Abstract
Introduction
It has been suggested that fluoroquinolone antibiotics increase the risk of developing collagen-associated adverse events such as aortic dissection and aortic aneurysm. These are life-threatening emergencies that need to be prevented.
Objectives
We performed this systematic review to clarify the association between fluoroquinolones and three collagen-associated adverse events: aortic aneurysm or aortic dissection, retinal detachment, and tendon disorders.
Methods
We searched PubMed, Embase, and Scopus for observational studies up to January 2019. Cohort and case–control studies were included if they reported data on the risk of collagen-related adverse events associated with fluoroquinolone exposure versus no exposure. We assessed the quality of the included studies using the Newcastle–Ottawa Scale. Effect statistics were pooled using random-effects models. Sensitivity and subgroup analyses were performed to identify any source of heterogeneity.
Results
After screening 2729 citations, we included 22 observational studies (12 cohort studies and ten case–control studies) with 19,207,552 participants. Current use of fluoroquinolones was significantly associated with aortic aneurysm and aortic dissection (odds ratio [OR] 2.20; 95% confidence interval [CI] 1.92–2.52), tendon disorders (OR 1.89; 95% CI 1.53–2.33), and retinal detachment (sensitivity analysis, OR 1.25; 95% CI 1.01–1.53). Past fluoroquinolone use (> 30 and ≤ 365 days) was associated with retinal detachment (OR 1.27; 95% CI 1.09–1.47).
Conclusions
Fluoroquinolone use incurs a risk of developing three collagen-associated diseases (aortic aneurysm or aortic dissection, retinal detachment, and tendon disorders). Patients at an increased risk of collagen-associated diseases should not use fluoroquinolones unless no other options are available.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Fluoroquinolone use has a positive relationship with the risk of three kinds of collagen-associated disorders: tendon disorders, retinal detachment, and aortic aneurysm or dissection. |
Fluoroquinolones should be used carefully in patients at high risk of collagen-associated diseases. Fluoroquinolones may promote the development of such diseases in this population. |
1 Introduction
Fluoroquinolones remain the most commonly prescribed broad-spectrum antibiotics, even though patients can experience rare adverse effects [1, 2]. They are used to treat various infections, including urinary tract, respiratory, skin, bone, and joint infections [3, 4]. Since the 1980s, a considerable number of articles have reported fluoroquinolone-associated tendinopathy [5,6,7,8,9,10,11,12,13,14,15,16,17], which was included in a black-box warning in 2008 [18]. Over the last decade, several observational studies have suggested that fluoroquinolone use is associated with an increased risk of retinal detachment [19,20,21,22] and aortic aneurysm or aortic dissection (AAD) [2, 23, 24]. These collagen-degeneration diseases can significantly affect the lives of patients. The collagen-associated adverse events are considered involve the non-antimicrobial properties of fluoroquinolones via activation of matrix metalloproteinases (MMPs) and oxidative stress in tendon cells [25, 26]. Biological experiments in rabbits have provided evidence for retinal damage and alterations in antioxidant status with fluoroquinolone injections [27]. Collagen loss and tissue degeneration are common pathological processes in these collagen-related disorders [23], which could be caused by fluoroquinolones [26, 28]. Therefore, the relationship between fluoroquinolones and collagen-related diseases must be elucidated.
An increasing number of clinical studies support the relationship between retinal detachment and fluoroquinolone use [21, 29, 30], which was originally proposed in a population-based pharmacoepidemiologic study in 2012 [19], but results have differed. Recently, a propensity-matched cohort study using a large national database in Sweden reported a hazard ratio (HR) of 1.66 (95% confidence interval [CI] 1.12–2.46) for an increased risk of AAD with fluoroquinolone prescription [2].
However, the relationship between fluoroquinolone antibiotics and collagen-associated events remains controversial. In this study, we summarize the available observational evidence in a systematic review and meta-analyses to quantify the association between fluoroquinolone use and AAD, retinal detachment, and tendon disorders.
2 Methods
2.1 Definition
Collagen-associated adverse events (AAD, retinal detachment, and tendon disorders) are defined according to the International Classification of Diseases and Related Health Problems (ICD) and other coding systems [19, 23, 31]. Every collagen-associated event consists of several diseases that share similar symptoms and correlative pathogenesis (e.g., collagen degeneration). Table A1 in the Electronic Supplementary Material (ESM) details the diagnosis codes for these diseases.
