Abstract
Background
Approximately one-third of patients with epilepsy presents seizures despite adequate treatment. Hence, there is the need to search for new therapeutic options. Cannabidiol (CBD) is a major chemical component of the resin of Cannabis sativa plant, most commonly known as marijuana. The anti-seizure properties of CBD do not relate to the direct action on cannabinoid receptors, but are mediated by a multitude of mechanisms that include the agonist and antagonist effects on ionic channels, neurotransmitter transporters, and multiple 7-transmembrane receptors. In contrast to tetra-hydrocannabinol, CBD lacks psychoactive properties, does not produce euphoric or intrusive side effects, and is largely devoid of abuse liability.
Objective
The aim of the study was to estimate the efficacy and safety of CBD as adjunctive treatment in patients with epilepsy using meta-analytical techniques.
Methods
Randomized, placebo-controlled, single- or double-blinded add-on trials of oral CBD in patients with uncontrolled epilepsy were identified. Main outcomes included the percentage change and the proportion of patients with ≥ 50% reduction in monthly seizure frequency during the treatment period and the incidence of treatment withdrawal and adverse events (AEs).
Results
Four trials involving 550 patients with Lennox–Gastaut syndrome (LGS) and Dravet syndrome (DS) were included. The pooled average difference in change in seizure frequency during the treatment period resulted 19.5 [95% confidence interval (CI) 8.1–31.0; p = 0.001] percentage points between the CBD 10 mg and placebo groups and 19.9 (95% CI 11.8–28.1; p < 0.001) percentage points between the CBD 20 mg and placebo arms, in favor of CBD. The reduction in all-types seizure frequency by at least 50% occurred in 37.2% of the patients in the CBD 20 mg group and 21.2% of the placebo-treated participants [risk ratio (RR) 1.76, 95% CI 1.07–2.88; p = 0.025]. Across the trials, drug withdrawal for any reason occurred in 11.1% and 2.6% of participants receiving CBD and placebo, respectively (RR 3.54, 95% CI 1.55–8.12; p = 0.003) [Chi squared = 2.53, degrees of freedom (df) = 3, p = 0.506; I2 = 0.0%]. The RRs to discontinue treatment were 1.45 (95% CI 0.28–7.41; p = 0.657) and 4.20 (95% CI 1.82–9.68; p = 0.001) for CBD at the doses of 10 and 20 mg/kg/day, respectively, in comparison to placebo. Treatment was discontinued due to AEs in 8.9% and 1.8% of patients in the active and control arms, respectively (RR 5.59, 95% CI 1.87–16.73; p = 0.002). The corresponding RRs for CBD at the doses of 10 and 20 mg/kg/day were 1.66 (95% CI 0.22–12.86; p = 0.626) and 6.89 (95% CI 2.28–20.80; p = 0.001). AEs occurred in 87.9% and 72.2% of patients treated with CBD and placebo (RR 1.22, 95% CI 1.11–1.33; p < 0.001). AEs significantly associated with CBD were somnolence, decreased appetite, diarrhea, and increased serum aminotransferases.
Conclusions
Adjunctive CBD in patients with LGS or DS experiencing seizures uncontrolled by concomitant anti-epileptic treatment regimens is associated with a greater reduction in seizure frequency and a higher rate of AEs than placebo.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Cannabidiol (CBD) has anti-seizure properties and lacks psychoactive effects. |
Adjunctive CBD is effective to reduce seizure frequency in patients with Lennox–Gastaut syndrome and Dravet syndrome. |
The most common adverse events are somnolence, decreased appetite, and increased serum aminotransferases. |
1 Introduction
Epilepsy is one of the most common chronic disorders of the brain. It affects around 65 million people worldwide, has an annual incidence of about 80 cases per 100,000 people and a point prevalence between four and ten cases per 1000 people [1, 2]. The majority of patients with epilepsy can reach sustained remission, while approximately one-third continues to present seizures despite adequate treatment [3,4,5]. Although the number of available therapies, including pharmacological, neuro-modulatory, surgical, and dietary interventions, has dramatically increased, the burden of treatment-resistant epilepsy has remained fairly stable over the years [6,7,8]. Hence, there is still the need to search for new therapeutic options.
The anti-epileptic activity of cannabinoids has attracted much attention in the last decade. Cannabidiol (CBD) is a major chemical component of the resin of Cannabis sativa plant, most commonly known as marijuana. In contrast to tetra-hydrocannabinol, CBD lacks psychoactive properties, does not produce euphoric or intrusive side effects, and is largely devoid of abuse liability [9]. The anti-seizure properties of CBD do not relate to the direct action on cannabinoid receptors, but are mediated by a multitude of mechanisms that include the agonist and antagonist effects on ionic channels, neurotransmitter transporters, and multiple 7-transmembrane receptors [10].
