze B, Blank N, Weissbarth-Riedel E, et al. Real-life effectiveness of canakinumab in cryopyrin-associated periodic syndrome. Rheumatology (Oxford). 2016;55(4):689–96.
Yokota S, Imagawa T, Nishikomori R, Takada H, Abrams K, Lheritier K, et al. Long-term safety and efficacy of canakinumab in cryopyrin-associated periodic syndrome: results from an open-label, phase III pivotal study in Japanese patients. Clin Exp Rheumatol. 2016 [Epub ahead of print, PMID 27974104].
Hoffman HM, Throne ML, Amar NJ, Sebai M, Kivitz AJ, Kavanaugh A, et al. Efficacy and safety of rilonacept (interleukin-1 Trap) in patients with cryopyrin-associated periodic syndromes: results from two sequential placebo-controlled studies. Arthritis Rheum. 2008;58(8):2443–52.
Hoffman HM, Throne ML, Amar NJ, Cartwright RC, Kivitz AJ, Soo Y, et al. Long-term efficacy and safety profile of rilonacept in the treatment of cryopryin-associated periodic syndromes: results of a 72-week open-label extension study. Clin Ther. 2012;34(10):2091–103.
Ozen S, Bilginer Y. A clinical guide to autoinflammatory diseases: familial Mediterranean fever and next-of-kin. Nat Rev Rheumatol. 2014;10(3):135–47.
Bulua AC, Mogul DB, Aksentijevich I, Singh H, He DY, Muenz LR, et al. Efficacy of etanercept in the tumor necrosis factor receptor-associated periodic syndrome: a prospective, open-label, dose-escalation study. Arthritis Rheum. 2012;64(3):908–13.
Nedjai B, Hitman GA, Quillinan N, Coughlan RJ, Church L, McDermott MF, et al. Proinflammatory action of the antiinflammatory drug infliximab in tumor necrosis factor receptor-associated periodic syndrome. Arthritis Rheum. 2009;60(2):619–25.
Gattorno M, Pelagatti MA, Meini A, Obici L, Barcellona R, Federici S, et al. Persistent efficacy of anakinra in patients with tumor necrosis factor receptor-associated periodic syndrome. Arthritis Rheum. 2008;58(5):1516–20.
Gattorno M, Obici L, Cattalini M, Tormey V, Abrams K, Davis N, et al. Canakinumab treatment for patients with active recurrent or chronic TNF receptor-associated periodic syndrome (TRAPS): an open-label, phase II study. Ann Rheum Dis. 2017;76(1):173–8.
Quillinan N, Mannion G, Mohammad A, Coughlan R, Dickie LJ, McDermott MF, et al. Failure of sustained response to etanercept and refractoriness to anakinra in patients with T50M TNF-receptor-associated periodic syndrome. Ann Rheum Dis. 2011;70(9):1692–3.
Torene R, Nirmala N, Obici L, Cattalini M, Tormey V, Caorsi R, et al. Canakinumab reverses overexpression of inflammatory response genes in tumour necrosis factor receptor-associated periodic syndrome. Ann Rheum Dis. 2017;76(1):303–9.
Vaitla PM, Radford PM, Tighe PJ, Powell RJ, McDermott EM, Todd I, et al. Role of interleukin-6 in a patient with tumor necrosis factor receptor-associated periodic syndrome: assessment of outcomes following treatment with the anti-interleukin-6 receptor monoclonal antibody tocilizumab. Arthritis Rheum. 2011;63(4):1151–5.
Akasbi N, Soyfoo MS. Successful treatment of tumor necrosis factor receptor-associated periodic syndrome (TRAPS) with tocilizumab: a case report. Eur J Rheumatol. 2015;2(1):35–6.
Hosoya T, Mizoguchi F, Hasegawa H, Miura K, Koike R, Kubota T, et al. A case presenting with the clinical characteristics of tumor necrosis factor (TNF) receptor-associated periodic syndrome (TRAPS) without TNFRSF1A mutations successfully treated with tocilizumab. Intern Med. 2015;54(16):2069–72.
Simon A, Drewe E, van der Meer JW, Powell RJ, Kelley RI, Stalenhoef AF, et al. Simvastatin treatment for inflammatory attacks of the hyperimmunoglobulinemia D and periodic fever syndrome. Clin Pharmacol Ther. 2004;75(5):476–83.
Vitale A, Rigante D, Lucherini OM, Caso F, Muscari I, Magnotti F, et al. Biological treatments: new weapons in the management of monogenic autoinflammatory disorders. Mediators Inflamm. 2013;2013:939847.
Bodar EJ, van der Hilst JC, Drenth JP, van der Meer JW, Simon A. Effect of etanercept and anakinra on inflammatory attacks in the hyper-IgD syndrome: introducing a vaccination provocation model. Neth J Med. 2005;63(7):260–4.
Kostjukovits S, Kalliokoski L, Antila K, Korppi M. Treatment of hyperimmunoglobulinemia D syndrome with biologics in children: review of the literature and Finnish experience. Eur J Pediatr. 2015;174(6):707–14.
Galeotti C, Meinzer U, Quartier P, Rossi-Semerano L, Bader-Meunier B, Pillet P, et al. Efficacy of interleukin-1-targeting drugs in mevalonate kinase deficiency. Rheumatology (Oxford). 2012;51(10):1855–9.
Shendi HM, Devlin LA, Edgar JD. Interleukin 6 blockade for hyperimmunoglobulin D and periodic fever syndrome. J Clin Rheumatol. 2014;20(2):103–5.
Arkwright PD, Abinun M, Cant AJ. Mevalonic aciduria cured by bone marrow transplantation. N Engl J Med. 2007;357(13):1350.
Neven B, Valayannopoulos V, Quartier P, Blanche S, Prieur AM, Debre M, et al. Allogeneic bone marrow transplantation in mevalonic aciduria. N Engl J Med. 2007;356(26):2700–3.
Chaudhury S, Hormaza L, Mohammad S, Lokar J, Ekong U, Alonso EM, et al. Liver transplantation followed by allogeneic hematopoietic stem cell transplantation for atypical mevalonic aciduria. Am J Transplant. 2012;12(6):1627–31.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Seza Ozen has previously received consultancy and speaker fees from Novartis, Sobi, and R-Pharma. Selcan Demir has no conflicts of interest.
Funding
No sources of funding were used to conduct this study or prepare this manuscript.
Rights and permissions
About this article
Cite this article
Ozen, S., Demir, S. Monogenic Periodic Fever Syndromes: Treatment Options for the Pediatric Patient. Pediatr Drugs 19, 303–311 (2017). https://doi.org/10.1007/s40272-017-0232-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40272-017-0232-6