2.2 Search Strategy
This systematic review and meta-analysis was not registered in the PROSPERO database. We conducted this study following the 2009 PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines [32]. We searched PubMed, EMBASE, and Scopus from inception to 25 January 2019 with no data or language limitations. We identified observational studies that evaluated the association between fluoroquinolone use and risks of AAD, retinal detachment, and tendon disorders. Our strategy included a combination of search terms, the details of which are included in the ESM. We manually checked original articles and removed duplicates. References of included studies were also searched for potential studies.
2.3 Selection Criteria
We included cohort or case–control studies that reported on the risk of tendon disorders, retinal detachment, and AAD in fluoroquinolone users compared with non-users. The effect sizes in the final results could be presented as relative risks (RRs), ORs, or HRs, and we required corresponding 95% CIs to be provided. We set no limits on the duration of follow-up in the analysis of AAD and retinal detachment. When we conducted the meta-analysis of fluoroquinolone use and tendon disorders, we only included studies that reported results with a follow-up duration of < 90 days, since most studies only reported the current use of fluoroquinolones (< 90 days).
2.4 Study Selection and Data Extraction
After duplicates were removed, two investigators (XY and DJ) reviewed the titles and abstracts of articles and excluded irrelevant studies. In the second review, the same investigators retrieved the abstracts and full reports and included potentially eligible studies, then included studies that met the inclusion criteria and provided qualified data. Disagreement was resolved in discussions with a third reviewer (SC).
Two reviewers (XY and DJ) independently extracted the following data from retrieved articles: study design, participants, exposures, outcomes, controls, specific fluoroquinolone use, risk rates, and corresponding 95% CIs. Adjusted rates were preferentially selected if available. Disagreements were resolved through discussion and by consulting a third investigator (SC). If included articles included more than one study, we analyzed each study as a single result and marked the results accordingly. We extracted effect estimates for different follow-up durations if studies included multiple reports. Study authors were not contacted for more information.
2.5 Quality Appraisal
We used the Newcastle–Ottawa Scale 10 [33], a scoring system used to assess the quality of non-randomized studies in terms of design, content, and ease of conducting a meta-analysis, to appraise the included articles. Studies were rated in three areas (selection of groups, comparability, and ascertainment of outcome and exposure). All items were independently assessed by two investigators (XY and DJ), with consensus reached after group discussion.
2.6 Statistical Analysis
Because the incidences for AAD, retinal detachment, and tendon disorders were low in the general population, we regarded RRs as approximations of ORs and merged them with HRs, producing a common estimated effect as an OR. We conducted random-effect meta-analyses to evaluate the combined estimates, and the I2 statistic was used to evaluate between-study heterogeneity. Outcomes for different follow-up durations were meticulously extracted, and separate results were pooled with a fixed-effect inverse variance method if necessary (e.g., when we analyzed a result for fluoroquinolone use in 1 month, we also combined the results for 1–10 days and 11–30 days). We conducted sensitivity analyses with a “leave-one-out” strategy to investigate the sources of heterogeneity in each meta-analysis. We performed subgroup analyses to evaluate potential factors that may have influenced pooled results. We performed a funnel plot, Begg’s test, and Egger’s test to assess publication bias in the meta-analysis of tendon disorders. All statistical analyses were conducted in Stata version 14.0.
3 Results
3.1 Literature Search
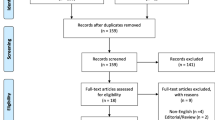
Searches identified 525 citations from PubMed, 573 from Embase, and 1631 from Scopus. After removing duplicates, we selected 2375 original articles for the first and second review (Fig. 1). Of these, 22 studies in 19 articles were appraised as eligible for the review. Two articles covered more than one study: one [29] included two case–control studies from two databases, and the other [23] contained a cohort with three collagen-associated diseases. In total, 12 cohort studies and ten case–control studies were eligible for this review.
