Abstract
Stevens–Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) are life-threatening mucocutaneous reactions, predominantly drug induced. The mortality rates for SJS and TEN are as high as 30 %, and short- and long-term morbidities are very common. SJS/TEN is one of the few dermatological diseases that constitute a true medical emergency. Early recognition and prompt and appropriate management can be lifesaving. In recent years, our understanding of the pathogenesis, clinical presentation, and management of SJS/TEN has improved. Nevertheless, in 2015, there are still no internationally accepted management guidelines. This review summarizes up-to-date insights on SJS/TEN and describes a protocol for assessment and treatment. We hope these suggested guidelines serve as a practical clinical tool in the management of SJS/TEN. The classic manifestation of SJS/TEN consists of initial “flu-like” symptoms (malaise, fever, anorexia) in the prodromal phase, followed by cutaneous and mucous membrane (ocular, oral, and genital) inflammation and pain, and other systemic involvement. Symptoms usually begin 4–28 days after the onset of drug intake. Treatment is multidisciplinary and includes identification and withdrawal of the culprit drug, transfer to a specialist unit, supportive care, medical treatment, communication, and provision of appropriate information and emotional support.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Stevens–Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) are considered the most severe types of cutaneous adverse reactions to drugs, with high morbidity and mortality rates. |
SJS/TEN mostly results from a cumulative effect of aligned risks related to the structure of a drug and to the patient’s genetic predisposition (human leukocyte antigen alleles, drug metabolism characteristics, and T cell clonotypes). |
Assessment can be summarized by the “5Ds”: Diagnosis, Drug exposure, Differential diagnosis, Determining the probabilities of causality, and Determining the severity of SJS/TEN. |
1 Introduction
Stevens–Johnson syndrome (SJS) was first described by A. M. Stevens and F. C. Johnson in 1922 [1]. The term “toxic epidermal necrolysis” (TEN) was coined in 1956 by A. Lyell [2]. Neither of those publications truly described what today is considered to be SJS or TEN. There are clinical and pathological variants of both SJS and TEN, but the terms are also used to define a condition that is a spectrum of disease from SJS to TEN. SJS and TEN represent different degrees of the same type of severe cutaneous adverse reaction. The mortality rates for SJS and TEN are high; that of TEN may approach 30 % [3]. In this manuscript, we refer to this specific disease spectrum as a single entity—namely, SJS/TEN.
In recent years, our understanding of the pathogenesis, clinical presentation, and management of SJS/TEN has been significantly enlightened. This review aims to summarize the most up-to-date insights.
2 Pathogenesis
SJS/TEN mostly results from a cumulative effect of aligned risks related to the structure of a drug and to the patient’s genetic predisposition (human leukocyte antigen [HLA] alleles, drug metabolism characteristics, and T cell clonotypes) (Fig. 1).
2.1 Genetic Associations of SJS/TEN
Over the past few decades, important progress has been made in understanding the pathogenic mechanisms of SJS/TEN—specifically, the important role of HLA alleles. The pathogenesis of inducing cytotoxic responses in SJS/TEN is generated by recognition of culprit drugs by specific HLA molecules. In 1987, the genetic susceptibility of HLA alleles was first proposed by Roujeau et al. [4], who identified weak associations of HLA-B*12 and oxicam- and sulfonamide-related TEN in Europeans. More notable evidence of genetic susceptibility to SJS/TEN was reported in 2004 by Chung et al. [5], who found that HLA-B*15:02 is strongly associated with carbamazepine (CBZ)-induced SJS/TEN. The early associations were made in an era of serological typing of HLA. Currently, in an era of sequence-based typing and deep sequencing, which allows resolution to four digits, identification of more specific and stronger associations are possible. Hence, many other HLA associations in SJS/TEN have been discovered [4, 6–21], as are detailed in Table 1.
HLA associations with specific drug-induced SJS/TEN can be restricted to certain ethnicities and phenotypes. The strength of HLA associations with specific drug-induced SJS/TEN has been found to be related to the prevalence of the susceptibility allele in the ethnic population. The association of HLA-B*15:02 and carbamazepine-induced SJS/TEN has been reported in Han Chinese, Thai, Indian [22], and Malaysian populations [23] but not in Europeans, who carry the HLA-B*15:02 allele in low frequency (<1 %) [24]. The association is also present in Han Chinese ancestry of Europeans. This ethnic difference can be tracked by historical evolution. The US Food & Drug Administration and Asian health administrations have recommended HLA-B*15:02 screening for new carbamazepine users of Asian ancestry since 2007 [25]. In contrast, the HLA-B*58:01 allele with allopurinol-induced SJS/TEN is common to all populations, being found in Han Chinese, Thai, Japanese, Korean, and European populations [26].
There are also phenotype-specific characteristics involved in carbamazepine hypersensitivity. HLA-B*15:02 and other B75 serotype HLA alleles are strongly associated with carbamazepine-induced SJS/TEN but are not associated with carbamazepine-induced drug reaction with eosinophilia and systemic symptoms (DRESS); however, HLA-A*31:01 is strongly associated with carbamazepine-induced DRESS but less so with carbamazepine-induced SJS/TEN [27].
Association with a specific HLA risk allele appears to be necessary but not sufficient for development of SJS/TEN. Additional factors such as individual differences in drug metabolism or clearance may also play an important role in SJS/TEN development, recovery, or prognosis. Drug clearance is known to be important for preventing exceptional damage, which can occur as a result of retention of the drug in the body. Shear and Spielberg first recognized potential pharmacogenetic associations and risks of altered drug metabolism for SJS/TEN and DRESS with anticonvulsants and sulfonamides [28, 29].
A recent genome-wide association study by Chung et al. [30] reported that genetic variants of cytochrome P450 2C (CYP2C) are strongly associated with phenytoin-related SJS/TEN. They identified 16 significant single-nucleotide polymorphisms (SNP) in CYP2C genes at 10q23.33. Further studies showed that CYP2C9*3 variants, which reduce CYP2C9 enzymatic activity, were significantly related to phenytoin-induced SJS/TEN. CYP2C9*3 has been known to be related to the drug’s metabolism and can attenuate the clearance of phenytoin [31, 32]. Patients with phenytoin-induced SJS/TEN who carried CYP2C9*3 showed delayed clearance of plasma phenytoin, resulting in increasing phenytoin toxicity in the body. Another study showing that genetic variability in a metabolizing enzyme can contribute to SJS/TEN examined nevirapine-induced SJS/TEN. CYP2B6 G516T and T983C SNPs were found to be associated with SJS/TEN susceptibility [33]. The issue of drug metabolism or clearance is another key factor in consideration of the risk of developing SJS/TEN.
2.2 Immunological Mechanisms of SJS/TEN
SJS/TEN is mostly recognized as an immune disorder elicited by drugs. SJS/TEN is a delayed-type drug hypersensitivity reaction, with a typical latency of 4–28 days and with cases rarely occurring as long as 8 weeks following initiation of the implicated drug [34].
The drugs are of low molecular weight and often serve as foreign antigens that are recognized by T cell receptors (TCRs) to activate adaptive immune responses. Several proposed concepts have been found to explain how small-molecule compounds are recognized by TCRs. In some cases, drugs interact directly with the TCRs involved in presenting to HLA molecules of antigen-presenting cells (APCs). This model is known as the p-i [pharmacological interaction of drugs with immune receptors] concept [35]. An example is that carbamazepine cannot bind covalently to peptides or proteins but is able to bind with low affinity to TCRs and provoke T cell activation [36, 37]. Drugs can also interact with TCRs by a drug–peptide complex presented to HLA molecules of APCs in what is known as the hapten concept. The β-lactams that form covalent binding to lysine residues are an example [38]. Upon noncovalent binding of specific drugs presented to HLA molecules and TCRs, the HLA–drug–TCRs may initiate a series of immune reactions, which result in activation of CD8+ cytotoxic T cell–mediated and natural killer (NK) cell–mediated cytotoxicity. Recently, the shared and restricted TCR usage subtype in carbamazepine-induced SJS/TEN was identified [39], and it was demonstrated that the endogenous peptide-loaded HLA-B*15:02 molecule presented carbamazepine to cytotoxic T cells without the involvement of intracellular drug metabolism or antigen processing (Fig. 2) [37].
Cytokines are involved in the pathogenesis of SJS/TEN. Several studies have shown that tumor necrosis factor (TNF)-α is strongly expressed in SJS/TEN lesions and correlates proportionally with disease development [40–42]. TNF-α can induce cell apoptosis, activation, and differentiation, and an inflammatory response [43, 44]. In addition, interferon (IFN)-γ is a common cytokine involved in delayed-type drug hypersensitivity, including SJS/TEN. IFN-γ is often expressed in the superficial dermis and epidermis of SJS/TEN lesions [41, 42]. IFN-γ is known to induce antigen presentation and thus stimulate cell-mediated immunity by upregulation of HLA molecules [45–47]. TNF-α, IFN-γ, several cytokines, and chemokine receptors that are responsible for proliferation, trafficking, and activation of T cells have been found in the skin lesions, blister fluids, blister cells, peripheral blood mononuclear cells, or plasma of SJS/TEN patients. These cytokines/chemokines include interleukin (IL)-2, IL-5, IL-6, IL-10, IL-12, IL-13, IL-15, IL-18, chemokine (C–C motif) receptor (CCR) 3, chemokine (C-X-C motif) receptor (CXCR) 3, CXCR4, and CCR10 [40–42, 48–50].
