Abstract
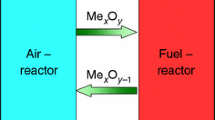
CaSO4 is an attractive oxygen carrier for chemical looping combustion(CLC) because of its high oxygen capacity and low price. The utilization of a CaSO4 oxygen carrier suffers the problems of sulfur release, and deactivation caused by sulfur loss. With respect to the fact that partial sulfur release could be recaptured and then recycled to CaSO4 by CaO sorbent, the mixture of CaSO4-CaO can be treated as an oxygen carrier. Thermodynamics of CaSO4 and CaSO4-CaO reduction by CO have been investigated in this study. The sulfur migrations, including the sulfur migration from CaSO4 to gas phase, mutual transformation of sulfur-derived gases and sulfur migration from gas phase to solid phase, were focused and elucidated. The results show that the releases of S2, S8, COS and CS2 from CaSO4 oxygen carrier are spontaneous, while SO2 can be released at high reaction temperatures above 884 °C. SO2 is the major emission source of sulfur at low CO/CaSO4 molar ratios, and COS is the major part of the byproducts as soon as the ratio exceeds 4 at 900 °C. Under CO atmosphere, all the sulfur-derived gases, SO2, S2, S8 and CS2, involved are thermodynamically favored to be converted into COS substance, and are spontaneously absorbed and solidified by CaO additive just into CaS species, which may be recycled to CaSO4 as oxygen carrier in the air reactor. But high reaction temperatures and high CO2 concentrations are adverse to sulfur capture.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Richter H. J., Knoche K. F., Reversibility of Combustion Processes, Efficiency and Costing, American Chemical Society, Washington DC, 1983
Lyngfelt A., Leckner B., Mattisson T., Chem. Eng. Sci., 2001, 56(10), 3101
Adanez J., Abad A., Garcia-Labiano F., Gayan P., Luis F., de Diego, Prog. Energy Combust. Sci., 2012, 38(2), 215
Jin H., Okamoto T., Ishida M., Energy & Fuels, 1998, 12(6), 1272
Yan C., Pan W. P., Energy & Fuels, 2006, 20(5), 1836
Anthony E. J., Ind. Eng. Chem. Res., 2008, 47(6), 1747
Shen L., Wu J., Xiao J., Song Q., Xiao R., Energy & Fuels, 2009, 23(5), 2498
Kvamsdal H. M., Jordal K., Bolland O., Energy, 2007, 32(1), 10
Qin W., Lin C. F., Cheng W. L., Xiao X. B., Chem. J. Chinese Uni-versities, 2015, 36(1), 116
Zeng L. P., Huang F., Zhu X., Zheng M., Li K. Z., Chem. J. Chinese Universities, 2017, 38(1), 115
Xiang W. G., Chen Y. Y., Energy & Fuels, 2007, 21(4), 2272
Adánez J., de Diego L. F., García-Labiano F., Gayán P., Abad A., Energy & Fuels, 2004, 18(2), 371
García-Labiano F., Adánez J., de Diego L. F., Gayán P., Energy & Fuels, 2006, 20(1), 26
Cho P., Mattisson T., Lyngfelt A., Ind. Eng. Chem. Res., 2006, 45(3), 968
Zhao H., Liu L., Wang B., Xu D., Jiang L., Zheng C., Energy & Fuels, 2008, 22(2), 898
Källén M., Rydén M., Lyngfelt A., Mattisson T., Appl. Energ., 2015, 157, 330
Zheng Y., Wang B. W., Song K., Zheng G. C., Eng. Thermophysics, 2016, 27(3), 531
Shen L., Zheng M., Xiao J., Xiao R., Combustion & Flame, 2008, 154, 489
Song Q., Xiao R., Deng Z., Zhang H., Shen L., Xiao J., Zhang M., Energ. Convers. Manage., 2008, 49(11), 3178
Song Q., Xiao R., Deng Z., Zheng W., Shen L., Xiao J., Energy & Fuels, 2008, 22(6), 3661
Song Q., Xiao R., Deng Z., Shen L., Xiao J., Zhang M., Ind. Eng. Chem. Res., 2008, 47(21), 8148
Tian H., Guo Q., Yue X., Liu Y., Fuel Process Technol., 2010, 91(11), 1640
Liu S., Lee D., Liu M., Li L., Yan R., Energy & Fuels, 2010, 24(12), 6675
Zheng M., Shen L., Xiao J., Int. J. Greenh. Gas Con., 2010, 4(5), 716
Xiao R., Song Q., Combust. Flame, 2011, 158(12), 2524
Ding N., Zheng Y., Luo C., Wu Q., Fu P., Zheng C., Chem. Eng. J., 2011, 171(3), 1018
Zheng M., Shen L., Feng X., Energ. Conver. & Manage., 2014, 83, 270
Wang J., Anthony E. J., Appl. Energ., 2008, 85(2/3), 73
Cheng J., Zhou J., Liu J., Zhou Z., Huang Z., Cao X., Zhao X., Cen K., Prog. Energy & Combust. Sci., 2003, 29, 381
Anthony E. J., Granatstein D. L., Prog. Energy Combust. Sci., 2001, 27(2), 215
Teaching and Research Group of General Chemistry, General Che-mistry 4th Ed., Higher Education Press, Beijing, 1995, 28
Mattisson T., Lyngfelt A., Energy & Fuels, 1998, 12(5), 905
Okumura S., Mihara N., Kamiya K., Ozawa S., Maurice S., Onyango, Kojima Y., Matsuda H., Ind. Eng. Chem. Res., 2003, 42(24), 6046
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(Nos.51306084, 51374004), the Scientific and Technological Leading Talent Projects in Yunnan Province, China(No.2015HA019), and the Natural Science Foundation of Kunming University of Science and Technology, China(No.KKZ3201352030).
Rights and permissions
About this article
Cite this article
Zhong, S., Zheng, M., Pu, S. et al. Thermodynamics on sulfur migration in CaSO4 oxygen carrier reduction by CO. Chem. Res. Chin. Univ. 33, 979–985 (2017). https://doi.org/10.1007/s40242-017-6457-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-017-6457-7