3.2 Characteristics and Quality of the Included Studies
In total, we included 22 observational studies involving 19,207,552 participants in our study (see Table A1 in the ESM). Eight studies involved adults aged > 18 years, one involved children aged < 19 years, and four involved patients aged > 50 years. The publication date was within the last 20 years, and the follow-up duration ranged from 30 days to 1 year. Most studies were conducted on clinical registry and administrative data (via ICD codes and additional coding systems) that supplied definite medical records. Exposure in our study was mainly determined by fluoroquinolone prescription. RRs, ORs, and HRs were displayed for different follow-up periods, and most were adjusted by multiple covariates (e.g., age, sex). Assessment of study quality using the Newcastle–Ottawa Scale is presented in Tables A2 and A3 in the ESM. Among 18 observational studies, 13 were considered high quality (score of ≥ 7), and all studies received a score > 5.
3.3 Outcome Measures
3.3.1 Fluoroquinolones and Aortic Aneurysm or Aortic Dissection (AAD)
Figure 2 shows the results of the random-effects analysis for the covariate-adjusted associations between fluoroquinolone and AAD, involving 2,613,713 participants and 19,972 identified cases. The final result for the three studies was an OR of 2.20 (95% CI 1.92–2.52, I2= 20.6%).
3.3.2 Fluoroquinolones and Retinal Detachment
Retinal detachment in relation to fluoroquinolone use involved 14,607,668 participants and 16,076 events over an average follow-up of 30–365 days (past use, presented in Fig. 3). The result indicates a positive relationship between past fluoroquinolone use and retinal detachment (OR 1.27; 95% CI 1.09–1.47, I2= 76.6%). In the other analysis (Fig. 4), follow-up was limited to 30 days, and the estimated OR was 1.37 (95% CI 0.73–2.55, I2= 91.4%), which was not statistically significant.
3.3.3 Fluoroquinolones and Tendon Disorders
The random-effects meta-analysis for tendon disorders is displayed in Fig. 5. The result indicates an increased risk for tendon disorders (OR 1.89; 95% CI 1.53–2.33, I2= 83.5%). The outcome was obtained from 11 individual studies with 67,946 identified cases.
3.4 Sensitivity and Subgroup Analyses
Sensitivity analysis using a “leave-one-out” strategy was performed in meta-analyses (Figs. A1–4 in the ESM). In the analysis of retinal detachment with a follow-up duration of < 30 days (see Figs. 6 and A3 in the ESM), when the original trial by Etminan et al. [19] was removed, no significant heterogeneity was found in the estimate (I2 = 0.0%, P = 0.469), and the result became statistically significant (OR 1.25; 95% CI 1.01–1.53). Subgroup analyses were conducted to evaluate potential factors that may have influenced the pooled results (Table 1).
3.5 Assessment of Publication Bias
We did not perform statistical tests for publication bias for AAD and retinal detachment, because the tests are underpowered when the number of studies is low. The funnel plots for fluoroquinolone use and tendon disorders showed some asymmetry (Fig. A5 in the ESM), but neither Egger’s test (P = 0.457) nor Begg’s test (P = 0.245) was statistically significant, which suggests no obvious publication bias.
4 Discussion
In this review, we systematically evaluated observational evidence for an association between fluoroquinolone use and collagen-associated adverse events (AAD, retinal detachment, and tendon disorders) for different follow-up durations. Current use of fluoroquinolones was significantly associated with an increased risk of AAD, retinal detachment, and tendon disorders. Past fluoroquinolone use (> 30 and ≤ 365 days) was associated with retinal detachment.
The AAD-related results of our study were consistent with those from a meta-analysis by Singh et al. [34]. The consequence of retinal detachment differs from the other two meta-analyses [35, 36], which included case series and no sensitivity analysis. The incidence of retinal detachment was low [21, 23, 30]. Our study excluded case series to improve the methodological quality of the evidence, and we conducted a sensitivity analysis to prevent the effects of uncertainty in minority research. In the sensitivity analysis of retinal detachment with a follow-up of < 30 days, the result was significantly altered when we excluded a nested case–control study in a Canadian population [19]. The study methods in this article differed from those of other studies, which may contribute considerably to the heterogeneity. For example, this study defined the current use of fluoroquinolone as the overlapping of the prescription termination date and the index date, which could overestimate the result. The analysis of tendon disorders found a positive association between tendon disorders and fluoroquinolone use, which has been proposed in a black box warning [37]. Since tendon disorders consist of a series of individual diseases (e.g., tendinitis, tendon rupture), the result covers an inevitable heterogeneity (I2 = 83.9).