The major theory to explain the severe epidermal detachment of SJS/TEN is CD8+ cytotoxic T cell–mediated and NK cell–mediated cytotoxicity. It has been well established that epidermal detachment in SJS/TEN is due to keratinocyte apoptosis. Recently, studies have shown that keratinocyte apoptosis in lesional skin and blister fluid in SJS/TEN patients is associated with greatly increased numbers of CD8+ cytotoxic T cells and NK cells [51, 52]. While CD8+ cytotoxic T cells and NK cells are activated, they subsequently carry out the cellular-mediated immune reactions directed at keratinocytes in an HLA class I–restricted manner. Upon activation of these responses, various cytotoxic signaling molecules, including granulysin, perforin/granzyme B, and Fas/Fas ligand, are relayed to the skin lesions to induce keratinocyte apoptosis [52–54]. More importantly, Chung et al. found that granulysin—a cytotoxic protein produced by cytotoxic T cells or NK cells—acts as a key mediator responsible for disseminated keratinocyte death [52, 55]. Furthermore, granulysin is not only a cytotoxic protein but also a chemoattractant and proinflammatory activator, which can promote monocyte expression of chemokine (C–C motif) ligand (CCL) 20 [56] and is capable of promoting antigen-presenting (dendritic) cell and leukocyte recruitment (specifically, a granulysin 15 kD subunit, which is largely produced by CD8+ T cells and NK T cells) [57]. Granulysin-positive cells in fixed drug eruptions have been found to be similar to those observed in SJS/TEN [58].
2.3 Environmental Factors
SJS/TEN can be secondary to infection with Mycoplasma pneumoniae or herpes simplex virus, but the full pathogenesis remains unclear [59–61]. Human enterovirus has not previously been recognized to be associated with SJS [59, 60]. By comparison, erythema multiforme major (EMM) is mainly caused by viruses, usually involving the palms and soles, and the patient experiences rapid healing without sequelae.
A recent study found that a new variant of coxsackie virus (CV) A6, which belongs to the human Enterovirus genus and causes severe mucocutaneous blistering reactions, mainly mediated by cytotoxic T cells and NK cells expressing granulysin, mimics the histopathological features of SJS or EMM in children [62]. In fact, there are still about 20 % of SJS/TEN cases without an identified causality [63, 64]. Potential risk factors for this unusual presentation of virus infection as a cause of SJS/TEN have not been elucidated.
3 Clinical Presentation
3.1 Acute Stage
Initially, SJS/TEN begins within 4 weeks (usually 4–28 days) after the onset of drug intake [65]. The disease can also occur a few days after the drug has been withdrawn in the case of long-half-life drugs. In very rare cases of rechallenge with the same drug, the disease appears more rapidly, within hours [66].
Initial symptoms are usually non-specific and can precede cutaneous manifestations by a few days (1–3 days) in one third of cases. Painful mucous membranes, stinging eyes, headache, rhinitis, malaise, cough, sore throat, and myalgias are frequently noticed. In the absence of cutaneous manifestations, these symptoms may contribute to an erroneous initial diagnosis and delayed specific management. In other cases, the disease may begin with mucous membrane involvement and/or non-specific exanthema. However, the addition of new clinical signs, severe pain, and rapid progression should alert the physician and may lead to consideration of a severe disease.
3.1.1 Cutaneous Lesions
Early sites of cutaneous involvement are the presternal region of the trunk, the face, and proximal parts of the limbs. Palms and soles may also be initially involved. The rash may spread to distal parts of the limbs and the rest of the body within a few days. Individual cutaneous lesions appear as erythematous, dusky dark red purpuric macules, which are irregularly shaped (Fig. 3) [67, 68]. These atypical targets, with two concentric rings, have a necrotic center and may tend to coalesce [68]. At this stage, involvement of at least two mucous membrane sites is observed in up to 90 % of cases, and this feature may help in diagnosis.
In the absence of spontaneous detachment, a positive Nikolsky sign should be sought by exertion of lateral mechanical pressure with a finger on an erythematous zone [67, 68]. This sign is considered positive if dermal–epidermal cleavage is induced and necrotic epidermis detachment on pressure points reveals large areas of exposed, red, sometimes oozing dermis. It can also be observed in autoimmune blistering diseases such as pemphigus. According to the percentage of epidermis that is detached or detachable, patients are classified into one of three groups: SJS involves <10 % of body surface area (BSA); SJS/TEN overlap involves between 10 % and 30 % of BSA; TEN involves >30 % of BSA [68].
Despite the obvious differences from erythema multiforme, occasional typical target lesions may appear [67, 69]. These lesions rapidly extend within 4 to 5 days. Skin involvement can be limited to the predilection sites at the SJS end of the spectrum (face, trunk) but are widespread in TEN. TEN displays areas of diffuse erythema, with individual macular lesions at the periphery. The epidermis detaches from the underlying dermis, leading to large, flaccid blisters (Fig. 4). The roofs of the blisters turn necrotic and display denudation of the epidermis in large sheets measuring >5 cm (Fig. 5). Tense bullous lesions are more frequently observed at the initial stage and on palmoplantar soles.
Thermal burn rules are currently used in SJS/TEN. Evaluation of the BSA involved may be difficult and is often overestimated, more particularly in the case of spotty lesions.
3.1.2 Mucous Membranes
Involvement of mucous membranes is noticed in more than 80 % of cases with at least two sites involved and may be inaugural in one third of cases [68]. Painful inflammation and erosions of mucosal surfaces occurs in 87 to 100 % of cases of TEN [70, 71]. Lesions usually begin with painful, burning sensations of the lips, conjunctivae, and genitalia, followed by edematous, erythematous, and flaccid bullous lesions. Blisters rupture and tend to extend. Bullous lesions become very painful and hemorrhagic erosions, coated by greyish-white pseudo-membranes of the oral cavity (Fig. 6), leading to impaired alimentation and hypersalivation. Genital erosions most often include painful erosions of the glans penis, vulva, and vagina, and may lead to “burning” on micturition, urinary retention, and synechiae [72]. Anal lesions are less frequent. Ocular involvement is mainly represented by conjunctival involvement in 80 % of cases [73, 74], including photophobia, pain, lacrimation, chemosis, and redness [73, 74]. However, more severe involvement leads to corneal ulceration, anterior uveitis, and purulent conjunctivitis. Ultimately, this can lead to blindness.
3.1.3 Systemic Manifestations
SJS/TEN corresponds to an acute skin failure associated with severe weakness, pain, and prolonged high fever [75]. Internal epithelial organ involvement is rare and mainly concerns the respiratory and gastrointestinal tracts. Early pulmonary dysfunction is observed in 25 % of cases, and the respiratory rate and blood oxygenation must be systematically evaluated [76]. Pulmonary involvement includes breathing difficulties, cough, bronchial obstruction, and respiratory distress. A recent retrospective study reported that 39 % of patients with SJS/TEN had specific endobronchial lesions and 25 % required mechanical ventilation [77]. Initial bronchial hypersecretion, laryngeal involvement, and dyspnea should alert the physician.
Less commonly, gastrointestinal involvement has been reported, including diarrhea, abdominal distention, and excretion of colonic intestinal epithelium, which can lead to bowel perforation [78, 79].
Renal involvement is currently observed in the acute phase, mainly represented by acute renal failure, proximal acute tubular necrosis, hematuria, and microalbuminemia [80]. Anemia, mild elevation in hepatic enzymes and amylase (mostly of salivary origin) are frequent, without any impact on prognosis. Neutropenia is rare and is considered a severity marker but is too rare to have an impact on the Severity of Illness Score for TEN (SCORTEN) (Table 2).
3.1.4 Prognosis
Progression lasts for about 4–5 days following admission. Next, patients enter a plateau phase, which corresponds to progressive re-epithelialization. Complete healing can take a few days to several weeks. This phase is associated with life-threatening complications such as sepsis due to epithelial loss [81, 82]. Staphylococcus aureus and Pseudomonas are the most frequent pathogens involved, both usually present in the skin and blood. Multisystem organ failure and pneumonitis are important causes of morbidity, with prevalence rates of 24 and 23.1 %, respectively [82].
In Europe, according to the RegiSCAR (European Registry of Severe Cutaneous Adverse Reactions) results, the mortality rate was 23 % at 6 weeks and 34 % at 1 year [83]. The severity of the reaction was a risk factor for mortality only in the 3 months after onset, whereas serious comorbidities and age influenced mortality beyond 90 days and up to 1 year after the onset of the reaction [83].
In parallel, it has already been demonstrated that prompt withdrawal of the offending agent may reduce the risk of death by 30 % [84].
A SJS/TEN-specific severity-of-illness score is proposed (SCORTEN), in which seven variables remain independent prognosis factors of death: age >40 years, heart rate >120 beats/min, cancer or hemopathy, BSA involvement >10 %, serum urea level >10 mmol/L, serum bicarbonate level <20 mmol/L, and serum glucose level >14 mmol/L (Table 2) [85]. The score’s ability to predict outcome and usefulness has been confirmed by several teams [86, 87], and a simplified score has recently been proposed for cases with missing laboratory data [88]. The prognostic value of SCORTEN is more accurate at day 3 of hospitalization [89].
3.2 Chronic Stage
During the past few decades, SJS/TEN has no longer been considered as only an acute disease. Specific follow-up of sequelae should be systematically planned and studied for better understanding of the pathophysiology, detection, and prevention of the reaction, and to reduce the health burden [90]. Dermatological sequelae frequently include hyper- and hypopigmentation (72 %), hypertrophic scars, and nail dystrophies (pigmentation in the nail bed, ridging, and permanent anonychia).
In 65–89 % of cases, patients develop late ocular complications, including dry eye syndrome, trichiasis, photophobia, symblepharon, corneal inflammation and neovascularization, and, in the most severe cases, reduced visual acuity and blindness [73, 91].
Buccal and dental complications in a study of 16 patients with TEN reported gingival synechiae, gingival recession, dental alteration, xerostomia, and increased saliva acidity [92].
Long-term follow-up of SJS/TEN patients can identify chronic lung disease, mostly bronchiolitis obliterans. Pulmonary function tests performed during the usual follow-up display abnormalities presenting mainly as asymptomatic diffusion impairment, with a risk proportional to BSA involvement [93].
Male genitalia synechiae may require circumcision. Strictures of vaginal mucosa and/or birth-canal stenosis [72] may be responsible for dyspareunia or vaginal dryness, and can complicate spontaneous vaginal delivery and sexual intercourse. Surgical, topical, physical, and laser treatments are often required.
The results of our recent study assessing the long-term emotional and physical sequelae of SJS/TEN [94] demonstrated major emotional complications (symptoms of anxiety, depression, and post-traumatic stress), impaired health-related quality of life, and long-term physical complications (most commonly cutaneous and ophthalmic).