CBD was approved in June 2018 by the US Food and Drug Administration (FDA) as add-on anti-epileptic drug (AED) in patients 2 years of age and older with Dravet syndrome (DS; previously known as severe myoclonic epilepsy of infancy) and Lennox–Gastaut syndrome (LGS). Currently, the drug is under regulatory review with the European Medicines Agency and a decision is expected in the first quarter of 2019 [11].
The aim of our study was to systematically evaluate the efficacy and safety of CBD as adjunctive treatment in patients with epilepsy uncontrolled by concomitant therapy. This could also allow differences in seizure control across the epileptic syndromes to be appreciated and assessment of the tolerability profile on the basis of a larger population dataset.
2 Materials and Methods
2.1 Search Strategy
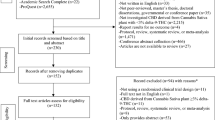
The results of this systematic review and meta-analysis were reported according to the recommendations of the preferred reporting items for systematic reviews and meta-analyses (PRISMA) statement [12]. We systematically searched MEDLINE (accessed by PubMed), the Cochrane Central Register of Controlled Trials (CENTRAL), and the US National Institutes of Health Clinical Trials Registry (http://www.clinicaltrials.gov); the searches were run in week 4 of June 2018 (search strategies are outlined in the Electronic Supplementary Material). There were no date limitations or language restrictions. The reference lists of retrieved studies were reviewed to identify additional reports of relevant trials. The protocol was not registered previously.
2.2 Eligibility Criteria
Studies were selected when they met the following entry criteria: randomized, double- or single-blinded, placebo-controlled, parallel-group add-on studies with active and control arms receiving oral CBD and matched placebo, respectively, in addition to an existing AED regimen at stable dose at the time of randomization. Participants had to meet the following criteria: any sex, any ethnicity, pediatric and/or adult age, diagnosis of epilepsy, and seizures uncontrolled by concomitant AEDs.
2.3 Outcome Measures
The efficacy outcomes were the percentage change and the proportion of patients with ≥ 50% reduction in monthly frequency of all types, convulsive (tonic, clonic, tonic–clonic, or atonic) and non-convulsive (myoclonic, focal, or absence) seizures from the pre-randomization baseline during the treatment period. The safety and tolerability outcomes included the proportions of participants who withdrew from treatment for any reason and for adverse events (AEs) and who experienced any AE, any treatment-related AE, any serious AE (SAE), and any treatment-related SAE. We also assessed the occurrence of the following AEs: somnolence, decreased appetite, diarrhea, fatigue, and increase in serum aminotransferase concentrations by three-fold or greater than the upper limit of the normal range (chosen by the review authors as found to be commonly related to CBD in the open-label Expanded Access Program [13]), and the five most frequent AEs in either trial group, if different from those listed already. Changes from baseline to the end of treatment in measures of global functioning, including Patient or Caregiver Global Impression of Change (P/CGIC), sleep disruption, daytime sleepiness, quality of life, and behavioral adaptation, as assessed by validated scales, were also reviewed.
2.4 Study Selection, Data Extraction, and Assessment of the Risk of Bias
Two review authors (SL and CC) independently assessed trials for inclusion and extracted the following information from included studies: main study author and age of publication, methodology and trial design (methods of randomization, allocation concealment and blinding, duration of baseline and treatment periods, dose(s) of CBD tested), number and demographics of participants (age, sex, ethnicity, number of previous and background AEDs, seizure frequency during the baseline period), change in baseline frequency of all/convulsive/non-convulsive seizures during treatment, and number of participants experiencing each outcome per randomized group. Any disagreement was resolved by discussion with a third review author (FB). The risk of bias of the identified studies was assessed in accordance with the recommendations of the Cochrane Collaboration [14].
2.5 Statistical Analysis
Dichotomous data were pooled through the inverse variance method and risk estimates synthesized by the risk ratios (RRs). Continuous outcome measures were analyzed by the generic inverse variance model as the overall estimates of effect from each study rather than summary data for each intervention group were available [14]. Differences in percentage changes in seizure frequencies between treatments were provided in original papers as medians [95% confidence intervals (CIs)], and approximation to normality was assumed due to the comparability between given values and estimated means. We assessed heterogeneity among the trials by using the Chi squared (χ2) test and the I2 statistics for heterogeneity [15, 16]. Provided no substantial heterogeneity was present (p > 0.10), results were synthesized using a fixed effect model; if the probability value was ≤ 0.10, heterogeneity determined the choice of a fixed- or random effects model for I2 < 40% or ≥ 40%, respectively [17,18,19,20,21,22]. We presented heterogeneity statistics for all analyses unless only one trial contributed data and heterogeneity was not applicable. The intent-to-treat (ITT) population data were used for the analyses. Results were presented according to CBD daily doses, where sufficient data were available. Data analysis was performed using STATA®/IC 13.1 (StataCorp LP, College Station, TX, USA).