Subgroup analyses were conducted to identify potential sources of heterogeneity according to various factors, such as sex, age, and type of drugs. The significant association did not differ in most subgroups, which indicates the finding is robust. However, some residual confounding may still affect the result. The administration of fluoroquinolones differs among the included articles. Most of the studies used oral fluoroquinolone medication as inclusion criteria, and whether this administration could affect the association remains unclear. Some factors (e.g., oral corticosteroid use) were analyzed in only a minority of included studies, and we could not conduct a subgroup analysis because sufficient data were lacking. Future studies need to focus on these issues to further confirm the population at high risk of collagen-associated diseases when using fluoroquinolones.
In the USA, an estimated 5.1% of adult fluoroquinolone prescriptions were unnecessary, and 19.9% could have been replaced by other antibiotic medications [38]. The US FDA has warned that fluoroquinolones should not be prescribed to patients at increased risk of AAD [39], which includes those with a family history of AAD, high blood pressure, specific genetic disorders, and the elderly [39]. The European Medicines Agency (EMA) has also advised that the use of fluoroquinolone antibiotics should be restricted because of the potential for permanent side effects [40]. Combined with the warnings from the FDA and EMA, we strongly suggest that fluoroquinolones should be carefully used in patients at high risk of collagen-associated diseases as they may promote the development of diseases in this population.
Several biological mechanisms have been hypothesized as explanations for these associations. Studies in tendons have indicated that specific MMPs known to degrade critical components of the extracellular matrix (ECM) are promptly upregulated following exposure to ciprofloxacin [8, 41]. In moderate, sporadic AAD mouse models, ciprofloxacin significantly increased the incidence of aortic dissection and rupture, which was related to increased MMP levels and decreased LOX expression and activity [42]. In patients with abdominal aortic aneurysm, ECM degradation was related to elevated MMP12 [43]. On the other hand, the mechanism underlying the development of retinal detachment with fluoroquinolone use is not yet well-established, but a direct toxicity to collagen has been suggested. The most common cause of rhegmatogenous retinal detachment is degeneration of the vitreous body, which is made up of 98% water and is stabilized by collagen fibrils that extend into the superficial layers of the retina [44]. The therapeutic dose of fluoroquinolone in the vitreous body may interrupt collagen synthesis or destroy the collagen fibrils, leading to the retinal detachment [45].
Our analysis has several limitations. First, we were unable to conduct meta-regression or dose–response analyses because of the lack of adequate data. Given this, we could not analyze the different durations and number of exposures. Second, the controls of included studies were distinct, and the pooled estimates could be affected by individual studies. For example, Daneman et al. [23] reported a positive relationship between fluoroquinolones and collagen-associated disease, but the control group (amoxicillin users) also had an increased risk in the same cohort, so the final result could be overestimated. Lastly, although the estimates were adjusted for a wide variety of confounders using propensity score methods, residual confounding is always possible.
Ethical issues mean that randomized controlled trials on this topic are lacking. We only included observational studies, so our findings should be interpreted with caution. Further worldwide observational studies are required to better analyze related risk factors.
5 Conclusion
In this article, we report statistically significant associations between fluoroquinolones and three rare but severe collagen-associated events: AAD, retinal detachment, and tendon disorders. This finding is consistent with those of most previous studies.
Although the results of our analysis support increasing concern, definitive proof of the causal association between fluoroquinolones and collagen-associated events remains limited. Additional biological experiments are required to elucidate the potential mechanisms by which fluoroquinolones influence human connective tissue, especially in high-risk groups. Additionally, further worldwide observational studies are strongly recommended to focus on identifying high-risk populations, which would be more instructive in the medical use of fluoroquinolone antibiotics.
References
Hicks LA, Taylor TH, Hunkler RJUS. Outpatient antibiotic prescribing, 2010. New Engl J Med. 2013;368(15):1461–2. https://doi.org/10.1056/NEJMc1212055.