4 Assessment and Treatment of SJS/TEN
SJS/TEN is one of the few dermatological diseases that constitute a true medical emergency. Early recognition and prompt appropriate management can be lifesaving. Nevertheless, there are no internationally accepted management guidelines. The following represents the authors’ protocol for assessment (Table 2) and treatment (Table 3) of patients with SJS/TEN, based on the most up-to-date literature and abundant experience in treating patients with SJS/TEN. We hope these suggested guidelines may serve as a practical clinical tool for physicians in the management of patients with SJS/TEN. While this is international by definition, it is not necessarily a consensus-based protocol. For the readers’ convenience, we have designed two tables: the assessment protocol (Table 2) and the treatment protocol (Table 3), which summarize the key elements.
4.1 Assessment
Assessment is conducted by the simple method of “5Ds by Dr. Shear”. Specifically, these are Diagnosis, Drug exposure, Differential diagnosis, Determining the probabilities of causality, and Determining the severity of SJS/TEN.
4.1.1 Diagnosis
The physician must determine whether the patient’s clinical symptoms are signs of SJS/TEN or of another skin disease. The diagnosis is based on three key clinical elements: cutaneous and mucous membrane manifestations, systemic involvement, and histological findings. A thorough history and a comprehensive physical examination are essential for accurate diagnosis of SJS/TEN. Basically, the classic manifestation of SJS/TEN consists of initial signs of “flu-like” symptoms (malaise, fever, anorexia) in the prodromal phase, followed by cutaneous and mucous membrane inflammation and pain (ocular, oral, and genital) and other systemic involvement (see elaboration in Sect. 3).
For cutaneous assessment, the classification of SJS/TEN defined by Bastuji-Garin et al. [69] includes the proper definition of the characteristic skin lesions. For BSA involvement, the Wallace rule of nines can be used; a value of 9 % of BSA is given for involvement of the head, each arm, the chest, and the abdomen; 18 % is given for each leg and the back; and 1 % is given for the groin. Also, the patient’s palm can serve as a reference point roughly equivalent to 1 % of BSA for total assessment. For children and infants, the Lund and Browder chart is used to assess the burned BSA. The extent of epidermal detachment should also be assessed. It should be emphasized that only necrotic skin that is already detached (e.g. blisters, erosions) or detachable skin (a positive Nikolsky sign, whereby slight rubbing of the skin results in exfoliation of the outermost layer) should be included in the evaluation of the extent of epidermal detachment [61].
Histological assessment is a key element in the diagnosis of SJS/TEN and to rule out diseases that may mimic SJS/TEN. Characteristic histological features include extensive keratinocyte destruction with separation of the epidermis from the dermis at the dermoepidermal junction [95]. A skin biopsy for direct immunofluorescence (DIF) from perilesional skin should also be conducted for the differential diagnosis. Blood work and consultation with specialists (ophthalmology, otolaryngology, and gynecology) for comprehensive assessment of mucous membrane involvement are to be conducted in the primary assessment of the patient, according to the clinical signs. During hospitalization, ocular assessment should be conducted on a daily basis, and consultation with other specialists is based on the patient’s clinical signs and symptoms.
A rapid immunochromatographic test for serum granulysin was found to be useful for the prediction of SJS/TEN [96] and, in the future, it may serve as a clinical tool for early diagnosis. However, it is still not known if granulysin may also be useful as a prognostic marker for SJS/TEN.
4.1.2 Drug Exposure (Timing)
All medications, regardless of the route of administration, must be considered, especially new drugs taken in the 8 weeks prior to the skin reaction. Drugs taken intermittently, such as vitamins, sedatives, pain relievers, laxatives, and natural products, must also be considered. It is also important to avoid a common mistake, which is to falsely implicate drugs that were introduced for early symptoms of SJS/TEN, such as antipyretics or antibiotics, in the causality of SJS/TEN. Assessment of the lag period—the time between initiation of the drug and the onset of the cutaneous reaction—is crucial in view of the different lag times for different cutaneous drug reactions. A recommended method for drug exposure analysis is to chart a timeline of the patient’s illness in order to visualize the chronology and facilitate comprehension of the event. The timeline includes the relevant information (the starting day, dosage, and discontinuing day) for each drug and the signs and symptoms throughout the period in question [97, 98]. In SJS/TEN, the lag period is 4–28 days. The median latency was found to be longer (>30 weeks) for drugs with no associated risk [65].
4.1.3 Differential Diagnosis
Establishing a differential diagnosis that takes into account all possible diagnoses is essential. Ranking of the approximate likelihood of each condition is encouraged. The major differential diagnoses for SJS/TEN are detailed in Table 2. The following clues may assist in differentiating them.
Staphylococcal Scalded Skin Syndrome (SSSS): In addition to the different clinical findings (typically the lack of mucous membrane involvement in SSSS), performing a frozen section to evaluate the level of epidermal separation, which is intra-epidermal in SSSS but sub-epidermal in TEN, is a quick tool for differentiating between these diseases [99].
Generalized Bullous Fixed Drug Eruption (GBFDE): The following typical clinical features of GBFDE may aid in differentiating it from SJS/TEN: (1) blistering usually affects only a small percentage of BSA, and between the large blisters there are sizable areas of intact skin; (2) erosive mucosal involvement is rare and, when it does occur, is rather mild; (3) patients usually do not feel sick or have fever, and generally they are in much better overall health than those with SJS/TEN; (4) most patients report a history of a similar, often local reaction [99].
Acute Graft-Versus-Host Reaction (aGVHR): aGVHR is a multi-organ disease process (major organs include the skin, liver, and gastrointestinal tract) resulting from interactions of donor-derived T cells against antigens expressed on the cells of the recipient host. Severe cases of aGVHR present with symptoms similar to those of SJS/TEN (clinical and histological): diffuse erythroderma, desquamation, bullae, a positive Nikolsky sign, and ulcerations of mucous membranes. Histologically, grade IV aGVHR involves full-thickness epidermal necrosis and has histological findings identical to those in SJS/TEN. The clinical setting assists in differentiation of aGVHR from SJS/TEN. aGVHR occurs in patients with bone marrow and allogeneic hematopoietic stem cell transplants, usually 2 weeks post-transplantation [100].
TEN-Like Lupus Erythematosus or Lupus-Associated TEN (TEN-Like LE): This was first described in 2003 [101]. It is a subacute or acute cutaneous rash, followed by bullous lesions and epidermal cleavage. It has been reported to occur in patients with established systemic lupus erythematosus, and it is considered to result from aggressive inflammatory epidermal basal layer damage due to increased extension of the interface dermatitis characteristic of lupus erythematosus. In distinction to SJS/TEN, there is a lack of an apparent trigger and an absence of systemic and mucous membrane involvement [102].
4.1.4 Determining the Probabilities of Causality by the Suspected Drugs
Drug exposure is the most common cause of SJS/TEN [61], with more than 200 culprit drugs identified [3]. The groups of medications associated with a high risk of inducing SJS/TEN vary according to the population (Table 4). Non-medication triggers, implicated mainly in SJS, include infections, contrast media, and vaccinations [59, 103, 104]. The most significant challenge in assessing SJS/TEN is determining the probabilities of causality between the suspected drugs and the untoward clinical event.
The following methods are helpful:
Patient History: The lag period (the time between initiation of the drug and the onset of the cutaneous reaction) and personal and familial previous cutaneous reactions to drugs are crucial factors in assessing likelihoods [97].
Analysis of the Literature: Search for information regarding the frequency with which the type of reaction is related to a particular drug (PubMed at http://www.ncbi.nlm.nih.gov/pubmed/; Litt’s Drug Eruptions and Reactions Manual and database at http://www.drugeruptiondata.com/).
Algorithm of Drug Causality for Epidermal Necrolysis (ALDEN): This is an algorithm for the assessment of drug causality in SJS/TEN, developed by the RegiSCAR Study Group, and consists of six parameters according to which the drug causality is classified as very unlikely, unlikely, possible, probable, or very probable [105].
Adjunctive Laboratory Assessments: In this decade, there has been a successful translation of the laboratory-based discovery of the HLA alleles associated with severe cutaneous adverse reactions to drugs to a clinical guideline-based test [25]. These tests are best utilized as genetic screening tools prior to drug prescription. However, they may also serve as a simple, fast, safe, and reliable method of establishing clear causality between a drug and a disease in cases of SJS/TEN (but should never be used in isolation as a single test in causality assessment) and are an important tool in counseling both the patient and his/her family members about drug avoidance [106]. The HLAs are specific to a drug and an ethnic background [107] (see elaboration in Sect. 2 and Table 1). In vitro assessments such as the lymphocyte transformation test (LTT) [108] and granulysin expression [109] have been reported. However, caution should always be exercised regarding false positive and false negative results. In addition, at this point in time, no laboratory tests should be used as justification for rechallenge when SJS/TEN associated with a drug is clinically possible.
4.1.5 Determining the Severity
The severity of SJS/TEN depends mostly on the haemodynamic status and the extent of systemic involvement, determined by epidermal barrier breakdown symptoms such as hypothermia, dehydration, secondary infection, and organ involvement induced by necrosis of epithelial lining, such as respiratory distress syndrome, colitis, hepatitis, and nephritis (see elaboration in Sect. 3) [110]. This assessment is conducted by taking a thorough history, a physical examination, blood work, and proper imaging (a chest X-ray is advised for all patients; further imaging is based upon clinical findings).
The following clinical and histological findings were found to be validated values for determination of severity.
SCORTEN: This scoring system was developed to stratify the severity of illness and predict mortality in patients with TEN. To optimize the predictive value of this tool, SCORTEN is to be performed on days 1 and 3 post-admission (see elaboration in Sect. 3 and Table 2) [85].
Histological Findings: In a retrospective analysis of the clinical records and histological material from 37 patients with TEN, the extent of dermal mononuclear inflammation was found to predict the clinical outcome approximately as well as SCORTEN. Increased inflammation correlated with a worse prognosis; a mean dermal mononuclear cell count >215 cells per high-power field in patients with 30 % or more total BSA sloughing predicted a worse prognosis (65 % mortality), compared with 24 % mortality in those with <215 cells per high-power field [95]. However, in a retrospective study analyzing clinical records and skin biopsies from 108 patients with SJS, SJS/TEN overlap, and TEN, dermal infiltrate severity was not associated with day 1 SCORTEN or hospital death, but full-thickness epidermal necrosis was associated with mortality [111].