3 Results
3.1 Results of the Search and Characteristics of Included Studies
One hundred and sixty-four records were identified by database and trial registers searching (Fig. 1). A total of four trials were eventually included in the review and meta-analysis. One study aimed to investigate the dose-ranging safety of add-on CBD in patients with DS and contributed only to safety outcomes [23]; three trials assessed either efficacy or safety of adjunctive CBD and contributed to both efficacy and safety analyses [24,25,26]. Details of the studies are provided in Table 1. In all trials, the active treatment was a plant-derived pharmaceutical formulation of purified CBD oral solution (100 mg/mL) (Epidiolex®, GW Pharmaceuticals), which was added twice daily to the pre-existing anti-epileptic regimen. The studies included 550 participants according to the ITT: 323 for CBD and 227 for placebo groups. Characteristics of the participants are summarized in Table 2. Patients with LGS were older, had a higher number of background AEDs, and higher total seizure frequency at baseline than patients with DS.
All trials used adequate methods of sequence generation and allocation concealment. We rated all included trials as having low risk of performance and detection bias since blinding was ensured by matching placebo, and neither the investigators nor the patients knew the identity of the study treatment being administered. The risks of attrition and selective reporting bias were judged low since patients lost to follow-up and withdrawals were documented, and there was no suspicion of selective outcome reporting. All trials were sponsored by the CBD manufacturer.
3.2 Change in Baseline Seizure Frequency
The pooled average difference in change in seizure frequency during the treatment period resulted 19.5 (95% CI 8.1–31.0; p = 0.001) percentage points between the CBD 10 mg and placebo groups and 19.9 (95% CI 11.8–28.1; p < 0.001) percentage points between the CBD 20 mg and placebo arms, in favor of CBD (Fig. 2a). Adjunctive CBD either at 10 or 20 mg/kg/day was associated with a significantly greater decrease in baseline frequency of convulsive seizures than the inactive treatment (Fig. 2b). There was no significant difference in the reduction of non-convulsive seizures frequency between the CBD 10 mg and placebo groups. High-dose CBD was overall more effective to reduce non-convulsive seizures frequency than placebo, but the effect did not reach statistically significance among patients with DS (Fig. 2c).
Percentage change in baseline seizure frequency: a all-types seizures; b convulsive seizures; and c non-convulsive seizures. Asterisk estimated median differences between cannabidiol and placebo groups in percentage changes from baseline in seizure frequency during the treatment period were pooled by the generic inverse variance model. CBD cannabidiol, CI confidence interval, df degrees of freedom
The reduction in all-types seizure frequency by at least 50% occurred in 37.2% of the patients in the CBD 20 mg group and 21.2% of the placebo-treated participants (RR 1.76, 95% CI 1.07–2.88; p = 0.025). The estimated RRs to achieve a 50% or greater reduction in frequency of convulsive and non-convulsive seizures for the CBD 20 mg group in comparison to placebo were 1.75 (95% CI 1.23–2.48; p = 0.002) [χ2 = 0.32, degrees of freedom (df) = 1, p = 0.575; I2 = 0.0%] and 1.42 (95% CI 0.95–2.11; p = 0.086), respectively (Table 3). Data on seizure response were not available for the CBD 10 mg daily dose and seizures other than convulsive in patients with DS.
3.3 Treatment Withdrawal
Across the trials, drug withdrawal for any reason occurred in 11.1% and 2.6% of participants receiving CBD and placebo, respectively (RR 3.54, 95% CI 1.55–8.12; p = 0.003) (χ2 = 2.53, df = 3, p = 0.506; I2 = 0.0%). The RRs to discontinue treatment were 1.45 (95% CI 0.28–7.41; p = 0.657) and 4.20 (95% CI 1.82–9.68; p = 0.001) for CBD at the doses of 10 and 20 mg/kg/day, respectively, in comparison to placebo. Treatment was discontinued due to AEs in 8.9% and 1.8% of patients in the active and control arms, respectively (RR 5.59, 95% CI 1.87–16.73; p = 0.002). The corresponding RRs for CBD at the doses of 10 and 20 mg/kg/day were 1.66 (95% CI 0.22–12.86; p = 0.626) and 6.89 (95% CI 2.28–20.80; p = 0.001) (Table 4). In the GWPCARE3 trial [25], six patients in the CBD 10 mg group temporarily received a dose that was above the target and were included in the CBD 20 mg group for the safety analysis.