Pasternak B, Inghammar M, Svanstrom H. Fluoroquinolone use and risk of aortic aneurysm and dissection: nationwide cohort study. BMJ. 2018;360:k678. https://doi.org/10.1136/bmj.k678.
Kaleagasioglu F, Olcay E. Fluoroquinolone-induced tendinopathy: etiology and preventive measures. Tohoku J Exp Med. 2012;226(4):251–8. https://doi.org/10.1620/tjem.226.251.
Redgrave LS, Sutton SB, Webber MA, Piddock LJV. Fluoroquinolone resistance: mechanisms, impact on bacteria, and role in evolutionary success. Trends Microbiol. 2014;22(8):438–45. https://doi.org/10.1016/j.tim.2014.04.007.
Akali AU, Niranjan NS. Management of bilateral Achilles tendon rupture associated with ciprofloxacin: a review and case presentation. J Plast Reconstr Aesthetic Surg. 2008;61(7):830–4. https://doi.org/10.1016/j.bjps.2006.08.005.
Stinner DJ, Orr JD, Hsu JR. Fluoroquinolone-associated bilateral patellar tendon rupture: A case report and review of the literature. Mil Med. 2010;175(6):457–9. https://doi.org/10.7205/MILMED-D-09-00142.
Shybut TB, Puckett ER. Triceps ruptures after fluoroquinolone antibiotics: a report of 2 cases. Sports Health. 2017;9(5):474–6. https://doi.org/10.1177/1941738117713686.
Tsai WC, Hsu CC, Chen CPC, Chang HN, Wong AMK, Lin MS, et al. Ciprofloxacin up-regulates tendon cells to express matrix metalloproteinase-2 with degradation of type i collagen. J Orthop Res. 2011;29(1):67–73. https://doi.org/10.1002/jor.21196.
Rampal S, Kaur R, Sethi R, Singh O, Sood N. Ofloxacin-associated retinopathy in rabbits: role of oxidative stress. Hum Exp Toxicol. 2008;27(5):409–15. https://doi.org/10.1177/0960327108092295.
Van der Linden PD, Sturkenboom MCJM, Herings RMC, Leufkens HGM, Stricker BHC. Fluoroquinolones and risk of Achilles tendon disorders: case-control study. Br Med J. 2002;324(7349):1306–7.
Van der Linden PD, Sturkenboom MCJM, Herings RMC, Leufkens HMG, Rowlands S, Stricker BHC. Increased risk of Achilles tendon rupture with quinolone antibacterial use, especially in elderly patients taking oral corticosteroids. Arch Intern Med. 2003;163(15):1801–7. https://doi.org/10.1001/archinte.163.15.1801.
Seeger JD, West WA, Fife D, Noel GJ, Johnson LN, Walker AM. Achilles tendon rupture and its association with fluoroquinolone antibiotics and other potential risk factors in a managed care population. Pharmacoepidemiol Drug Saf. 2006;15(11):784–92. https://doi.org/10.1002/pds.1214.
Van der Linden PD, Van de Lei J, Nab HW, Knol A, Stricker BHC. Achilles tendinitis associated with fluoroquinolones. Br J Clin Pharmacol. 1999;48(3):433–7. https://doi.org/10.1046/j.1365-2125.1999.00016.x.
Yee CL, Duffy C, Gerbino PG, Stryker S, Noel GJ. Tendon or joint disorders in children after treatment with fluoroquinolones or azithromycin. Pediatr Infect Dis J. 2002;21(6):525–9. https://doi.org/10.1097/00006454-200206000-00009.
Sode J, Obel N, Hallas J, Lassen A. Use of fluroquinolone and risk of Achilles tendon rupture: a population-based cohort study. Eur J Clin Pharmacol. 2007;63(5):499–503. https://doi.org/10.1007/s00228-007-0265-9.
Hori K, Yamakawa K, Yoshida N, Ohnishi K, Kawakami J. Detection of fluoroquinolone-induced tendon disorders using a hospital database in Japan. Pharmacoepidemiol Drug Saf. 2012;21(8):886–9. https://doi.org/10.1002/pds.3285.
Morales DR, Slattery J, Pacurariu A, Pinheiro L, McGettigan P, Kurz X. Relative and absolute risk of tendon rupture with fluoroquinolone and concomitant fluoroquinolone/corticosteroid therapy: population-based nested case-control study. Clin Drug Investig. 2019;39(2):205–13. https://doi.org/10.1007/s40261-018-0729-y.