Early Withdrawal and Half-Life of the Causative Drug: A 10-year observational study including 113 patients with TEN or SJS found that the earlier the causative drug was withdrawn, the better the prognosis was (odds ratio 0.69 for each day; 95 % confidence interval 0.53–0.89). Patients exposed to causative drugs with long half-lives had an increased risk of dying (odds ratio 4.9; 95 % confidence interval 1.3–18.9) [84].
4.2 Treatment
The treatment of SJS/TEN consists of a multidisciplinary approach, which includes the following important aspects (Table 3):
4.2.1 Identification and Withdrawal of the Culprit Drug
The medication history during the previous 2 months is documented and all suspected and non-essential medications are withdrawn [99].
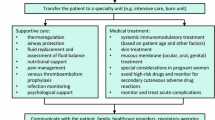
4.2.2 Transfer of the Patient to an Appropriate Unit
The patient is transferred to intensive care, a burn unit, or another specialty unit [112, 113].
4.2.3 Supportive Care
Thermoregulation: Cutaneous thermoregulation is impaired by the compromised skin barrier; thus, increasing the room temperature to 28–32 °C is important, especially for patients with large amounts of epidermal detachment [99, 114].
Airway Protection: The decision to intubate and ventilate is made on the basis of the following considerations: the extent of upper airway mucosal involvement, potential for airway obstruction, severity of respiratory distress, and anticipated analgesia and sedation requirements.
Fluid Replacement and Assessment of Fluid Balance: Fluid management differs from that of patients with burn injuries; fluid and electrolyte requirements are less than for burns of the same extent [115]. Fluid replacement with electrolyte solution (0.7 mL/kg/% affected area) and albumin solution (5 % human albumin, 1 mL/kg/% affected area) is advised [99]. The goal is to maintain a urinary output of 0.5–1 mL/kg/h [115].
Nutritional Support: Enteral nutritional hypercaloric and high-protein diet support is advised to prevent protein loss and promote healing. Enteral nutrition through a nasogastric tube should be considered in patients unable to ingest food. Nasogastric feeding should be initiated cautiously because of possible gastrointestinal involvement and potential difficulties in placing a tube in the presence of oropharyngeal involvement [116]. Parenteral nutrition is not recommended, because it is commonly poor tolerated and is associated with an increased risk of sepsis [114]. Patients with SJS/TEN require fewer calories per day than patients with burns. A recommended equation for the estimation of energy requirements in pediatric SJS/TEN patients is (24.6 × weight in kg) + (% wound × 4.1) + 940 [117]. Other guidelines advise the following protocol for nutrition through a gastric tube: 1500 calories in 1500 mL over the first 24 h, increasing by 500 calories daily to 3500–4000 calories daily [99].
Pain Management: Management of pain is a major point, often underestimated, and necessitates assessment and medical treatment accordingly. Use of morphine should be considered, with appropriate respiratory monitoring. If available, patient-controlled analgesia is advised [114]. Placement of the patient on an alternating pressure air mattress may also reduce the pain [99].
Venous Thromboembolism Prophylaxis: Standard prophylactic anticoagulation therapy is advised for patients who are bedridden [114].
Monitoring for Infection: Antibiotic prophylaxis is not recommended. However, monitoring for infection is mandatory, and if clinical suspicion arises, bacterial cultures should be obtained from the skin, urine, and blood, and empirical treatment with antibiotics given until culture results are available. [99] Some guidelines advise performing bacterial and fungal cultures from the skin, urine, and blood on a regular basis (2–3 times per week) [112–114].
Psychological Support: Professional psychological support for the patient and family members is important [99].
4.2.4 Medical Treatment
Systemic Immunomodulatory Treatment in Adults: The optimal therapeutic regimen has yet to be established, but, according to recent publications, the following conclusions can be drawn.
-
1.
Intravenous immunoglobulin (IVIg): Use of IVIg does not yield survival benefits in adults with SJS/TEN [118–120].
-
2.
Cyclosporine: In an open-label, phase II trial to determine the safety and possible benefit of cyclosporine, cyclosporine was found to decrease the death rate and the progression of detachment in adults (dosage: 3 mg/kg/day for 10 days and tapered over a month) [121]. Other publications have also reported beneficial use of cyclosporine in SJS/TEN [122, 123].
-
3.
Systemic Corticosteroids: These were associated with clinical benefit according to the EuroSCAR (European Study of Severe Cutaneous Adverse Reactions) study [124] and were reported to be the most common treatment for SJS/TEN in a recent survey of 50 drug hypersensitivity experts from 20 countries [125]. One of the suggested protocols is intravenous dexamethasone 1.5 mg/kg pulse therapy (given for 30–60 min) for three consecutive days [126].
-
4.
TNF Inhibitors: Treatment with anti-TNF biologic agents seems to be very promising for the management of SJS/TEN [127, 128]. In a recent study in ten consecutive patients with TEN, 50 mg of etanercept was administered in a single subcutaneous injection. All patients responded promptly to treatment, reaching complete re-epithelialization without complications or side effects [129]. In addition, the preliminary results of a prospective, randomized, open-label trial currently underway in Taiwan [125], comparing etanercept with systemic corticosteroids in patients with SJS/TEN, demonstrated that the average time to reach maximal skin detachment and complete skin healing was shorter in the etanercept group. Also, in vitro investigations demonstrated that etanercept, corticosteroids, and thalidomide significantly decreased granulysin expression of blister cells. Etanercept did not, however, increase the cytotoxic effect on keratinocytes found with thalidomide [125].
Systemic Immunomodulatory Treatment in Children: The bulk of the literature on the management of SJS/TEN includes adults, and these findings may not be simply extrapolated to children. On the basis of the scant quality literature in children, both IVIg and systemic corticosteroids seem to improve the outcome of children with SJS/TEN [130–132].
Skin Treatment: There are no clinical guidelines for the skin care of patients with SJS/TEN. Debridement of the necrotic epidermis was recommended in past publications [112, 133]. Recent publications advise avoiding debridement (which may cause hypertrophic scars) and recommend considering the detached epidermis as a natural biological dressing that favors re-epithelialization [64, 114, 125]. Various topical treatments that have been reported include bioactive skin substitutes, semi-synthetic and synthetic dressings, and topical antimicrobials [112, 133]. A recent report on the management of SJS/TEN in an experienced French referral center described the following treatment: wound care once daily with minimal manipulation to prevent skin detachment, including a bath containing a solution of chlorhexidine 1/5000 (morphine is given prior to the bath and/or an equimolar mix of oxygen and nitrogen monoxide during the bath); if bathing is not possible, the chlorhexidine solution is sprayed 2–3 times daily on the skin; blister fluid is aspirated while maintaining the blister roof; petrolatum is systematically applied over all detached skin areas; topical sulfa-containing medications are avoided; and hydrocellular or absorbent nonadhesive dressings are applied at least once daily to cover pressure points [114]. Another recently published protocol [134] based the wound management algorithm on the stage of denudation and skin loss, categorizing four stages of skin with specialized treatments accordingly.
Mucous Membrane Treatment: Specialized care is essential to prevent lifelong complications [64].
-
1.
Ocular management: Although there is no standardized care for ocular management, the following supportive local treatment is advised: tear replacement solutions; removal of peudomembranes; lysis of symblepharon; debridement of loosened epithelium; topical antibiotics to prevent secondary infection; topical corticosteroids to prevent scar formation; and cycloplegic drops to relieve pain, photophobia, and ciliary spasm [135]. Amniotic membrane transplantation has been found to be effective in the acute and chronic stages of SJS/TEN [136, 137]. A “Triple-TEN” protocol for severe ocular cases was recently reported [138], comprising the following:
-
(a)
Subconjunctival triamcinolone (Kenalog™ 20 mg) is administered into each of the fornices to curb the local inflammatory response without compromising systemic immunity.
-
(b)
Amniotic membrane tissue mounted on a polycarbonate skirt (ProKera®) is placed over the corneal and limbal regions to facilitate re-epithelialization of the ocular surface.
-
(c)
A steeply curved acrylic scleral shell spacer (Technovent, SC21) is inserted to vault each lid away from the globe and provide a barrier to symblephara formation. This treatment offers an effective therapeutic option without the need for microsurgical equipment, a microscope, or sutures in the critical care setting.
-
(a)
-
2.
Oropharyngeal Management: The mouth should be rinsed several times daily with an antiseptic or antifungal solution, and the lips should be lubricated with an ointment such as dexpanthenol [99].
-
3.
Genital Management: Vulvovaginal sequelae of SJS/TEN are well documented in the literature, though little consensus exists about effective preventive strategies. The goals of therapy should be to protect vaginal function by decreasing adhesion formation and agglutination, as well as limiting vulvovaginal adenosis with potentially neoplastic changes in affected tissue [72, 139–141]. Some treatment protocols suggest wet dressings or sitz baths and lubrication with emollient to avoid adhesions and strictures of genital erosions in females [99, 114]. In a recent expert opinion publication [141], the authors advised using the following protocol:
-
(a)
Early initiation of intravaginal steroids: Betamethasone valerate 0.1 % cream applied every 12 h to the vulva externally, and betamethasone valerate 0.1 % ointment applied every 12 h to the internal vaginal mucosa via a Milex® dilator (Milex Products Inc., Chicago, IL, USA); regular application of antifungal creams as well. Treatment was advised to be given in alternating courses to prevent tachyphylaxis (for example, 3 days on, 4 days off), and treatment with topical steroids should be continued until resolution of the acute phase of the illness.
-
(b)
Vaginal molds: In addition to topical steroids, a soft vaginal mold should be placed prophylactically as early as possible during the acute phase of the illness and used regularly until complete healing of ulcerative lesions has occurred. The mold can be coated with topical steroids, and patients are advised to wear it for most of the day, removing it only for cleansing, medication application, and toileting. The author advised using Milex® vaginal dilators; they are made of latex-free, hypoallergenic silicone and come in various lengths and widths. They are available to purchase from online distributors (e.g. CooperSurgical, Trumbull, CT, USA). Special consideration is to be given to virginal patients, in whom use of vaginal molds would be inadvisable. Instead, the steroid formulation can be applied via a standard vaginal applicator. Also, use of a dilator is not advised in the pediatric population, to prevent physical discomfort and emotional trauma.