3.4 Adverse Events
AEs were reported in 87.9% and 72.2% of the patients during treatment with CBD and placebo, respectively (RR 1.22, 95% CI 1.11–1.33; p < 0.001) (χ2 = 3.39, df = 3, p = 0.336; I2 = 11.5%). The treatment with CBD was associated with a higher incidence of treatment-related AEs (55.7% vs. 26.9%), SAEs (18.6% vs. 6.7%), and treatment-related SAEs (7.7% vs. 0.4%) in comparison to placebo (Table 5). One death unrelated to treatment occurred in the CBD 20 mg/kg group due to respiratory failure [26]; there were no deaths among the placebo-treated patients.
The incidence rates of selected AEs in the CBD- versus placebo-treated participants were as follows: somnolence 24.5% versus 8.4%, decreased appetite 20.1% versus 4.8%, diarrhea 18.2% versus 8.6%, fatigue 7.5% versus 4.0%, increased alanine or aspartate aminotransferases more than three times the upper normal limit 16.1% versus 0.9%, upper respiratory tract infection 10.5% versus 10.0%, pyrexia 13.0% versus 10.6%, vomiting 10.8% versus 11.5%, and sedation 9.7% versus 1.1%. The AEs significantly associated with CBD in the overall analysis were somnolence, decreased appetite, diarrhea, and increased transaminases levels (Table 5). The results by daily dosages are reported in Table 6.
3.5 Global Functioning Measures
The improvement (slightly, much, or very much improved) in baseline status according to the P/CGIC at the last visit was reported in 177 of 292 (60.3%) and 82 of 218 (38.8%) patients in the CBD and placebo arms, respectively (RR 1.57, 95% CI 1.30–1.91; p < 0.001) (χ2 = 1.27, df = 2, p = 0.531; I2 = 0.0%). Compared with placebo, the RRs for P/CGIC improvement were 1.49 (95% CI 1.10–2.03; p = 0.010) and 1.55 (95% CI 1.26–1.90; p < 0.001] (χ2 = 1.90, df = 2, p = 0.386; I2 = 0.0%) for CBD at the daily dosages of 10 and 20 mg/kg/day, respectively. There were no significant differences between the treatments in changes from baseline to the end of treatment in scores evaluating sleep disruption, daytime sleepiness, quality of life, and behavioral adaption (Table 7).
4 Discussion
4.1 Summary of Main Results
Three randomized controlled trials provided substantial evidence of the effectiveness of the pharmaceutical formulation of purified oral CBD to treat seizures when added to other anti-epileptic therapies in patients with LGS or DS [24,25,26].
Adjunctive CBD showed a broad-spectrum anti-epileptic profile and improved the control of either convulsive or non-convulsive seizures in patients with LGS. In patients with DS, the treatment with CBD was not associated with a greater reduction of non-convulsive seizures than placebo. These findings suggest that the anti-seizure activity of CBD may be specific to convulsive seizures in DS or the trial was underpowered to detect a difference between arms in the change in non-convulsive seizures frequency, which was a secondary endpoint and not part of the primary efficacy assessment [23]. It is also to be noted that non-convulsive seizures could not reliably be counted by caregivers in developmentally delayed children.
The global impression of change is an endpoint used in epilepsy studies to provide information on the clinical relevance of decline in seizure frequency [24]. Findings from the P/CGIC questionnaire showed that a significantly higher proportion of patients and caregivers in the CBD than placebo group perceived the patients’ condition as improved. The high rate of enrollment into the open-label extension phase of the blinded trials (GWPCARE5 trial; ClinicalTrials.gov identifier NCT02224573) further supported the overall positive effect of the treatment with CBD.
Clinically meaningful and statistically significant improvements were observed in a very difficult-to-treat population. Both LGS and DS are severe forms of epilepsy, and the inclusion criteria ensured the recruitment of highly treatment-resistant patients: at baseline, they were taking an average of three concomitant AEDs, had a history of exposure to four or more anti-epileptic treatments, and presented a high seizure frequency. Patients with better disease control were enrolled only in the GWPCARE1-part A trial to avoid the need to change dosages of concomitant AEDs and, hence, monitor the dose-ranging safety rather than efficacy of CBD [23]. In this context of difficult-to-treat epilepsy, it is worth noting that 5.5% of all patients with LGS randomized to CBD were reported to be free from drop seizures during the maintenance period in comparison with 0.6% in the placebo group [27], and 5% of the participants with DS treated with CBD attained complete freedom from convulsive seizures versus none in the inactive-treatment arm [24]. Genetic variations may explain the remarkable response observed in a minority of patients despite their very refractory status and should deserve further exploration.