Tanne JH. FDA adds “black box” warning label to fluoroquinolone antibiotics. BMJ. 2008;337:a816. https://doi.org/10.1136/bmj.a816.
Etminan M, Forooghian F, Brophy JM, Bird ST, Maberley D. Oral fluoroquinolones and the risk of retinal detachment. J Am Med Assoc. 2012;307(13):1414–9. https://doi.org/10.1001/jama.2012.383.
Pasternak B, Svanström H, Melbye M, Hviid A. Association between oral fluoroquinolone use and retinal detachment. J Am Med Assoc. 2013;310(20):2184–90. https://doi.org/10.1001/jama.2013.280500.
Choi SY, Lim HA, Yim HW, Park YH. Administration of oral fluoroquinolone and the risk of rhegmatogenous retinal detachment: a nationwide population-based study in Korea. PLoS One. 2018;13(4):e0195563. https://doi.org/10.1371/journal.pone.0195563.
Eftekhari K, Ghodasra DH, Haynes K, Chen J, Kempen JH, Vanderbeek BL. Risk of retinal tear or detachment with oral fluoroquinolone use: a cohort study. Pharmacoepidemiol Drug Saf. 2014;23(7):745–52. https://doi.org/10.1002/pds.3623.
Daneman N, Lu H, Redelmeier DA. Fluoroquinolones and collagen associated severe adverse events: a longitudinal cohort study. BMJ Open. 2015;5(11):e010077. https://doi.org/10.1136/bmjopen-2015-010077.
Lee CC, Gabriel Lee MT, Chen YS, Lee SH, Chen YS, Chen SC, et al. Risk of aortic dissection and aortic aneurysm in patients taking oral fluoroquinolone. JAMA Intern Med. 2015;175(11):1839–47. https://doi.org/10.1001/jamainternmed.2015.5389.
Pouzaud F, Bernard-Beaubois K, Thevenin M, Warnet JM, Hayem G, Rat P. In vitro discrimination of fluoroquinolones toxicity on tendon cells: involvement of oxidative stress. J Pharmacol Exp Ther. 2004;308(1):394–402. https://doi.org/10.1124/jpet.103.057984.
Baugé C, Leclercq S, Conrozier T, Boumediene K. TOL19-001 reduces inflammation and MMP expression in monolayer cultures of tendon cells. BMC Complement Altern Med. 2015;15(1):217. https://doi.org/10.1186/s12906-015-0748-7.
Kumbhar GB, Khan AM, Rampal S. Evaluation of gatifloxacin for its potential to induce antioxidant imbalance and retinopathy in rabbits. Hum Exp Toxicol. 2015;34(4):372–9. https://doi.org/10.1177/0960327114530743.
Belavic JM. Fluoroquinolone-induced tendinopathy. Nurse Pract. 2009;34(1):17–8. https://doi.org/10.1097/01.NPR.0000343751.83969.1b.
Fife D, Zhu V, Voss E, Levy-Clarke G, Ryan P. Exposure to oral fluoroquinolones and the risk of retinal detachment: retrospective analyses of two large healthcare databases. Drug Saf. 2014;37(3):171–82. https://doi.org/10.1007/s40264-014-0138-y.
Kuo SC, Chen YT, Lee YT, Fan NW, Chen SJ, Li SY, et al. Association between recent use of fluoroquinolones and rhegmatogenous retinal detachment: a population-based cohort study. Clin Infect Dis. 2014;58(2):197–203. https://doi.org/10.1093/cid/cit708.
Corrao G, Zambon A, Bertù L, Mauri A, Paleari V, Rossi C, et al. Evidence of tendinitis provoked by fluoroquinolone treatment a case-control study. Drug Saf. 2006;29(10):889–96. https://doi.org/10.2165/00002018-200629100-00006.
Moher D, Liberati AF, Tetzlaff J, Tetzlaff JF, Altman DG, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. (1539-3704 (Electronic)).
Hartling L, Milne A, Hamm MP, Vandermeer B, Ansari M, Tsertsvadze A, et al. Testing the Newcastle Ottawa Scale showed low reliability between individual reviewers. J Clin Epidemiol. 2013;66(9):982–93. https://doi.org/10.1016/j.jclinepi.2013.03.003.