-
(c)
Menstrual suppression: To decrease the risk of vaginal adenosis, menstrual suppression in the acute phase and healing process is advised. Detailed information on treatment of confirmed adenosis and the importance of close colposcopic surveillance, given the association of this process with subsequent malignancy, is described in this report [141].
-
(a)
Special Considerations in Pregnant Women: Many cases of SJS/TEN occurring during pregnancy have been reported, with high survival rates of both the mothers and the newborns [142–144].
The following major aspects are of special concern when treating a pregnant woman with SJS/TEN:
-
1.
Fetal status: Fetal stress due to maternal disease may lead to premature labor.
-
2.
Delivery methods: Although vaginal deliveries have been reported [145], Caesarean section has been used in many cases [142, 144] because of genital mucous membrane involvement of TEN. Risk management for labor includes frequent obstetric examinations to assess possible vaginal strictures, which may impair spontaneous delivery. If a spontaneous delivery is planned, preparations for a Caesarean section should be made, and anesthetic and neonatology support should be available [142].
-
3.
Involvement of the disease within the fetus/newborn: SJS/TEN may develop during pregnancy in the mother alone or simultaneously in the mother and fetus; it can be lethal for the fetus [142]. In cases reported in the literature, most babies presented as healthy [142, 144]. However, a few cases of TEN in newborns, including one case of simultaneous TEN in a mother and her stillborn fetus, have been reported [143].
-
4.
HIV: Pregnancy increases the chances of developing SJS 14-fold in HIV-infected pregnant women treated with nevirapine [146]. A recently published retrospective study of patients admitted to a university hospital with SJS/TEN over a 3-year period in Cape Town, South Africa [147], found that nevirapine-based antiretroviral therapy—a regimen for women in the first trimester of pregnancy and those planning a pregnancy to prevent mother-to-child transmission of HIV infection—placed pregnant women at a disproportionally higher risk of developing SJS/TEN.
Avoiding High-Risk Drugs and Monitoring for Secondary Cutaneous Adverse Drug Reactions: Avoiding high-risk drugs (Table 4) is important, as SJS/TEN predisposes the patient to developing additional cutaneous adverse drug reactions.
Monitoring and Treating Acute Complications: The most common cause of death in SJS/TEN in the acute stage is septicemia. Nonetheless, other possible complications induced by changes in the hemodynamic status and systemic involvement of SJS/TEN are to be monitored and treated.
4.2.5 Communication with the Patient, Family, Health Care Providers and Regulatory Agencies
Patient and Family: Good communication strategies will aid in the interactions with the patient and family following SJS/TEN and will decrease the likelihood of lawsuits. Physicians are advised to follow these steps:
-
1.
Express empathy and say “sorry” according to the “apology laws” in an honest and respectful fashion, and in a way that protects the physician from having an apology used against him in case of legal action. See http://www.sorryworks.net/.
-
2.
Provide disclosure in a “disclosure meeting” planned according to the acronym CONES (Context: arrange the setting for a quiet, uninterrupted meeting, and decide on the participants; Opening shot: the first sentence in the meeting explains the aim of the conversation; Narrative: lay out the facts, and avoid using the words “error” and “mistake”, since the SJS/TEN is a result of multiple factors, particularly when the facts are not completely known; Emotions: provide an empathic environment; Summary).
-
3.
Provide the patient with clear information about SJS/TEN and its cause, the name of the offending drug, potential cross-reacting drugs, and drugs that can be safely taken as an alternative to the offending drug. In addition, provide the results of the HLA genetic screening, if conducted, and advise the patient to wear a MedicAlert® bracelet.
-
4.
Family counseling is part of the management plan because of genetic predisposition. The authors’ protocol is to advise all first-degree family members to avoid use of the drug that caused the SJS/TEN in their family member (unless past treatment was safe) [3], and to provide information on HLA genetic screening for the relevant drug if available.
Health Care Providers: Information on the adverse event must be provided to the family physician and entered into the patient’s records.
Regulatory Agencies: Report the cutaneous adverse drug reaction to the manufacturer and regulatory agencies [148]. International reporting systems include MedWatch, the FDA Safety Information and Adverse Event Reporting Program (http://www.fda.gov/safety/MedWatch/default.htm), and the WHO Uppsala Monitoring Centre (UMC; http://www.who-umc.org).
4.2.6 Prior to Discharge
Information on the Culprit Drug: This should be provided to the patient and their primary physicians. Appropriate labeling should be applied to the medical record, according to the most likely causative drugs. Care should be taken to verify that the appropriate coding has been applied (e.g. International Classification of Diseases, Tenth Revision [ICD-10] for future reference and studies.
Medical Recommendations: Advise on medical treatment, if still necessary, and provide clear recommendations on sun protection to prevent post-inflammatory hyperpigmentation.
Explanation of the Possible Long-Term Medical Complications and Information on Medical Follow-Up: Long-term physical complications of patients surviving SJS/TEN are common (see elaboration in Sect. 3). Hence, it is highly important to explain to the patient the prognosis and the need for follow-up.
Offering Emotional Support: We recommend using the General Health Questionnaire-12 (GHQ-12) [149]—the most commonly used screening instrument for psychiatric disorders in various settings—as a quick and valid screening instrument for psychological distress. Patients should be asked to complete the scale at discharge, and scores of 2 or greater should trigger a referral to a psychiatrist or psychologist [94].
Referral to a Support Group: Several support groups have been established in different countries, such as the Stevens–Johnson Syndrome Foundation in the USA, CAST (Canadians Against Stevens Johnson Syndrome & Toxic Epidermal Necrolysis) in Canada, and Amalyste in France. More information on support groups in different countries can be found at http://www.sjsupport.org/sjsupport_group_facilitators.shtml.
5 Conclusion
SJS/TEN is one of the few dermatological diseases that constitute a true medical emergency.
In the last decade, important progress has been achieved in our understanding of the pathogenesis, clinical presentation, and treatment of SJS/TEN. Furthermore, laboratory-based discovery of the HLA alleles associated with SJS/TEN induced by drugs has been translated into a clinical guideline-based test, which serves as a practical clinical tool for genetic screening to prevent SJS/TEN.
However, there is still no international consensus on the management of patients with SJS/TEN, and genetic screening is not well implemented in most countries.
This review has summarized up-to-date insights on SJS/TEN, including new discoveries on the genetic associations of SJS/TEN, and described a protocol for assessment and treatment. We hope that this summary will increase the awareness among physicians regarding the importance of genetic screening for decreasing the incidence of SJS/TEN and that our suggested management protocol will serve as a practical clinical tool for physicians.
Further research is needed to generate adequate in vitro and in vivo models for SJS/TEN, to develop novel treatments, to study the involvement of unknown pathogens, and to further investigate the mechanism of keratinocyte death.
References
Stevens A, Johnson F. A new eruptive fever associated with stomatitis and ophthalmia: report of two cases in children. Arch Pediatr Adolesc Med. 1922;24(6):526.
Lyell A. Toxic epidermal necrolysis: an eruption resembling scalding of the skin. Br J Dermatol. 1956;68(11):355–61.
Knowles S, Shear NH. Clinical risk management of Stevens–Johnson syndrome/toxic epidermal necrolysis spectrum. Dermatol Ther. 2009;22(5):441–51.
Roujeau JC, Huynh TN, Bracq C, et al. Genetic susceptibility to toxic epidermal necrolysis. Arch Dermatol. 1987;123(9):1171–3.
Chung WH, Hung SI, Hong HS, et al. Medical genetics: a marker for Stevens–Johnson syndrome. Nature. 2004;428(6982):486.
Kaniwa N, Saito Y, Aihara M, et al. HLA-B*1511 is a risk factor for carbamazepine-induced Stevens–Johnson syndrome and toxic epidermal necrolysis in Japanese patients. Epilepsia. 2010;51(12):2461–5.
Kim SH, Lee KW, Song WJ, et al. Carbamazepine-induced severe cutaneous adverse reactions and HLA genotypes in Koreans. Epilepsy Res. 2011;97(1–2):190–7.
Shi YW, Min FL, Qin B, et al. Association between HLA and Stevens–Johnson syndrome induced by carbamazepine in southern Han Chinese: genetic markers besides B*1502? Basic Clin Pharmacol Toxicol. 2012;111(1):58–64.
Hsiao YH, Hui RC, Wu T, et al. Genotype–phenotype association between HLA and carbamazepine-induced hypersensitivity reactions: strength and clinical correlations. Journal of dermatological science. 2014;73(2):101–9.
Kaniwa N, Saito Y, Aihara M, et al. HLA-B locus in Japanese patients with anti-epileptics and allopurinol-related Stevens–Johnson syndrome and toxic epidermal necrolysis. Pharmacogenomics. 2008;9(11):1617–22.
Kang HR, Jee YK, Kim YS, et al. Positive and negative associations of HLA class I alleles with allopurinol-induced SCARs in Koreans. Pharmacogenet Genomics. 2011;21(5):303–7.
Tassaneeyakul W, Jantararoungtong T, Chen P, et al. Strong association between HLA-B*5801 and allopurinol-induced Stevens–Johnson syndrome and toxic epidermal necrolysis in a Thai population. Pharmacogenet Genomics. 2009;19(9):704–9.
Hung SI, Chung WH, Liou LB, et al. HLA-B*5801 allele as a genetic marker for severe cutaneous adverse reactions caused by allopurinol. Proc Natl Acad Sci USA. 2005;102(11):4134–9.
Ikeda H, Takahashi Y, Yamazaki E, et al. HLA class I markers in Japanese patients with carbamazepine-induced cutaneous adverse reactions. Epilepsia. 2010;51(2):297–300.
Niihara H, Kakamu T, Fujita Y, et al. HLA-A31 strongly associates with carbamazepine-induced adverse drug reactions but not with carbamazepine-induced lymphocyte proliferation in a Japanese population. J Dermatol. 2012;39(7):594–601.
