Abstract
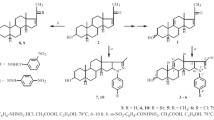
Three series of novel steroidal thiazole derivatives were synthesized by microwave assisted one-pot reaction from pregnenolone, testosterone and estrone, respectively. Their structures were characterized by IR, NMR and HRMS, and the antiproliferative activities of all the synthesized compounds against human cervical carcinoma (HeLa), human liver carcinoma(HepG2), human lung carcinoma(A549), nasopharyngeal carcinoma(CNE2) and normal kidney epithelial cells(HEK293T) were screened. Among the compounds, 1-estron-17′-ylidene-2-[4′-(p-bromophenyl)-2′-thiazol] hydrazone(12) displayed distinct antiproliferative activity against all the tested cancer cell lines and was almost inactive to normal kidney epithelial cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Singh H., Jindal D. P., Yadav M. R., Kumar M.; Eds. Ellis G. P., West G. B., Progress in Medicinal Chemistry, Vol. 28, Elsevier Science Publishers, Amsterdam, 1991, 233
Singh H., Kapoor V. K., Paul D.; Eds. Ellis G. P., West G. B., Progress in Medicinal Chemistry, Vol. 16, Springer, Amsterdam, 1979, 135
Cui J. G., Liu L., Gan C. F., Xiao Q., Huang Y. M., Progr. Chem., 2014, 26(2/3), 320
Cui S. F., Wang Y., Lv J. S., Damu G. L. V., Zhou C. H., Scientia Si-nica Chimica, 2012, 42(8), 1105
Lesyk R., Zimenkovsky B., Atamanyuk D., Jensen F., Kononowicz K. K., Gzella A., Bioorg. Med. Chem., 2006, 14, 5230
Kashfi K., Adv. Pharmacol., 2009, 57, 31
Sun L. P., Jiang Z., Gao L. X., Liu X. F., Quan Y. C., Zheng G. H., Li J., Piao H., Chin. J. Org. Chem., 2013, 33, 1496
Mohareb R. M., Al-Omran F., Steroids, 2012, 77(14), 1551
Chen C. J., Song B. A., Chinese J. Pesticides., 2005, 44(2), 53
Mohareb R. M., Wardakhan W. W., Elmegeed G. A., Ashour R. M. S., Steroids, 2012, 77(14), 1560
Bao M. K., Yang Y. Q., Gu W., Xu X., Cao M. Z., Wang S. F., Chin. J. Org. Chem., 2014, 34, 2146
Mohareb R. M., Zaki M. Y., Abbas N. S., Steroids, 2015, 98, 80
Mohareb R. M., Al-Omran F., Azzam R. A., Steroids, 2014, 84, 46
Cui J. G., Qi B. B., Gan C. F., Liu Z. P., Huang H., Lin Q. F., Zhao D. D., Huang Y. M., Mar. Drugs, 2015, 13, 2488
Cui J. G., Liu L., Zhao D. D., Gan C. F., Huang X., Xiao Q., Qi B. B., Yang L., Huang Y. M., Steroids, 2015, 95, 32
Ahluwalia V. K., Chibber S. S., Goyal B., Indian J. Chem. Sec. B, 1996, 35(8), 856
Yuan M. Y., Liu W. H., Zhang Y. Q., Yan H. F., Zhang D. N., Liu H. M., Wang J. P., Chin. J. Org. Chem., 2013, 33, 1108
Cui J. G., Zhao D. D., He D. M., Huang Y. M., Liu Z. P., Lin Q. F., Shi H. S., Gan C. F., Chin. J. Org. Chem., 2016, 36(3), 630
Cui J. G., Fan L., Huang L. L., Liu H. L., Zhou A. M., Steroids, 2009, 74(1), 62
Cui J. G., Fan L., Huang Y. M., Xin Y., Zhou A. M., Steroids, 2009, 74(12), 989
Gan C. F., Cui J. G., Su S. Y., Lin Q. F., Jia L.Y., Fan L. H., Huang Y. M., Steroids, 2014, 87, 99
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(Nos.21462009, 21562007) and the Foundation of Guangxi Colleges and University Key Laboratory of Beibu Gulf Oil and Natural Gas Resource Effective Utilization, China(No. 2016kLOG10).
Rights and permissions
About this article
Cite this article
Huang, Y., Zhao, D., Liu, C. et al. Rapid microwave assisted synthesis and antiproliferative evaluation of novel steroidal thiazole derivatives. Chem. Res. Chin. Univ. 33, 206–212 (2017). https://doi.org/10.1007/s40242-017-6372-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-017-6372-y