Although there were no sufficient data to undertake a regression analysis, the positive dose–response correlation may be suggested by the greater reduction observed in non-convulsive seizure frequency with CBD at the daily dose of 20 mg/kg rather than 10 mg/kg. Furthermore, in the pooled analysis of the GWPCARE3 and GWPCARE4 trials there was evidence of a significant increase in the responder rate for drop seizures with increasing exposure of either CBD or its active metabolite 7-hydroxy-CBD, and the predicted probability of response resulted in the order of 80% for the highest values of the area under the plasma drug concentration–time curve [28]. Therefore, in patients who are tolerating CBD at 10 mg/kg/day and require further reduction of seizures, titration up to the maximum recommended maintenance dose of 20 mg/kg can be considered. Dose is advised to be increased through weekly increments of 2.5 mg/kg twice daily and no more frequently than every other day for patients needing a more rapid up-titration [29].
The overall rates of patients withdrawing CBD for any reason and AEs were similar or lower than those associated with other AEDs in LGS or DS [30,31,32], and a meaningful difference with placebo emerged when CBD was administered at the higher daily dosage. The most common AEs associated with CBD were somnolence, decreased appetite, and diarrhea. They were mostly mild to moderate in severity, dosage related, and consistent with the tolerability profile emerged during the open-label administration of CBD in severe refractory epilepsies [13]. There were no AEs related to the so-called stoned-like effects and no discernible influences on sleep and behavioral domains such as language, socialization, self-care, and motor skills.
CBD was causally associated with increases of serum transaminases and a signal for hepatic toxicity was identified. Most transaminases elevations occurred in the first 30 days, almost all in the first 90 days of treatment, and only a few after 100 days; the risk window was wider for patients taking concomitant valproate [33]. All cases resolved either spontaneously during the treatment period or open-label extension trial, or after the dose of a concomitant AED was reduced, or after CBD was tapered or discontinued. Transaminases elevations represented the most frequent SAE and accounted for half of the drug discontinuations across the phase III trials. However, no events suggested lasting liver damage and no patients met the criteria for severe drug-induced liver injury as coincidentally substantial rises of serum bilirubin did not occur (Hy’s law) [34]. Close monitoring of liver enzyme levels and signs or symptoms suggestive of hepatic dysfunction, above all during the initial stages of treatment, is highly recommended to appropriately manage the risk of liver toxicity.
4.2 Pharmacological Interactions
Pharmacological interactions can explain some effects associated with CBD and have relevant implications in clinical practice. Somnolence, which was the most frequent AE observed during CBD treatment, had a higher incidence among patients concomitantly taking clobazam and reduction of clobazam dosage occurred more frequently in CBD- than placebo-treated participants. Notably, CBD can inhibit the catalytic activity of the cytochrome P450 (CYP) 2C19 and determine a three-fold increase in plasma concentrations of N-desmethylclobazam (N-CLB), the biologically active metabolite of clobazam [23, 35]. In parallel, clobazam can raise the exposure to 7-hydroxy-CBD through the inhibition of CYP2D6. Therefore, it would be advisable to strictly observe patients on concomitant clobazam and adjust doses as necessary to manage AEs. The elevation in N-CLB levels did not occur in patients taking stiripentol, which is a potent CYP2C19 inhibitor, suggesting prior saturation of the cytochrome isoenzyme [23].
Given the pharmacokinetics of CBD and clobazam, the influence of clobazam co-administration on CBD efficacy might be also hypothesized. In this respect, the pooled post hoc analysis of the GWPCARE3 and GWPCARE4 trials revealed significant reductions in drop seizures frequency with add-on CBD versus add-on placebo, regardless of concomitant use of clobazam. Despite the limits inherent to the re-assessment of non-randomized subgroups differing in baseline characteristics, these data provided further support for an intrinsic anti-seizure effect independent of baseline medication [36].
Concomitant valproate emerged as the most common risk factor for hepatic injury during CBD treatment. More than two-thirds of the patients presenting transaminases elevations were co-medicated with valproate, and some cases resolved while on CBD after the decrease in dose of concomitant valproate. Therefore, in addition to the hepatotoxic profile of CBD alone, there can be an additive toxic effect when the AEDs are combined. The interaction between these two drugs has been shown at the level of hepatic mitochondria in vitro [33], and it could represent the mechanism underlying the clinical observations. The combined effects of CBD and valproate on liver injury signaling pose an important clinical challenge and further investigations may be required with regard to agent-specific sequencing of dose adjustment, drug discontinuation, and patient observation when monitoring indicates acute hepatotoxicity. The co-administration of CBD with clobazam or felbamate has been also associated with slightly increased incidence of transaminases elevations, which, however, appeared to be driven by concomitant valproate.