Ozeki T, Mushiroda T, Yowang A, et al. Genome-wide association study identifies HLA-A*3101 allele as a genetic risk factor for carbamazepine-induced cutaneous adverse drug reactions in Japanese population. Hum Mol Genet. 2011;20(5):1034–41.
Cheung YK, Cheng SH, Chan EJ, et al. HLA-B alleles associated with severe cutaneous reactions to antiepileptic drugs in Han Chinese. Epilepsia. 2013;54(7):1307–14.
Hung SI, Chung WH, Liu ZS, et al. Common risk allele in aromatic antiepileptic-drug induced Stevens–Johnson syndrome and toxic epidermal necrolysis in Han Chinese. Pharmacogenomics. 2010;11(3):349–56.
Locharernkul C, Loplumlert J, Limotai C, et al. Carbamazepine and phenytoin induced Stevens–Johnson syndrome is associated with HLA-B*1502 allele in Thai population. Epilepsia. 2008;49(12):2087–91.
Kim SH, Kim M, Lee KW, et al. HLA-B*5901 is strongly associated with methazolamide-induced Stevens–Johnson syndrome/toxic epidermal necrolysis. Pharmacogenomics. 2010;11(6):879–84.
Lonjou C, Borot N, Sekula P, et al. A European study of HLA-B in Stevens–Johnson syndrome and toxic epidermal necrolysis related to five high-risk drugs. Pharmacogenet Genomics. 2008;18(2):99–107.
Mehta TY, Prajapati LM, Mittal B, et al. Association of HLA-B*1502 allele and carbamazepine-induced Stevens–Johnson syndrome among Indians. Indian J Dermatol Venereol Leprol. 2009;75(6):579–82.
Tangamornsuksan W, Chaiyakunapruk N, Somkrua R, et al. Relationship between the HLA-B*1502 allele and carbamazepine-induced Stevens–Johnson syndrome and toxic epidermal necrolysis: a systematic review and meta-analysis. JAMA Dermatol. 2013;149(9):1025–32.
Chung WH, Hung SI. Recent advances in the genetics and immunology of Stevens–Johnson syndrome and toxic epidermal necrosis. J Dermatol Sci. 2012;66(3):190–6.
Chen P, Lin JJ, Lu CS, et al. Carbamazepine-induced toxic effects and HLA-B*1502 screening in Taiwan. N Engl J Med. 2011;364(12):1126–33.
Hershfield MS, Callaghan JT, Tassaneeyakul W, et al. Clinical Pharmacogenetics Implementation Consortium guidelines for human leukocyte antigen-B genotype and allopurinol dosing. Clin Pharmacol Ther. 2013;93(2):153–8.
Genin E, Chen DP, Hung SI, et al. HLA-A*31:01 and different types of carbamazepine-induced severe cutaneous adverse reactions: an international study and meta-analysis. Pharmacogenomics J. 2014;14(3):281–8.
Shear NH, Spielberg SP. Anticonvulsant hypersensitivity syndrome: in vitro assessment of risk. J Clin Investig. 1988;82(6):1826–32.
Shear NH, Spielberg SP, Grant DM, et al. Differences in metabolism of sulfonamides predisposing to idiosyncratic toxicity. Ann Intern Med. 1986;105(2):179–84.
Chung WH, Chang WC, Lee YS, et al. Genetic variants associated with phenytoin-related severe cutaneous adverse reactions. JAMA. 2014;312(5):525–34.
Kesavan R, Narayan SK, Adithan C. Influence of CYP2C9 and CYP2C19 genetic polymorphisms on phenytoin-induced neurological toxicity in Indian epileptic patients. Eur J Clin Pharmacol. 2010;66(7):689–96.
Depondt C, Godard P, Espel RS, et al. A candidate gene study of antiepileptic drug tolerability and efficacy identifies an association of CYP2C9 variants with phenytoin toxicity. Eur J Neurol Off J Eur Fed Neurol Soc. 2011;18(9):1159–64.
Ciccacci C, Di Fusco D, Marazzi MC, et al. Association between CYP2B6 polymorphisms and nevirapine-induced SJS/TEN: a pharmacogenetics study. Eur J Clin Pharmacol. 2013;69(11):1909–16.
Lerch M, Pichler WJ. The immunological and clinical spectrum of delayed drug-induced exanthems. Curr Opin Allergy Clin Immunol. 2004;4(5):411–9.
Pichler WJ. Pharmacological interaction of drugs with antigen-specific immune receptors: the p-i concept. Curr Opin Allergy Clin Immunol. 2002;2(4):301–5.
Yang CW, Hung SI, Juo CG, et al. HLA-B*1502-bound peptides: implications for the pathogenesis of carbamazepine-induced Stevens–Johnson syndrome. J Allergy Clin Immunol. 2007;120(4):870–7.
Wei CY, Chung WH, Huang HW, et al. Direct interaction between HLA-B and carbamazepine activates T cells in patients with Stevens–Johnson syndrome. J Allergy Clin Immunol. 2012;129(6):1562–9 (e5).
Torres MJ, Mayorga C, Garcia JJ, et al. New aspects in betalactam recognition. Clin Exp Allergy. 1998;28(Suppl 4):25–8.
Ko TM, Chung WH, Wei CY, et al. Shared and restricted T-cell receptor use is crucial for carbamazepine-induced Stevens–Johnson syndrome. J Allergy Clin Immunol. 2011;128(6):1266–76 (e11).
Posadas SJ, Padial A, Torres MJ, et al. Delayed reactions to drugs show levels of perforin, granzyme B, and Fas-L to be related to disease severity. J Allergy Clin Immunol. 2002;109(1):155–61.
Nassif A, Moslehi H, Le Gouvello S, et al. Evaluation of the potential role of cytokines in toxic epidermal necrolysis. J Investig Dermatol. 2004;123(5):850–5.
Caproni M, Torchia D, Schincaglia E, et al. Expression of cytokines and chemokine receptors in the cutaneous lesions of erythema multiforme and Stevens–Johnson syndrome/toxic epidermal necrolysis. Br J Dermatol. 2006;155(4):722–8.
Liu ZG. Molecular mechanism of TNF signaling and beyond. Cell Res. 2005;15(1):24–7.
Chavez-Galan L, Arenas-Del Angel MC, Zenteno E, et al. Cell death mechanisms induced by cytotoxic lymphocytes. Cell Mol Immunol. 2009;6(1):15–25.
Schroder K, Hertzog PJ, Ravasi T, et al. Interferon-gamma: an overview of signals, mechanisms and functions. J Leukoc Biol. 2004;75(2):163–89.
Steimle V, Siegrist CA, Mottet A, et al. Regulation of MHC class II expression by interferon-gamma mediated by the transactivator gene CIITA. Science. 1994;265(5168):106–9.
Fruh K, Yang Y. Antigen presentation by MHC class I and its regulation by interferon gamma. Curr Opin Immunol. 1999;11(1):76–81.
Paquet P, Paquet F, Al Saleh W, et al. Immunoregulatory effector cells in drug-induced toxic epidermal necrolysis. Am J Dermatopathol. 2000;22(5):413–7.
Correia O, Delgado L, Barbosa IL, et al. Increased interleukin 10, tumor necrosis factor alpha, and interleukin 6 levels in blister fluid of toxic epidermal necrolysis. J Am Acad Dermatol. 2002;47(1):58–62.
Tapia B, Padial A, Sanchez-Sabate E, et al. Involvement of CCL27–CCR10 interactions in drug-induced cutaneous reactions. J Allergy Clin Immunol. 2004;114(2):335–40.
Chung WH, Hung SI. Genetic markers and danger signals in Stevens–Johnson syndrome and toxic epidermal necrolysis. Allergol Int. 2010;59(4):325–32.
Chung WH, Hung SI, Yang JY, et al. Granulysin is a key mediator for disseminated keratinocyte death in Stevens–Johnson syndrome and toxic epidermal necrolysis. Nat Med. 2008;14(12):1343–50.
Viard I, Wehrli P, Bullani R, et al. Inhibition of toxic epidermal necrolysis by blockade of CD95 with human intravenous immunoglobulin. Science. 1998;282(5388):490–3.
Nassif A, Bensussan A, Dorothee G, et al. Drug specific cytotoxic T-cells in the skin lesions of a patient with toxic epidermal necrolysis. J Investig Dermatol. 2002;118(4):728–33.
Wang CW, Chung WH, Cheng YF, et al. A new nucleic acid–based agent inhibits cytotoxic T lymphocyte-mediated immune disorders. J Allergy Clin Immunol. 2013;132(3):713–22 (e11).
Finn MW, Clayberger C, Krensky AM. Expression and purification of 15 kDa granulysin utilizing an insect cell secretion system. Protein Expr Purif. 2011;75(1):70–4.
Tewary P, Yang D, de la Rosa G, et al. Granulysin activates antigen-presenting cells through TLR4 and acts as an immune alarmin. Blood. 2010;116(18):3465–74.
Schlapbach C, Zawodniak A, Irla N, et al. NKp46+ cells express granulysin in multiple cutaneous adverse drug reactions. Allergy. 2011;66(11):1469–76.
Fournier S, Bastuji-Garin S, Mentec H, et al. Toxic epidermal necrolysis associated with Mycoplasma pneumoniae infection. Eur J Clin Microbiol Infect Dis. 1995;14(6):558–9.
Mulvey JM, Padowitz A, Lindley-Jones M, et al. Mycoplasma pneumoniae associated with Stevens Johnson syndrome. Anaesth Intensive Care. 2007;35(3):414–7.
Harr T, French LE. Toxic epidermal necrolysis and Stevens–Johnson syndrome. Orphanet J Rare Dis. 2010;5:39.
Chung WH, Shih SR, Chang CF, et al. Clinicopathologic analysis of coxsackievirus a6 new variant induced widespread mucocutaneous bullous reactions mimicking severe cutaneous adverse reactions. J Infect Dis. 2013;208(12):1968–78.
Rzany B, Mockenhaupt M, Baur S, et al. Epidemiology of erythema exsudativum multiforme majus, Stevens–Johnson syndrome, and toxic epidermal necrolysis in Germany (1990–1992): structure and results of a population-based registry. Journal of clinical epidemiology. 1996;49(7):769–73.