Singh S, Nautiyal A. Aortic dissection and aortic aneurysms associated with fluoroquinolones: a systematic review and meta-analysis. Am J Med. 2017;130(12):1449. https://doi.org/10.1016/j.amjmed.2017.06.029.
Chui CSL, Wong ICK, Wong LYL, Chan EW. Association between oral fluoroquinolone use and the development of retinal detachment: a systematic review and meta-analysis of observational studies. J Antimicrob Chemother. 2015;70(4):971–8. https://doi.org/10.1093/jac/dku507.
Alves C, Penedones A, Mendes D, Batel Marques F. A systematic review and meta-analysis of the association between systemic fluoroquinolones and retinal detachment. Acta Ophthalmol. 2016;94(5):e251–9. https://doi.org/10.1111/aos.12931.
Jupiter DC, Fang X, Ashmore Z, Shibuya N, Mehta HB. The relative risk of achilles tendon injury in patients taking quinolones. Pharmacotherapy. 2018;38(9):878–87. https://doi.org/10.1002/phar.2162.
Etminan M, Maberley DAL. Improving reporting quality in ophthalmologic observational studies that use big data: the case of retinal detachment associated with fluoroquinolone use. (2168-6173 (Electronic)).
FDA warns about increased risk of ruptures or tears in the aorta blood vessel with fluoroquinolone antibiotics in certain patients. https://www.fda.gov/Drugs/DrugSafety/ucm628753.htm.
Disabling and potentially permanent side effects lead to suspension or restrictions of quinolone and fluoroquinolone antibiotics. https://www.ema.europa.eu/en/documents/press-release/disabling-potentially-permanent-side-effects-lead-suspension-restrictions-quinolone-fluoroquinolone_en.pdf. Accessed 1 Mar 2019.
Corps AN, Harrall RL, Curry VA, Fenwick SA, Hazleman BL, Riley GP. Ciprofloxacin enhances the stimulation of matrix metalloproteinase 3 expression by interleukin-1beta in human tendon-derived cells. A potential mechanism of fluoroquinolone-induced tendinopathy. Arthritis Rheum. 2002;46(11):3034–40. https://doi.org/10.1002/art.10617.
LeMaire SA, Zhang L, Luo W, Ren P, Azares AR, Wang Y, et al. Effect of ciprofloxacin on susceptibility to aortic dissection and rupture in mice. JAMA Surg. 2018;153(9):e181804. https://doi.org/10.1001/jamasurg.2018.1804.
Amin M, Pushpakumar S, Muradashvili N, Kundu S, Tyagi SC, Sen U. Regulation and involvement of matrix metalloproteinases in vascular diseases. Front Biosci (Landmark edition). 2016;21:89–118.
Feltgen N, Walter P. Rhegmatogenous retinal detachment–an ophthalmologic emergency. Dtsch Arztebl Inter. 2014;111(1–2):12–22. https://doi.org/10.3238/arztebl.2014.0012.
Baek YH, Park SJ, Jeong S, Oh IS, Jeong HE, Park KH, et al. Signal detection between fluoroquinolone use and the risk of rhegmatogenous retinal detachment: sequence symmetry analysis using nationwide South Korean healthcare database between 2004 and 2015. Clin Drug Investig. 2018. https://doi.org/10.1007/s40261-018-0708-3.
Acknowledgements
The authors thank all the authors of the original articles. We also appreciate Dr. Mouniir SA Durgahee for editing work.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
Xin-yu Yu, Ding-sheng Jiang, Jing Wang, Rui Wang, Tai-qiang Chen, Kan Wang, Xiang Wei, and Shi-yi Cao have no conflicts of interest that are directly relevant to the content of this study.
Funding
This review was supported by the National Natural Science Foundation of China (NSFC, 71603091).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yu, X., Jiang, Ds., Wang, J. et al. Fluoroquinolone Use and the Risk of Collagen-Associated Adverse Events: A Systematic Review and Meta-Analysis. Drug Saf 42, 1025–1033 (2019). https://doi.org/10.1007/s40264-019-00828-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-019-00828-z