4.3 Strengths and Limitations
This meta-analysis is the most comprehensive and updated quantitative synthesis of the efficacy and safety of CBD in patients with epilepsy published so far. In comparison to studies that previously addressed the same question [37, 38], this analysis pooled a larger population, assessed drug efficacy according to seizure subtypes and daily dosages, and also evaluated global functioning endpoints. The included studies had similar design characteristics and the low between-trial heterogeneity strengthened the accuracy of estimates [39]. Nonetheless, shortcomings should be considered when interpreting the findings. Only four trials met the eligibility criteria, and a single pharmaceutical company funded all of them. The ethnic diversity of the trials’ population was low, with more than 90% of patients being Caucasian, efficacy of CBD administered at 10 mg/kg/day was explored in one study [25], and seizure response rates were available only for the higher drug dose. Due to the limited treatment periods of the trials, conclusions on long-term therapy cannot be drawn. In this respect, sustained reductions in seizure frequency and improvements in overall condition up to 60 weeks have been observed in pre-specified interim analyses of the open-label extension of the randomized studies [40, 41]; add-on CBD continued to be generally well-tolerated and the reduction in seizure frequency remained stable through 96 weeks among patients with treatment-resistant epilepsies enrolled in the Expanded Access Program [42]. Additional data from post-marketing surveillance will, however, be needed to evaluate the incidence of rare AEs and the long-term effects of continuous CBD exposure on growth, childbearing potential, brain development, and learning. Likewise, this meta-analysis cannot provide information on the effectiveness of CBD in comparison with other AEDs, as monotherapy, and in epilepsies other than LGS and DS.
4.4 Conclusions
The approval of CBD represents a milestone in the history of medical use of cannabinoids to treat seizure disorders. CBD is the first FDA-authorized product made directly from the cannabis plant rather than created synthetically, and the first in a new class of AEDs. Randomized clinical trials are ongoing to evaluate CBD in orphan seizure disorders such as tuberous sclerosis complex and infantile spasms, and controlled studies in other types of epilepsy, including refractory focal epilepsy, may be warranted to fully determine its therapeutic potentialities.
References
Hirtz D, Thurman DJ, Gwinn-Hardy K, Mohamed M, Chaudhuri AR, Zalutsky R. How common are the “common” neurologic disorders? Neurology. 2007;68:326–37.
Cagnetti C, Lattanzi S, Foschi N, Provinciali L, Silvestrini M. Seizure course during pregnancy in catamenial epilepsy. Neurology. 2014;83:339–44.
Cockerell OC, Johnson AL, Sander JW, Hart YM, Shorvon SD. Remission of epilepsy: results from the national general practice study of epilepsy. Lancet. 1995;346:140–4.
Lattanzi S, Zaccara G, Giovannelli F, Grillo E, Nardone R, Silvestrini M, et al. Antiepileptic mono-therapy in newly diagnosed focal epilepsy. A network meta-analysis. Acta Neurol Scand. https://doi.org/10.1111/ane.13025 (Epub 2018 Sep 8).
Lattanzi S, Cagnetti C, Foschi N, Provinciali L, Silvestrini M. Lacosamide monotherapy for partial onset seizures. Seizure. 2015;27:71–4.
Kwan P, Brodie MJ. Early identification of refractory epilepsy. N Engl J Med. 2000;342:314–9.
Lattanzi S, Cagnetti C, Foschi N, Lorusso A, Provinciali L, Silvestrini M. Eslicarbazepine acetate as adjunctive treatment in partial-onset epilepsy. Acta Neurol Scand. 2018;137:29–32.
Lattanzi S, Cagnetti C, Matricardi S, Silvestrini M. Palliative non-resective surgery for drug-resistant epilepsy. Brain Dev. 2018;40:512–3.
Devinsky O, Cilio MR, Cross H, Fernandez-Ruiz J, French J, Hill C, et al. Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia. 2014;55:791–802.
Ibeas Bih C, Chen T, Nunn AV, Bazelot M, Dallas M, Whalley BJ. Molecular targets of cannabidiol in neurological disorders. Neurotherapeutics. 2015;12:699–730.
GW Pharmaceuticals plc and its U.S. subsidiary Greenwich Biosciences announce FDA approval of EPIDIOLEX® (cannabidiol) oral solution—the first plant-derived cannabinoid prescription medicine. https://www.gwpharm.com/about-us/news/gw-pharmaceuticals-plc-and-its-us-subsidiary-greenwich-biosciences-announce-fda. Accessed Sep 2018.
Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097.
Devinsky O, Marsh E, Friedman D, Thiele E, Laux L, Sullivan J, et al. Cannabidiol in patients with treatment-resistant epilepsy: an open-label interventional trial. Lancet Neurol. 2016;15:270–8.
Cochrane handbook for systematic reviews of interventions. Version 5.1.0 [updated March 2011]. Higgins JPT, Green S, editors. The Cochrane Collaboration, 2011. http://handbook-5-1.cochrane.org/. Accessed Jun 2018.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–60.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–58.
Lattanzi S, Cagnetti C, Foschi N, Provinciali L, Silvestrini M. Brivaracetam add-on for refractory focal epilepsy: a systematic review and meta-analysis. Neurology. 2016;86:1344–52.
Lattanzi S, Brigo F, Grillo E, Cagnetti C, Verrotti A, Zaccara G, et al. Adjunctive eslicarbazepine acetate in pediatric patients with focal epilepsy: a systematic review and meta-analysis. CNS Drugs. 2018;32:189–96.
Lattanzi S, Grillo E, Brigo F, Silvestrini M. Efficacy and safety of perampanel in Parkinson’s disease. A systematic review with meta-analysis. J Neurol. 2018;265:733–40.
Lattanzi S, Cagnetti C, Danni M, Provinciali L, Silvestrini M. Oral and intravenous steroids for multiple sclerosis relapse: a systematic review and meta-analysis. J Neurol. 2017;264:1697–704.
Lattanzi S, Brigo F, Cagnetti C, Di Napoli M, Silvestrini M. Patent foramen ovale and cryptogenic stroke or transient ischemic attack: to close or not to close? A systematic review and meta-analysis. Cerebrovasc Dis. 2018;45:193–203.
Lattanzi S, Brigo F, Di Napoli M, Cagnetti C, Corradetti T, Silvestrini M. Endovascular treatment of symptomatic vertebral artery stenosis: a systematic review and meta-analysis. J Neurol Sci. 2018;391:48–53.
Devinsky O, Patel AD, Thiele EA, Wong MH, Appleton R, Harden CL, et al.; GWPCARE1 Part A Study Group. Randomized, dose-ranging safety trial of cannabidiol in Dravet syndrome. Neurology. 2018;90:e1204–11.
Devinsky O, Cross JH, Laux L, Marsh E, Miller I, Nabbout R, et al.; Cannabidiol in Dravet Syndrome Study Group. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N Engl J Med. 2017;376:2011–2020.
Devinsky O, Patel AD, Cross JH, Villanueva V, Wirrell EC, Privitera M, et al.; GWPCARE3 Study Group. Effect of cannabidiol on drop seizures in the Lennox–Gastaut syndrome. N Engl J Med. 2018;378:1888-1897.
Thiele EA, Marsh ED, French JA, Mazurkiewicz-Beldzinska M, Benbadis SR, Joshi C, et al.; GWPCARE4 Study Group. Cannabidiol in patients with seizures associated with Lennox–Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2018;391:1085–96.
Lattanzi S, Brigo F, Cagnetti C, Trinka E, Silvestrini M. Efficacy and safety of adjunctive cannabidiol in patients with Lennox–Gastaut syndrome: a systematic review and meta-analysis. CNS Drugs. https://doi.org/10.1007/s40263-018-0558-9 (Epub 2018 Aug 21).
Morrison G, Sardu M, Rasmussen C, Sommerville K, Roberts C, Blakey GE. Exposure-response analysis of cannabidiol oral solution for the treatment of Lennox–Gastaut syndrome [abstract no. 2.281]. The American Epilepsy Society Annual Meeting; 1–5 Dec 2017; Washington, DC. https://www.aesnet.org/meetings_events/annual_meeting_abstracts/view/344717. Accessed Jul 2018.
FDA. Cannabidiol oral solution. Full prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/210365lbl.pdf. Accessed Jul 2018.
Ng YT, Conry JA, Drummond R, Stolle J, Weinberg MA; OV-1012 Study Investigators. Randomized, phase III study results of clobazam in Lennox–Gastaut syndrome. Neurology. 2011;77:1473–81.
Glauser T, Kluger G, Sachdeo R, Krauss G, Perdomo C, Arroyo S. Rufinamide for generalized seizures associated with Lennox–Gastaut syndrome. Neurology. 2008;70:1950–8.
Chiron C, Tonnelier S, Rey E, Brunet ML, Tran A, d’Athis P, et al. Stiripentol in childhood partial epilepsy: randomized placebo-controlled trial with enrichment and withdrawal design. J Child Neurol. 2006;21:496–502.