Mockenhaupt M. The current understanding of Stevens–Johnson syndrome and toxic epidermal necrolysis. Expert review of clinical immunology. 2011;7(6):803–13 (quiz 14–5).
Mockenhaupt M, Viboud C, Dunant A, et al. Stevens–Johnson syndrome and toxic epidermal necrolysis: assessment of medication risks with emphasis on recently marketed drugs. The EuroSCAR-study. J Investig Dermatol. 2008;128(1):35–44.
Huang LY, Liao WC, Chiou CC, et al. Fatal toxic epidermal necrolysis induced by carbamazepine treatment in a patient who previously had carbamazepine-induced Stevens–Johnson syndrome. J Formos Med Assoc Taiwan yi Zhi. 2007;106(12):1032–7.
Auquier-Dunant A, Mockenhaupt M, Naldi L, et al. Correlations between clinical patterns and causes of erythema multiforme majus, Stevens–Johnson syndrome, and toxic epidermal necrolysis: results of an international prospective study. Arch Dermatol. 2002;138(8):1019–24.
Assier H, Bastuji-Garin S, Revuz J, et al. Erythema multiforme with mucous membrane involvement and Stevens–Johnson syndrome are clinically different disorders with distinct causes. Arch Dermatol. 1995;131(5):539–43.
Bastuji-Garin S, Rzany B, Stern RS, et al. Clinical classification of cases of toxic epidermal necrolysis, Stevens–Johnson syndrome, and erythema multiforme. Arch Dermatol. 1993;129(1):92–6.
Revuz J, Penso D, Roujeau JC, et al. Toxic epidermal necrolysis: clinical findings and prognosis factors in 87 patients. Arch Dermatol. 1987;123(9):1160–5.
Rajaratnam R, Mann C, Balasubramaniam P, et al. Toxic epidermal necrolysis: retrospective analysis of 21 consecutive cases managed at a tertiary centre. Clin Exp Dermatol. 2010;35(8):853–62.
Meneux E, Wolkenstein P, Haddad B, et al. Vulvovaginal involvement in toxic epidermal necrolysis: a retrospective study of 40 cases. Obstet Gynecol. 1998;91(2):283–7.
Gueudry J, Roujeau JC, Binaghi M, et al. Risk factors for the development of ocular complications of Stevens–Johnson syndrome and toxic epidermal necrolysis. Arch Dermatol. 2009;145(2):157–62.
Sotozono C, Ueta M, Koizumi N, et al. Diagnosis and treatment of Stevens–Johnson syndrome and toxic epidermal necrolysis with ocular complications. Ophthalmology. 2009;116(4):685–90.
Pereira FA, Mudgil AV, Rosmarin DM. Toxic epidermal necrolysis. J Am Acad Dermatol. 2007;56(2):181–200.
Lebargy F, Wolkenstein P, Gisselbrecht M, et al. Pulmonary complications in toxic epidermal necrolysis: a prospective clinical study. Intensive care Med. 1997;23(12):1237–44.
de Prost N, Mekontso-Dessap A, Valeyrie-Allanore L, et al. Acute respiratory failure in patients with toxic epidermal necrolysis: clinical features and factors associated with mechanical ventilation. Crit Care Med. 2014;42(1):118–28.
Michel P, Joly P, Ducrotte P, et al. Ileal involvement in toxic epidermal necrolysis (Lyell syndrome). Dig Dis Sci. 1993;38(10):1938–41.
Powell N, Munro JM, Rowbotham D. Colonic involvement in Stevens–Johnson syndrome. Postgrad Med J. 2006;82(968):e10.
Hung CC, Liu WC, Kuo MC, et al. Acute renal failure and its risk factors in Stevens–Johnson syndrome and toxic epidermal necrolysis. Am J Nephrol. 2009;29(6):633–8.
Weinand C, Xu W, Perbix W, et al. 27 years of a single burn centre experience with Stevens–Johnson syndrome and toxic epidermal necrolysis: analysis of mortality risk for causative agents. Burns. 2013;39(7):1449–55.
de Prost N, Ingen-Housz-Oro S, Duong T, et al. Bacteremia in Stevens–Johnson syndrome and toxic epidermal necrolysis: epidemiology, risk factors, and predictive value of skin cultures. Medicine (Baltimore). 2010;89(1):28–36.
Sekula P, Dunant A, Mockenhaupt M, et al. Comprehensive survival analysis of a cohort of patients with Stevens–Johnson syndrome and toxic epidermal necrolysis. J Investig Dermatol. 2013;133(5):1197–204.
Garcia-Doval I, LeCleach L, Bocquet H, et al. Toxic epidermal necrolysis and Stevens–Johnson syndrome: does early withdrawal of causative drugs decrease the risk of death? Arch Dermatol. 2000;136(3):323–7.
Bastuji-Garin S, Fouchard N, Bertocchi M, et al. SCORTEN: a severity-of-illness score for toxic epidermal necrolysis. J Investig Dermatol. 2000;115(2):149–53.
Zhu QY, Ma L, Luo XQ, et al. Toxic epidermal necrolysis: performance of SCORTEN and the score-based comparison of the efficacy of corticosteroid therapy and intravenous immunoglobulin combined therapy in China. J Burn Care Res. 2012;33(6):e295–308.
Cartotto R, Mayich M, Nickerson D, et al. SCORTEN accurately predicts mortality among toxic epidermal necrolysis patients treated in a burn center. J Burn Care Res. 2008;29(1):141–6.
Sekula P, Liss Y, Davidovici B, et al. Evaluation of SCORTEN on a cohort of patients with Stevens–Johnson syndrome and toxic epidermal necrolysis included in the RegiSCAR study. J Burn Care Res. 2011;32(2):237–45.
Guegan S, Bastuji-Garin S, Poszepczynska-Guigne E, et al. Performance of the SCORTEN during the first five days of hospitalization to predict the prognosis of epidermal necrolysis. J Investig Dermatol. 2006;126(2):272–6.
Fellahi A, Zouhair K, Amraoui A, et al. Stevens–Johnson and Lyell syndromes: mucocutaneous and ocular sequels in 43 cases [in French]. Ann Dermatol Venereol. 2011;138(2):88–92.
Morales ME, Purdue GF, Verity SM, et al. Ophthalmic manifestations of Stevens–Johnson syndrome and toxic epidermal necrolysis and relation to SCORTEN. Am J Ophthalmol. 2010;150(4):505–10 (e1).
Gaultier F, Rochefort J, Landru MM, et al. Severe and unrecognized dental abnormalities after drug-induced epidermal necrolysis. Arch Dermatol. 2009;145(11):1332–3.
Duong TA, de Prost N, Ingen-Housz-Oro S, et al. Stevens–Johnson syndrome and toxic epidermal necrolysis: follow-up of pulmonary function after remission. Br J Dermatol. 2015;172(2):400–5.
Dodiuk-Gad RP, Olteanu C, Hashimoto R, et al. Long-term emotional and physical complications of Stevens–Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN). 23rd World Congress of Dermatology; Vancouver; 2015.
Quinn AM, Brown K, Bonish BK, et al. Uncovering histologic criteria with prognostic significance in toxic epidermal necrolysis. Arch Dermatol. 2005;141(6):683–7.
Fujita Y, Yoshioka N, Abe R, et al. Rapid immunochromatographic test for serum granulysin is useful for the prediction of Stevens–Johnson syndrome and toxic epidermal necrolysis. J Am Acad Dermatol. 2011;65(1):65–8.
Nigen S, Knowles SR, Shear NH. Drug eruptions: approaching the diagnosis of drug-induced skin diseases. J Drugs Dermatol. 2003;2(3):278–99.
Eisen ER, Fish J, Shear NH. Management of drug-induced toxic epidermal necrolysis. J Cutan Med Surg. 2000;4(2):96–102.
Mockenhaupt M. Stevens–Johnson syndrome and toxic epidermal necrolysis: clinical patterns, diagnostic considerations, etiology, and therapeutic management. Semin Cutan Med Surg. 2014;33(1):10–6.
Chavan R, el-Azhary R. Cutaneous graft-versus-host disease: rationales and treatment options. Dermatol Ther. 2011;24(2):219–28.
Mandelcorn R, Shear NH. Lupus-associated toxic epidermal necrolysis: a novel manifestation of lupus? J Am Acad Dermatol. 2003;48(4):525–9.
Paradela S, Martinez-Gomez W, Fernandez-Jorge B, et al. Toxic epidermal necrolysis-like acute cutaneous lupus erythematosus. Lupus. 2007;16(9):741–5.
Baldwin BT, Lien MH, Khan H, et al. Case of fatal toxic epidermal necrolysis due to cardiac catheterization dye. J Drugs Dermatol. 2010;9(7):837–40.
Ball R, Ball LK, Wise RP, et al. Stevens–Johnson syndrome and toxic epidermal necrolysis after vaccination: reports to the vaccine adverse event reporting system. Pediatr Infect Dis J. 2001;20(2):219–23.
Sassolas B, Haddad C, Mockenhaupt M, et al. ALDEN, an algorithm for assessment of drug causality in Stevens–Johnson syndrome and toxic epidermal necrolysis: comparison with case–control analysis. Clin Pharmacol Ther. 2010;88(1):60–8.
Heelan K, Shear NH. Cutaneous drug reactions in children: an update. Paediatr Drugs. 2013;15(6):493–503.
Pavlos R, Mallal S, Ostrov D, et al. Fever, rash, and systemic symptoms: understanding the role of virus and HLA in severe cutaneous drug allergy. J Allergy Clin Immunol Pract. 2014;2(1):21–33.
Pichler WJ, Tilch J. The lymphocyte transformation test in the diagnosis of drug hypersensitivity. Allergy. 2004;59(8):809–20.
Porebski G, Pecaric-Petkovic T, Groux-Keller M, et al. In vitro drug causality assessment in Stevens–Johnson syndrome—alternatives for lymphocyte transformation test. Clin Exp Allergy. 2013;43(9):1027–37.
Schwartz RA, McDonough PH, Lee BW. Toxic epidermal necrolysis: part I. Introduction, history, classification, clinical features, systemic manifestations, etiology, and immunopathogenesis. J Am Acad Dermatol. 2013;69(2):173 e1–13 (quiz 85–6).
Valeyrie-Allanore L, Bastuji-Garin S, Guegan S, et al. Prognostic value of histologic features of toxic epidermal necrolysis. J Am Acad Dermatol. 2013;68(2):e29–35.
Endorf FW, Cancio LC, Gibran NS. Toxic epidermal necrolysis clinical guidelines. J Burn Care Res. 2008;29(5):706–12.
Fromowitz JS, Ramos-Caro FA, Flowers FP, et al. Practical guidelines for the management of toxic epidermal necrolysis and Stevens–Johnson syndrome. Int J Dermatol. 2007;46(10):1092–4.
Valeyrie-Allanore L, Ingen-Housz-Oro S, Chosidow O, et al. French referral center management of Stevens–Johnson syndrome/toxic epidermal necrolysis. Dermatol Sin. 2013;31(4):191–5.
Shiga S, Cartotto R. What are the fluid requirements in toxic epidermal necrolysis? J Burn Care Res. 2010;31(1):100–4.
Fernando SL. The management of toxic epidermal necrolysis. Australas J Dermatol. 2012;53(3):165–71.
Mayes T, Gottschlich M, Khoury J, et al. Energy requirements of pediatric patients with Stevens–Johnson syndrome and toxic epidermal necrolysis. Nutr Clin Pract. 2008;23(5):547–50.
Bachot N, Revuz J, Roujeau JC. Intravenous immunoglobulin treatment for Stevens–Johnson syndrome and toxic epidermal necrolysis: a prospective noncomparative study showing no benefit on mortality or progression. Arch Dermatol. 2003;139(1):33–6.
Faye O, Roujeau JC. Treatment of epidermal necrolysis with high-dose intravenous immunoglobulins (IV Ig): clinical experience to date. Drugs. 2005;65(15):2085–90.
Lee HY, Lim YL, Thirumoorthy T, et al. The role of intravenous immunoglobulin in toxic epidermal necrolysis: a retrospective analysis of 64 patients managed in a specialized centre. Br J Dermatol. 2013;169(6):1304–9.
Valeyrie-Allanore L, Wolkenstein P, Brochard L, et al. Open trial of ciclosporin treatment for Stevens–Johnson syndrome and toxic epidermal necrolysis. Br J Dermatol. 2010;163(4):847–53.
Arevalo JM, Lorente JA, Gonzalez-Herrada C, et al. Treatment of toxic epidermal necrolysis with cyclosporin A. J Trauma. 2000;48(3):473–8.
Kirchhof MG, Miliszewski MA, Sikora S, et al. Retrospective review of Stevens-Johnson syndrome/toxic epidermal necrolysis treatment comparing intravenous immunoglobulin with cyclosporine. J Am Acad Dermatol. 2014;71(5):941–7.
Schneck J, Fagot JP, Sekula P, et al. Effects of treatments on the mortality of Stevens–Johnson syndrome and toxic epidermal necrolysis: a retrospective study on patients included in the prospective EuroSCAR study. J Am Acad Dermatol. 2008;58(1):33–40.
Dodiuk-Gad RP, Chung WH, Yang CH, et al. The 8th International Congress on Cutaneous Adverse Drug Reactions, Taiwan, 2013: focus on severe cutaneous adverse reactions. Drug Saf. 2014;37(6):459–64.
Kardaun SH, Jonkman MF. Dexamethasone pulse therapy for Stevens–Johnson syndrome/toxic epidermal necrolysis. Acta dermato-venereol. 2007;87(2):144–8.
Scott-Lang V, Tidman M, McKay D. Toxic epidermal necrolysis in a child successfully treated with infliximab. Pediatr Dermatol. 2014;31(4):532–4.
Gubinelli E, Canzona F, Tonanzi T, et al. Toxic epidermal necrolysis successfully treated with etanercept. J Dermatol. 2009;36(3):150–3.
Paradisi A, Abeni D, Bergamo F, et al. Etanercept therapy for toxic epidermal necrolysis. J Am Acad Dermatol. 2014;71(2):278–83.
Del Pozzo-Magana BR, Lazo-Langner A, Carleton B, et al. A systematic review of treatment of drug-induced Stevens–Johnson syndrome and toxic epidermal necrolysis in children. J Popul Ther Clin Pharmacol. 2011;18:e121–33.
Koh MJ, Tay YK. Stevens–Johnson syndrome and toxic epidermal necrolysis in Asian children. J Am Acad Dermatol. 2010;62(1):54–60.
Hamilton GM, Fish J. Pediatric toxic epidermal necrolysis: an institutional review of patients admitted to an intensive care unit. J Burn Care Res. 2013;34(6):e351–8.
Paquet P, Pierard GE. Topical treatment options for drug-induced toxic epidermal necrolysis (TEN). Expert Opin Pharmacother. 2010;11(15):2447–58.
Abela C, Hartmann CE, De Leo A, et al. Toxic epidermal necrolysis (TEN): the Chelsea and Westminster Hospital wound management algorithm. J Plast Reconstr Aesthet Surg. 2014;67(8):1026–32.
Fu Y, Gregory DG, Sippel KC, et al. The ophthalmologist’s role in the management of acute Stevens–Johnson syndrome and toxic epidermal necrolysis. Ocul Surf. 2010;8(4):193–203.
Shay E, Kheirkhah A, Liang L, et al. Amniotic membrane transplantation as a new therapy for the acute ocular manifestations of Stevens–Johnson syndrome and toxic epidermal necrolysis. Surv Ophthalmol. 2009;54(6):686–96.
Hsu M, Jayaram A, Verner R, et al. Indications and outcomes of amniotic membrane transplantation in the management of acute Stevens–Johnson syndrome and toxic epidermal necrolysis: a case–control study. Cornea. 2012;31(12):1394–402.
Tomlins PJ, Parulekar MV, Rauz S. “Triple-TEN” in the treatment of acute ocular complications from toxic epidermal necrolysis. Cornea. 2013;32(3):365–9.
Meneux E, Paniel BJ, Pouget F, et al. Vulvovaginal sequelae in toxic epidermal necrolysis. J Reprod Med. 1997;42(3):153–6.
Niemeijer IC, van Praag MC, van Gemund N. Relevance and consequences of erythema multiforme, Stevens–Johnson syndrome and toxic epidermal necrolysis in gynecology. Arch Gynecol Obstet. 2009;280(5):851–4.
Kaser DJ, Reichman DE, Laufer MR. Prevention of vulvovaginal sequelae in Stevens–Johnson syndrome and toxic epidermal necrolysis. Rev Obstet Gynecol. 2011;4(2):81–5.
Struck MF, Illert T, Liss Y, et al. Toxic epidermal necrolysis in pregnancy: case report and review of the literature. J Burn Care Res. 2010;31(5):816–21.
Rodriguez G, Trent JT, Mirzabeigi M, et al. Toxic epidermal necrolysis in a mother and fetus. J Am Acad Dermatol. 2006;55(5 Suppl):S96–8.
Claessens N, Delbeke L, Lambert J, et al. Toxic epidermal necrolysis associated with treatment for preterm labor. Dermatology. 1998;196(4):461–2.
Vasicka AI, Lin TJ, Das BK. Erythema multiforme exudativum (Stevens–Johnson syndrome) at term pregnancy: report of one case. Obstet Gynecol. 1958;12(2):225–9.
Dube N, Adewusi E, Summers R. Risk of nevirapine-associated Stevens–Johnson syndrome among HIV-infected pregnant women: the Medunsa National Pharmacovigilance Centre, 2007–2012. S Afr Med J. 2013;103(5):322–5.
Knight L, Muloiwa R, Dlamini S, et al. Factors associated with increased mortality in a predominantly HIV-infected population with Stevens Johnson syndrome and toxic epidermal necrolysis. PloS One. 2014;9(4):e93543.
Mittmann N, Knowles SR, Gomez M, et al. Evaluation of the extent of under-reporting of serious adverse drug reactions: the case of toxic epidermal necrolysis. Drug Saf. 2004;27(7):477–87.
Sanchez-Lopez MP, Dresch V. The 12-Item General Health Questionnaire (GHQ-12): reliability, external validity and factor structure in the Spanish population. Psicothema. 2008;20(4):839–43.
Man CB, Kwan P, Baum L, et al. Association between HLA-B*1502 allele and antiepileptic drug–induced cutaneous reactions in Han Chinese. Epilepsia. 2007;48(5):1015–8.
Carr DF, Chaponda M, Jorgensen AL, et al. Association of human leukocyte antigen alleles and nevirapine hypersensitivity in a Malawian HIV-infected population. Clin Infect Dis. 2013;56(9):1330–9.
Levi N, Bastuji-Garin S, Mockenhaupt M, et al. Medications as risk factors of Stevens–Johnson syndrome and toxic epidermal necrolysis in children: a pooled analysis. Pediatrics. 2009;123(2):e297–304.
Saka B, Barro-Traore F, Atadokpede FA, et al. Stevens–Johnson syndrome and toxic epidermal necrolysis in sub-Saharan Africa: a multicentric study in four countries. Int J Dermatol. 2013;52(5):575–9.
Acknowledgments
We would like to thank Dr. Michael Shier (Colposcopy Unit, Gynaecology Services, Sunnybrook Health Sciences Centre, Toronto, ON, Canada) for his valuable advice regarding the optimal treatment of the gynecological complications of females with SJS/TEN.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for the preparation of this review.
Conflict of interest
Roni P. Dodiuk-Gad, Wen-Hung Chung, Laurence Valeyrie-Allanore, and Neil H. Shear have no conflicts of interest—including financial interests, relationships, and affiliations—that are relevant to the subject of this submission.
Rights and permissions
About this article
Cite this article
Dodiuk-Gad, R.P., Chung, WH., Valeyrie-Allanore, L. et al. Stevens–Johnson Syndrome and Toxic Epidermal Necrolysis: An Update. Am J Clin Dermatol 16, 475–493 (2015). https://doi.org/10.1007/s40257-015-0158-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40257-015-0158-0