FDA briefing document. Peripheral and central nervous system drugs. Advisory Committee Meeting. April 19, 2018. NDA 210365. Cannabidiol. https://www.fda.gov/downloads/advisorycommittees/committeesmeetingmaterials/drugs/peripheralandcentralnervoussystemdrugsadvisorycommittee/ucm604736.pdf. Accessed Jul 2018.
US Department of Health and Human Services. Guidance for industry. Drug-induced liver injury: premarketing clinical evaluation. 2009. https://www.fda.gov/downloads/Guidances/UCM174090.pdf. Accessed Jul 2018.
Geffrey AL, Pollack SF, Bruno PL, Thiele EA. Drug-drug interaction between clobazam and cannabidiol in children with refractory epilepsy. Epilepsia. 2015;56:1246–51.
Thiele EA, Devinsky O, Checketts D, Knappertz V. Cannabidiol (CBD) treatment responders analysis in patients with Lennox–Gastaut syndrome (LGS) on and off clobazam (CLB) [abstract no. 1.436]. The American Epilepsy Society Annual Meeting; 1–5 Dec 2017; Washington, DC. https://www.aesnet.org/meetings_events/annual_meeting_abstracts/view/381224. Accessed Jul 2018.
Gloss D, Vickrey B. Cannabinoids for epilepsy. Cochrane Database Syst Rev. 2014;(3):CD009270.
Stockings E, Zagic D, Campbell G, Weier M, Hall WD, Nielsen S, et al. Evidence for cannabis and cannabinoids for epilepsy: a systematic review of con-trolled and observational evidence. J Neurol Neurosurg Psychiatry. 2018;89:741–53.
Rhodes KM, Turner RM, Savović J, Jones HE, Mawdsley D, Higgins JPT. Between-trial heterogeneity in meta-analyses may be partially explained by reported design characteristics. J Clin Epidemiol. 2018;95:45–54.
Devinsky O, Nabbout R, Miller I, Laux L, Zolnowska M, Wright S, et al. Maintenance of long-term safety and efficacy of cannabidiol (CBD) treatment in Dravet syndrome (DS): results of the open-label extension (OLE) trial (GWPCARE5) [abstract no. 1.289]. The American Epilepsy Society Annual Meeting; 1–5 Dec 2017; Washington, DC. https://www.aesnet.org/meetings_events/annual_meeting_abstracts/view/343046. Accessed Jul 2018.
Marsh ED, Mazurkiewicz-Beldzinska M, Halford JJ, Gunning B, Checketts D, Nichol K, et al. Maintained safety and efficacy of cannabidiol (CBD) in a long-term open-label trial in patients with Lennox–Gastaut syndrome (LGS) (GWPCARE 5) [abstract no. 2.271]. The American Epilepsy Society Annual Meeting; 1–5 Dec 2017; Washington, DC. https://www.aesnet.org/meetings_events/annual_meeting_abstracts/view/344387. Accessed Jul 2018.
Szaflarski JP, Bebin EM, Comi AM, Patel AD, Joshi C, Checketts D, et al.; CBD EAP study group. Long-term safety and treatment effects of cannabidiol in children and adults with treatment-resistant epilepsies: expanded access program results. Epilepsia. 2018;59(8):1540–8. https://doi.org/10.1111/epi.14477.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding has been received for the conduct of this study.
Conflict of interest
Simona Lattanzi, Claudia Cagnetti, Cinzia Del Giovane and Mauro Silvestrini have no conflicts of interest directly relevant to the content of this study. Francesco Brigo acted as a consultant for Eisai. Eugen Trinka received speaker’s honoraria from UCB, Biogen, Gerot-Lannach, Bial, Eisai, Takeda, Newbridge, Sunovion Pharmaceuticals Inc., LivaNova, and Novartis; consultancy funds from UCB, Biogen, Gerot-Lannach, Bial, Eisai, Takeda, Newbridge, GW Pharmaceuticals, Sunovion Pharmaceuticals Inc., and Novartis; and directorship funds from Neuroconsult GmbH. Eugen Trinka’s institution received grants from Biogen, Red Bull, Merck, UCB, the European Union, FWF Österreichischer Fond zur Wissenschaftsförderung, and Bundesministerium für Wissenschaft und Forschung. Gaetano Zaccara received speaker’s or consultancy fees from Eisai, Sanofi-Aventis, and UCB Pharma.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lattanzi, S., Brigo, F., Trinka, E. et al. Efficacy and Safety of Cannabidiol in Epilepsy: A Systematic Review and Meta-Analysis. Drugs 78, 1791–1804 (2018). https://doi.org/10.1007/s40265-018-0992-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-018-0992-5